CHAPTER 122 Complications of Neck Surgery
Complications of neck surgery affect every surgeon regardless of experience and technical skill. Despite our best intentions, complications arise due to the intricate anatomy of the neck and complexities of the diseases and patients treated. Factors such as medical comorbidities (hypertension, hepatitis, etc.), substance abuse, prior radiation therapy, aerodigestive tract entry, prolonged anesthesia times, the volume of intraoperative fluid administered, and the need for microvascular free flap reconstruction have been associated with head and neck surgical complications in various studies.1–4 Prevention and management of complications that occur both intraoperatively and postoperatively are essential to proper head and neck surgical care.
The history of neck dissection reveals a continuing quest to reduce complications and their sequelae.5 The results of a systematic approach to cervical lymphadenectomy for the treatment of a series of patients with head and neck cancer was first reported by George Crile, MD, in 1906.6 In that era, radical neck dissection (RND), with removal of the sternocleidomastoid muscle (SCM), the internal jugular vein (IJV), and the spinal accessory nerve (SAN), was considered the standard of care for patients with neck metastasis but resulted in significant functional and cosmetic morbidities.7 Introduction of the modified radical neck dissection (MRND) and proof that it was oncologically sound led to improved functional outcomes without sacrifice of oncologic effectiveness.8–11 Further technique refinements that established selective neck dissections (SND) were based on predictable patterns of spread from specific primary tumor locations.12,13 This approach is highly effective for management of the clinically negative neck and may have applications in clinically positive necks as long as postoperative radiation therapy is administered.14–17 In addition, SND may be the preferred approach for planned neck dissection after concurrent chemoradiation for advanced head and neck cancer because these patients have a low incidence of recurrence in the neck.18 As head and neck surgical techniques have evolved, so have the type and frequency of complications. Shoulder disability from spinal accessory nerve injury was an expected sequela of RND but is now considered a potential complication of MRND. Internal jugular vein thrombosis and hemorrhage may occur more frequently in the modern era because this structure is more commonly preserved with MRND and SND.19
Wound Complications
Incision Planning
Carefully designed neck incisions must take into account the location and extent of the primary tumor; adequate exposure for the type of neck dissection planned; carotid artery coverage; excision of previously violated skin, when applicable; and cosmesis. Preferred incisions combine limbs that meet at right angles to create skin flaps that preserve the blood supply to neck skin, which includes branches of the facial and occipital arteries superiorly and branches of the transverse cervical and suprascapular arteries inferiorly.20 Subplatysmal flap elevation should be used in all approaches to preserve the skin flap blood supply.
Numerous neck incisions for neck dissection have been described. Early designs such as the Crile single-Y and the Martin double-Y provided excellent exposure but placed a trifurcation over the carotid artery. Furthermore, they created tenuous posterior skin flaps and anterior skin flaps that relied partially on blood flow from branches of the contralateral external carotid artery. The Conley-modified Schobinger approach was an improvement, maintaining excellent exposure for comprehensive neck dissection with creation of a large, robust anterior flap and placement of the trifurcation posterior to the carotid artery.20 MacFee introduced the use of two transverse incisions that appear to transect the dominant blood supply, but studies have shown that the central, bipedicled skin flap is resistant to ischemia and may be the preferred approach in the previously irradiated patient.21,22 Certain incisions are well-suited to accompany the extirpation of specific primary tumors such as the apron flap for neck dissection with laryngectomy and the Attie incision for neck dissection with thyroidectomy.23
Wound Dehiscence and Flap Necrosis
Flap survival depends on incision design, surgical technique, and factors intrinsic to the patient. A study of 184 neck dissections using triradiate, modified MacFee, or apron incisions showed a wound dehiscence rate of 11%, 8%, and 0%, respectively, for each approach.24 The same study showed a statistically significant increase in the overall rate of wound dehiscence in previously irradiated necks, particularly for the triradiate group, leading the authors to recommend the apron flap for neck dissection in patients who have received prior radiation therapy.24 Whether or not preoperative radiation therapy increases the complication rate of neck dissection continues to be debated, but several reports indicate that higher total radiation doses are associated with a higher rate of wound complications.25,26 Skin loss from involvement with tumor or excision of violated skin may lead to increased tension on the closure. Poor microvasculature, secondary to malnutrition, active tobacco use, or uncontrolled diabetes mellitus, contributes to the lack of tissue tolerance for surgical manipulation and a higher rate of wound dehiscence or flap necrosis.
Seroma
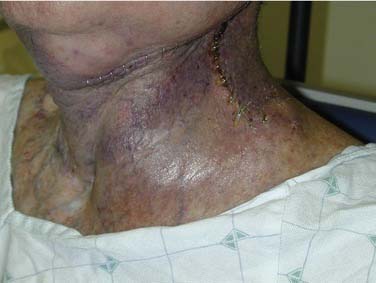
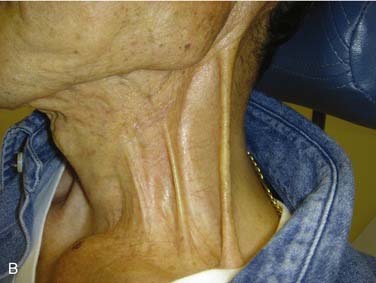
Division of lymphatic and adipose tissue during neck dissection allows for formation of seroma, the collection of serous fluid in the potential space between cervical skin flaps and underlying structures. Seromas present as enlarging masses under the skin, with or without overlying induration or erythema. Loss of the expected postoperative scaphoid appearance of the neck following neck dissection is cause for suspicion (Fig. 122-1). If left untreated, a seroma may lead to flap necrosis or infection. Many case series report seroma and hematoma formation together, with rates ranging from 3% to 10% with RND to as high as 20% with functional neck dissection.27–29 These differences may be explained by the increased amount of native tissue preserved in MRND or SND compared with RND that can weep serous fluid into the wound bed. Causes of seroma include incorrect drain placement, drain failure, or early drain removal. Prevention consists primarily of proper management of closed suction drains that are left in place until the total output per drain falls below 25 mL in a 24-hour period. Some authors also advocate the routine use of fibrin glue, which has been shown in animal models to prevent seroma formation.30 Some small seromas can be observed with anticipation of gradual resorption. In most cases, however, management of seroma includes needle aspiration and, in select patients, drain replacement. Pressure dressings do not appear to prevent fluid reaccumulation.
Wound Infection
Aerodigestive tract entry is the single most important factor that contributes to the risk of wound infection. Neck dissection without exposure to oral flora is considered a clean procedure. In a study of 438 patients undergoing parotidectomy, thyroidectomy, or submandibular gland excision, three (0.7%) patients developed a wound infection, one of whom had received perioperative erythromycin.31 Administration of prophylactic antibiotics for clean neck dissections is controversial. One study showed a 10% rate (9/92) of infection for those not receiving antibiotics compared with 3.3% (3/93) of patients receiving first-generation cephalosporins, clindamycin, or pencillin.32 Although the difference did not reach statistical significance, the trend may justify its use.32 Antibiotic prophylaxis in this setting has been shown to be cost-effective.33 In terms of duration, no difference in the rate of infection was seen with 1-day versus 4- or 5-day antibiotic regimens for clean procedures.34,35 Interestingly, significantly higher infection rates for RND compared with other clean surgical procedures have been documented, for reasons that remain unclear.35
The risk of wound infection increases significantly with entry into the oral cavity, pharynx, or larynx, ranging from 15% to 87%.36,37 Factors associated with wound infection include the performance of bilateral neck dissections, total laryngectomy, advanced-stage tumors, and in some studies, a history of prior tracheotomy.35 The benefit of antibiotic prophylaxis in surgery that required aerodigestive tract entry was demonstrated convincingly in a well-designed prospective clinical trial in which 20/23 (87%) patients who received a placebo developed an infection compared with 12/32 (38%) in a cefazolin-treated group.36 In another prospective, randomized clinical trial of four antibiotic regimens, statistically significant reductions in the infection rate were seen with gentamicin-clindamycin (4% to 7%) compared with cefazolin (20% to 33%), demonstrating the importance of broad-spectrum coverage that includes gram-negative and anaerobic organisms.37 A trend toward fewer infections with a 5-day course compared with 1 day of antibiotics was found.37 Cefazolin-metronidazole prophylaxis is a suitable alternative regimen to gentamicin-clindamycin and avoids the potential ototoxicity associated with gentamicin.38
Salivary Fistula
Saliva may enter the wound from either aerodigestive tract communication or parotid leak (Fig. 122-2). With associated infection, an inflammatory response in adjacent tissue rapidly occurs. Erythema, edema, and induration of skin flaps, as well as a change in the character of drain output to that of saliva or purulent discharge, are common signs.
The risk of pharyngocutaneous fistula formation with aerodigestive tract entry during surgery varies from 10% to 30%.39,40 Prevention consists of water-tight, inverted, and tension-free closure during the primary surgical procedure and consideration of vascularized tissue coverage if the closure appears tenuous. Conservative management with broad-spectrum antibiotics and continued closed drainage is usually successful, although one should have a low threshold for opening a suture line to divert drainage away from the major blood vessels and gently packing the wound.19 Nutritional status and glucose control should be optimized and thyroid-stimulating hormone levels checked, particularly in previously irradiated patients. Fistulae refractory to conservative measures require closure with vascularized tissue, either local-regional flaps or free flaps.
Postparotidectomy fistula does not carry a risk of major blood vessel rupture but can be annoying to patients. Most are small-volume leaks that exit the divided inferior cervical portion of the parotid gland. These normally respond well to conservative management.41 Although numerous treatment modalities for persistent leaks have been proposed, injection of the parotid gland with botulinum toxin is usually effective.42,43
Scar Formation
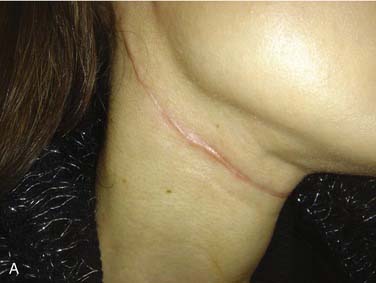
Cosmetic principles that apply to neck surgery include placement of incisions in natural skin creases or relaxed skin tension lines, using gently curved vertical limbs to avoid linear scar contracture, and performance of a tension-free, everted skin closure.44 Lapses in meticulous technique or postoperative wound infection may lead to the development of a hypertrophic scar (Fig. 122-3A). Formation of a keloid—scar tissue that exceeds the boundaries of the original incision—usually occurs in patients with a personal or family history of exuberant scar formation. Web formation along linear vertical limbs in the lateral neck may also occur (see Fig. 122-3B). Unilateral deep plane neck lift and midline platysmaplasty can be considered for rehabilitation of post–neck dissection asymmetry.45
Vascular Complications
Carotid Artery Hemorrhage
Acute postoperative carotid artery rupture, or “blowout,” occurs in 3% to 4% of RNDs and is associated with a mortality rate of approximately 50%.46–48 Factors associated with carotid artery hemorrhage include wound breakdown, necrosis, and infection; pharyngocutaneous fistula; prior radiation therapy; tumor involvement of the arterial wall; and rough handling of the adventitia during dissection.47,48 Close surveillance of dehiscent or infected wounds for lack of improvement and signs of impending carotid rupture such as carotid exposure or a sentinel bleed is critical to postoperative management of the at-risk patient. This is because the mortality of elective carotid artery ligation is significantly lower than for emergency ligation,49 rising from 14% to 64%, respectively, in one series.50
Emergent treatment consists of control of hemorrhage with application of direct pressure to the area of rupture and expeditious return of the patient to the operating room for definitive control. Fluid resuscitation with blood products and crystalloid is performed. Separate incisions are made inferior and superior to the region of infected or necrotic tissue, and proximal and distal vessel control is obtained.51 Vessel ligation away from the contaminated wound is preferred in order to prevent rebleeding. The central wound is left packed or is covered with vascularized tissue. Endovascular techniques including angiography with selective embolization and temporary stent grafts for patients at high risk for stroke have been described.52,53
Internal Jugular Vein Complications
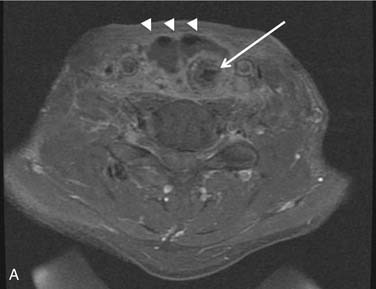
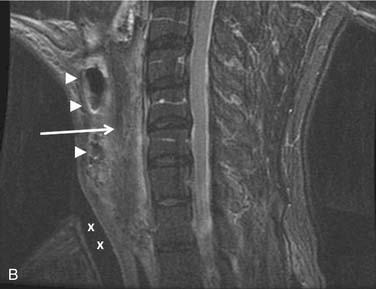
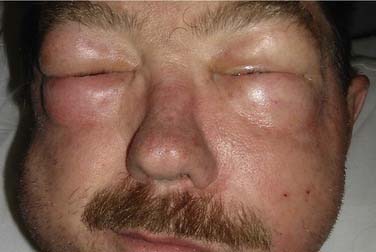
The theoretic benefits of preserving the internal jugular vein (IJV) during unilateral MRND or SND include decreased incidence of edema and less morbidity if a contralateral RND becomes necessary in the future,54 although this practice has not been evaluated by any outcome studies. Preservation of at least one IJV during bilateral neck dissection is clearly beneficial to avoid facial or laryngeal edema, intracranial pressure elevations, stroke, and blindness (Fig. 122-4).55 Techniques for reconstruction of the IJV from either the external jugular vein or a greater saphenous vein graft have been described56 but are not used routinely. The widespread use of MRND and SND with IJV preservation has made possible other complications including IJV thrombosis and hemorrhage.
The IJV remains patent in approximately 86% of patients after MRND in studies using preoperative and postoperative imaging with CT or MRI.54,57 Factors that predispose to immediate IJV thrombosis include damage to the adventitia, desiccation, and improper ligation of IJV branches intraoperatively, as well as the presence of central venous catheters.19,58 One study of patients undergoing MRND examined with retrograde venography within 1 month postoperatively demonstrated IJV occlusion in 8/27 (29.6%) of operated necks.59 Five of these patients had postoperative courses complicated by wound infection or salivary fistula, indicating that wound complications may lead to a high rate of IJV thrombosis.59 Furthermore, postoperative radiation therapy may contribute to IJV thrombosis. An ultrasound study of patients undergoing MRND alone, radiation therapy alone, or MRND plus postoperative radiation therapy showed a normal-appearing vein when compared with the contralateral, untreated IJV in 88%, 57%, and 18% of patients, respectively.60 In the long term, the rate of IJV thrombosis may be significantly lower, only 5.8% in one ultrasound study, indicating that many thrombosed veins will recanalize with reestablishment of patency.61
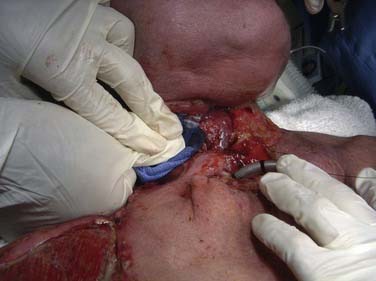
Internal jugular vein hemorrhage is rare, occurring in less than 1.3% of MRND with concurrent laryngectomy or pharyngectomy.62 Although often less severe than arterial bleeding, this event can be just as life-threatening (Fig. 122-5). Similar to carotid artery bleeding, the major risk factors for this complication include salivary fistula, significant tobacco use, and poor nutritional status.63 Careful handling of the IJV and avoidance of circumferential dissection in cases involving aerodigestive tract entry may help in its prevention.63
Blindness, an exceedingly rare complication of neck dissection, may occur following unilateral or, more commonly, bilateral RND.64,65 Prolonged intraoperative hypotension, increased cerebrospinal fluid pressure, and venous pressure on the optic nerve may be causative factors.66
Hematoma
Hematoma occurs in approximately 1% of neck dissections and 4% of major head and neck surgeries.67,68 Hematoma can usually be differentiated from seroma by the presence of skin ecchymosis, firmness to palpation, or clotted drain output, although both complications can cause elevation of skin flaps and loss of the expected scaphoid contour of a dissected neck. Failure to recognize and properly treat a neck wound hematoma results in increased wound complications.67 Even a small venous bleed or hematoma in an undesirable location, for example, in proximity of the vascular pedicle of a free flap reconstruction, can have serious consequences. Prevention consists of preoperative avoidance of anticoagulants and antiplatelet agents and meticulous intraoperative hemostasis. Treatment should include prompt evacuation of clotted blood, at the bedside if necessary, to prevent upper airway edema, and return of the patient to the operating room for wound exploration and control of any bleeding vessels.