Retinopathy with Focal or Mottled Lesions
Causes of focal infiltration (“foci”) of the fundus:
- drusen
- cotton-wool spots
- hard exudates
- focal anomalies
- focal retinal and choroidal scars
- toxoplasmosis
- tuberculosis
- mycoses
- incipient toxocariasis
- focal scarring from laser photocoagulation
- acute posterior multifocal placoid pigment epitheliopathy (APMPPE)
- early phase of birdshot retinochoroidopathy.
Retinopathy with focal or mottled lesions often occurs in relation to anatomic vascular structures such as in the retinal capillary network or as “lobes” in the choroidal terminal vascular bed. These disorders should be differentiated from primary vascular disorders such as vasculitis even if symptoms of secondary vasculitis are observed in severe cases of focal inflammation.
 White Mottled Deposits
White Mottled Deposits
These findings are characteristic of degenerative changes in the retina in systemic disease and a few specific diseases of the retina. Leukocytic infiltrates occurring with lymphomas and tumor metastases also lead to focal retinal changes.
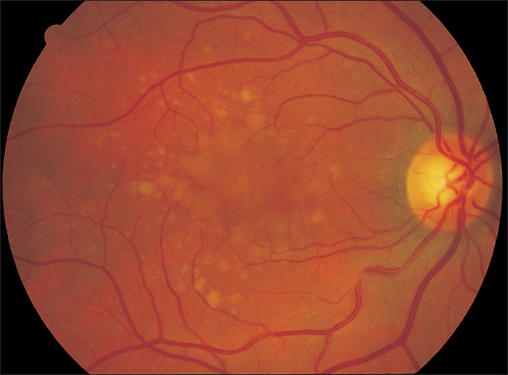
Drusen
- gray punctate deposits
- dull = “soft” drusen
- crystalline, <30 μm = “hard” drusen
- histology: hyaline deposits
- retinal pigment epithelium (RPE)
displaced or atrophic
- fluorescein angiography
- hard drusen:
hyperfluorescent in the early phase
- soft drusen:
hyperfluorescent in the late phase
Drusen in the Retinal Pigment Epithelium
Drusen are defined as gray punctate deposits beneath the retina visible on direct ophthalmoscopy or with the 78-diopter lens.
One differentiates between “hard,” “soft,” and “reticular” drusen. Soft drusen are not sharply demarcated and can be confluent.
Histologic examination of drusen reveals hyaline deposits beneath the retinal pigment epithelium. This applies especially to eyes in which Bruch membrane is thickened and exhibits increased lipid staining upon histochemical examination. Therefore, it is thought that drusen result from impaired diffusion through Bruch membrane. The retinal pigment epithelium is largely displaced at the site of the drusen, creating an epithelial defect. For this reason, hard drusen appear luminescent with the choroid in the early phase of fluorescein angiography. During angiography, soft drusen are initially hypofluorescent due to a blockade, whereas in the late phase they increasingly appear hyperfluorescent as a result of “staining.” As these drusen are not associated with exudation, their hyperfluorescence remains undiminished during the late phase of the angiogram. Drusen, especially soft drusen, are precursors of both dry and wet forms of age-related macular degeneration (p. 48).
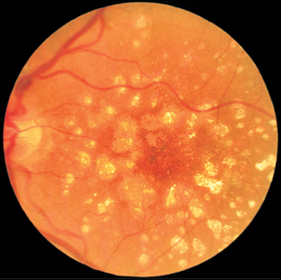
Fig. 3.2 Crystalline drusen of a 67-year-old woman with normal visual acuity. Drusen are confluent and exhibit pigment epithelium defects indicative of the transition to dry age-related macular degeneration.
Treatment: see p. 48
Hard Exudates
- irregularly shaped but sharply demarcated
- shiny
- lipid-storing macrophages
Hard Exudates Occurring Secondary to Vascular Disorders—Fatty Degeneration
These reversible deposits occur within the retinal parenchyma. Because they are beneath the internal limiting membrane, they appear shiny.
Hard exudates (fatty degeneration) occur with:
- hypertensive retinopathy
- diabetic retinopathy
- Coats disease
- chronic ischemia
- vascular processes such as retinal vasculitis
- retinal edema
- exudative age-related macular degeneration
- tumors.
- exudative age-related macular degeneration
Treatment:as hard exudates are secondary signs, they can disappear when the underlying disorder is treated.
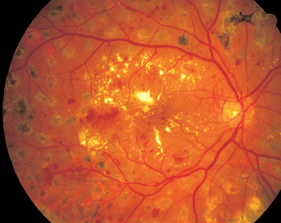
Fig. 3.3 Hard exudates in hypertensive retinopathy occurring in a pattern known as circinate retinopathy in the setting of treated advanced diabetic retinopathy.
Canthaxanthin Retinopathy
The carotenoid preparation canthaxanthin is used orally for endogenous cosmetic skin tanning. After a while, crystalline deposits appear in the retinal pigment epithelium at the posterior pole of the eye. Defects in the pigment epithelium occur with chronic use and can lead to permanent visual impairment. In some European countries, the preparation has been taken off the market.
 Sharply Demarcated, Bright, and Pigmented Areas
Sharply Demarcated, Bright, and Pigmented Areas
Posterior Staphyloma
- in pathologic myopia
- monocular funduscopy: large myopic cone
- binocular funduscopy: bright, nearly
- white posterior evagination of the eyeball
- marked decrease in visual acuity if macula is involved
Posterior Staphyloma in Myopia
In severe myopia, the peripapillary crescent develops into a marked posterior evagination of the eyeball. This “staphyloma” can extend past the macular region. This area appears bright, nearly white, because the choroid and retina lining the cavity are greatly stretched and atrophic or have become very thin. There is a high risk of retinal tear with subsequent retinal detachment. In monocular funduscopy, this change appears as a large myopic cone. Binocular ophthalmoscopy reveals the full extent of the posterior evagination.
Treatment : there is no known effective treatment. If both eyes are affected, it is recommended that the patient be referred to a specialist for the visually impaired.
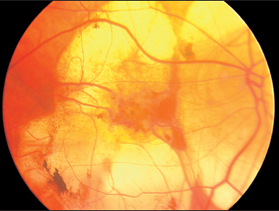
Fig. 3.4 Posterior staphyloma in severe myopia. Evagination of the outer layer of the globe leads to retraction of the choroid and retina, exposing the white sclera.
Coloboma
- dominant inheritance
- white sector inferiorly
- sclera visible through choroidal defect
- highly variable appearance
Choroidal Coloboma
Colobomas can vary widely in size and shape. They may be rudimentary, in the form of a small white lesion on the inferior optic disc, but can also extend far into the peripheral fundus as far as the ora serrata or beyond. Their width is variable. They may form narrow white sectors or may be so wide that they cover the entire inferior half of the fundus. Rarely, a portion of normal fundus with choroid is located between the defects. Such findings indicate a “bridge” coloboma. This form can be confused with a coloboma of the macula secondary to fetal retinochoroiditis (p. 30). Often the fovea lies in the marginal portion of the coloboma. In many cases, a coexisting iris coloboma is the impetus for the fundus examination that reveals the choroidal coloboma.
Treatment: if vision is impaired, see Treatment above.
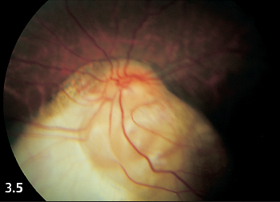
Fig. 3.5 Large choroidal coloboma in the inferior fundus of a 12-year-old boy. Both eyes exhibited the same findings and subsequently suffered retinal detachment.
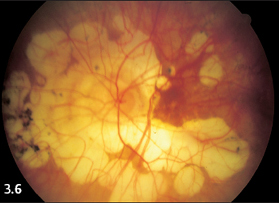
Fig. 3.6 Extensive, partially confluent focal choroidal atrophy at the posterior pole of a severely myopic 75-year-old woman.
Choroidal atrophy in Myopia
- in severely myopic eyes
- round to oval defects with smooth margins
- luminous white sclera
- highly variable size
 Choroidal Atrophy in Myopia (White)
Choroidal Atrophy in Myopia (White)
In severely myopic eyes, the elongation of the eyeball stretches the retina and choroid. The resulting tension creates round or oval retinal and choroidal defects at the posterior pole of the eye and in the periphery of the fundus. These defects may remain very small (as small as the diameter of a retinal arteriole) or they may attain a size of several disc diameters. The defects have smooth margins; the sclera appears luminous and white.
Treatment: if vision is impaired, see Treatment above.
Chorioretinal Scars
- sharply demarcated without any ill-defined infiltrates
- yellowish-white
- retinal and choroidal defect
- size and location variable
- particles or clumps of pigment as remains of RPE
- no light perception in scarred area
Scars from Chorioretinitis (Yellowish-White)
Scars from chorioretinitis are sharply demarcated and yellowish-white when the underlying sclera shines through. If remnants of the choroid are present, a few choroidal vessels will be visible.
Lesions may include small round spots, larger scars, and broad defects. Areas of scarring can occur at any location on the fundus and may be isolated, clustered, or diffusely disseminated.
Treatment: if vision is impaired, see Treatment p. 23
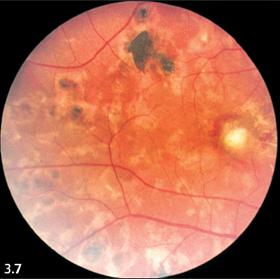
Fig. 3.7 Multiple scars from chorioretinitis.
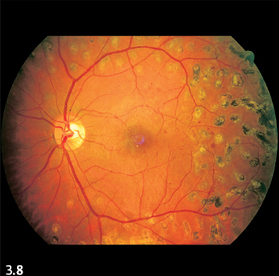
Fig. 3.8 Focal scarring from laser treatment in diabetic retinopathy.
| Disseminated, isolated | Disseminated retinochoroiditis, for example secondary to toxoplasmosis |
| Center of retina, large, pigmented halo | Congenital (fetal) toxoplasmosis |
| In the macula, small | Solar retinopathy, for example secondary to exposure to intense light |
| Small, disseminated over entire fundus | Laser coagulations, ocular histoplasmosis syndrome (POHS) |
| Small, disseminated, in the middle of the fundus, indistinct | Acute posterior multifocal placoid pigment epitheliopathy (APMPPE) |
| Small, disseminated, periphery of the fundus | Rheumatic retinochoroiditis |
| Small, disseminated in the whole fundus | Scars in the final stages of birdshot retinochoroidopathy, intermediate choroiditis |
| Maplike pigmented margins | Serpiginous choroiditis |
| Broad area | Necrotizing retinitis |
 Blurred, Fluffy, White Focal Lesions
Blurred, Fluffy, White Focal Lesions
Recent Scars from Laser, Diathermy, or Cryotherapy
- luminous, white, ill-defined, and slightly prominent focal lesions
- after about two weeks, these become yellowish-white chorioretinal scars
Recent Scars from Laser, Diathermy, or Cryotherapy
The thermal effect of laser photocoagulation and diathermy and the cold of cryotherapy all produce white spots on the fundus. Ill defined and slightly prominent, these spots closely resemble focal chorioretinitis. The luminous white lesions fade within about two weeks and assume the appearance of a yellow-white and pigmented chorioretinitis scar of undefined origin (p. 25).
Myelinated Nerve Fibers
- congenital
- irregular, not entirely sharply demarcated patches
- luminous white
- finely striped pattern
- relative visual field defects
Myelinated Nerve Fibers
Nerve fibers extending from retinal ganglion cells to the optic disc generally lack a myelin sheath. They form a transparent layer of the inner retina that appears as a fine, glistening striped pattern corresponding to the course of the nerve fibers. Normally, the retinal nerve fibers only exhibit a myelin sheath within the optic nerve posterior to the lamina cribrosa.
Myelinated nerve fibers can occur in the retina as a congenital anomaly. They appear luminous and white against the fundus, and occur in broad, irregular, not entirely sharply demarcated patches on the optic disc and in the vascular arcades, rarely further peripherally. They are recognizable by their finely striped pattern that reflects the course of the nerve fibers. The retinal vessels may be partially obscured by myelinated nerve fibers. These fibers are harmless and remain constant throughout the patient’s lifetime. However, this white, thickened layer reduces the sensitivity of the retina to light, and myelinated nerve fibers produce relative visual field defects.
Fig. 3.9 Recent laser burns following photocoagulation of a retinal hole.
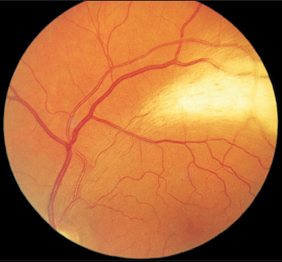
Fig. 3.10 Myelinated nerve fibers in the superior temporal vascular arcade.
Treatment: there is no known effective treatment.
Cotton-Wool Spots
- ill-defined, dull white spots measuring one-quarter of a disc diameter at maximum
- localized ischemia
- swelling of nerve fibers
- fluorescein angiography lesions masked (which remain dark) in contrast to retinochoroiditis
Cotton-Wool Spots of Varying Etiology
These spots are observed at the posterior pole of the eye and in the vascular arcades, often in close proximity to the retinal vessels. They are produced by edematous nerve fibers in the retinal layer of ganglion cells that are the result of localized ischemia in the retinal capillaries. These are typical concomitant signs of other disease processes and are therefore regarded as a diagnostic feature. Soft exudates appear transiently and can disappear after a few days.
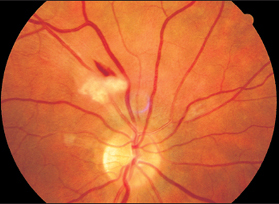
Fig. 3.11 Cotton-wool spots and retinal hemorrhage in eclampsia.
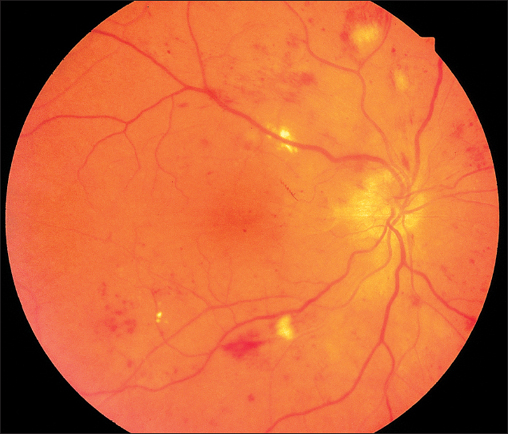
Fig. 3.12 Cotton-wool spots in hypertensive retinopathy with round and linear intraretinal hemorrhages. Note the narrow caliber in the arteries and the optic disc edema.
Cotton-wool spots are important diagnostic signs in:
- hypertensive retinopathy (p. 135)
- diabetic retinopathy (p. 138)
- occlusion of the central retinal vein (p. 146)
- branch retinal vein occlusion (p. 148)
- retinal microemboli
- HIV retinopathy (p. 73)
- eclampsia (p. 137)
- severe anemia
- pancreatic diseases.
Treatment: as these are secondary symptoms, they can disappear when the underlying disease is treated.
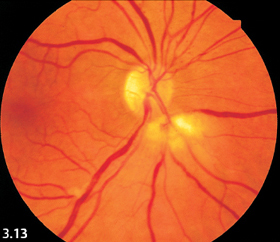
Fig. 3.13 Cotton-wool spots in acute pancreatitis.
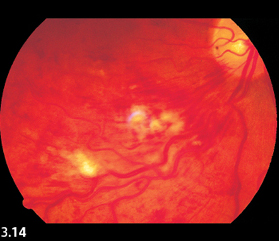
Fig. 3.14 Cotton-wool spots in ischemic branch retinal vein occlusion. Findings include engorged veins and numerous hemorrhages.
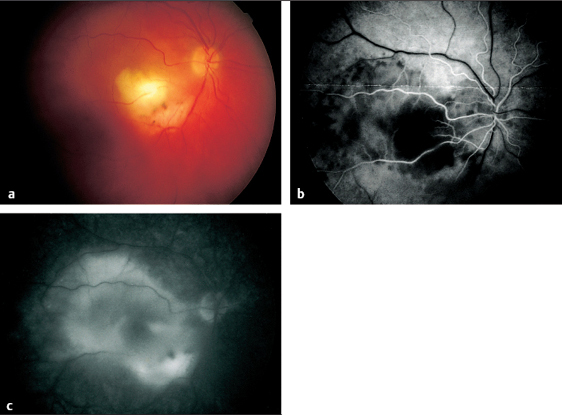
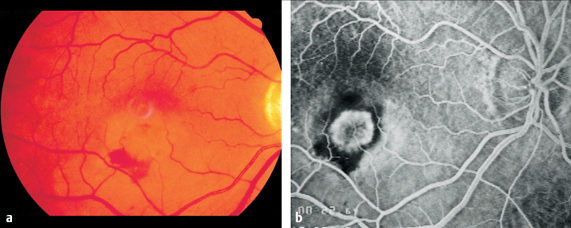
Fig. 3.15 a-c Disseminated retinochoroiditis in toxoplasmosis. a Large, acute focal lesion at the margin of a scar in the inferior temporal vascular arcade. b Arterial phase of fluorescein angiography (11 seconds). The focal retinochoroiditis lesion masks the fluorescence. c Late phase of fluorescein angiography (445 seconds). The focal inflammatory lesion now exhibits extensive exudation.
Acute Lesion in Retinochoroiditis
- white and flaky
- slightly prominent
- ill-defined
- size one half to one disc diameter
Chronologic Course of a Lesion in Retinochoroiditis
- four weeks: acute and flaky
- four more weeks: increasingly sharp margins and decreasing blurriness of edema, yellowish color, clumps of pigment visible
- eight to 16 weeks: sharply demarcated scar
Symptoms in Retinochoroiditis
- veil or shadow with peripheral lesions; often no visual impairment
- acute loss of visual acuity with macular involvement
- sector field defect with lesions close to the optic disc
Diagnosis of Toxoplasmosis
- focal retinochoroiditis lesion
- simultaneously acute lesions, healing foci, and scars
- cells in the vitreous body
- isolated cells in the aqueous humor
- associated vasculitis
- clinical findings often unequivocal
- frequently recurrent
Disseminated Retinochoroiditis
If many such lesions appear on the fundus simultaneously, one also refers to multifocal retinochoroiditis (p. 83). This is merely a descriptive term used to characterize the appearance of findings; it does not define a specific disease entity. Extensive inflammatory lesions are associated with cells in the vitreous body and aqueous humor (p. 172).
After two to six weeks, the lesions turn a more yellowish color and their margins become more sharply demarcated. As the focal lesions become less dense, clumps of pigment appear. A sharply defined, yellowish-white choroidal scar develops within another two to four months. Pigmented remnants of the choroid and the white sclera are visible through the resulting retinal defect.
Toxoplasmosis
Local infections with Toxoplasma gondii produce the classic clinical picture of disseminated retinochoroiditis. The pathogen is occasionally detected in the human eye by histologic examination. Experimental models can also produce this clinical picture. However, the diagnosis is made on the basis of the clinical findings described above and the typical clinical course. Antitoxoplasma antibodies are often not positive proof of infection. An increase in the complement-fixation test supports the clinical diagnosis. A high ratio between the titers in serum and aqueous humor is even better proof.
Forms of retinochoroiditis in ocular toxoplasmosis:
- disseminated
- central
- fetal (featuring so-called macular coloboma)
- juxtapapillary
- recurrent.
Laboratory Tests to detect Toxoplasma Gondii Infection:
- fluorescent antibodies
- complement-fixation test
- polymerase chain reaction
- ratio of blood-aqueous humor titers
- vitreous body biopsy.
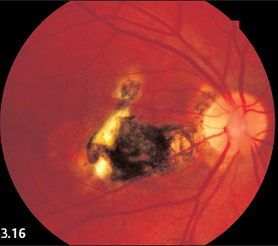
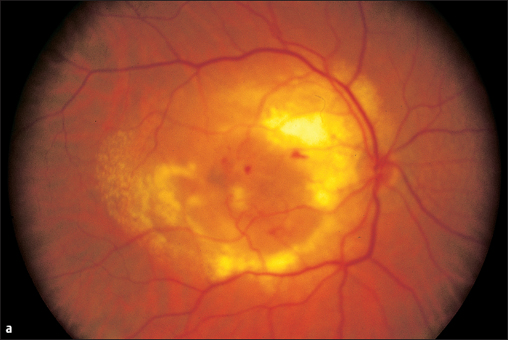
Fig. 3.16 Right eye with a large central scar secondary to fetal retinochoroiditis (macular coloboma).
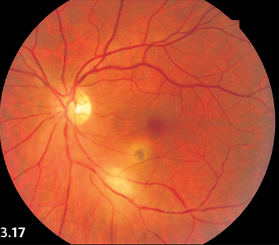
Fig. 3.17 Acute central retinochoroiditis. Small healing lesion near to an acute lesion are visible immediately inferior to the macula.
Central Fetal Toxoplasmosis
The fovea centralis is rarely affected in adults. Central, usually bilateral, involvement in toxoplasmosis is typical of fetal infection during the sixth to eighth month of pregnancy. The mother usually contracts a lung infection that goes unnoticed. The newborn’s eyes will already have developed extensive scars in the central retina, visible as large white defects with a pigmented halo. This means that visual acuity is greatly reduced right from birth, leading to secondary strabismus.
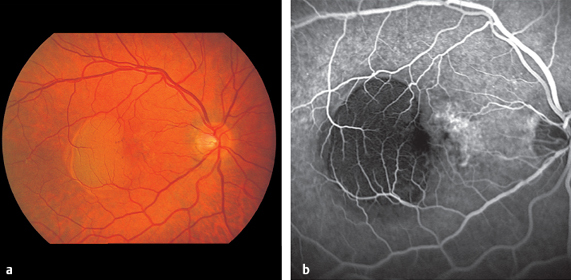
Retinochoroiditis Juxtapapillaris
The patient perceives lesions in close proximity to the optic disc as dark fields in front of the eye. These correspond to the typical sectoral visual field defects that result from interruption of nerve conduction due to focal pathology (Jensen disease Figs. 3.18 and 3.19).
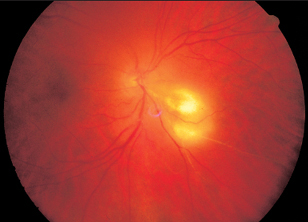
Fig. 3.18 Retinochoroiditis juxtapapillaris.
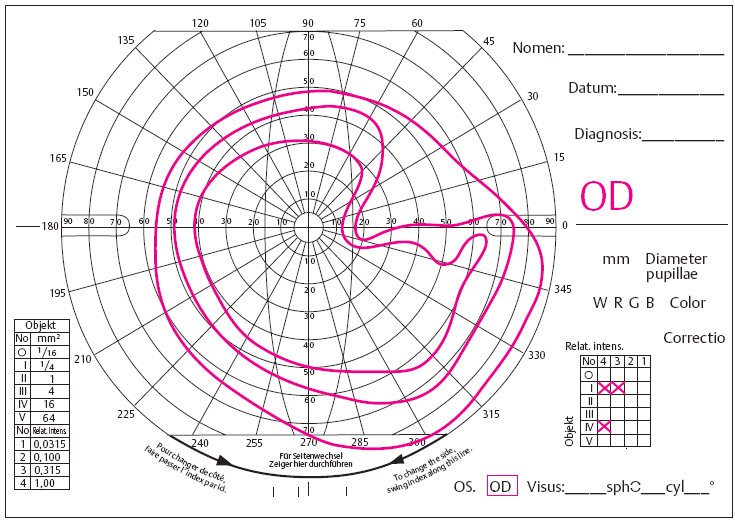
Fig. 3.19 Retinochoroiditis juxtapapillaris with typical sectorial visual field defect
Congenital Central Toxoplasmosis
- mother infected during the sixth to eighth month of pregnancy
- bilateral involvement
- occasionally asymmetrical
- large, white defect with pigmented halo in the central retina
- so-called “macular coloboma”
- severe loss of visual acuity, secondary strabismus
Retinochoroiditis Juxtapapillaris
- focal lesion adjacent to optic disc
- sectoral visual field defect
Differential Diagnosis of Retinochoroiditis
- toxoplasmosis, acute lesions and scars simultaneously
- miliary tuberculosis—many small lesions
- tuberculoma—large, isolated, focal lesion with cellular infiltration of the vitreous body
- mycosis—satellite lesions often present
- discontinuous arteriopathy—“shield” on arteriole, dense homogeneous vitreous opacification
- consider the possibility of HIV as the underlying infection
- systemic immune disorders, such as sarcoidosis
Ocular Manifestation of Sarcoidosis
- frequently recurrent iridocyclitis
- retinochoroiditis
- retinal vasculitis (see p. 163)
- scleritis
Inflammatory Granulomas
- optic nerve
- conjunctiva
- orbit
- lacrimal gland
Signs of General Sarcoidosis
- young adults aged 20–40
- women affected more often than men
- ocular involvement in 25–50% of all cases
- iridocyclitis usually present
- less often conjunctiva, sclera, orbita, or lacrimal gland involved
- posterior uveitis can also occur
- chorioretinal granuloma appears as retinochoroiditis with vitreous opacity
- retinal vasculitis (see p. 163)
Systemic Diagnosis of Sarcoidosis
- chest radiograph
- serum ACE level
- biopsy of an enlarged lymph node
- epithelioid cells
- giant cells
- macrophages
- epithelioid cells
Differential Diagnosis of Sarcoidosis
- tuberculosis
- all other causes of retinochoroiditis
- lupus erythematosus
- Wegener granulomatosis
- lymphomas
Other Features of Ocular Toxoplasmosis
Acute focal lesions appear increasingly luminous, whereas chronic scars exhibit the consistent white gleam of the sclera and choroidal vessels. In rare cases, the chorioretinitis is severe enough to produce extensive edema, or extensive lesions give rise to a diffuse and occasionally very dense vitreous opacification. In these cases, one should consult an internist and look for a systemic infection and other general diseases.
The cellular vitreous opacification is often accompanied by signs indicative of secondary vasculitis. These include ill-defined margins of the adjacent retinal vessels and fine gray-white longitudinal stripes along the vessels. Where these longitudinal stripes lie on both sides of the vessel, they are referred to as “vascular sheathing.” In discontinuous reversible arteriopathy (Kyrieleis disease), it is this vasculitis rather than the focal lesion that is the prominent characteristic of the disorder (p. 163).
Treatment: often clinical findings are unequivocal so that prolonged oral therapy with clindamycin is the current treatment of choice. Pyrimethamine (Daraprim) and sulfonamides (trimethoprim and sulfamethoxazole) are also effective. Corticosteroids should be used sparingly.
Retinochoroiditis in Sarcoidosis
Sarcoidosis is a systemic granulomatous inflammation that can occur in any organ. Histologically, the granulomas are characterized by accumulations of macrophages, by typical epithelioid cells, and by multinucleated giant cells. Sarcoidosis most often manifests itself in the eye as acute recurrent iridocyclitis. However, inflammatory granulomas can cause various clinical syndromes in any part of the eye. Young adults between the ages of 20 and 40 often develop sarcoidosis. Women are affected two to three times more often than men. Ocular involvement is present 25–50 % of all sarcoid disease. Conversely, about 3–7% of all cases of uveitis are attributable to sarcoidosis.
A choroidal or retinal granuloma appears clinically as retinochoroiditis with focal lesions of varying size. Fig. 3.20a shows multiple ill-defined confluent infiltrates in the early stages of acute inflammation. Usually, these inflammatory granulomas are accompanied by circumscribed or diffuse cellular opacification of the vitreous body. Because of this, the focal lesions on the fundus often appear blurred. Retinochoroiditis in sarcoidosis heals slowly over a period of months. Often it takes one or two years before chorioretinal scars develop (Fig. 3.20b). The diagnosis is made on the basis of comprehensive examinations by a general practitioner or, better, a specialized internist. Classic findings in sarcoid disease include a chest radiograph demonstrating typical hilar adenopathy in the lungs. Lymph nodes must be located and a biopsy obtained wherever possible because histologic diagnosis is still regarded as highly reliable. Other common general findings included increased level of serum angiotensin converting enzyme (ACE), increased immunoglobulins, and a negative tuberculin skin test.
Treatment: this consists of local and/or systemic corticosteroid therapy over a period of months. As this is invariably a systemic disease, treatment should be rendered in close consultation with an internist.
Ocular Tuberculosis
- large, focal retinochoroiditis lesion: tuberculoma.
- many small focal lesions: miliary tuberculosis
- look for systemic disease
Tuberculosis
A retinochoroiditis lesion can also be caused by a tuberculoma. Ophthalmoscopy cannot distinguish this infection from other infections such as toxoplasmosis. Miliary tuberculosis is characterized by multiple, disseminated focal lesions on the fundus that are fluffy and less compact than those in toxoplasmosis. Tuberculous retinochoroiditis produces hardly any symptoms. However, in the context of the severe primary disease, it should be evaluated and treated in close cooperation with an internist. All forms of ocular tuberculosis are recurrent. When in doubt, a polymer chain reaction (PCR) with blood or, better yet, with a vitreous body biopsy specimen is recommended in addition to examination by an internist.
Treatment: once the diagnosis has been confirmed, tuberculostatic treatment must be performed.
Mycotic Retinochoroiditis
- veil or shadow in front of the affected eye as in disseminated retinochoroiditis
Signs
- focal lesions are more compact and more sharply demarcated
- arranged in groups with small satellite lesions
- spherical focal lesions in vitreous body as well, often aligned like strings of pearls
- clinical diagnosis often clear
- uncertain sensitivity to chemotherap
Laboratory Studies
- in blood: antibodies occasionally found in serum
- vitreous biopsy: direct identification of bacterial or mycotic pathogen
- polymerase chain reaction
Mycoses
Patients at high risk for intraocular mycoses:
- drug addicts
- patients infected with HIV
- immunosuppression
- patients with massive blood loss
- patients in poor general health
- patients with massive infections.
The lesions resemble those in disseminated retinochoroiditis although they are more compact and often exhibit diffuse infiltration into the surrounding region. Candida infections appear whiter whereas aspergillosis tends to be more yellowish. Typical findings in mycotic retinochoroiditis include small satellite lesions in the retina and, crucial to the diagnosis, in the adjacent vitreous body. There they often form patterns resembling strings of pearls (p. 175).
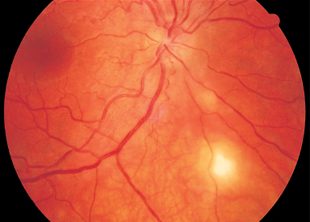
Fig. 3.21 Retinochoroiditis due to Candida albicans in a 20-year-old woman with contact to the drug scene. A small satellite lesion is visible above the main lesion.
Treatment: antimycotics poorly penetrate the blood-retina barrier to pass into the vitreous body. Therefore, intravitreous injections and intravenous infusions of antimycotics are recommended.
- amphotericin-B
- fluconazole
- voriconazol.
Caution: 1ocal and systemic toxicity.
Small Retinal Tumors
Incipient Retinoblastoma
- occurs in very young children
- white focal lesion on the fundus
- ill-defined
Incipient Retinoblastomas
Retinoblastomas begin as small white focal lesions, often simultaneously at several sites. The lesions are ill defined, and are initially difficult to distinguish from retinochoroiditis. Such findings should be carefully observed in newborns and infants until a positive differential diagnosis has been made. Retinoblastomas later develop into tuberous growth. See pages 123, 168, and 173 for a detailed description.
Incipient “Mulberry” Tumors
- typical berrylike structure
- white focal lesion on the fundus
- sharply defined margin
- hereditary disorder (family history is important!)
Incipient Tumors in Tuberous Sclerosis—Bourneville Disease
The small tumor foci are white and sharply demarcated. From the beginning, they exhibit a typical berrylike, multilobulated structure. There is no associated cellular infiltration of adjacent tissue. A family history of this hereditary astrocytic retinal hamartoma often leads to the correct diagnosis. See p. 124 for a detailed description of this disorder.
Treatment: very small tumors can be destroyed by laser photocoagulation.
Structured Focal Lesions due to Parasites
Parasites have few, but specific intermediate hosts. There are geographic regions where they are highly prevalent. Therefore, an important step in making the diagnosis is to obtain a detailed history. Parasites that occur primarily in the eyes are often phototrophic. Often one must also consider involvement of other organs as well.
Toxocara Canis
- foci often temporal or nasal to the optic disc
- children (ages 9–16) often affected
- history of contact with dog and soil
- ELISA for antibodies against pathogen capsule
- eosinophilia in blood and granulomas
Differential Diagnosis
- retinoblastoma
- Coats disease
- endophthalmitis
- retinochoroiditis
Toxocara Canis (Subspecies of Nematodes)
This parasite is ubiquitous. About 50 % of all dogs are carriers of Toxocara canis. Infection occurs by ingestion of the eggs of the parasite. These develop into larvae that penetrate the wall of the bowel and spread hematogenously in large numbers. They have a life span ranging from months to years and rarely develop into adult worms.
Life cycle of Toxocara canis (nematode):
- infection with worm eggs from soil contaminated with dog feces
- larvae disseminate through the body; sickle-shaped larva migrans measures 1–2 disc diameters
- inflammatory infiltrates on fundus, vitreous involvement.
Other organs are often infested as well (visceral larva migrans). In the eye, focal chorioretinitis occurs at the posterior pole. Gray–white infiltrates measuring one disc diameter indicative of subretinal eosinophilic granulomas appear on the fundus. Infiltration of the vitreous body occurs.
Treatment
Caution: the destroyed parasites can elicit a severe immune reaction. Therefore, corticosteroids are also indicated.
Echinococcus
- rare in the eye
- hydatid cysts with solid capsule
- subretinal or intravitreal
- slight inflammatory reaction
- ultrasound detects cysts
- no definitive laboratory tests
- eosinophilia in blood
Oocysts on the Fundus
- larvae grow slowly, approximately 12 mm in two to four years
- worm is occasionally visible and mobile
- slight inflammatory reaction
- capsule: calcifications
- CT and ultrasound
Echinococcus (Dog Tapeworm)
As in other worm disorders, the larvae disseminate through the body from the bowel. They then form hydatid cysts that gradually enlarge. Diagnosis is usually made based on histologic examination demonstrating typical heads (scolices) with a double hooked ring.
Treatment : surgical.
Cysticercosis—Taenia Solium (Pork Tapeworm)
In this disorder, infection occurs by ingesting tapeworm eggs. This may be the result of contamination with the feces of infected humans or animals, such as in raw manure. The larvae disseminate through the bloodstream, often infesting the eyes as well. There they form isolated subretinal oocysts that enlarge. Once they have attained a certain size, the growing organism becomes visible. As the antigenicity of these parasites is low, serologic studies are not very helpful in making a diagnosis. The adult tapeworm only arises when the meat of its host is eaten. The parasite passes to humans in food such as beef tartar or undercooked pork.
Treatment : surgical.
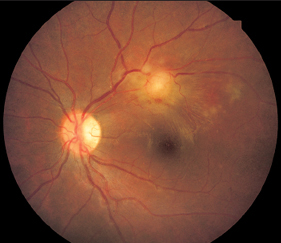
Fig. 3.22 Echinococcus cyst on the fundus of a 14-year-old girl. The patient and her father had pronounced eosinophilia. The father died with liver cysts confirmed in pathologic examination.
Internal Ophthalmomyiasis
- history
- elongated infiltrates
- maggots on the surface of the conjunctiva
- sequelae including areas of retinal and choroidal atrophy, exudative retinal detachment, and subretinal scars
Internal Ophthalmomyiasis
Certain species of tropical flies deposit their eggs or hatched maggots on the moist surface of the conjunctiva in flight like a dive-bomber. Occasionally, the maggots migrate into the inner eye. They can pass beneath the conjunctival epithelium and from there actively migrate into deeper tissue as far as the subretinal space, where they continue to grow. They produce elongated infiltrates with a shape resembling a worm and later lead to scars of the same shape. The diagnosis is made based on the patient’s history and, possibly, from maggots found on the surface of the conjunctiva. Sequelae include areas of retinal and choroidal atrophy, exudative retinal detachment, and subretinal scars with tractional retinal detachment.
Treatment : surgical removal of the fly larvae.
Diffuse Unilateral Subacute Neuroretinitis (DUSN)
Treatment: there is no known effective treatment.
Major Ophthalmic Lymphomas
- chronic myelogenous leukemia
- Hodgkin disease
- non-Hodgkin lymphomas
Diagnostic Procedures
- diagnostic aspiration or vitrectomy
- choroid biopsy if indicated
Chronic Myelogenous Leukemia
- hemorrhages on the fundus with white centers
Multifocal Yellowish-White Flat Infiltrations
Infiltrations of such appearance may be caused by retinal lymphomas, which occasionally invade the choroid and the retina. Retinal oculo-cerebral lymphomas are very rare. A differential diagnosis must exclude many other disorders. However, retinal lymphomas exhibit a few characteristics that are helpful in making a diagnosis.
The major ophthalmic lymphomas include Hodgkin disease, non-Hodgkin lymphomas (oculo-cerebral lymphoma), and chronic myelogenous leukemia.
Hodgkin Disease
- subretinal granulomas as in retinochoroiditis
Hodgkin Disease
Hodgkin lymphomas produce granulomatous choroidal infiltrates that resemble focal lesions in chorioretinitis. Isolated or multiple nodules of this sort may be present. This disorder can only be distinguished from other inflammatory disorders on the basis of systemic findings.
Non-Hodgkin Lymphomas
- B cell: oculo-cerebral
- T cell: oculo-visceral
- yellowish, subretinal infiltrates measuring one-quarter to one disc diameter, smooth margins
- pseudouveitis
- cellular infiltrates in the vitreous body
- fluorescein angiography: permanent masking of choroidal fluorescence
Differential Diagnosis of Lymphomas
- metastases of other tumors (breast carcinoma, bronchial carcinoma, lung carcinoma)
- uveitis
- diffuse panuveitis
- disseminated retinochoroiditis
- Harada syndrome
- acute posterior multifocal placoid pigment epitheliopathy (APMPPE)
Non-Hodgkin Lymphomas—Diffuse Malignant Lymphomas (Ocular-Cerebral Lymphoma)
T-cell and B-cell lymphomas can involve the eye, and appear as oculovisceral or oculocerebral lymphomas, respectively. They produce focal accumulations of cells in the subretinal space. Ocular involvement is often noticed before other organs become affected. The size and localization of the lymphomas can change over a period of weeks and months, and they can even disappear. These lesions permanently mask the fluorescence of the choroid on fluorescein angiography. Occasionally cellular infiltration is observed in the vitreous body (and to a slight extent in the aqueous humor as well). This often mimics uveitis (p. 172). When in doubt, diagnostic vitreous aspiration or vitrectomy is indicated. Cytologic examination can lead to a diagnosis.
These tumors were once referred to as reticulum cell sarcomas, a term that aptly described their malignancy and their pathologic and anatomic appearance. Current nomenclature classifies them as non-Hodgkin lymphomas. Ophthalmologic examination can only provide the basis for clinical suspicion. The diagnosis is confirmed by pathologic examination.
Treatment: cytostatic treatment of the underlying disorder.
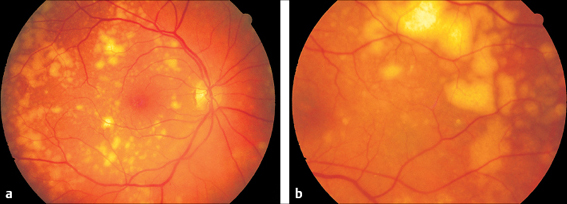
Fig. 3.23 a and b Malignant oculovisceral lymphoma. a Yellow–white, sharply demarcated infiltrates in the fundus of the right eye of a 77-year-old woman. b Left eye of the same patient. Here, the lymphoma lesions lie further in the periphery and are confluent.
Choroidal Metastases
- visual field defects, loss of visual acuity
- round, yellowish-white protrusions
- fluorescein angiography: early and persistent fluorescence typical of tumor
- systemic findings and imaging modalities
Early Tumor Metastases, Initially Still Flat
Choroidal metastases can occur with the following carcinomas:
- breast carcinoma
- bronchial carcinoma
- lung carcinoma.
Treatment: surgical and cytostatic treatment of the underlying disorder.
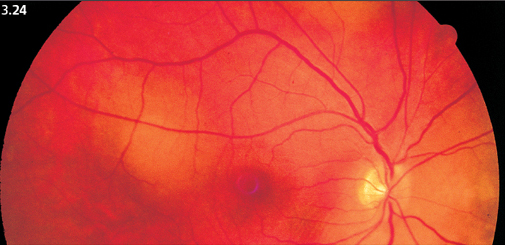
Fig. 3.24 Small choroidal metastases of a breast carcinoma, still flat at this stage.
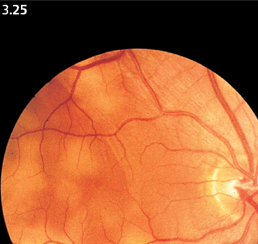
Fig. 3.25 Multiple metastases of small-cell lung carcinoma, appearing as flat focal lesions. The 43-year-old patient died eight weeks later. The diagnosis was made through autopsy.
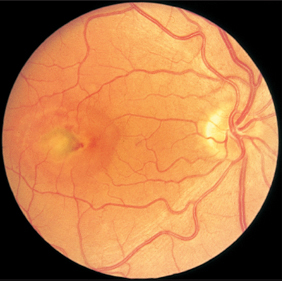
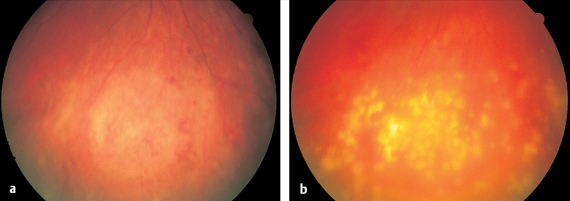
Fig. 3.26a–c Ocular histoplasmosis syndrome (POHS).a Dirty gray macular edema. A small retinal hemorrhage is visible at the margin.
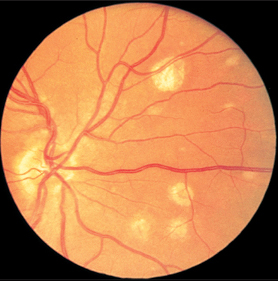
Fig. 3.26c Round scars further in the periphery of the fundus so called “histo spots.”
Presumed Ocular Histoplasmosis Syndrome (POHS)
- distorted vision
- decrease in visual acuity to 20/40–20/200
- often bilateral, lasting several months
- usually affects women between 20 and 40
- retinal edema in or near macula (gray spot) with crescent-shaped or round marginal hemorrhages
- fluorescein angiography: “classic” neovascularization appears early during the choroidal filling phase
- focal peripheral scars form “histo spots”
Presumed Ocular Histoplasmosis Syndrome (POHS)
The disorder first manifests itself as distorted vision and a decrease in visual acuity to between 20/40 and 20/200. Findings on the fundus include a retinal edema in or adjacent to the macula with crescent shaped or round hemorrhages at its margin.
Fluorescein angiography reveals sharply demarcated (classic) choroidal neovascularization similar to that seen in age-related macular degeneration. Occasionally visual acuity will continue to decrease only gradually or not at all, and the patient will not necessarily lose the ability to read. Histoplasma infection is detectable by a skin test in the United States. In Europe, no pathogens have yet been identified (except for isolated cases of Borrelia burgdorferi). A chorioretinal scar develops after two to three months. Similar new lesions appear immediately or within a period of weeks to months. These new lesions may be observed directly adjacent to the scar, in the broader vicinity of the posterior pole of the eye, or further in the periphery of the fundus. They, too, heal with small yellow atrophic scars known as “histo spots.” The initial central retinal involvement and diffuse scarring represent a typical clinical syndrome.
Treatment: prolonged administration of systemic corticosteroids.
APMPPE
Generalized Symptoms
- flulike infection
- neurologic deficits
- encephalitic signs
- frequently distorted vision
- shadows in visual field
- occasionally cerebral vasculitis with Lyme disease
On the Fundus
- 10 to 20 fine, slightly prominent gray–white focal lesions, at the posterior pole, size one-quarter to one-half of a disc diameter
- fluorescence initially masked, luminous in late phase
- visual acuity temporarily reduced, rarely permanently
- choroidal scars with fine pigment clumping appear within a few weeks
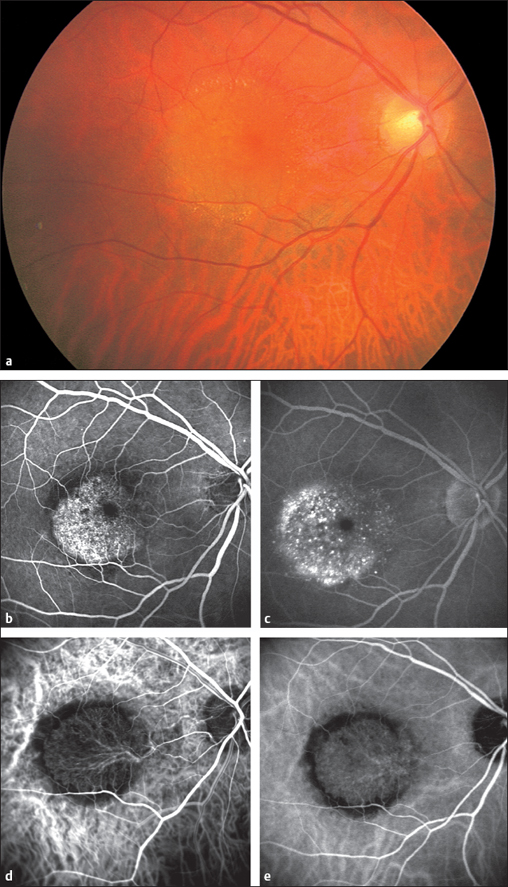
Acute Posterior Multifocal Placoid Pigment Epitheliopathy (APMPPE)
If one of these focal lesions lies directly on the fovea, visual acuity can decrease to 20/200 or less. Less often, slit lamp examination will reveal a fine cellular exudation in the vitreous body. The diagnosis is made by fluorescein angiography: the focal lesions initially remain dark, masking the fluorescence of the choroid and retinal capillaries. The exudation in the inflammatory lesions becomes luminous only in the late phase of the angiogram and remains visible for a long time. Choroidal scars with fine pigment clumping develop within a few weeks. Visual acuity often recovers, and permanent impairments are rare.
Treatment: there is no known effective treatment.
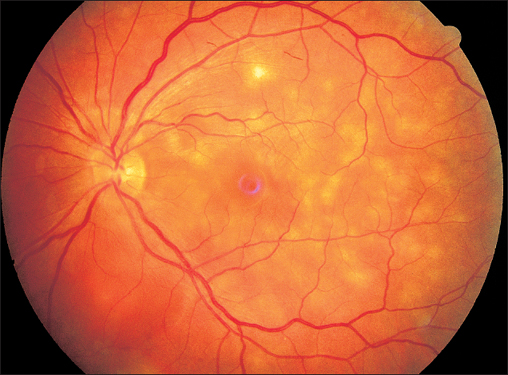
Fig. 3.a–c Acute posterior multifocal placoid pigment epitheliopathy (APMPPE). a Disseminated indistinct lesions.
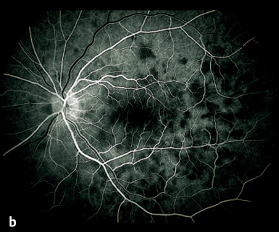
Fig. 3.27b b Fluorescein angiography of APMPPE: lesions mask fluorescence in the early phase (after eight seconds).
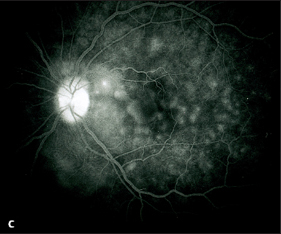
Fig. 3.27c Fluorescein angiography of APMPPE: the inflammatory lesions appear luminescent in the choroid in the late phase (after four minutes).
Birdshot Retinochoroidopathy
- commonly occurs in women between 20 and 40
Early Phase of the Disease
- broad areas of retinopathy
- bilateral
- peripapillary infiltrates similar to grains of rice
- rapidly forming chorioretinal scars, increasing centripetally toward the periphery
- visual acuity slightly reduced (such as 20/40)
- ERG: b-wave reduced
Late Phase of the Disease
- HLA-A29 positive vasculitis
- vascular sheathing with retinal and vitreous bleeding
- irregular visual field defects
- decrease in visual acuity
Birdshot-Retinopathie
Birdshot retinochoroidopathy begins with peripapillary gray–white round to oval retinal infiltrates the size of grain of rice. These eruptions disseminate toward the periphery in a pattern resembling a shotgun blast, hence the name of the disease. The infiltrates are transient and soon turn into many small chorioretinal scars. Severe retinal vasculitis develops within a period of years and may be accompanied with vitreous bleeding and perivascular infiltrates. The optic disc becomes pale, and visual acuity decreases. Visual field defects also occur. In the final phase, vascular sheathing occurs in the peripheral retinal vessels. The strong association between retinal vasculitis and the HLA-A29 antigen is thought to be indicative of its pathogenesis (p. 165). However, there is also HLA-A29 positive vasculitis that does not produce birdshot lesions on the fundus.
Treatment: immunosuppressive treatment over a period of months and years is indicated at the first sign of retinal vasculitis. Neovascularization is treated by local laser photocoagulation.
Multiple Evanescent White Dot Syndrome
- blurred vision
- decrease in visual acuity to 20/80 or less
- multiple, dull, white ill-defined confluent focal lesions measuring one-tenth to one-quarter of a disc diameter
- enlarged blind spot often present in visual field
- pathologic ERG and EOG
- fluorescein angiography: mottling of the retinal pigment epithelium leakage from optic disc and retinal capillaries.
- indocyanine green angiography: hyperfluorescent spots visible in late phase
- resolves spontaneously
Multiple Evanescent White Dot Syndrome
This disease of the fundus is frequently unilateral but occasionally bilateral. Symptoms include blurred vision and a decrease in visual acuity to about 20/80 or worse. Fundus findings include disseminated, dull white, round, ill-defined lesions 1/10 –1/4 of a disc diameter. These lesions are minimally or not at all prominent and confluent.
Fluorescein angiography reveals mottling, a diffuse pattern of focal exudation, in the retinal pigment epithelium. Notable findings include additional exudation of fluorescein from the optic disc and retinal capillaries. Hyperfluorescent focal lesions appear in the late phase of indocyanine green angiography after about 20–30 minutes. Some of these may be confluent with the optic disc. These findings largely correlate with the enlargement of the blind spot in the visual field. Symptoms of the disease disappear within a few weeks without treatment. Visual acuity, electroretinography, electro-oculography, and fluorescein angiography findings then return to normal. The disorder may recur or form central neovascular subretinal membranes.
Treatment: there is no known effective treatment.
Acute Retinal Pigment Epithelitis
- flulike infection
- bilateral decrease in visual acuity
- small perimacular focal lesions measuring one-tenth of a disc diameter, initially gray later depigmented with a spot of pigment in the center
- resolves spontaneously; usually with good visual acuity
Acute Retinal Pigment Epithelitis
Onset is often bilateral and is accompanied by flulike complaints. Symptoms include decreased visual acuity, metamorphopsia, and central scotomas. The center of the focal lesion is initially masked in the fluorescein angiogram; the irregularly demarcated pigment epithelium appears luminescent all around it. All symptoms remit within six to eight weeks.
Treatment: there is no known effective treatment. Systemic corticosteroids are used to treat severe exudations.
Multifocal Inner Choroiditis (MIC)
- decrease in visual acuity
- metamorphopsia
- commonly younger myopic women
- multiple small, round to oval lesions, clearly demarcated
- initially grayish-yellow, later yellowish-white
- in RPE and choriocapillaris
- cells in the vitreous body and aqueous humor (in contrast to punctate inner choroidopathy)
- fluorescein angiography: acute and healed lesions hyperfluorescent
Multifocal Inner Choroiditis (MIC)
Other names for multifocal inner choroiditis and punctate inner choroidopathy:
- chorioretinopathy with anterior uveitis
- multifocal choroiditis with panuveitis
- progressive subretinal fibrosis and uveitis
- multifocal inner choroiditis with disciform macular degeneration
- inflammatory pseudohistoplasmosis.
The inflammatory process occurs within the retinal pigment epithelium and in the choriocapillaris.
One possible cause is sarcoidosis. Subclinical infections with tuberculosis, EpsteinBarr virus, syphilis, and histoplasmosis have also been discussed. Between five and 20 focal lesions appear nearly simultaneously over the entire fundus, both in the periphery and at the posterior pole. They measure from 100 to 200μm in diameter, are round to oval, sharply demarcated, and initially gray-yellow, later turning yellow-white.
The healed lesions appear as spots of retinal and choroidal atrophy (see Fig. 3.76 on page 83). They are often surrounded by a pigmented ring. Some of these scars are shaped more like bands and exhibit subretinal fibrosis. Often, in about 45 % of reported individuals, choroidal neovascularization and peripapillary atrophy are present. Genuine disciform lesions (p. 54) have also been observed. New lesions occur when the old ones have healed. This is why the disorder is also known as recurrent multifocal inner choroiditis. The electro-oculogram and electroretinogram are correspondingly reduced depending on the size of the diseased area of the fundus.
Complications of multifocal inner choroiditis:
- frequent recurrence
- subretinal fibrosis
- choroidal neovascularization
- disciform lesions.
The prognosis is poor since there are frequent recurrences. The fovea itself is rarely completely destroyed so that visual acuity is often preserved. Multifocal inner choroiditis and punctate inner choroidopathy can be distinguished by characteristic clinical findings. Punctate inner choroidopathy does not include cells in the vitreous body and aqueous humor whereas these signs of inflammation are invariably present in multifocal inner choroiditis.
Treatment: corticosteroids are administered to improve visual acuity. Systemic antibiotics are indicated if infectious etiology is suspected.
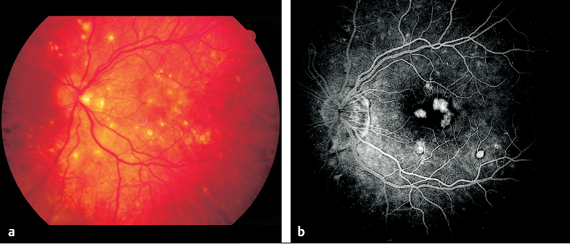
Fig. 3.28 a and b Punctate inner chorioretinopathy (PIC) in a 39-year-old woman. a Numerous sharply demarcated round to oval yellow-white lesions are present. b Fluorescein angiography: the lesions exhibit fluorescence in the later venous phase but little exudation (after 37 seconds).
Punctate Inner Choroidopathy (PIC)
Sympathetic Ophthalmia
- latency days to years after penetrating injuries or surgical procedures
- decreased visual acuity
- sensitivity to glare
- anterior uveitis, disseminated white spots on the fundus
- cells in the vitreous body
Sympathetic Ophthalmia
This severe panuveitis can occur secondary to penetrating injury or surgical procedures of one eye and leads to uveitis in the fellow eye within a period of a few days to several years. In addition to diminished visual acuity and increased sensitivity to glare, the disorder is primarily characterized by anterior uveitis. Fundus findings include cells in the vitreous body and disseminated white spots on the fundus. Refinements in modern surgical techniques have made the disease rare.
Treatment: high doses of systemic corticosteroids and immunosuppressive agents.
 Literature
Literature
Gass JDM. Macular diseases. Saint Louis: The CV Mosby Company; 1977.
Remky H. Kammerwasseranalytik bei intraokularer Toxoplasmose. Ophthalmologe. 1998;95:120–1.
 Prominence of the Macula
Prominence of the Macula
 Secondary Macular Edema
Secondary Macular Edema
Causes of macular edemas:
- age-related macular degeneration
- Irvine–Gass syndrome
- retinal vein occlusion
- diabetic retinopathy
- arterial hypertension
- uveitis
- Leber miliary aneurysms
- Coats disease
- Behçet disease
- Eales disease
- retinal periphlebitis
- discontinuous reversible arteriopathy (Kyrieleis disease)
- retinitis pigmentosa
- birdshot retinochoroidopathy
- inflammatory processes
- retinal detachment
- macular pucker
- possibly idiopathic.
Diagnosis of Macular Edema
- decrease in visual acuity
- history: see list above
- ophthalmoscopy: contour of cysts visible near focal light
Angiography is Diagnostic Proof
- angiographic findings often present without significantly diminished visual acuity
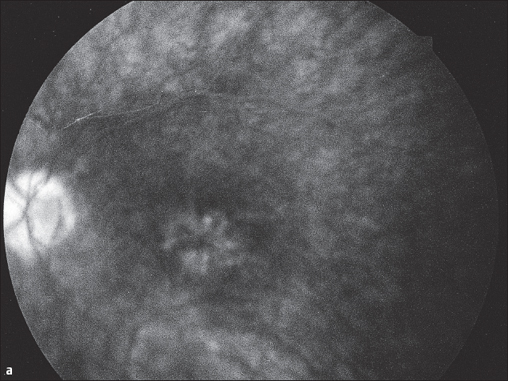
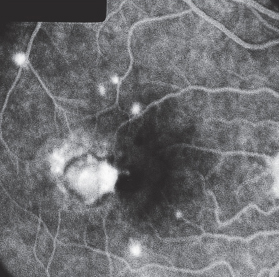
Fig. 3.29a and b Cystoid macular edema following to cataract surgery (Irvine-Gass syndrome). a Typical stellate exudation around the fovea centralis appears in the late phase of fluorescein angiography (after five minutes).
The cystoid macular edema occurs relatively often together with:
In a few of the disorders mentioned above, the cystoid macular edema gradually develops over a period of weeks to months. Visual acuity gradually decreases until the patient is unable to read. It is highly probable that cystoid macular edemas occur under the influence of prostaglandins. Once the frequently associated inflammatory symptoms have disappeared, a cystoid macular edema can resolve over a period of months and visual acuity can improve, especially in juvenile patients.
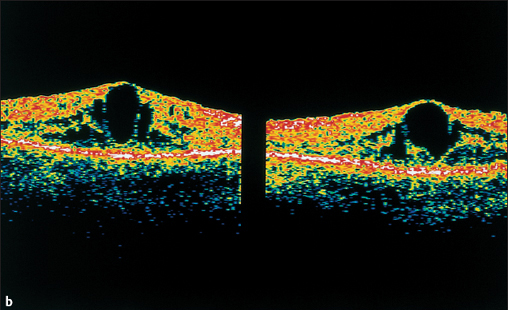
Fig. 3.29b Optical coherence tomography (OCT) demonstrates the anatomic position of the cysts.
Macular Edema with Aphakia or Pseudophakia (Irvine-Gass Syndrome)
Cystoid macular edema occurs in 10–20 % of all cases following intracapsular cataract operations, less frequently after extracapsular operations (2 %). Its incidence increases significantly secondary to rupture of the posterior capsule of the lens. Cystoid macular edema also occurs secondary to other procedures that lead to decreased intraocular pressure (paracentesis effect).
Macular Edema with Posterior Uveitis
Cystoid macular edema is a long-term complication of chronic inflammation. It often manifests itself with the symptoms described above after an insidious clinical course of several months. Fluorescein angiography is indicated if visual acuity decreases without correlating ophthalmoscopic findings.
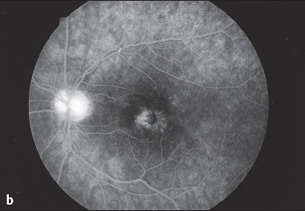
Fig. 3.30b Late phase of fluorescein angiography (after five minutes) showing the parafoveal fluorescence typical of macular edema. The hyperfluorescence of the optic disc is a sign of the inflammatory process..
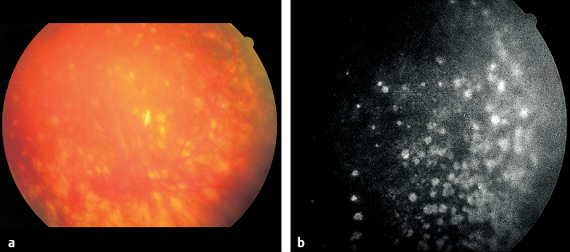
Fig. 3.31a and b Intermediate uveitis—pars planitis. a The inferior periphery of the fundus exhibits typical preretinal “snowball” infiltrates also retinal and choroidal lesions posterior to them. b Fluorescein angiography: the retinal and choroidal lesions appear brightly luminous.
Cystoid Macular Edema with Pars Planitis—Intermediate Uveitis
- cells in the vitreous body
- peripheral “snowball” infiltrates
- retinal and choroidal atrophy anterior to the ora serrata
- often extending to the equator
Macular Edema with Intermediate Uveitis—Pars Planitis
Cystoid macular edema often determines the prognosis for the underlying disorder with respect to visual acuity.
Treatment
- treatment of the underlying disorder
- cyclo-oxygenase inhibitors
- systemic and peribulbar corticosteroids where indicated.
 Macular Edema with Retinal Vascular Disease
Macular Edema with Retinal Vascular Disease
Macular Edema in Diabetic Retinopathy
Leakage and occlusion of the perifoveal capillaries often leads to exudative and later on to ischemic macular edema (p. 143).
Macular Edema in Central Retinal Vein Occlusion
Cystic macular edema that decreases visual acuity occurs not only with truncal occlusion of the retinal veins, but also with occlusion of minor branches of the perimacular veins. In complete retinal vein occlusion, these edemas are often irreversible.
Treatment
- treatment of the underlying disorder
- adjunctive administration of systemic carbonic anhydrase inhibitors if necessary.
Central Serous Chorioretinopathy
- clear retinal edema
- commonly affects middle-aged men
- cause unknown but apparently stress-related
- slightly diminished visual acuity, blurred vision
- fine, glassy retinal edema
- hyperopia phenomenon
- fluorescein angiography: characteristic “smokestack” sign
- frequently recurrent
- often bilateral
- often progresses to chronic serous chorioretinopathy
Central Serous Chorioretinopathy
This disorder commonly affects middle-aged men. Patients complain of blurred vision. Findings in the macular region of the fundus include a fine, glassy retinal edema. Circular glistening is usually observed around the foveal reflex. The brown macular pigment appears less intense. The examination with the slit lamp and three-mirror lens reveal a fine gray line in front of the fundus indicative of serous elevation of the central retina. Often there will also be small circumscribed detachments of the pigment epithelium in the vicinity of the neuroretinal edema. The fluorescein angiogram is typical (Figs. 3.32b, c).
Treatment: there is no known effective medical treatment. Reduction of stress is indicated. Steroids are contraindicated.
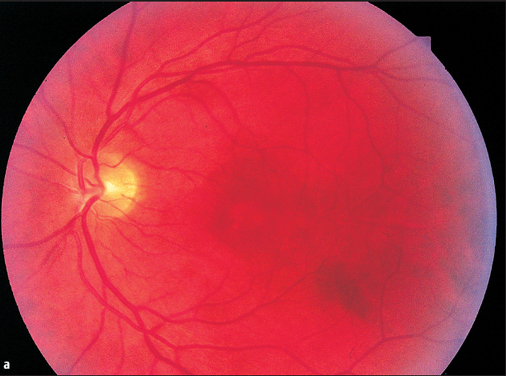
Fig. 3.32a-c Central serous chorioretinopathy.a Circumscribed clear transudate in the macula, easily missed ophthalmoscopically.
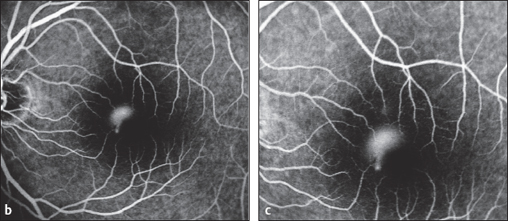
Fig. 3.32b-c entral serous chorioretinopathy. b Fluorescein angiography: a focal area of hyperfluorescence appears in the venous phase. c Fluorescein angiography: within a few minutes, the fluorescence expands upward in the classic “smokestack” sign.
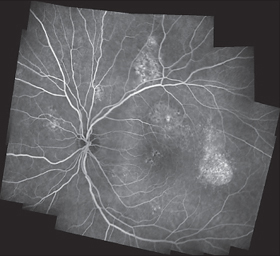
Fig. 3.33 Chronic central serous chorioretinopathy. Fluorescein angiography: defects in the pigment epithelium with slow exudation.
Diffuse Retinal Pigment Epitheliopathy (Chronic Serous Chorioretinopathy)
- irregular pigmentation in the macula
- fluorescein angiography: fine circumscribed defects in the RPE
Diffuse Retinal Pigment Epitheliopathy (Chronic Serous Chorioretinopathy)
Treatment: there is no known effective treatment.
 Macular Edema from Choroidal Neovascularization
Macular Edema from Choroidal Neovascularization
Choroidal neovascularization (CNV) primarily leads to the wet form of age-related macular degeneration (AMD). It is the most common cause of significant loss of visual acuity involving the central retina in patients over the age of 50. This is a disorder of the central retina, which is crucial to the most important visual functions such as reading, recognition of fine detail, and color vision. Age-related macular degeneration (AMD) may be subclassified into an early degenerative stage with drusen and changes in the retinal pigment epithelium and a late atrophic or edematous stage. The early stage is also known as age-related maculopathy. In this stage of the disorder, visual acuity is usually minimally impaired. The late stage of age-related macular degeneration is characterized by either areolar atrophy of the central retinal pigment epithelium or exudative changes as a result of choroidal neovascularization. The disorder is accordingly divided into a “dry” form and a “wet” or exudative form. The following sections are based on the classification system in Fig. 3.34.
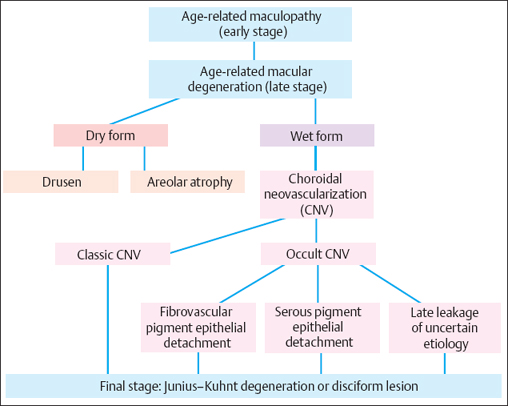
Fig. 3.34 Classification of age-related macular degeneration (AMD
Drusen
- between RPE and Bruch membrane
- lipid deposit in Bruch membrane
- impaired diffusion in the RPE
- visual acuity minimally impaired
Age-related Maculopathy—Drusen at the Posterior Pole of the Eye
Drusen are yellow–white subretinal deposits visible on ophthalmoscopy. Sometimes they appear dull, sometimes shiny, and crystalline. They may occur as isolated lesions or as multiple and confluent lesions. Drusen are classified as “hard,” “soft,” or “reticular.” Today, hard drusen are no longer regarded as a sign of disease as they occur in 90 % of all patients above the age 60. Soft drusen are not very sharply demarcated and may be confluent (for detailed description see Chapter 3.1, Figs. 3.1 and 3.2).
Aside from drusen, typical changes in age-related maculopathy include hyperpigmentation or depigmentation. The disorder can progress to age-related macular degeneration.
Treatment: 1ong-term oral therapy with alpha-tocopherol (vitamin E), ascorbic acid (vitamin C), zinc, and beta-carotene reduces the risk of progression by 25%. Some authors recommend treating the pigment epithelium adjacent to the drusen with low-dose argon laser photocoagulation.
Dry AMD
- atrophy of the retinal pigment
- epithelium (RPE)<180 μm
- as disease progresses:
- areolar atrophy
Areolar Atrophy
- sharply demarcated
- homogeneous red
- granular pigmentation
- central scotoma
- loss of visual acuity often late
- increased autofluorescence at the margin
Age-Related Macular Degeneration (AMD)
Dry Form of AMD—Areolar Atrophy
The dry form is characterized by progressive atrophy of the retinal pigment epithelium, followed by degeneration of the retina and choriocapillaris. One refers to dry age-related macular degeneration once the atrophy has reached a diameter of 180μm.
The atrophic areas usually enlarge during the further course of the disorder. This may then be referred to as areolar atrophy. The lesion consists of an irregularly demarcated area of retinal pigment epithelium atrophy, usually involving the macula. The lesion is sharply demarcated and often surrounded by a fine, pigmented halo. The retina has degenerated to a thin layer of glial cells, and the retinal pigment epithelium has disappeared. Often a small area of intact pigment epithelium will remain in the foveal region, and the patient will retain good visual acuity for a long time. Autofluorescence imaging reveals a lack of autofluorescence at the site of the atrophy and usually increased autofluorescence at the margin of the atrophic area. Often the atrophic area will expand toward the region of increased autofluorescence.
Treatment: there is no known effective medical treatment.
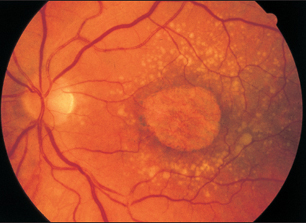
Fig. 3.35 Areolar macular atrophy, surrounded by numerous softdrusen, some of which are confluent.
Wet AMD
- choroidal neovascularization (CNV)
- serous pigment epithelial detachments
Course of Symptoms in CNV
- metamorphopsia for two to four weeks
- relative central scotoma for a few weeks
- absolute central scotoma after six to eight weeks
- visual acuity decreases to less than 20/400
Expansion of CNV
- initially punctate
- after six months often > 1 disc diameter
Wet Form of Age-Related Macular Degeneration (AMD)
Clinical signs of exudative age-related macular degeneration include subretinal bleeding, hard exudates, fibrovascular or serous pigment epithelium detachment, and fibrovascular scars. Choroidal neovascularization is occasionally detectable by ophthalmoscopy as a greenish-gray prominence with surrounding edema and hemorrhages or as hyperpigmentation. A positive diagnosis can be made by fluorescein or indocyanine green angiography (Dobberstein and coworkers 1998). Choroidal neovascularization may be angiographically subdivided into so-called classic and occult forms.
Ophthalmoscopy of CNV
- greenish-gray protrusion (edema) in the macula, circumscribed blurriness of structures
- small retinal hemorrhages at its margin
- later hard exudates
- end stage: fibrovascular scars
- pseudotumor of the macula occasionally present
Classic CNV Fluorescein Angiography
- early phase: circumscribed hyperfluorescence, vascular network often visible
- late phase: sharply demarcated increasing leakage
difference between Classic and Occult CNV by Fluorescein Angiography
Classic CNV
- early phase: well-demarcated areas of hyperfluorescence
- late phase: homogenous leakage Occult CNV
- early phase: irregular fluorescence
- late phase: in-homogenous leakage
Classic Choroidal Neovascularization
In the classic form, the early phase of fluorescein angiography reveals a focal area of hyperfluorescence and occasionally a fine vascular network. In the late phase of angiography, increasing homogeneous leakage is observed in the vicinity of the choroidal neovascularization. This form of choroidal neovascularization is further subdivided according to its position relative to the center of the fovea. It is specified as extrafoveal (more than 200 μm from the center), juxtafoveal (1–199 μm from the center), or subfoveal (beneath the center). Extrafoveal or juxtafoveal classic choroidal neovascularization is accessible to thermal (argon) laser photocoagulation, whereas photodynamic therapy may be considered for subfoveal choroidal neovascularization.
Localization of classic choroidal neovascularization relative to the fovea (distance from the center of the macula):
- extrafoveal: more than 200 μm
- juxtafoveal: 1–199 μm
- subfoveal: directly beneath the macula.
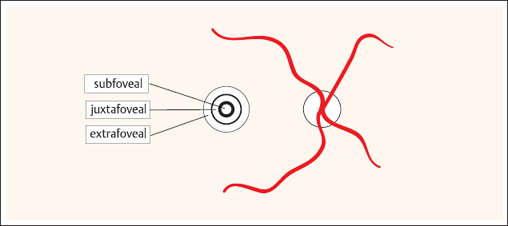
Fig. 3.36 Sketch of localization of macular lesions: subfoveal, juxtafoveal, and extrafoveal.
Fibrovascular RPE Detachment
- irregular thickening of RPE
- fluorescein angiography: early phase: focal hyperfluorescence; late phase: pin-point hyperfluorescence
- indocyanine green angiography: early phase: often CNV visible (see Fig. 3.38)
Serous RPE Detachment
- sharply demarcated, dome-shaped detachment of the retinal pigment epithelium
- visual acuity often still good
- fluorescein angiography: slow, diffuse, and late filling of space beneath detached RPE-flap (more than 10 minutes), persistent hyperfluorescence (see Fig. 3.39 on next page)
- indocyanine green angiography: occasionally, CNV is seen, hypofluorescence during entire angiographic examination
Occult Choroidal Neovascularization and Pigment Epithelial Detachments
Occult choroidal neovascularization cannot be distinguished in the early phase of fluorescein angiography.
This form of age-related macular degeneration may be further subdivided as follows:
- fibrovascular pigment epithelial detachments
- serous pigment epithelial detachments
- late leakage of undetermined source.
Fibrovascular Pigment Epithelial Detachments
Fibrovascular pigment epithelial detachments are characterized by irregular thickening of the retinal pigment epithelium. The early phase of angiography demonstrates focal areas of hyperfluorescence that exhibit irregular leakage in the late phase. In this form of occult choroidal neovascularization, a vascular network is often distinguishable in the early phase of indocyanine angiography.
Serous Pigment Epithelial Detachments
Serous pigment epithelial detachments are sharply demarcated, dome-shaped brownish elevations, of the retinal pigment epithelium. There is no visible choroidal structure on ophthalmoscopy; in the early phase of fluorescein angiography the area of RPE detachment is hypofluorescent. Only in the late phase, a gradually increasing uniform hyperfluorescence appears, which persists for more than 15 minutes.
Late Leakage of Undetermined Source
- often only a discrete edema on ophthalmoscopy
- soft drusen
- fluorescein angiography: early phase: no abnormal hyperfluorescence
- late phase: increasing leakage
- Indocyanine green angiography: choroidal neovascularization (CNV) often seen
Late Leakage of Undetermined Source (LLUS)
In occult choroidal neovascularization characterized by late leakage of undetermined source, there is no circumscribed hyperfluorescence in the early phase of fluorescein angiography. Increasing leakage and hyperfluorescence are only observed as the angiography progresses. This hyperfluorescence steadily increases and is not sharply demarcated. Ophthalmoscopic examination of this form usually reveals only a discrete edema or small subretinal hemorrhage with confluent soft drusen.
Treatment: there is no known effective medical treatment. Local laser photocoagulation may be considered in a few cases of extrafoveal choroidal neovascularization. Photodynamic therapy has been found to be effective with subfoveal choroidal neovascularization. Photodynamic therapy competes with surgical treatment options. New antangiogenic drugs are being tested.
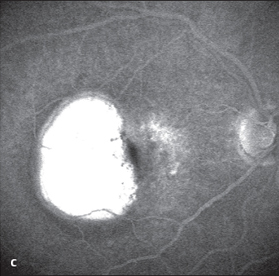
Fig. 3.39c Late phase (after 5-10 minutes): the serous fluid absorbed fluorescein and remains fluorescent for a long time.
Disciform Lesion (Junius–Kuhnt Degeneration)
- fibrovascular scar at the posterior pole of the eye
- greatly protruding glial proliferation (pseudotumor)
- large absolute central scotoma
- greatly reduced visual acuity
End Stage of the Wet Form of Age-related Macular Degeneration (AMD)
Left untreated, exudative age-related macular degeneration develops into a fibrovascular scar that can cover the entire posterior pole of the eye (pseudotumor). This end stage of the disorder is referred to as a disciform lesion or Junius–Kuhnt degeneration. This stage is usually associated with a large absolute central scotoma and greatly reduced visual acuity.
Treatment: there is no effective treatment for the end stage.
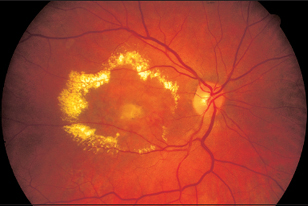
Fig. 3.40 The advanced stage of age-related macular degeneration (disciform lesion or Junius–Kuhnt degeneration). Glial scar surrounded by hard exudates.
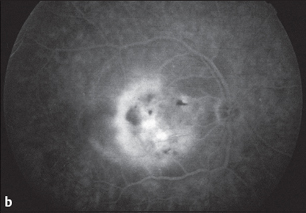
Fig. 3.41b Fluorescein angiography (late phase, after 10 minutes): exudation over a broad area.
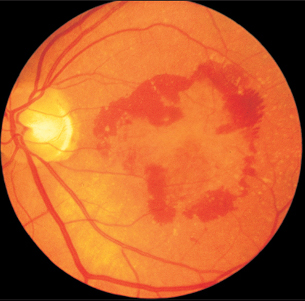
Fig. 3.42 The advanced stage of untreated age-related macular degeneration. Retinal hemorrhages are surrrounding the macular edema.
 Choroidal Neovascularization of Other Causes
Choroidal Neovascularization of Other Causes
- primary disorders
- presumed ocular histoplasmosis syndrome (POHS)
- secondary disorders
- severe myopia
- angioid streaks
- punctate inner choroidopathy
- multifocal inner choroiditis
- serpiginous choroiditis
- adult vitelliform dystrophy
- severe myopia
 Literature
Literature
Hogg R.U., Chakravarthy, AMD and micro-nutrient antioxidants. Curr Eye Res 2004. 29(6): p. 387-401.
 Proliferation, Scarring, and Holes in the Macula
Proliferation, Scarring, and Holes in the Macula
 Epiretinal Membranes (Macular Pucker)
Epiretinal Membranes (Macular Pucker)
Epimacular membrane formation after posterior vitreous separation is termed macular pucker. This term is used to describe wrinkles of the surface of the retina as a result of contracting epiretinal membranes. The membranes consist of macrophages, pigment epithelial cells, glia, fibroblasts, and myofibroblasts. The pathogenesis of macular pucker is not understood. Epimacular formation of contractile membranes without posterior vitreous separation is called “vitreo-macular traction syndrome”. More subtle expressions of epimacular traction are clinically termed: “cellophane or surface wrinkling maculopathy”.
Causes of macular pucker:
- Retinal tear without or without retinal detachment
- Severe ocular trauma
- Retinal vein occlusion
- Minor hemorrhages
- Other vascular processes
- Idiopathic
The fine gray strands indicative of thickening of the preretinal structures are not always immediately visible. A typical finding is distortion of the perimacular blood vessels, which may occasionally include the major temporal vessels. Contraction of the scar-like tissue causes the retinal vessels to follow abnormal, often bizarre courses. This also places traction on the macula, which in turn appears torn and often displaced in the direction of the bizarre retinal vessels. The retinal structures often appear ill-defined due to edema.
Macular pucker cannot cause retinal detachment. It often occurs secondary to other retinal disorders and vitreoretinal surgery. It can be readily managed by vitrectomy.
Treatment: Cyclooxygenase inhibitors and corticosteroids have only limited effect. The method of choice is surgical removal of the connective tissue membrane from the surface of the retina.
Macular Pucker
Symptoms
- distorted vision, metamorphopsia
- visual acuity reduced
- patients often unable to read
Signs
- contorted vascular structures next to elongated vessels
- gray strands in front of the retina
- distortion of the macula
- angiography: edema, contorted blood vessels good visible
 Idiopathic Macular Hole
Idiopathic Macular Hole
Idiopathic macular holes are presumably caused by tractional forces in the vitreo–retinal interface at the posterior pole. The causative mechanism is thought to initially involve posterior–anterior tractional forces and later tangential tractional forces within the cortex of the vitreous. If such forces produce a macular hole, the foveola initially becomes detached (stage I according to Gass) and later the foveal tissue will radially retract (stage II and higher). Despite a clinically visible hole, there is no loss of retinal tissue. These holes practically never give rise to a retinal detachment. The edges of the hole are usually slightly raised. Yellow pigments will often be visible on the floor of the macular hole.
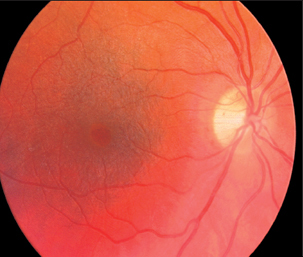
Fig. 3.43 Vitreo-macular traction syndrome, so-called cellophane maculopathy. Visual acuity is 20/40.
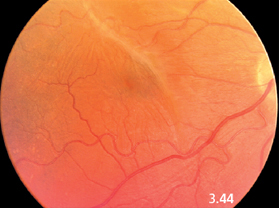
Fig. 3.44 A macular pucker in a 64-year-old woman.
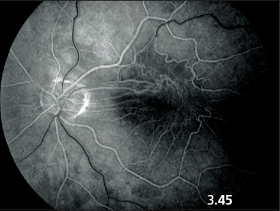
Fig. 3.45 Fluorescein angiography in macular pucker. The early venous phase demonstrates bizarre vascular structures. Exudation appears only in the late phase after 5–10 minutes.
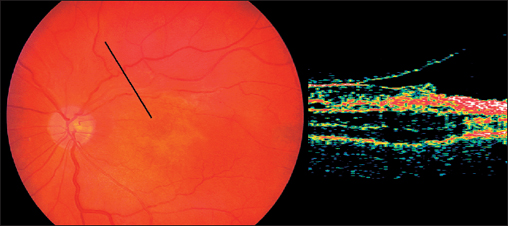
Fig. 3.46 Optical coherence tomography (OCT) with macular pucker.
Gass classification of macular holes:
- Stage 1A: imminent hole with detachment of the foveola.Ophthalmoscopic findings include a small yellow spot.
- Stage 1B: imminent hole with detachment of the foveola.Ophthalmoscopic findings include a small yellow ring.
- Stage 2: foveal hole, (less than 200 μm in diameter). The defect is often found on the border of the fovea and may be round, arched, or horseshoe-shaped.
- Stage 3:

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree