CHAPTER 146 Cochlear Transduction and the Molecular Basis of Auditory Pathology
Deafness, in all of its forms, affects an estimated 186 of every 100,000 newborns in the United States372 and increases in prevalence with advancing age. For example, by the age of 60 years, one of every three people in the United States experiences communication difficulties as a consequence of lost hearing, and by the age of 85 years, one half of the population has some degree of hearing disability. In most cases, peripheral auditory disease involves some form of cochlear abnormality that disrupts sensory transduction, and various etiologic factors have been recognized, including genetic abnormalities, trauma (acoustic and mechanical), ototoxic agents, bacterial and viral infections, and disturbances in other organ systems that influence cochlear physiology indirectly. This chapter describes the physiologic mechanisms underlying cochlear transduction at both the systemic and molecular levels. In addition, basic mechanisms of cochlear transduction are correlated with a subset of inner ear disorders that are representative of various classes of disease.
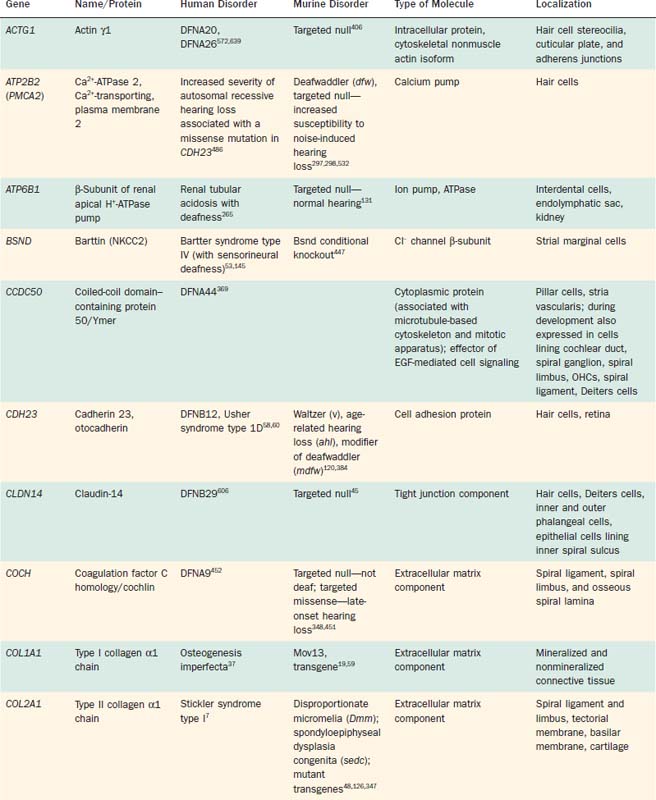
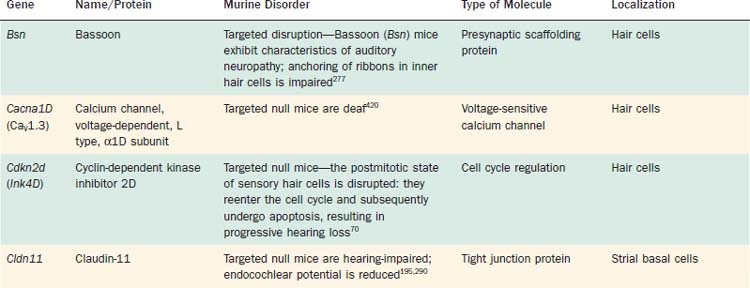
Significant breakthroughs in the identification of genes and gene products that serve as integral elements of cochlear transduction have been made beginning in the mid to late 1990s. Clearly, pathogenic mutations of many of these genes are implicated in various forms of deafness (Table 146-1). Although numerous cases of inherited deafness had been identified historically on the basis of phenotype, the etiopathogenic mechanisms responsible for deafness remained unknown before technologic advances allowed for the identification and cloning of genes associated with normal hearing. By comparing human genes with orthologous genes that are known to cause deafness in animals, the cell biology and disease associated with particular gene mutations can be studied, leading to an understanding of the role of gene products in sensory transduction. Although a variety of nonhuman species have played indispensable roles in the study of cochlear transduction and sensory pathology, in recent years the mouse model has emerged as particularly useful (Table 146-2; see also Table 146-1). This movement is fueled, in large part, by the highly successful, comprehensive investigation of the murine genome and the ever-expanding availability of tools that allow the targeting and manipulation of specific genes, technologic changes that greatly extend the capability of researchers to examine the role of gene products in the transduction process. However, although much has been learned about the molecular and genetic basis of cochlear function, or malfunction as the case may be, much work remains to be done in the ongoing effort to unravel the intricacies associated with cochlear transduction and deafness.
Table 146-2 Selected Genes Underlying Murine Deafness for Which Human Deafness Has Not Been Determined
Approximately 68% of all cases of congenital hearing loss are inherited, and of these, at least 70% are nonsyndromic—that is, auditory disability is the only manifestation of the disorder.372 Nonsyndromic hearing losses are grouped according to their inheritance mode: DFNA refers to autosomal dominant cases, DFNB to autosomal recessive, and DFN to X-linked or mitochondrial cases; a unique numeral following each acronym has been used to identify each chromosomal locus discovered in chronologic order. As a consequence of a major effort to identify and classify inherited hearing disabilities, it is clear that the nonsyndromic basis of inherited deafness is highly heterogeneous. Many loci known to produce autosomal dominant forms of deafness when mutated have been genetically mapped, although only a subset of the genes responsible for autosomal dominant forms have been identified and cloned. Autosomal recessive forms of inherited hearing loss account for most nonsyndromic abnormalities (approximately 80%). It has become increasingly clear, however, that different mutations in a given gene may be responsible for differences in the severity of hearing loss, different patterns of inheritance, and susceptibility to environmental factors such as noise or aminoglycoside antibiotics, as well as the involvement of other organ systems. For example, mutations in a number of genes (e.g., COL11A2, ESPN, GJB2, GJB3, GJB6, MYO6, MYO7A, PJVK, TECTA, TMC1) are responsible for both autosomal dominant and autosomal recessive forms of nonsyndromic deafness. Moreover, mutations in several genes (e.g., CDH23, COL11A2, GJB2, GJB3, GJB6, MYH9, MYO7A, MYO15A, PCDH15, SLC26A4, USH1C, WFS1, WHRN) are responsible for both nonsyndromic and syndromic forms of deafness (see Table 146-1). The phenotypic spectrum produced by mutations in a gene has been attributed to the severity of the mutation, allelic heterogeneity, alternative splicing, and the contribution of modifier genes.60,136,171,254,336,362,486
This chapter reviews basic mechanisms underlying sensory transduction associated with hearing and integrates contemporary models of transduction with the latest acquired information regarding the molecular structure and organization of key inner ear systems. Because of the sheer number of factors now known to be necessary for normal inner ear development, maintenance, and function, a comprehensive accounting is not possible here—not all such factors are addressed. Of note, the stated biologic role of most of the proteins discussed here remains speculative, and substantial work is needed to determine their precise role in the inner ear, as well as the nature of their interactions with other proteins. Excellent reviews that are relevant to the subject of this chapter are listed in the Bibliographic References.29,61,170,187,215,236,310,320,443,453,585,588,631
Passive Cochlear Mechanics
The cochlea, and the organ of Corti in particular, constitute an evolutionary marvel of biologic engineering as intricate, microscaled mechanical systems. Although the hallmark of peripheral auditory function in mammals may be the evolutionary adaptation of a relatively simple, passive resonant system into an active one that consumes energy and efficiently detects and amplifies vibratory energy, it is useful to note that the fundamental nature of cochlear mechanics is evident even in cadavers, as so elegantly demonstrated by Nobel prize winner Georg von Békésy.586 Because cochlear mechanics at this level of function are independent of other factors—neither requiring nor consuming adenosine triphosphate (ATP), for example—the sound-driven motion of the organ of Corti observed by von Békésy and others is commonly referred to as passive.
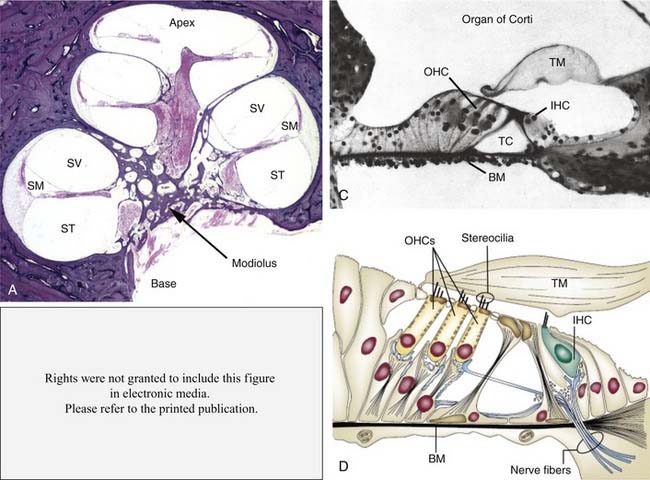
The primary elements of passive cochlear transduction are hydromechanical in character, and the integrated contributions of many structural elements are known to determine the resonant character of cadaveric and therefore mechanically passive cochleae (Fig. 146-1). The cochlear partition is composed of an epithelial sheet known as the tympanic cover layer that supports the fibroelastic basilar membrane, itself the central structure of the end organ both functionally and anatomically. This partition supports, in turn, both the sensory and nonsensory supporting epithelium. The fluid-filled major chambers of the cochlea—specifically, the scala vestibuli and scala tympani, which are filled with perilymph, and the scala media, which is filled with endolymph—also play an important role in transduction. These structural elements, together with the tectorial membrane (an extracellular gelatinous structure secreted by nonsensory inner ear epithelial cells) that extends from the spiral limbus radially over the apical surface of the organ of Corti, comprise the principal elements that give rise to passive mechanical transduction. Although Reissner’s membrane separates scala media from scala vestibuli and therefore plays an important homeostatic role in the maintenance of cochlear electrochemistry, it does not seem to play a significant role as an element of passive cochlear mechanics.
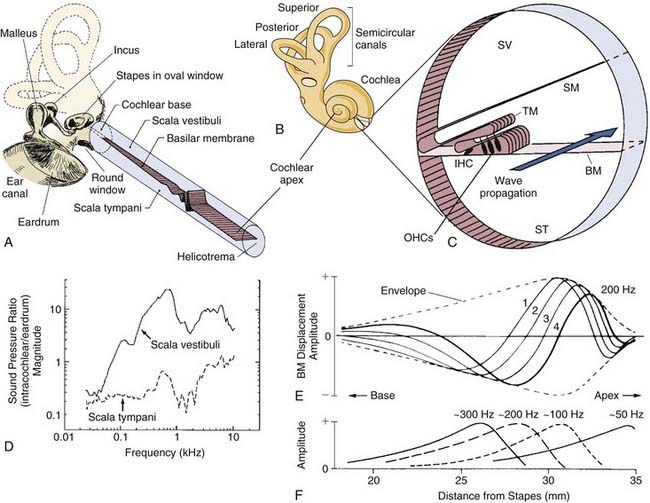
Because the focus of this chapter is on cochlear transduction, we will only remind the reader that sound waves collected in the external ear canal produce vibrations of the tympanic membrane, which in turn vibrate middle ear ossicles (Fig. 146-2A). It is, of course, the vibratory motion of the stapedial footplate that transmits the mechanical energy of the ossicular chain directly through the oval window of the cochlea, effectively delivering sound pressure waves to the scala vestibuli and translating mechanical motion into pressure waves that propagate through the virtually incompressible cochlear fluids at a velocity approximating 1.5 km/sec. At this rate of propagation, the spread of the pressure wave throughout the volume of the cochlea is nearly instantaneous. Because the bony walls of the cochlea are rigid and its fluid contents incompressible, the higher pressure (or lower, depending on the direction of motion of the stapedial footplate) in scala vestibuli relative to scala tympani produces a pressure differential across the cochlear partition that creates intracochlear forces that set the partition into motion (see Fig. 146-2D). As described in more detail in a later section, the physical makeup of the organ of Corti, and of the basilar membrane in particular, establishes a space-frequency map that determines the limits of hearing and the capacity of the subject to resolve frequency differences among vibrations that contain energy in the audible range.
Physical Dimensions
At the basal end of the cochlea near the round window, the width of the basilar membrane, as measured between the tympanic lip of the spiral limbus and the basilar crest of the spiral ligament, is narrow relative to dimensions representing more apical locations (Fig. 146-3). In fact, the width of the basilar membrane progressively increases along a base-to-apex gradient in mammals, such that in humans, for example (see Fig. 146-3 E), the width at the base is approximately 100 µm and approaches 500 µm near the apex.604 In association with graded basilar membrane width, the number of mesothelial cells, or tympanic border cells, lining the basilar membrane increases along the basoapical length of the end organ, as does the length of outer hair cells (OHCs) and associated stereocilia. Conversely, the thickness of the basilar membrane and the density of radial filaments embedded in the homogenous ground matrix of the partition decrease progressively from base to apex (see Fig. 146-3C). It is notable that radial filaments are composed primarily of collagen type II,132,545,558 whereas the ground matrix of the partition itself is composed primarily of fibronectin, tenascin, proteoglycans containing keratin sulfate and chondroitin sulfate, and emilin-2 (elastin microfibril interface-located protein-2).21,375,475,539,611 Consequently, the basilar membrane and organ of Corti complex is stiffer and less massive at the base than at the apex (see Fig. 146-3F). The result is an end organ with resonant properties that are continuously distributed from base to apex that supports a mechanical system that separates, or filters, complex fluid waves into sinusoidal constituents. As a result, high-frequency acoustic events are preferentially transduced in the base (see Fig. 146-3G), because it is stiff and less massive. The inverse is true in the case of low-frequency acoustic events.
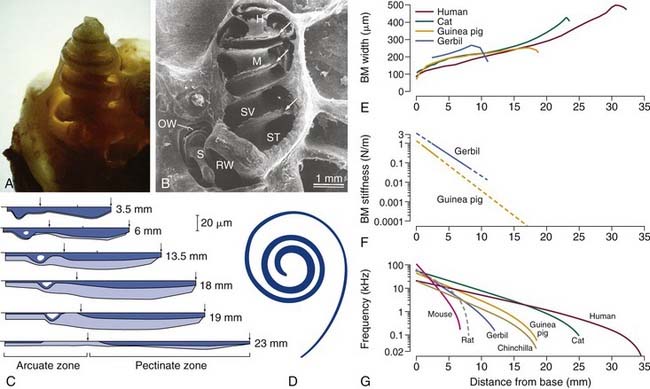
Figure 146-3. A, Photograph of the cochlea of a guinea pig after the outer bony shell has been thinned. B, Scanning electron micrograph (SEM) of a chinchilla cochlea after the bone on one side has been removed. The bony modiolus (M) is shown in the center, and the fluid-filled scala tympani (ST) and scala vestibuli (SV) are indicated in the basal turn. The helicotrema (H) is shown at the apex, and the arrows indicate the osseous spiral lamina to which the cochlear partition is attached at its medial extent. The round window (RW) and the stapes (S) are indicated, and the footplate of the stapes has been pulled away slightly, exposing the oval window (OW). C, Schematic illustration of the variation in thickness of the basilar membrane (dark blue area) and the associated mesothelial layer (light blue area) and the width of the basilar membrane (horizontal axis) at six locations along the cochlear spiral of the cat; specified locations are relative to the base. The demarcation between the arcuate and pectinate zones is indicated by the left arrow at each location; the arcuate zone (pars tecta) extends from the lip of the osseous spiral lamina to the foot of the outer pillar cell (OPC) and the pectinate zone from the OPC to the basilar crest of the spiral ligament (right arrows). D, Scaled representation of the ribbon-like nature of the human basilar membrane indicating its width relative to its length. E, Quantitative relationship between the width of the basilar membrane (BM) and the distance from the base for the human,604 cat,64 guinea pig,154 and gerbil.445 F, Estimates of the stiffness of the basilar membrane in the guinea pig205 and gerbil139 as a function of location. G, Maps of the characteristic frequency (CF) of a cochlear location (i.e., peak of the envelope of the traveling wave) as a function of position along the basilar membrane for several species. Note that the lengths of the basilar membrane for each species can be estimated by the distance that each curve occupies along the x-axis. Frequency-position maps were based on a formulation developed by Greenwood196 using empirical fits to data for the species shown here, except for rat, which was developed by Muller.374
(A, From Wasterstrom SA. Accumulation of drugs on inner ear melanin. Therapeutic and ototoxic mechanisms. Scand Audiol Suppl. 1984;23:1; B, from Harrison RV, Hunter-Duvar IM. An anatomical tour of the cochlea. In: Jahn AF, Santos-Sacchi J, eds. Physiology of the Ear. New York: Raven Press; 1988:160; C, from Cabezudo LM. The ultrastructure of the basilar membrane in the cat. Acta Otolaryngol. 1978;86:160; D, from Wever EG. Theory of Hearing. New York: Dover Publications; 1949.)
Another physical characteristic of the cochlea that exerts influence over the mechanics of transduction is the relative volume of perilymphatic fluid compartments that are larger at the base than at the apex (see Fig. 146-1). Consequently, there is a clear difference in fluid-mass loading of basilar membrane motion along the length of the end organ. The magnitude of the intracochlear fluid load is thought to dominate longitudinal coupling along the length of the cochlea. Of note, very little coupling occurs otherwise between adjacent regions of the basilar membrane—beyond, that is, simple stretching and compression forces.204,394,395,583 At rest, therefore, the basilar membrane seems to be under very little tension, and without direct mechanical coupling, the traveling wave cannot propagate further apically after reaching its resonant peak, or place. Moreover, the viscous friction between the basilar membrane and cochlear fluids produces a damping effect that limits its displacement.
The Traveling Wave
The hydromechanical consequences of introducing a pressure pulse, or wave, into perilymphatic fluids is a traveling wave that propagates along the organ of Corti from the base to the apex (see Fig. 146-2). The traveling wave propagates basoapically, because the stiffness of the cochlear partition decreases longitudinally from the base to the apex (see Fig. 146-3F) as its mass increases. The motion and direction of the wave that travels along the basilar membrane are independent of the location of the vibratory source. von Békésy586 observed this feature of cochlear mechanics by introducing a pressure pulse through an artificial window carved into the bony labyrinth near the cochlear apex. Traveling waves created under these conditions are essentially indistinguishable from traveling waves triggered by ossicular vibration. The basoapical propagation of the traveling wave is an outcome of the end organ’s physical makeup, with the stiffer and less massive elements forming the base of the organ of Corti being set into motion by an instantaneous pressure difference between the major scalae before the more compliant and massive sections of the apical cochlea. The resultant traveling wave reflects the more or less independent, yet orderly, sequential movement of loosely coupled basilar membrane segments that form a frequency decomposition system generally thought of as the space-frequency or tonotopic map (see Fig. 146-3G).
As the traveling wave propagates toward the apex, traveling initially at a high rate of speed, the velocity of the wave decreases, with production of progressively shorter wavelengths for any given input frequency as a consequence (see Fig. 146-2E). As the amplitude of the propagating wave steadily increases as it passes toward the apex, partition displacement reaches a maximum value at its characteristic place—where intrinsic resonance meets, and nearly matches, the vibratory frequency of the triggering acoustic event (see Fig. 146-2F). As pointed out previously, the stiffness of the basilar membrane decreases exponentially with distance from the stapes,205,394,395,586 so it is not surprising that the resonant frequency also decreases exponentially as a function of distance from the stapes, because the characteristic place is determined by the stiffness of the cochlear partition. Thus, the aforementioned space-frequency, or tonotopic, map is created, whereby the peak in vibration amplitude is located at the base of the cochlea for high frequencies and at progressively apical regions for lower frequencies.
At the apical end of the cochlea, the endolymphatic space terminates just short of the bony labyrinthine wall and maintains a firm physical boundary between endolymph and perilymph, allowing the fluids in the scala vestibuli and scala tympani to communicate directly by way of a small duct called the helicotrema (see Fig. 146-3B). The helicotrema acts as an acoustic shunt across the cochlear partition that reduces the pressure difference between the scalae produced by very-low-frequency stimulation.96 The size of the helicotrema is thought to determine the low frequency cutoff of the system; that is, large openings shunt pressure waves extending to higher frequencies than those associated with small helicotrema. The shunting effect of helicotrema reduces the pressure differential across the cochlear partition, and it is this aspect of cochlear design that is thought to decrease damage to the cochlea produced by intense low-frequency pressure fluctuations.403
Active Cochlear Mechanics
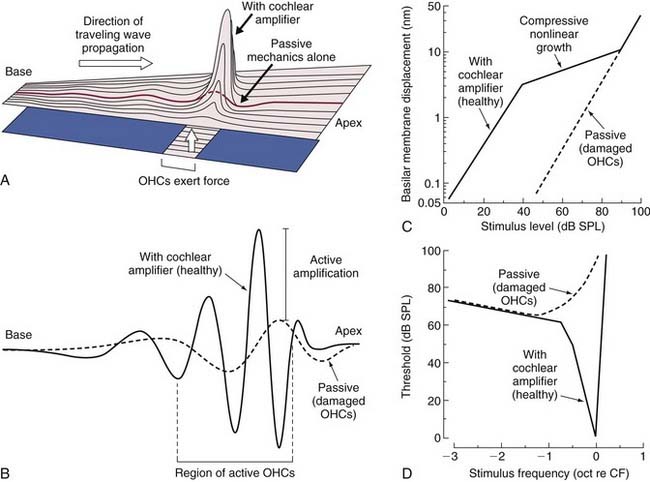
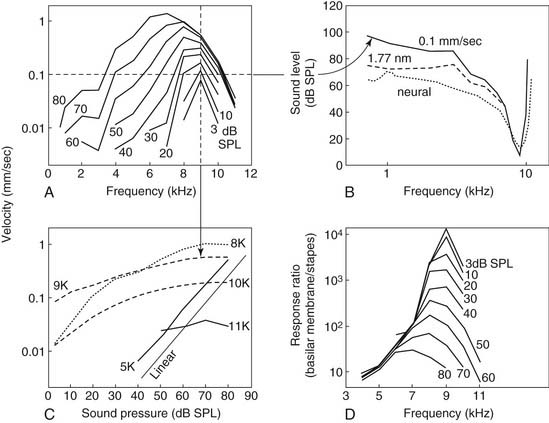
Von Békésy recognized more than half a century ago that the envelope of the traveling wave measured in cochleae from human cadavers is broadly tuned—so broadly tuned as to be inconsistent with a living person’s ability to discriminate closely spaced frequencies. Numerous groups of investigators have since confirmed the findings of von Békésy and have shown with clarity that sharp tuning and high sensitivity are lost in physiologically compromised cochleae or cochleae from cadavers.432,494 Only after mechanical measurements were made in vivo with nonhuman species did it become clear that the peak of the traveling wave in living animals is, in fact, very sharply tuned at low levels of stimulation and exhibits nonlinear growth as sound levels increase.274,314,433,454,494 The seminal study of Rhode,433 which showed the amplitude of basilar membrane vibration relative to stapes displacement to be as much as two to three orders of magnitude greater under low-stimulus-level conditions than at higher levels in normal animals, led most directly to the contemporary understanding of cochlear mechanics as a nonlinear phenomenon (i.e., high sensitivity to changes in stimulus pressure at low levels, marked compression in the midlevel range, and linear growth at high levels) (Fig. 146-4).
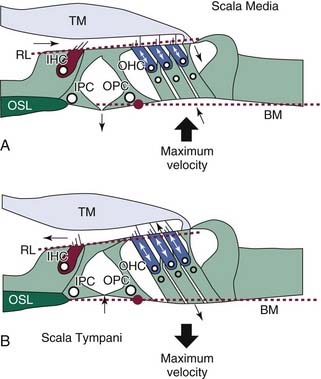
Active mechanics associated with basilar membrane amplification is a metabolically labile, energy-consuming process that is, consequently, physiologically vulnerable. It is of particular importance to recognize that the active mechanical event underlying amplification is highly localized. That is, the principle of active mechanics applies to a circumscribed portion of the cochlear partition that is limited to the immediate vicinity of the characteristic place for a given stimulus frequency (see Fig. 146-4A and B). At frequencies removed from the characteristic place, the properties of cochlear partition vibration in a living animal follow linear rules of growth, as in cadaverous tissue (Fig. 146-5).179 Another important point is that the traveling wave peaks at increasingly apical regions of the cochlear partition with increasing stimulus level, shifting the cochlear place-frequency map toward lower frequencies at any given place.
Role of Outer Hair Cells in Active Mechanics
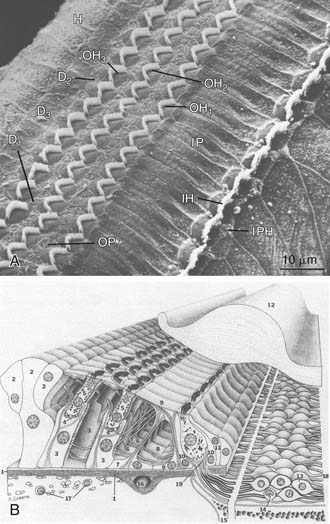
Many early studies indicated that OHCs play a fundamental role in active cochlear mechanics, as indicated by estimates of diminished cochlear sensitivity and frequency selectivity and loss of nonlinear operating properties when OHCs are missing from the end organ (see Fig. 146-4). The identification of specializations that distinguish inner hair cells (IHCs) from OHCs of the mammalian cochlea offered early clues that the contribution of OHCs to the transduction process is unique (Fig. 146-6). For example, only approximately 5% of all auditory nerve fibers innervate OHCs,371,525,524 yet there are at least three times as many OHCs as IHCs (Fig. 146-7). These findings suggested that the transmission of sensory information to the central nervous system (CNS) probably is not the primary function of OHCs. In studies of acute preparations, investigators noted that the cochlea, and OHCs in particular, are highly vulnerable to hypoxia or other forms of insult.76,148,293,432,433,448,449,494 In animal models of chronic damage, OHCs were found to be more susceptible to noise-induced or aminoglycoside-induced damage than IHCs, and when OHCs were lost, with IHCs appearing unaffected, cochlear sensitivity was significantly reduced, as was frequency selectivity, and input-output curves acquired a more linear character in the region of OHC damage.77,97,101,149,208,278,279,329,450,465 In addition, other cochlear nonlinearities, such as two-tone suppression (i.e., the reduction in the response to one tone elicited by the presence of another) and distortion tones (i.e., the appearance of additional tones not present in the stimulus produced by the interaction of two or more primary tones, such as 2f1-f2 or f2-f1, where f1 and f2 represent the primary tones, with f1 less than f2), are either lost or diminished when OHCs are traumatized.102,480 The discovery of sound-evoked otoacoustic emissions, sounds that are generated by the end organ during transduction and are measured in the external ear canal,270 along with spontaneous otoacoustic emissions,608 and their dependence on OHCs provided additional evidence that an active, OHC-driven mechanism operates within the cochlea, which is capable of producing energy and propagating it in the reverse direction. The influence of OHCs on cochlear mechanics was further suggested on the basis of observations made after stimulating the crossed olivocochlear bundle (OCB), a brainstem pathway populated by large, myelinated fibers that arise among neurons located in the medial region of the superior olivary complex that cross the brainstem at the floor of the fourth ventricle and terminate in calyx-like presynaptic terminals on OHCs.200,287,427,428,518,597,598 Specifically, OCB activation altered cochlear output,118,155,156,176,605 as well as otoacoustic emissions373,505 and basilar membrane displacement,376 supporting the notion that OHCs directly influence cochlear mechanics. Further support for the view that OHCs play an essential role in active mechanics comes from the work of Mammano and Ashmore,349 who demonstrated that outer hair cell motility induced by electrical stimulation of the excised cochlea displaced both the reticular lamina and the basilar membrane.
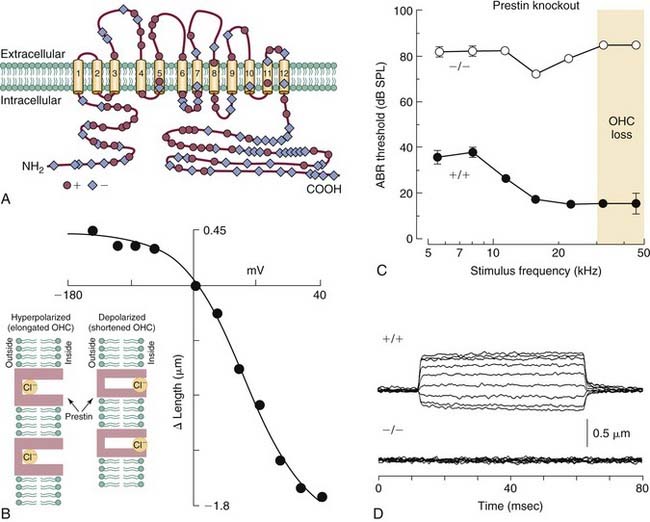
Prestin (SLC26A5) Is Essential for Outer Hair Cell Motility and Active Transduction
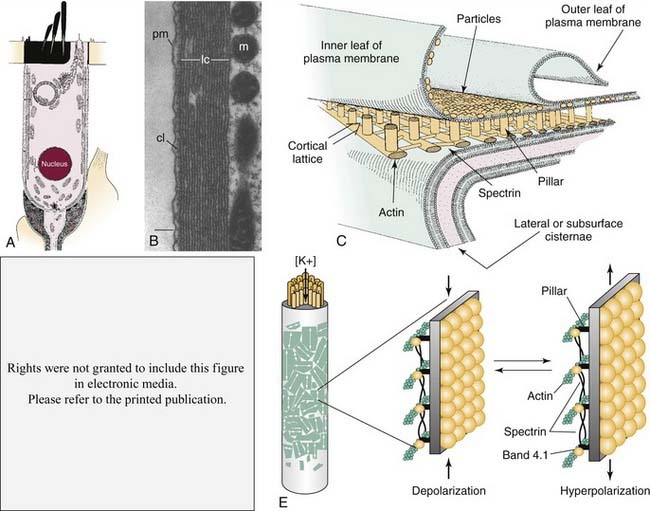
The finding that OHCs contract or elongate at very high rates (up to at least 70 kHz) along the length of their longitudinal axes when depolarized or hyperpolarized, respectively,30,62,63,98,99,168,177,261,478,630 led to the suggestion that OHCs may amplify the displacement of the basilar membrane if forces generated by their motility are produced on a cycle-by-cycle basis and precisely timed relative to passive mechanical events (see Fig. 146-6E). Somatic length changes of up to 5% of the total length of OHCs are determined directly by the magnitude of the transmembrane voltage and not by ionic currents. In addition, the voltage-motility relationship observed among OHCs is highly nonlinear (Fig. 146-8B). Unlike in IHCs, the lateral wall of OHCs is densely packed with particles that are integral components of the plasma membrane,165,202,264,470 and these particles are thought to be oligomers of the protein recently identified as the molecular motor underlying fast somatic motility (see Fig. 146-6C and E).
The discovery that OHC motility is accompanied by gating currents that reflect a nonlinear capacitance led to the suggestion that it was the movement of charged particles within the domain of the plasma membrane that operates as the motive force of contractility.31,476 The voltage-sensitive motor protein underlying the process was identified in 2000 and given the name prestin (SLC26A5). Prestin (Fig. 146-8A) is a modified anion-transporter protein belonging to the solute carrier 26 (SLC) family of proteins.633 Antibodies to prestin have been localized in OHC membranes, and developmental changes in the expression of prestin immunoreactivity follow a temporal profile that overlaps the development of electromotility, providing further support for the view that prestin is the motor protein.42 Although the precise location of the voltage sensor remains unknown, and although it may be an integral component of the prestin molecule itself, findings from recent studies suggest that intracellular Cl− ions act as extrinsic charged voltage sensors (see Fig. 146-8B, inset). In this model, Cl− ions occupy a position in the intracellular domain of the membrane-bound prestin protein and move along an inward electrical gradient away from the extracellular domain during depolarization. This charge movement produces, in turn, a conformational change in the protein that reduces its surface area and shortens the cell; the opposite action occurs during hyperpolarization, when Cl− ions move toward the extracellular space within the domain of the protein (although the anion is never translocated to the extracellular space), causing an increase in surface area and lengthening of the cell.100,391 Salicylates, which are known to cause temporary hearing loss when administered at high doses,535 are permeable to the plasma membrane and act as competitive antagonists of Cl−, thereby reducing electromotility and, presumably, diminishing sensitivity.391 The role of prestin in transduction was studied directly in a mouse strain in which prestin was deleted. The knockout mouse exhibited a loss of OHC electromotility in homozygous mutant animals (see Fig. 146-8D), a 40- to 60-dB loss in sensitivity, and loss of OHCs in the base of the cochlea (see Fig. 146-8C).328 In addition, the frequency selectivity of individual auditory nerve fibers is diminished in knockout mice.68 Although the length and stiffness of OHCs are reduced in the knockout mouse, these findings collectively suggest that active transduction is a direct product of OHC somatic motility. Confirmation of prestin’s role in cochlear amplification was recently established in a study of a “knock-in” mouse model, in which the protein’s function was diminished but OHCs exhibited normal length and stiffness.103 Natural mutations of the prestin gene have been identified in humans, and the resulting condition is transmitted as a recessive, nonsyndromic form of congenital deafness. In this case, however, hearing loss is in the severe to profound category (i.e., on the order of 90 dB or greater) and exists as a bilateral form of deafness among persons who are homozygous for the trait. The degree of hearing loss is more variable among heterozygotes, ranging from normal in one case to mild (40 dB) and even moderate to profound (60 dB) in others, suggesting the semidominance of the abnormal allele through haploinsufficiency.337
Conversion of Basilar Membrane Displacement to Radial Shearing Forces
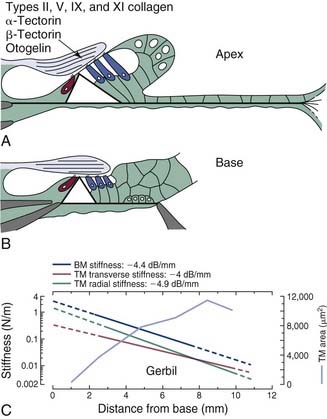
All existing models of transduction center on the idea that displacement of the basilar membrane results in a radial shearing motion between the reticular lamina and the tectorial membrane, a motion that serves as the mechanical trigger of transduction currents. The reticular lamina consists of the interdigitating heads of the inner and outer pillar cells, the apical surfaces of the phalangeal processes of Deiters cells, and the apical surfaces of the hair cells through which the stereocilia project (see Fig. 146-6D). The apical surfaces of the supporting cells are flattened and are composed of microtubules and actin microfilaments, lending support to the idea that the reticular lamina is a relatively rigid structure that moves in concert with basilar membrane motion. The tectorial membrane is firmly attached to the spiral limbus and overlies the reticular lamina, with its tip or marginal zone connected to Hensen cells. The tallest row of stereocilia protrude from the apical surface of each outer hair cell and embed within the inferior surface of the tectorial membrane along the entire length of the cochlear spiral.288,333 Bundles of radially oriented filaments, consisting of type II, V, IX, and XI collagens, are embedded within the gelatinous matrix of the mammalian tectorial membrane (see Fig. 146-10A and B).209,361,444,514,515,545,546 The presence of collagen enhances the rigidity of the structure, making it less compressible and less elastic, particularly in the radial dimension.3,199,446 These features support a system in which the shearing motion between the reticular lamina and the tectorial membrane causes stereocilia to bend in the direction of the modiolus or the spiral limbus, depending on whether the basilar membrane is displaced toward the scala tympani or scala vestibuli, respectively.
In contrast with OHCs, the stereocilia of IHCs do not appear to firmly contact the tectorial membrane (see Fig. 146-1D). Therefore, during basilar membrane vibration, the relevant mechanical stimulus to IHC stereocilia is thought to be the flow of endolymph within the subtectorial space, the narrow channel between the reticular lamina and tectorial membrane into which the stereocilia protrude.387 Fluid coupling is then thought to lead directly to the displacement of IHC stereocilia in proportion to basilar membrane velocity at low frequencies (less than 500 Hz) and to basilar membrane displacement at higher frequencies; sustained displacements of the basilar membrane, or very-low-frequency displacements, do not effectively bend IHC stereocilia.403
Radial Displacement Patterns of the Basilar Membrane
The dynamics of basilar membrane displacement at the characteristic frequency place are more complex when examined radially. Inner and outer pillar cells that form the fluid-filled tunnel of Corti are packed with bundles of microtubules and actin microfilaments arranged in parallel that are cross-linked and are thought to produce structural rigidity within the cochlear partition.25,140,244 The foot of the inner pillar cell lies near the bony spiral lamina, whereas the foot of the outer pillar cell lies over the basilar membrane and is not supported by bone (Fig. 146-9). Therefore, when the basilar membrane is displaced in a passive cochlea, movement occurs maximally near the foot of the outer pillar cell, with the basilar membrane pivoting at the foot of the inner pillar cell.104 When OHCs contract, the reticular lamina pivots at the apex of the tunnel of Corti, and the basilar membrane and reticular lamina are pulled together, enhancing overall basilar membrane displacement.168,349,350,383 Furthermore, unlike in passive mechanical events, in which the full width of the basilar membrane moves uniformly (synchronously), during active mechanics, the magnitude and phase of displacement vary in the radial dimension.83,383,388,395,396,431,495,563,609,622 Although differences among studies from different laboratories are significant regarding the precise pattern of basilar membrane movement in the radial direction after displacement, many workers suggest that the position of greatest motion is in the domain of the OHC, and that motion in the OHC region is out of phase relative to the foot of the outer pillar cell. This finding has significant implications in the ongoing effort to understand the relationship between inner and outer hair cell responses during the transduction process.
Tectorial Membrane Pathophysiology
The tectorial membrane is composed of two primary matrix elements: a group of radially oriented collagen fibrils and a striated sheet matrix that is made up of tightly packed, small-diameter filaments that are interlinked by cross-bridges to give it a striated appearance.209,288,300,333,559 In addition to the variety of collagens making up the core structure, a collection of glycoproteins—α-tectorin, β-tectorin, and otogelin—also are constituent elements of the tectorial membrane.78,193,285,319,444 Otogelin, a glycoprotein that is found in essentially all of the acellular membranes of the inner ear, is associated with the large collagen fibrils, whereas α-tectorin and β-tectorin are components of the striated sheet matrix.
The mass and stiffness of the tectorial membrane are important variables that together determine the resonant properties of the structure that, in turn, affects its displacement pattern during acoustic stimulation. Much like dimensional changes that have been measured along the length of the basilar membrane, the cross-sectional area of the tectorial membrane has been shown to increase from the base to the apex as indicated by morphometric measurements of fully hydrated tissue specimens (Fig. 146-10C).445 Moreover, base-to-apex stiffness gradients in both the transverse and radial axes of the tectorial membrane have been observed,199,446 and they are similar to those reported for basilar membrane stiffness gradients (see Fig. 146-10C).139,205 Taken together, basoapical gradients in the physical properties of the tectorial membrane suggest the possibility that the structure may support longitudinally propagating traveling waves,181 giving rise to a second cochlear frequency-place map. Some investigators have suggested that the resonant frequency of the tectorial membrane lies approximately one-half octave below that of the basilar membrane’s characteristic or resonant frequency at any given position along the cochlear spiral.17,18,203,641 However, until the opportunity to study mutant mice in which the tectorial membrane is completely detached from the spiral limbus and the surface of the organ of Corti317 presented itself, direct evidence regarding the precise role of the tectorial membrane as an element of transduction was largely unavailable.