CHAPTER 199 Vascular Anomalies of the Head and Neck
Vascular anomalies encompass vascular tumors and malformations and involve the head and neck 70% to 80% of the time. Clinical and radiographic findings, and occasionally histologic evidence, are used to distinguish among various vascular anomaly subtypes. A vascular anomaly classification system has been developed based on these factors1,2 (Table 199-1). Hemangiomas of infancy (HOIs) were the first vascular lesions to be distinguished from other vascular anomalies, based on their natural history.3 HOIs, unlike other vascular anomalies, are absent at birth, appear during infancy, grow rapidly within the first year of life, and then undergo a variable period of involution. Numerous recent discoveries have further solidified the distinction between vascular tumors and vascular malformations.4–6
Table 199-1 Vascular Anomaly Classification*
| Vascular Malformation | Vascular Tumor |
|---|---|
* Binary classification system adopted by the International Society for the Study of Vascular Anomalies (ISSVA) in 1996.
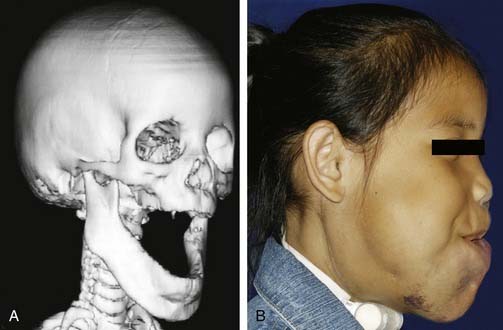
Vascular tumors usually are benign, have neoplastic characteristics, and represent disordered angiogenesis.4 Radiographically these tumors can be characterized by describing lesion size, blood flow, and location.7,8 Certain clinical conditions, such as Kasabach-Merritt phenomenon and PHACES syndrome (posterior fossa abnormalities, hemangiomas, arterial anomalies, cardiac anomalies, eye abnormalities, sternal or abdominal clefting), are associated with vascular tumors and not vascular malformations.9–11 Vascular malformations often contain all vessel types—arterial, venous, and lymphatic—and seem to represent disordered vasculogenesis. Most vascular malformations are present at birth and demonstrate growth parallel to the child’s growth. These lesions have a distinct radiographic appearance in their location, size, and blood flow—in particular, that through the lesion. Additionally, certain types of vascular malformations are characterized on the basis of the radiologic architecture of the lesion. This characterization has been most useful in identifying lymphatic malformations. These malformations also are associated with either local tissue or bony overgrowth (Fig. 199-1). Since the original distinction of hemangiomas from other types of vascular tumors and malformations, molecular characteristics as well as radiographic features of individual vascular anomaly subtypes have allowed further distinction between various types of vascular anomalies. This chapter outlines these distinctions and explains how these distinctions have led to treatment improvements, potentially leading to better clinical outcomes.
A genetic and molecular basis for some vascular anomalies is being investigated.12–14 The most apparent link between genetic defect and disease is seen in inherited venous malformations, in which a mutation in the protein receptor for tyrosine kinase (Tie2), which normally allows endothelial–smooth muscle communication essential for venous morphogenesis.15 Defects in the smooth muscle layer of vessels with venous malformations result from this mutation. Similarly, vascular malformations in hereditary hemorrhagic telangiectasia types 1 to 3 are associated with gene mutations that adversely affect endothelial function.16 Familial HOIs are linked to defects in the fifth chromosome.17 Sporadic HOI endothelium is clonal.18,19 Glomangiomas have defective “glomulin” function, distinguishing them from other venous malformations.20 Ateriovenous malformations occurring with atypical capillary malformations have been linked to a gene RASA1.21 Hypertrophy of tissues near vascular malformations has been linked in certain syndromes to a mutation in the PTEN tumor suppressor gene.22,23 This information creates a basis for understanding the mechanisms behind the pathogenesis of vascular anomalies.
Vascular Tumors
Hemangiomas of Infancy
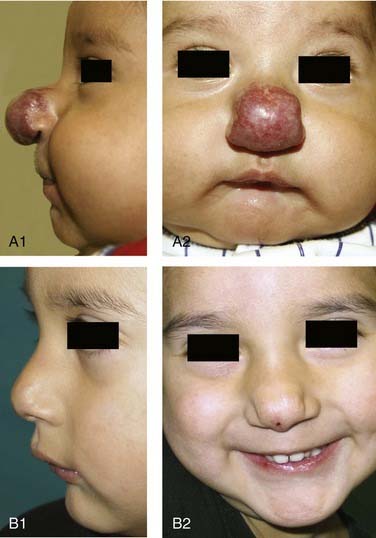
HOIs are absent at birth and appear in infancy; they proliferate for 6 to 9 months and then involute partially or completely over several years24–26 (Fig. 199-2). Vascular lesions not following this pattern should be considered for alternative diagnoses.6 In addition to clinical diagnosis, HOI diagnosis can be further confirmed through molecular staining and imaging.27,28 HOI endothelium has several unique surface markers detected by immunohistochemistry. The first marker for HOI described was glucose transporter-1 (GLUT-1); others have since been described. GLUT-1 detection separates HOIs from other vascular anomalies and congenital hemangioma.28 Improvements in image acquisition with both computed tomography (CT) scanning and magnetic resonance imaging (MRI) allow differentiation between HOIs and other vascular anomalies8,29,30 (Fig. 199-3).
Clinical Presentation
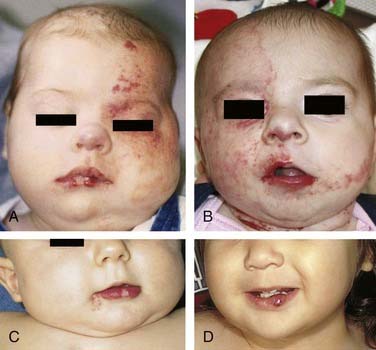
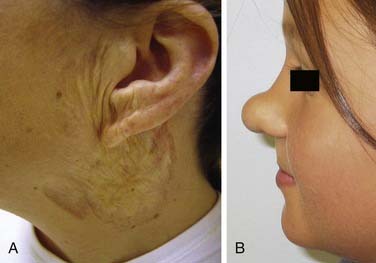
At birth, HOI can be completely absent or may be present as a slight redness or pallor in the area in which a HOI subsequently appears.24 In the first several months of life, HOIs grow rapidly and can involve any area of the head and neck. Often multiple hemangiomas are present in areas outside the head and neck. Two thirds of HOIs occur in girls. HOIs are more prevalent in premature infants and infants born to mothers of advanced maternal age, with a history of chorionic villus sampling. Terminology to describe types of HOIs and their appearances and locations31 are illustrated in Figure 199-4. Segmental HOIs usually are superficial, variably involve cutaneous dermatomes, and have more associated morbidity than focal HOIs. These lesions are thought to occur in embryologic prominences that are related to neural crest cells, which influence vascular patterning.32 In the head and neck, focal HOIs occur in identifiable patterns that are thought to be associated with lines of embryonic tissue fusion.31,32 Building on the concept of embryonic fusion planes, several groups of investigators have observed that midface HOIs do not predictably involute.
PHACES Syndrome
An association between some HOIs and ventral clefting, congenital cardiac malformations, intracranial malformations, and ocular abnormalities has been described; the specific manifestations collectively make up the PHACES syndrome11 (Fig. 199-5). Central nervous system symptoms include developmental delays, seizures, and congenital stroke. Patients suspected to have PHACES syndrome should undergo head imaging, ophthalmologic examination, and cardiac evaluation. Some clinicians recommend antiplatelet therapy for patients with abnormal cerebral vasculature, to prevent congenital stroke.33,34 Further analysis of HOI biology is necessary to elucidate these associations.
Other Types of Vascular Tumors
Congenital Hemangioma
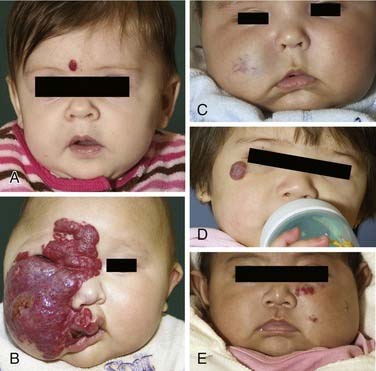
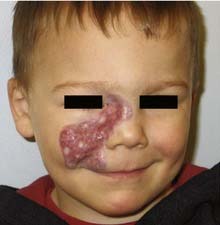
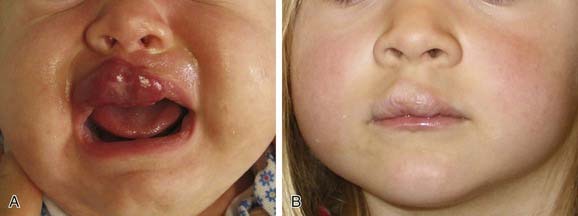
High-flow vascular tumors manifesting at birth that are histologically identical to HOIs except for positive GLUT-1 staining are called congenital hemangiomas.73,74 There are two types of congenital hemangiomas—those that rapidly resolve over the span of a year and those that persist. Accordingly, they are named rapidly involuting congenital hemangioma (RICH) and non-involuting congenital hemangioma (NICH), respectively. These lesions often are difficult to distinguish from HOIs unless a careful history is taken and histologic analysis is performed. Often the diagnosis remains uncertain until the clinical behavior of a given lesion is observed (Fig. 199-6). Medical therapy is ineffective for congenital hemangiomas. Treatment for RICH usually is limited to observation, whereas NICH may require laser or surgical therapy.
Kaposiform Hemangioendothelioma and Tufted Angioma
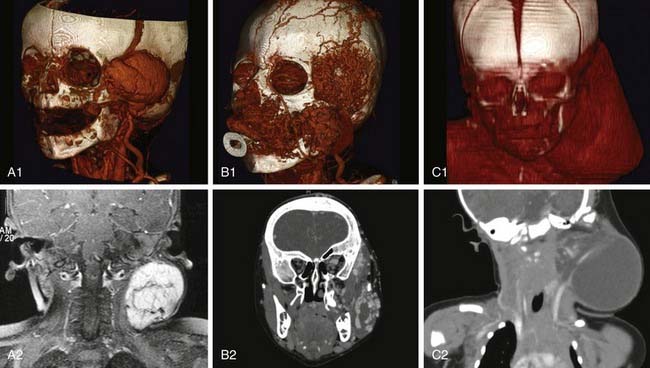
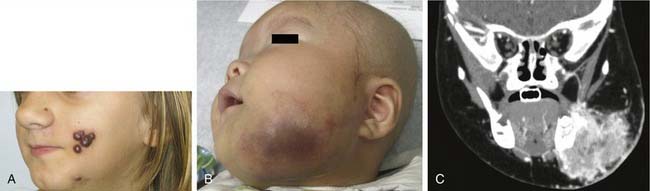
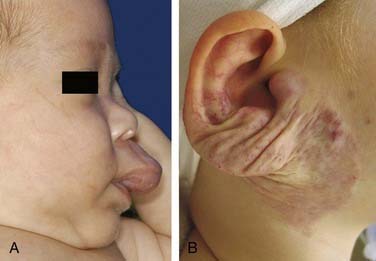
Most nonhemangioma vascular tumors are benign. These tumors occur rarely in the head and neck. The two most common tumors are kaposiform hemangioendothelioma (KHE) and tufted angioma (TA). KHEs occur anywhere in the head and neck and have a significant lymphatic component in addition to blood vascular endothelium.75 Clinically, KHEs appear as violaceous cutaneous nodules extending into deep tissues locally (Fig. 199-7). Their radiographic appearance is one of diffuse infiltrative vascular process (see Fig. 199-7). Careful assessment through imaging and diagnostic biopsy should guide KHE therapy. TAs are more localized and may or may not involve the skin. Treatment for both KHE and TA is centered on proper diagnosis, prevention of complications, and tumor control. Diagnosis requires an incisional biopsy with possible immunohistochemical staining. Curative surgical resection is possible and advisable for some localized tumors, but for large lesions, surgery may not be curative. With extensive KHEs or TAs that are causing pain, loss of function, or significant disfigurement, antiangiogenic chemotherapy may be necessary. The natural history of these lesions in the head and neck often is unclear; therefore, treatment needs to be individualized.
Common Complications of Vascular Tumors
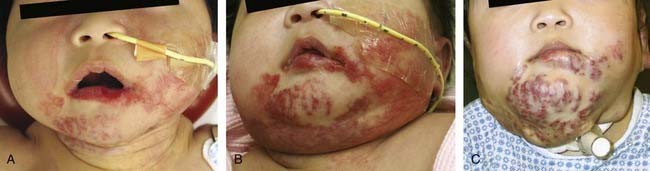
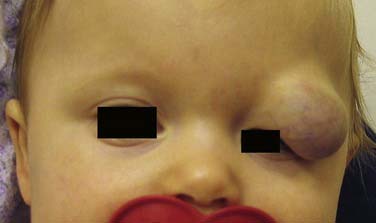
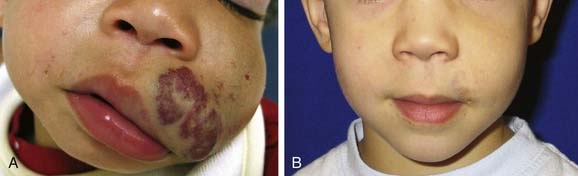
Because most vascular tumors are HOIs, which proliferate rapidly, complications occur most frequently with these lesions. HOI complications occur primarily from lesion-induced functional compromise and cutaneous ulceration. Breathing can become compromised and obstructed from airway HOI. HOI can cause biphasic stridor by narrowing the infant’s subglottic airway35 (Fig. 199-8). Obstruction of the visual axis by a proliferating HOI occurs with eyelid lesions, inducing deprivation amblyopia36 (Fig. 199-9). Deformation of the eye from lesion compression or periocular tissue distortion before the age of 6 months induces astigmatic amblyopia.37 Proliferating HOI located in places of repetitive skin trauma such as the lips and neck skinfolds can ulcerate, resulting in scarring. The reasons for ulceration are unclear, but when present, ulcerations begin as brown crusts, become painful, bleed, and require local wound care for healing. Extensive ulcerations of the hemangioma can become infected and result in tissue loss, further complicating healing and treatment38 (Fig. 199-10). Ulceration involving the lip often makes oral feeding challenging because of pain and hemangioma-induced lip distortion. Large, deep head and neck HOIs involving the parapharyngeal and scalp regions often are associated with high-output cardiac failure, due to the increased blood flow in these vascular lesions.39 The psychosocial consequences of HOI for the family and patient are important to consider in treatment planning.40
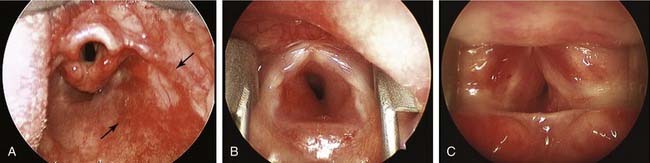
Figure 199-8. Endoscopic appearance of airway hemangioma of infancy. A, Diffuse hypopharyngeal mucosal involvement (arrows). B, Unilateral posterior focal hemangioma; left subglottis (see also Fig. 199-16A and B). C, Circumferential, segmental hemangioma; subglottis (see also Fig. 199-16C and D).
Unusual Conditions Associated with Vascular Tumors
Kasabach-Merritt Phenomenon
Kasabach-Merritt phenomenon is the occurrence of profound thrombocytopenia in association with vascular tumors. For unknown reasons, KHE and TA, not HOI, are the vascular tumors most frequently associated with Kasabach-Merritt phenomenon.9,10,76 This occurs in infancy and requires extensive chemotherapy to reduce the risk of bleeding associated with thrombocytopenia. The mechanisms leading to thrombocytopenia are unknown. Before the capability of histologic distinction between the endothelium of KHE, TA, and HOI, many vascular tumors were called simply “hemangiomas,” and Kasabach-Merritt phenomenon was associated erroneously with HOI. Newly described vascular entities, not classified as vascular tumors, also are associated with thrombocytopenia. The spectrum of this phenomenon as it relates to vascular anomalies is still being defined.77
Treatment Overview
Analysis and Treatment of Facial Hemangiomas
A complete discussion of all aspects of facial hemangioma treatment is beyond the scope of this chapter.72 Some general concepts and principles useful in management decisions follow:
Medical Therapy
Medical therapy for proliferating HOI is directed at stopping angiogenesis and decreasing peritumoral inflammation; in contrast, medical therapy for nonproliferating HOI is ineffective.4,41,42 Although controversial, the mainstay of medical therapy has been systemic corticosteroid administration, which is antiangiogenic and anti-inflammatory. The major indication for corticosteroid use depends on the presence of HOI complications or the attempt to prevent such complications, which occur more frequently with extensive segmental lesions.26 For extensive proliferating HOI, prednisolone is given orally with doses ranging from 1 to 5 mg/kg/day in treatment courses that begin with an empirical burst at a high dose and then a slow 4- to 6-week taper off the medication. In theory, this minimizes the systemic side effects of steroid usage, but no long-term efficacy studies have been done.43 Effectiveness of therapy is monitored throughout therapy; live vaccine administration is avoided, and blood pressure is monitored. Focal HOI of the head and neck can be injected with corticosteroids to localize drug delivery.44,45 This practice is controversial and specialty specific, owing to the risk of blindness, training differences, and uncertain efficacy. Despite this, it is frequently done under general anesthesia for eyelid HOI while directly visualizing the retina.36,46 In this location steroids are very effective in reducing HOI size, thereby decreasing the risk of HOI-induced visual impairment, but intralesional steroid injections can induce measurable adrenal suppression.46 The role of topical medications to treat hemangiomas is being defined.47 Other forms of antiangiogenic chemotherapy, such as interferon, have been used to treat HOIs.48,49 The initial optimism for interferon as an effective agent for treatment of extensive HOI was subdued by the realization that before the age of 1 year, the central nervous system appears to be sensitive to this medication; neurologic complications such as spastic dysplegia were frequent.50 For this reason, interferon is not used without serious consideration. However, it is still used in selected patients during times of rapid HOI proliferation. The proper dose of interferon is controversial, because previously reported complications were attributed to inappropriately high interferon dosing. More recently, vincristine has been investigated as a medication that can be used safely for treatment of extensive HOI.51,52 Trials are under way to determine its efficacy in comparison with corticosteroids. Most recently, propranolol has been found to have an effect on reduction of HOI size. Further study is necessary to refine its use in the treatment of HOI.78