CHAPTER 62 Benign Vocal Fold Mucosal Disorders
Benign vocal fold mucosal disorders are common. More than 50% of patients seeking medical attention because of voice change have a benign mucosal disorder. Even before the laryngeal videostroboscopy era, when subtle and small lesions may have been missed, Brodnitz1 reported that 45% of 977 patients had a diagnosis of nodules, polyps, or polypoid thickening. From the same era (1964-1975), Kleinsasser2 reported that slightly more than 50% of 2618 patients seen for a voice complaint had one of these benign entities.
Anatomy and Physiology
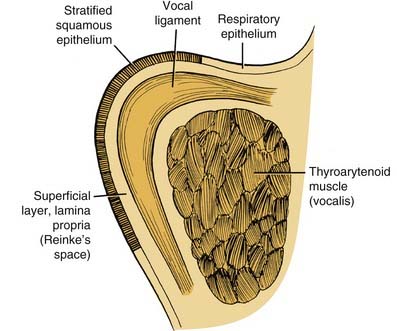
The anatomy most relevant to the benign vocal fold mucosal disorders is the microarchitecture of the vocal folds, as seen on whole-organ coronal sections in a study of cancer growth patterns3,4 and in the work of Hirano.5 Medially to laterally, the membranous vocal fold is made up of squamous epithelium, Reinke’s potential space (superficial layer of the lamina propria), the vocal ligament (elastin and collagen fibers), and the thyroarytenoid muscle. Perichondrium and thyroid cartilage provide the lateral boundary of the vocal fold (Fig. 62-1).
The vocal folds move as a whole between abducted and adducted positions for breathing and phonation, respectively. The mucosa (epithelium and superficial layer of the lamina propria [Reinke’s potential space]), which covers the vocal folds, is the chief oscillator during phonation (continuous adduction during expiratory flow of pulmonary air); thus, one might speak of vocal fold mucosal vibration rather than vocal fold vibration. In a canine study that supports this idea, Saito and associates6 placed metal pellets at varying depths into the vocal fold (e.g., epithelially, subepithelially, intramuscularly) and used radiographic stroboscopy to trace their coronal plane trajectories during vibration. Pellet trajectories of the mucosa were far wider than those of the ligament or the muscle. Thus, primarily the vocal fold mucosa oscillates to produce sound.
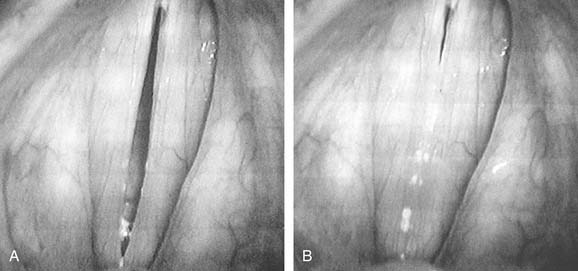
’The work of Hirano7 provides an explanation for these observations. Hirano described the vocal fold muscle as the body of the fold, the epithelium and superficial layer of the lamina propria (Reinke’s potential space) as the cover, and the intermediate layers of collagenous and elastic tissue (vocal ligament) as the transitional zone (see Fig. 62-1). Because of the different physiologic stiffness characteristics of these layers, they are somewhat decoupled mechanically from each other during phonation. Decoupling is graphically illustrated in Figure 62-2 (mucosa being stretched). This decoupling allows the mucosa to oscillate with some freedom from the ligament and muscle. An analogy can be made to the relative freedom from the paddle experienced by the red rubber ball and elastic band in a child’s paddleball toy. During phonation, pulmonary air power supplied to adducted vocal folds is transduced into acoustic power. To accomplish this, pulmonary air is passed between appropriately adducted vocal folds. At this point, the vocal fold mucosa vibrates passively according to the length, tension, and edge configuration determined by the intrinsic muscles and elastic recoil forces of the vocal fold tissues. Figure 62-3 shows the maximum open and closed phases of one vibratory cycle, as seen during laryngeal videostroboscopy. Further details concerning the mucosa’s vibratory behavior can be found in the works of Baer8 and Hirano5 and in Chapter 57.
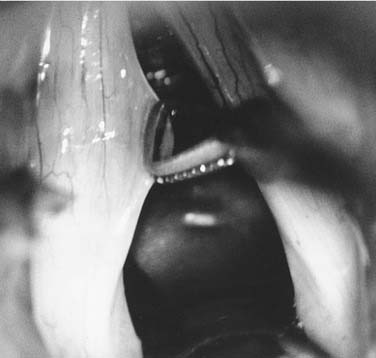
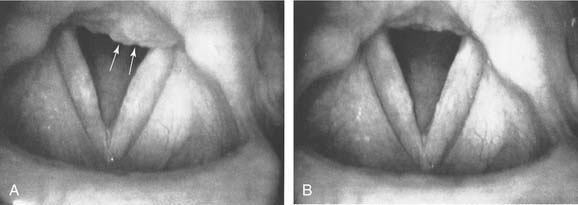
Figure 62-2. Gentle medial retraction shows the relative decoupling of the mucosa from the underlying, nondeformed vocal ligament.
Evaluation of the Patient: General Principles
History
Besides the usual items in the general medical history, the voice history should focus in particular on the following list of items, which may be best captured via use of a questionnaire9,10:
Vocal Capability Battery
The basic vocal capabilities and phenomena to be tested are (1) average or anchor speech frequency, (2) maximum frequency range, (3) projected voice and yell, (4) very-high-frequency, very-low-intensity tasks that detect mucosal disturbances,11 (5) register use and phenomena, (6) maximum phonation time, and (7) instability and tremors.
As stated, the vocal capability battery—coupled with the initial voice history and then subsequent laryngeal examination—is crucial in diagnosing a voice disorder and in directing subsequent management. For example, signs of a mucosal disturbance (e.g., detection of air escape, onset delays, loss of clarity and range) during assessment of the ability to perform high-frequency, low-intensity tasks minimize the possibility that a singer’s normal-sounding speaking voice during history-taking will subconsciously bias the clinician to selectively perceive normal vocal folds when, in fact, small vocal nodules are present.11a The vocal capability battery also provides insight into the severity of the patient’s vocal limitations, which can then be correlated with the visual examination to help determine, along with the patient’s needs and motivation, the intensity and direction of management.
Office Examination of the Larynx
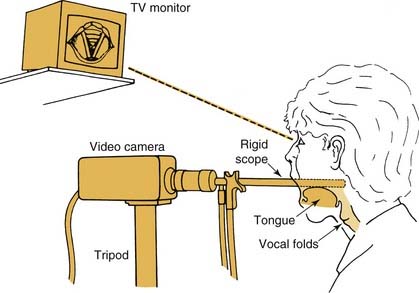
The laryngeal mirror should provide three-dimensional viewing and good color resolution but, in practice, offers poor visualization in many cases. In other cases, visualization is good, but only during phonation, because the view is obstructed by the epiglottis during respiration. In addition, no permanent image of the larynx results from this examination technique. Because the physician must therefore remember the lesion or document it with a simple sketch, precise critique of the effectiveness of the therapy chosen may not be possible. Rigid and also flexible laryngeal scopes often allow a clearer view, particularly during respiration. When used with the naked eye, however, they have similar disadvantages to those of the mirror. The fiberoptic nasolaryngoscope or a newer “chip-tip” video-endoscope is especially important in a patient who is difficult to examine because of unusual anatomy or an exceptional gag reflex. Even with these technologies, however, it is possible to overlook subtle-to-small mucosal changes unless the larynx is topically anesthetized to allow a close approach of the tip of the fiberscope to the vocal folds. With topical anesthesia, the vocal folds, subglottis, and trachea can be examined easily (Fig. 62-4).12,13
General Management Options
Sinonasal Management
Patients often incorrectly attribute chronic hoarseness to sinonasal conditions. Existing sinonasal problems should be managed on their own merits; however, the clinician may need to help diminish the patient’s perception of the contribution of these problems to a voice disorder in favor of more likely behavioral causes. When optimal laryngeal function is of concern, as in a vocal performer, nasal conditions should be managed locally (topically) when possible. The reason is that many systemic drugs (e.g., oral decongestants, antihistamine-decongestant combinations) dry not only nasal secretions but also secretions in the larynx, where a continuous secretional flow is important for proper vibratory function and mucosal endurance, particularly under demanding phonatory conditions. Medications that affect voice minimally are the topical nasal decongestants, which should be used for only a few days before the nasal mucosa is allowed to rest, so as to avoid rhinitis medicamentosa. Profuse rhinorrhea that accompanies the common cold can also be managed with ipratropium bromide inhalations.14,15 Corticosteroid inhalers are invaluable for the management of nasal allergies. Activating pump-action nasal inhalers without any inspiratory airflow avoids the alleged risk of the effects of nasally applied corticosteroid on the vocal folds.
Management of Acid Reflux Laryngopharyngitis
In a person with an incompetent lower esophageal sphincter or hiatal hernia, acid reflux into the pharynx and larynx during sleep can lead to chronic laryngopharyngitis. Such persons may or may not experience one or more of the following symptoms: exaggerated “morning mouth,” excessive phlegm, scratchy or dry throat irritation that is usually worse in the morning, habitual throat clearing, and huskiness or lowered pitch of the voice in the morning. The larynx may show characteristic erythema of the arytenoid mucosa, interarytenoid pachyderma, or contact ulcers; laryngeal findings may, however, be subtle (Fig. 62-5).
Voice Therapy
Finally, in this technologic era, voice clinicians increasingly document various aspects of vocal tract output, using acoustic analysis, spirometric measures to test respiratory adequacy, frequency and loudness measures, translaryngeal airflow rates, and other measures under various conditions. Speech pathologists may use this equipment for biofeedback (e.g., using a visual electronic frequency readout to modify average pitch for speech in a tone-deaf patient). For obligate false vocal fold phonation and intractable psychogenic disorders of voice production with visible vocal fold posture abnormalities, therapy room videoendoscopy can also be converted into an effective biofeedback tool (Fig. 62-6).11,16
Surgery
A set of laryngoscopes, microlaryngeal forceps, scissors, dissectors, and knives should be on hand. In the face of the plethora of instruments currently available, the comment by Kleinsasser2 that a relatively simple set suffices the experienced surgeon remains true (Fig. 62-7)!
The carbon dioxide laser has become an important part of the surgeon’s armamentarium, and many have discussed its application to benign laryngeal disorders. Tissue effects of the laser depend on spot size and focus, wattage, duration of beam activation, waveform mode (pulsed vs. continuous), and, perhaps most important, surgical precision. Cold microdissection may be safer than laser techniques, provided that the surgeon is equally proficient in both. Norris and Mullarky,17 comparing continuous-mode carbon dioxide laser with the cold scalpel for incising pig skin, reported that a short-term advantage resulted after laser incision with regard to the speed of reepithelialization; no long-term difference in healing was noted. However, although the fact was not noted in their report, these investigators’ histologic sections clearly showed a wider zone of tissue destruction beneath the epithelium with laser than with scalpel. Duncavage and Toohill18 compared healing response in dogs after traditional fold stripping and after carbon dioxide mucosal vaporization. They concluded that, until late in healing, more edema and giant-cell reactions to bits of charred debris, and greater subepithelial fibrosis occurred with the laser technique than with the cup forceps alone. Manipulation of wattage, focus, and mode of laser irradiation of tissues may decrease thermal injury, charring, and other adverse effects of the laser.
The preceding studies date from the early era of the carbon dioxide laser. The microspot carbon dioxide laser appears to diminish these disadvantages,19,20 although a systematic comparison of functional results (including vocal capabilities and videostroboscopy) is not available to guide the surgeon in choosing between laser and microdissection methods. With a caseload approaching 1000 singers and double or triple that number of non-singers—for whom laser and non-laser methods have been used on an individualized basis—I believe that surgical technique and skill are preeminent over the specific tools used.
After surgery, vocal quality and capabilities should show good to excellent improvement; however, patients should be counseled preoperatively what the risk of worsening the voice is predicted to be: For nodules it may be appropriate to say, “This surgery typically restores the voice to ‘original equipment status,’ but there is a small risk that you will experience a large improvement but not to fully normal; and there is a remote, rare risk that your voice will be worse after surgery.” By contrast, one may say to the person with bilateral sulci in whom mucosa is thin, “I am expecting a modest improvement of your voice, but it will take many months to experience this improvement, and there is a quite significant chance your voice will be no better, and even possibly worse.” For the experienced surgeon who uses dissection, rather than microavulsion techniques, along with preoperative and postoperative videostroboscopy as his or her “teacher,” the question in the general case becomes not so much one of possibly making the voice worse, but rather, “Can I make this patient’s speaking and singing capabilities normal, and if not, how close can I come?” Cornut and Bouchayer’s21 experience operating on 101 singers and Bastian’s22 experience in the same population established a role for laryngeal microsurgery in restoring vocal capabilities and in abolishing or diminishing limitations.
Specific Benign Vocal Fold Mucosal Disorders
Vocal Nodules
Pathophysiology and Pathology
Only the anterior two thirds (membranous portion) of the vocal folds participates in vibration because the arytenoid cartilages lie within the posterior third of the glottic aperture. Vibration that is too forceful or prolonged causes localized vascular congestion with edema at the midportion of the membranous (vibratory) portion of the vocal folds, where shearing and collisional forces are greatest. Fluid accumulation in the submucosa from acute abuse or overuse results in submucosal swelling (sometimes unwisely called incipient or early nodules). Long-term voice abuse leads to some hyalinization of Reinke’s potential space and, in a subset of cases, some thickening of the overlying epithelium. This pathophysiologic sequence explains the easily reversible nature of most acute nonhemorrhagic swellings, in contrast to the slower, incomplete, or failed resolution of chronic vocal nodules. Whether acute edema or more chronic nodules, the change in mucosal mass, lessened ability to thin the free margin, and incomplete glottic closure caused by the nodules account for a constellation of vocal symptoms and limitations that is characteristic of mucosal swelling.11,22
Diagnosis
Vocal Capability Battery
In patients with moderate to large vocal nodules, the speaking voice is usually lower than expected, husky, breathy, or harsh. Patients with subtle to moderate swellings often have speaking voices that sound normal; the speaking voice is an insensitive indicator of mucosal disorders in comparison with the singing voice. In patients with subtle or small swellings (usually only singers present with small mucosal disturbances), vocal limitations (e.g., delayed phonatory onset with preceding momentary air escape, diplophonia, inability to sing softly at high frequencies) may become evident only when high-frequency, low-intensity vocal tasks for detecting swelling are elicited.11a At high frequency, short-segment vibration may occur; in other words, the nodules stop vibrating and the short segments of mucosa anterior or posterior to them, or both, vibrate.
Many patients with nodules may have undergone indirect laryngoscopy and have been told that their vocal folds were normal or have been given a nonspecific diagnosis such as “laryngeal irritation.” Use of vocal tasks that detect swellings and videostroboscopy, when indicated (see Figs. 62-3 and 62-4), protect the laryngologist from missing the most subtle vocal fold swellings. The ability to diagnose tiny nodules is crucial, because failure to make such a diagnosis can have serious consequences for the professional voice user.
Management
Behavioral
Vocal nodules arise from the vocal overdoer profile, so speech (voice) therapy plays a primary role initially. Typically, the nodules and their more obvious symptoms regress, particularly if the patient is not a singer. However, the most skilled behavioral (voice) therapy sometimes fails to achieve complete visual resolution of nodules that have been present for many months to years. Sensitive singing tasks that detect impairment (not the size of persistent swellings) are generally more helpful in the decision whether to consider their surgical removal.11,22
Surgical
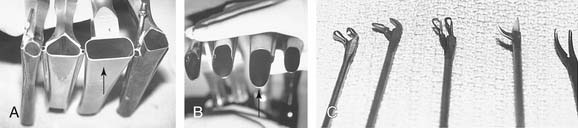
Surgical removal becomes an option when nodules of whatever size persist and the voice remains unacceptably impaired (from the patient’s perspective) after an adequate trial of therapy (generally a minimum of 3 months). Some writers prefer precise removal using microdissection techniques (Fig. 62-8). Vocal fold stripping has no place in the surgery of nodules. The proper duration of voice rest is controversial; some writers prefer a relatively short period. Typically, the patient is asked not to speak for 4 days, although sighing sounds begin 1 day after surgery. Beginning on the fourth day, the patient gradually progresses over 4 weeks to full voice use under a speech pathologist’s supervision. Early return to nonstressful voice use, as described in Table 62-1, seems to promote dynamic healing. The results of precision surgery are typically remarkably good, even in singers. Cornut and Bouchayer21 stated, in their study of approximately 160 singers treated with surgery, “As long as certain management principles are followed in a majority of cases, laryngeal microsurgery enables the singing voice to regain the whole of its functioning.”
Table 62-1 General Guidelines for Initial Voice Use after Vocal Fold Microsurgery
| Time After Surgery* | Talking† | Singing (for Singers) |
|---|---|---|
| Days 1 to 4 | None | Gentle attempts at yawn or sigh for approximately 30 sec, 6-8 times per day‡ |
| Week 2 (begins day 5) | 3 | Same exercise 5 min twice per day (after first postoperative exam) |
| Week 3 | 4 | Same exercise 10 min twice per day§ |
| Week 4 | 5 | Same exercise 15 min twice per day§ (after second postoperative exam) |
| Week 5 | 4 or 5 | Same exercise 20 min twice per day§ |
| Weeks 6-8 | 4 or 5 | Same exercise, up to 20 min three times per day| |
* After fourth examination, return to performance should be considered.
† Based on a 7-point talkativeness scale, in which 1 = very untalkative, 4 = average, and 7 = extremely talkative.
‡ Accept what comes out, even if it is only air or is very hoarse.
§ With emphasis on ease, clarity, and agility, not voice building. The entire expected range should be practiced in each session, with gentle insistence on high notes that do not want to sound. In general, practice mostly a mezzo piano dynamic and only occasionally a mezzo forte.
| Same as preceding footnote, with the addition of gradually increasing the dynamic range and insistence.
Capillary Ectasia
Epidemiology
Capillary ectasia seems to happen most often with vocal overdoers (Figs. 62-9 to 62-11). Because of the female preponderance of this disorder, some writers have speculated about an estrogen effect.
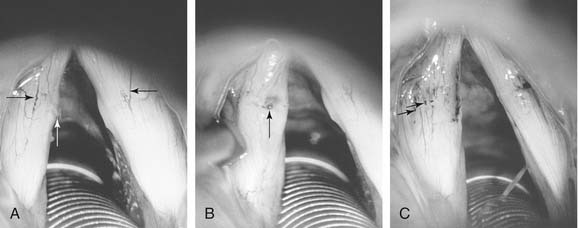
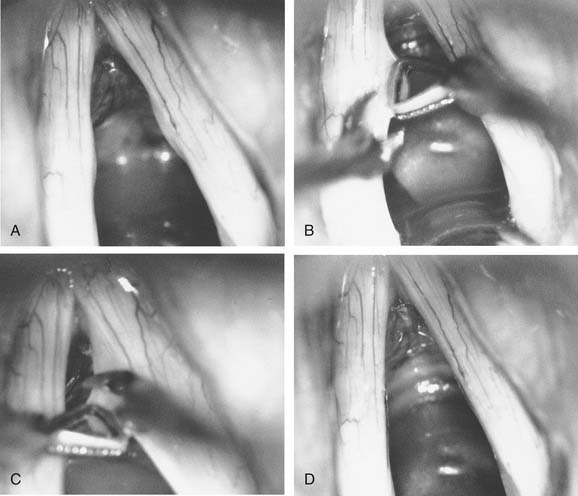
Figure 62-9. Vocal nodules with associated capillary ectasia. A, Superficial ectasia is seen (black arrows); the white arrow indicates deeper ectasia seen within left-sided nodule. B, The left vocal fold margin is rolled up onto the superior surface of the fold so that ectasia within the nodule can be seen more clearly (arrow), particularly the knuckle variant. C, Nodules have been removed (see Fig. 62-8), but spot-coagulation of dilated capillaries is present at approximately 2-mm intervals (arrows).
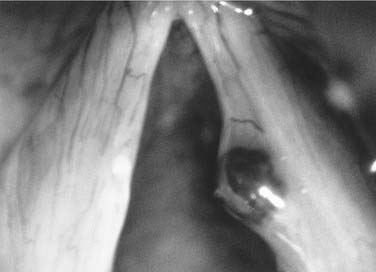
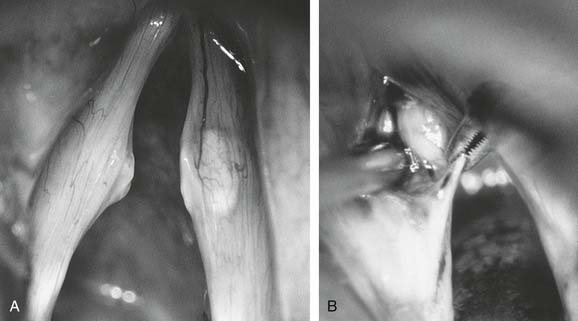
Figure 62-10. Operative view of a hemorrhagic polyp on the right true vocal fold of 14 months’ duration. Note the capillary ectasia.