13 Tumors of the Middle Ear and Temporal Bone Benign tumors of the middle ear and temporal bone are rare. However, with conditions including glomus tumors, acoustic neuroma (acoustic schwannoma), and meningioma—and with tumor-like lesions such as petrous bone cholesteatoma and cholesterol granuloma, in particular—any otology service can expect to deal with quite a few cases every year, requiring expensive diagnostic procedures and surgical treatment. Table 13.1 shows the most frequent benign types. Benign forms of neoplasia, such as adenoma and fibroma, usually originate from the tympanic cavity, but facial nerve schwannomas and glomus tympanicum chemodectomas are also found here. Depending on their location, they produce different symptoms. Schwannomas and glomus tumors are discussed in separate sections below. Symptoms. Exophytic benign tumor growth within the tympanic cavity soon creates a feeling of ear closure or fullness, and leads to conductive hearing loss. When the lesion originates at the tubal orifice, it causes secretory otitis media. Tinnitus and autophony may also be presenting symptoms, but these may not appear for a long time. Pain or giddiness are rare. However, osteomas in the mastoid or in the petrous apex can soon lead to dull internal aural pain. Diagnosis and differential diagnosis. Otoscopy can help identify a tumor mass within or behind the drum membrane. If this is intensely red and pulsating, it can justifiably be assumed to be a glomus tumor. A hemangioma may have a similar appearance. A displaced internal carotid artery, persistent stapedial artery, or high jugular bulb also need to be considered. Pure tone audiometry and impedance measurements will detect impaired sound conduction. Plain radiography—e.g., with the Schüller and Stenvers projections—is only able to image larger tumors, but high-resolution computed tomography (CT) can delineate small lesions very precisely. Magnetic resonance imaging (MRI) or selective angiography are important tools for identification and differential diagnosis of vascular tumors, malformations, or tumor-like lesions such as aneurysms, granulomas, cholesteatomas, etc. Table 13.2 outlines the rules for radiodiagnostic interpretation. While benign soft-tissue lesions are usually marked by sharp borders, the margins of osteomas may be blurred, leading to recurrent growth after incomplete surgical removal. Check-up imaging is therefore indicated one year after surgery. Indications for surgery. Benign space-occupying lesions in the middle ear generally require complete surgical removal in order to prevent further destruction and functional deficits. For facial nerve schwannomas, however, individualized treatment is recommended. Curative radiotherapy is reserved for malignancies, if it is indicated at all. Palliative radiotherapy for benign tumors is discussed in the sections on glomus tumors and acoustic neuromas below.
 Benign Neoplasias and Tumor-like Lesions
Benign Neoplasias and Tumor-like Lesions
General Remarks
| Neoplasias | Tumor-like lesions |
| Adenoma | Exostoses |
| Angioma | Cholesteatoma |
| Schwannoma nerves VII, VIII, IX, X, chorda tympani) | Cholesterol granuloma Histiocytosis |
| Fibroma, ossifying fibroma | Giant cell granuloma |
| Osteoma | Fibrous dysplasia of bone (Jaffé-Lichtenstein disease, Uehlinger disease) |
| Chemodectoma | Paget’s disease |
| Meningioma | Osteopetrosis |
| Transmeatal/transtympanic | Middle cranial fossa Internal auditory canal (transotic) Petrous apex (limited) |
| Transmastoid | Middle cranial fossa Posterior cranial fossa Internal auditory canal, Cerebellopontine angle (translabyrinthine) Sigmoid sinus, jugular bulb |
| Transtemporal/infratemporal | Middle cranial fossa Petrous apex, clivus Internal carotid artery Internal auditory canal, Cerebellopontine angle |
| Suboccipital | Petrous bone/petrous apex Internal auditory canal, Cerebellopontine angle |
| Transcervical | Jugular foramen Internal carotid artery Petrous apex |
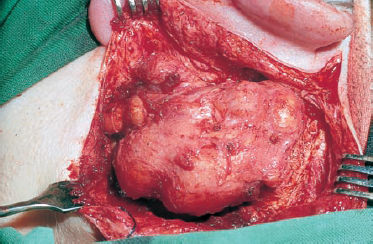
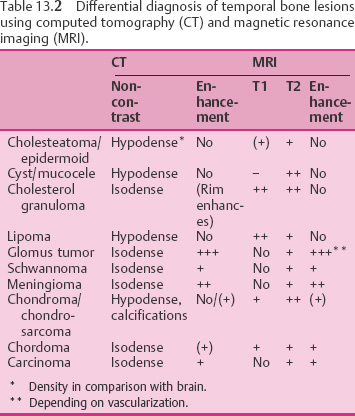
Fig. 13.1 Tumor-like invasion of the left temporal bone by a giant cholesteatoma; postauricular exposure.
Surgical techniques. All of the surgical principles involved in functional middle ear surgery, as described in Chapter 10, are applicable in the removal of benign middle ear tumors. Depending on the size and location of the lesion, an endaural or postauricular approach is adequate. These can both be adapted to the individual situation in order to ensure complete exposure of the mass and allow immediate reconstruction of the middle ear walls and of the sound-conducting apparatus. Destructive operations are not appropriate in most cases (except for invasive hemangiomas and glomus jugulare tumors, which are discussed below).
However, surgery for advanced neoplasia of the temporal bone, whether it is histopathologically benign or malignant, belongs to the demanding field of skull base surgery. Lesions of this type require an intimate knowledge of both the topographical anatomy and of the specific ranges of the various surgical approaches. Table 13.3 provides an overview of the relevant access routes to the various targets.
Petrous Bone Cholesteatoma
As soon as a cholesteatoma has progressed beyond the natural confines of the middle ear, it becomes a space-occupying process and is defined as a tumor-like lesion. Smaller expansions with osteoclastic destruction are common—e.g., with erosion of the labyrinth or fallopian canal. The discussion of surgery for tumors of the temporal bone given here does not deal with these, but focuses instead on larger destructive processes reaching beyond the geniculate ganglion into the petrous apex, or invading through or between the labyrinthine structures into the areas of the internal auditory canal—coming into broad direct contact with the temporal or cerebellar dura and potentially protruding into the cerebellopontine angle or into the occiput (Fig. 13.1). This group of lesions is termed “petrous bone cholesteatomas.” The fact that they often destroy the compartments of the inner ear or compromise the facial nerve is not part of their definition, but it is an imminent danger.
Clinical Entity
Petrous bone cholesteatomas develop from common middle ear cholesteatomas. Histopathological evaluations have so far failed to identify petrous bone cholesteatoma as a distinct pathological entity; the texture of the matrix and of the underlying connective tissue is identical with that of middle ear cholesteatomas. It has often been assumed that these lesions originate from congenitally dystopic squamous epithelium behind an intact drum membrane (Peron and Schuknecht 1975, Nager 1982, Michaels 1988), but this is still speculative. Cases of cholesteatoma without perforation or retraction pockets have been described. I have seen two such cases, in both of which it was possible to follow an epithelial cord to the roof of the external auditory meatus. However, the pathogenesis of the lesions is not relevant in the surgical management of petrous bone cholesteatoma. The classic theories that the basal layer cells of the epithelium are particularly invasive (Ruedi 1963), that subepithelial inflammation has a stimulating effect (Nager 1925, Lange 1925), and that the lesion is metaplastic in origin (Sadé et al. 1983) will not therefore be discussed here. Our own strategies, in agreement with Tumarkin (1961), are based on the concept that the expansion of the matrix is a simple consequence of biomechanical factors. As soon as the epidermal layer of Shrapnell’s membrane or of the drumhead is retracted by a subepidermal inflammatory mucosal adhesion, or is pressed into the attic or tympanic cavity by a pressure difference, a “retraction pocket” is created. Once this process of invagination has progressed to a certain point, the permanent physiologic production of squamous cells and of keratin cannot longer be depleted by the natural outflow (migration) into the external auditory canal. A congestive squamous mass then develops, exercising continuous pressure on the underlying bone. The self-cleaning mechanism, which functions well on flat surfaces, is easily obstructed by the formation of mechanical barriers such as bony crests, or by fibrous adhesions. This type of occurrence is often observed during ear microscopy after initial retractions, and over a period of months the consecutive phases of secondary cholesteatoma formation can be seen. On the other hand, if this type of concave epidermal surface is flattened by microsurgery, the pseudotumorous properties will disappear, and a cholesteatoma matrix can in this way turn into the healthy, harmless surface of a flat radical cavity.
The growth pattern of petrous bone cholesteatoma is also in accordance with these assumptions. Invaginated matrix is always found in continuity, and single, solid tumor extensions (which are common in basalcell carcinomas, for example), are not seen. This particular feature is challenging for the surgeon, who needs to be able to follow surfaces consistently in order to achieve complete dissection. This can become very difficult in narrow bony tunnels, or with an enveloped facial nerve (see below). In principle, the expansion of a petrous bone cholesteatoma shows a preference for the weaker parts of the temporal bone. It penetrates the better pneumatized areas first, and follows the cell tracts. A frequent path is the superior tract from the anterior attic into the apex, with early resorption of the thin floor of the middle cerebral fossa, bringing the matrix into close contact with the dura. Another site of predilection is the subarcuate tract, leading from the mastoid to the internal auditory canal. A third, usually broader, route of invasion runs along Citelli’s sinus—dura angle to the posterior cerebral fossa. Less frequently, the hypotympanum forms a gateway for sublabyrinthine expansion, compromising the jugular bulb and the lower internal carotid artery. The hard bone of the labyrinth and cochlea resists for a longer period, but it is ultimately eroded or even invaded from above or below. In both instances, it is rare for the fallopian canal not to be involved.
Symptoms, Signs, Diagnosis
In patients with petrous bone cholesteatoma, the history and presenting symptoms do not differ very much from those in patients with chronic otitis media. Discharge from the ear, conductive or combined hearing losses (including total deafness), and giddiness or vertigo are prevalent. Unilateral headache and facial paralysis are common, and pareses of the abducent nerve have also been observed. Gacek (1980) and Glasscock et al. (1989) have described representative cases. Late development of such sequelae may occur even after earlier radical mastoidectomies.
Ear microscopy, or endoscopy for examining hidden recesses in cavities and epitympanic defects, reveals epidermized bone defects and niches with keratin debris and fetid secretion, often framed by soft granulations. A small defect in the lateral attic wall above an intact pars tensa is often virtually closed by a keratinous pearl, which may be covered by a thin, filmy layer of mucosal epithelium (Fig. 13.2). Applying gentle suction can reopen the cholesteatoma sac. There may be a positive-pressure fistula symptom. If there is the slightest suspicion that the lesion extends endocranially, this test and the Politzer maneuver must be carried out only at low pressure (less than 0.3 bar). The necessary vestibular examination must also be performed very carefully: thermal stimulation should be carried out without irrigation, and preferably with air insufflation. Electromyography is indicated to detect and classify any impairment of the facial nerve or other nerves. Schirmer’s test and a check on gustatory sensation complete the neuro-otological investigation. An ophthalmologist and a neurologist should be consulted if there are related ocular or central nervous symptoms.
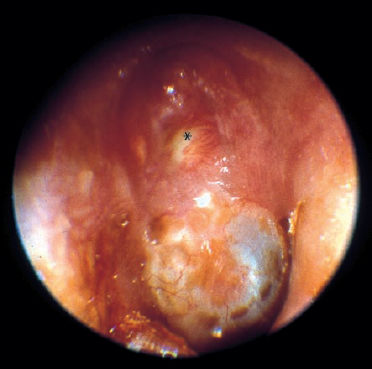
Fig. 13.2 A large petrous bone cholesteatoma. Otoscopy had only shown an intact pars tensa, with a virtually closed defect (*) in the lateral attic wall (right ear).
High-resolution CT is the best tool for precise imaging of petrous bone cholesteatoma. A CT can delineate the extent of bone destruction and reveal any labyrinthine involvement (Fig. 13.3). MRI studies may help identify the nature of an apical lesion. Cholesteatomas give positive signals on T1 and T2 sequences, but they are not enhanced by gadolinium. Fat-saturating sequences produce negative shadows. MRI is particularly helpful for detecting recurrences in previously resected temporal bones, as CT scanning is not capable of distinguishing between residual cholesteatoma and scar formation in a bone defect.
|
Differential Diagnosis
The differential diagnosis includes all types of lesion in the petrous apex, such as granulomas, cysts, neuromas, chondromas, etc. There are often positive MRI signals with apical secretory mucositis (not destructive apicitis), but this usually produces an image of liquid-filled cells. Cholesterol granulomas can also have a similar appearance on a CT scan. They are less prominent in the T1 phase, and are also negative with gadolinium enhancement. True neoplasias such as neuromas or chondromas, however, show marked enhancement. Metastases be have similarly in radiological investigations, but rather produce a round configuration with blurred margins. In doubtful cases in which a clear pre-treatment decision is needed, transtympanic or transtemporal exploration may be necessary to allow a biopsy to be taken. We have done this, for instance, in children in whom there was a suspicion of rhabdomyosarcoma of the temporal bone.
Indications for Surgery
Petrous bone cholesteatomas are indications for surgery on principle, with the aim being to achieve complete removal of the invaded matrix. Since any delay will increase the danger of further destruction, with endocranial invasion or penetration into the adjacent sphenoid compartments and a loss of inner ear function in particular, surgery is urgently required. Only high-risk patients should undergo palliative decompression procedures, opening up the routes of epidermoid proliferation into a radical cavity. In infants as well, one may be inclined to avoid any unnecessary destruction resulting from the transtemporal or radical transmastoid approaches, and a staged intervention without reconstructive closure during the first session may be preferable. In normal conditions, however, complete exenteration of the cholesteatoma is combined with restoration of an approximately natural configuration of the middle ear. Purely destructive removal of walls to allow permanent drainage of a persistent cholesteatoma cavity appears inappropriate. This view is not yet commonplace in the contemporary literature, in which radical operations and transotic or translabyrinthine approaches are reported even in ears with preoperative hearing ability (Atlas et al. 1992). Such large ablative interventions are, of course, sometimes indispensable for the management of giant lesions, or to solve specific problems when it is not possible to proceed with a less invasive access route that has been planned.
Dissection problems of this type that may affect the surgical technique are listed in Table 13.4. Residual matrix in remote, difficult to reach areas must be removed at all costs, possibly with the sacrifice of healthy bone that would otherwise be used for restoration. Although they consist of benign tissue, basal cells must be regarded as clinically deleterious. An extension of the initial approach or a second bidirectional access route may therefore become necessary. Dilemmas of this type are presented, for example, when strong matrix is found clinging to soft dura mater. It is unlikely that it will be possible to remove this along a cleavage plane, and it may be prudent to lacerate any part of the dura that is doubtful and to graft the defect. This problem is most difficult to deal with when there is matrix attached to thin meatal dura over or around the internal auditory canal. As soon as cerebrospinal fluid inundates the surgical field, further striping of the matrix becomes hazardous. It may then become necessary to continue with a translabyrinthine route instead of persisting with a middle fossa approach. Similar difficulties may arise with cholesteatoma invasion of a cochlea or vestibule that are still functional, requiring the surgeon to carry out destructive procedures in order to achieve complete removal of the matrix. It should be emphasized that incomplete ablation is not an acceptable alternative.
In our own group of 27 patients with petrous bone cholesteatomas, bidirectional surgery was carried out in 14 (50%), and a three-directional access route was used in seven (27%). These multiple exposures were often carried out using staged interventions, to allow safe incorporation of grafts as a precondition for subsequent resection of the adjacent structures.
Surgical Techniques
Depending on the site of cholesteatomatous invasion of the petrous bone, four main routes of access are indicated:
- The transmeatal approach to the tympanic cavity and its near vicinity. This approach can be modified as an endaural exposure, particularly for restoring a functioning middle ear as the last stage of a combined approach.
- The transmastoid approach to the posterior compartments of the temporal bone, possibly continued as a translabyrinthine exposure of the internal auditory canal, in cases of total deafness.
- The transtemporal middle cerebral fossa approach to the petrous bone. This exposure of the pyramid from above is particularly valuable for exenteration of the petrous apex, with dissection of the horizontal portion of the internal carotid artery. It is the access route of first choice for all attempts to clean the petrous tip without sacrificing the middle ear and cochlea. It is especially appropriate for patients with facial nerve impairment in the supralabyrinthine portion of the fallopian canal. It may not be possible to expose these areas around the geniculate ganglion otherwise, without destruction of the attic and superior semicircular canal. The frequently observed invasion of the basal turn of the cochlea and of the labyrinth from above by a petrous bone cholesteatoma can also be managed from this access viewpoint with the best chances for nondestructive dissection.
- The anterolateral approach. With cholesteatomas that have extensively destroyed the anterior pyramidal segments around the internal carotid artery, one of the infratemporal approaches can be chosen.
The approaches listed above need not be discussed in detail here. They are described and illustrated in Chapter 4, and are explained with in vivo procedures in the sections on chronic otitis media (transmastoid and transmeatal approaches, pp. 133–155), on acoustic neuromas (middle fossa approach, pp. 230–241), and on glomus jugulare tumors (infratemporal, lateral approaches, pp. 268–277). However, some supplementary information can be given here that is relevant to the peculiarities of petrous bone cholesteatoma.
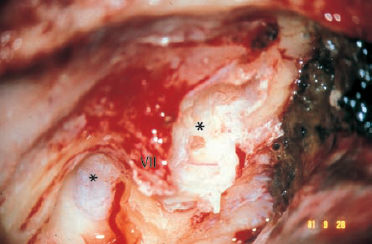
Fig. 13.4 A petrous bone cholesteatoma exposed from above via a middle fossa approach. The cochlea and labyrinth (asterisks) are covered, and the facial nerve is enveloped with cholesteatoma matrix (left ear).
VII Facial nerve
Dissection of the cholesteatoma matrix in petrous bone cholesteatoma can follow the same principles as described for middle ear cholesteatomas. Gentle elevation from the underlying mucosa, bone, dura, or facial nerve is best achieved by following a cleavage plane with a blunt instrument—e.g., the ball-tipped probe. Intracapsular debulking can considerably facilitate this type of stripping. Removal of debris is carried out with suction, and forceps are used only to tear off the already lifted parts of the sac. Still adherent parts of the matrix have to be mobilized in continuity. When these precautions are taken, prophylactic removal of the underlying mucosa or superficial milling of the eroded bone does not appear to be necessary.
Dissection of an enveloped facial nerve is easy on the surface, but may become difficult at the sides, where fibrous tissue (epineurium) is fixed to the bone. A sharp needle may be indispensable here. Problems arise if the nerve is completely covered with matrix, although this is rare (Fig. 13.4). Removal of the matrix from the undersurface of the nerve may require mobilization of the nerve. Great care needs to be taken to follow the continuity of the epidermal envelope. This is even more difficult with the greater petrosal nerve or the inferior cranial nerves. Whether or not to sacrifice these nerves has to be weighed up against the likelihood of recurrence.
Dissection of the matrix from an arroded labyrinth or cochlea is the most delicate phase of the operation. It should be remembered that below the matrix there is always an endoperiosteal layer that protects the perilymphatic space. However, it is very fragile and can easily be torn by a needle. If the fistula is located in the antrum or tympanic cavity, carrying out a circular incision around it is reasonable. The preserved matrix can then be removed from an uninfected middle ear after an interval of a few weeks. However, matrix on cranial fistulas has to be removed during the first, transtemporal, intervention. Elevation of this matrix in continuity from a fistula, and even out of an open semicircular canal, from a congested vestibulum, or even out of a cochlea, can be successfully achieved without loss of function (Fig. 13.5). If a leak occurs, immediate closure with connective tissue (I always use fibrin glue as well) can save the hearing. If the continuity of the matrix is lost and there is pre-sumably residual cholesteatoma within the inner ear, destructive extirpation is unavoidable.
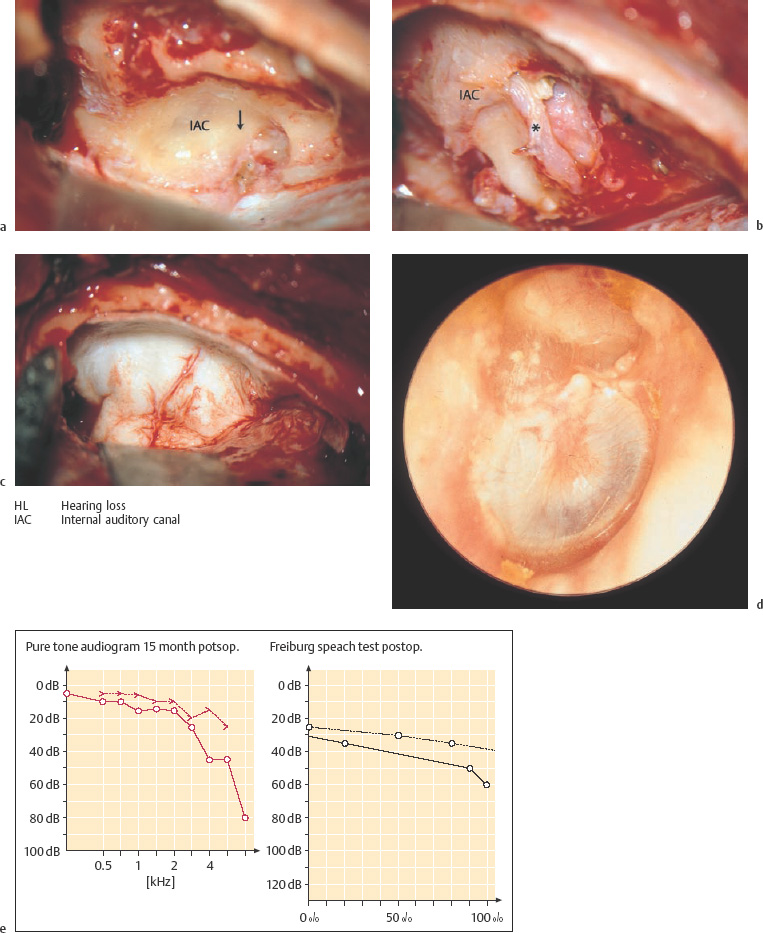
Fig. 13.5 Invasion of the labyrinth by a large petrous bone cholesteatoma (right ear; same patient as in Fig. 13.2). a Exposure of the filled superior semicircular canal (arrow, yellowish shadow) via a middle fossa approach. b The skeletonized floor of the middle fossa after dissection of the matrix. The superior semicircular canal (*) is now open. c The new floor of the middle fossa, consisting of homologous dura. d The reconstructed tympanic membrane and meatal wall over the reventilated middle ear, after additional endaural tympanomeatoplasty. e The postoperative audiogram.
Cleaning of involved dura or of the internal carotid artery is another substantial problem during dissection. As mentioned above, a switch may need to be made from a less invasive procedure to a broader, more destructive intervention. It is sometimes not possible to distinguish between a thin, gray layer of squamous epithelium and the fibrous surface of the dura. The outer dural layer can then be stripped away (Fig. 13.6), or it may even be reasonable to carry out limited resection of the doubtful area, with immediate repair using an overlay graft of periosteum or Tutoplast.
Expansion into the clivus and sphenoid bone, with involvement of large basal vessels and the cavernous sinus, is the most challenging problem—requiring cooperation between very experienced otosurgeons and neurosurgeons. Depending on the area opened up by the cholesteatoma, a total petrosectomy carried out via the translabyrinthine–transcochlear (transotic) approach may be adequate. As soon as the prepontine cistern, basal artery, or chiasma are covered by matrix, temporo-occipital neurosurgical exposure becomes necessary. It is doubtful whether complete removal is possible in cases of this type, but incomplete exenteration with drainage of the cavity is an option.
Results in Our Own Series
In our own series of 26 petrous bone cholesteatomas, the lesions were confined to the pyramid and did not penetrate beyond the petrous apex. The otosurgical methods used were limited to the transmastoid, transmeatal, and transtemporal approaches. Tables 13.5a and 13.5b list the distribution of approaches used in these patients. They also show that no additional permanent facial palsy occurred, and that the incidence of total deafness increased from 57% preoperatively to 71% postoperatively. This is an example of restitutional surgery, confirming the possibility of preserving hearing even in the presence of labyrinthine fistulas. In comparison, all 14 ears in the series reported by Atlas et al. (1992) were submitted to translabyrinthine or transotic approaches, with inevitable total deafness.
Cholesterol Granuloma of the Petrous Pyramid
Cholesterol granuloma is one of the most devastating tumor-like lesions of the temporal bone. Its local behavior is expansive and destructive, it has a uniform texture, it is difficult to handle, and its cause remains a mystery. The first descriptions of it were published around a century ago (Manasse 1894, Wagner 1907). The lesion is still a rare one; a review by Lorenz et al. (1996) listed 40 cases in the available literature. The term “cholesterol granuloma” was introduced by Friedman (1959). Graham et al. (1985) drew a distinction between cholesterol granulomas and giant cholesterol cysts, but the practical relevance of this in terms of diagnosis and treatment is difficult to see.
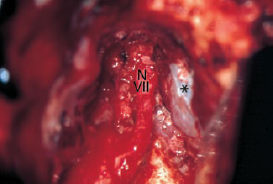
Fig. 13.6 Transmastoid dissection of a cholesteatoma matrix (*) from the cerebellar dura, splitting an external dural layer away to allow complete eradication of the cholesteatoma.
VII Facial nerve
| Transmastoid | Transmeatal | Transtemporal | Total | |
| One-stage | 6 | – | – | 6 |
| Two-stage | 12 | 6 | 8 | 13 |
| Three or more stages | 10 | 6 | 5 | 7 |
| Preoperative | Postoperative | |
| Deafness (n = 14) | 8 | 10 |
| Facial palsy (n = 15) | 4 | 4 |
Clinical Entity
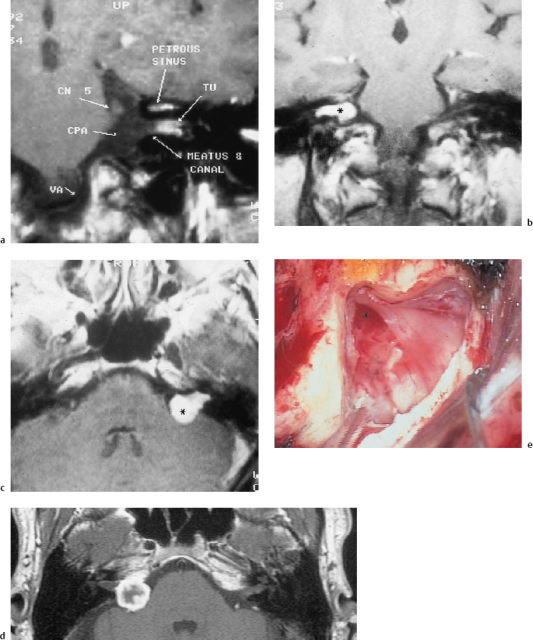
Friedman (1974) stated that cholesterol granuloma is not a clinical entity, but part of the pathology of otitis media. In fact, this is true only of lesions that are found in connection with chronic mastoiditis, or those located in tympanomastoid cavities. A similar type of pathology is occasionally located in the maxillary or frontal sinus. Cholesterol granuloma in the petrous apex, however, has mainly been found in patients with no history of otitis media, and with a healthy pneumatization. The morphology of the lesions is always the same: there is a thin, fibrous capsule filled with soft granulation tissue, well vascularized and saturated with fresh or older hemorrhage. In parts, the texture may be stiffened by fibrous components. The typical color is dark red and brown, and the lesion is translucent through thin bone and mucosa (Fig. 13.7). Histological sections reveal cholesterol crystals and the brown blood pigment hemosiderin, with many foreign-body giant cells immersed in it. The granuloma surface is free of epithelial cells—and in relation to incomplete removals, it is important to know this.
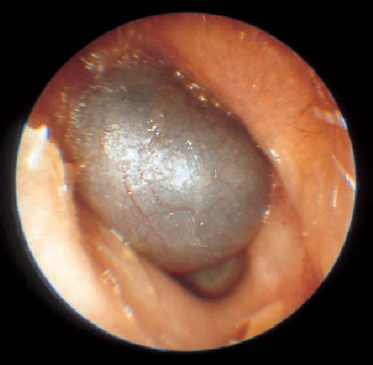
Fig. 13.7 Cholesterol granuloma of the petrous bone, penetrating into the external auditory canal (left ear)
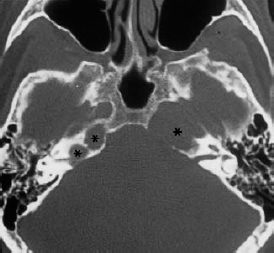
Fig. 13.8 Bilateral cholesterol granulomas of the petrous pyramids (*). The axial CT shows complete destruction of the right petrous apex and two smaller lesions in the left pyramid, one of which has affected the internal auditory canal (59-year-old man).
The fact that cholesterol granulomas consist of blood-related constituents suggests that hemorrhage, or at least microhemorrhage, is a major causative factor. It has not yet been determined whether the cholesterol crystals serve as a stimulating irritant for the formation of growing, tumor-like granulomas. The occurrence of bilateral cholesterol granuloma at corresponding sites in the skull base (Fig. 13.8) may support the assumption that trauma is involved, causing rupturing of the equivalent vessels on each side. With regard to the typical topographical locations of most apical cholesterol granulomas, I would speculate that the site of origin is usually the apical bone marrow. The reason for the individual direction in which the lesion expands—backward toward the internal auditory canal, for instance—remains enigmatic. Mini-trauma in connection with inflammatory fragility of small vessels would also explain the occurrence of cholesterol granuloma in ears that have previously undergone surgery for chronic otitis media. The recurrence of such granulomas in the neighborhood of submucosal hematomas in the attic or mastoid is a strong argument for this type of pathogenesis. After tympanoplasty procedures, I have repeatedly observed hematomas of the canal skin that contained the typical, glistening cholesterol crystals.
Symptoms and Signs
Depending on the site of the space-occupying granuloma, the presenting symptoms are conductive hearing loss, giddiness and aural pain or pressure (in the middle ear), facial nerve impairment or spasm, progressive sensorineural hearing loss, tinnitus, and vertigo (involvement of the inner ear or internal auditory canal), headache, trigeminal impairment, diplopia, or abducent nerve palsy (petrous apex syndrome). Usually, these symptoms are not perceived before the granuloma has reached a considerable size and has produced a certain amount of pressure due to lack of space.
Diagnosis and Differential Diagnosis
Inspection or ear microscopy can directly raise a suspicion of cholesterol granuloma only in external auditory canals or cavities in which there has been prior surgery. Audiometry and vestibular testing are indicated for ear-related symptoms. Ophthalmological investigations are often the first examination that is carried out in patients with apical tumors, and provide sufficient grounds for requesting an otological consultation. In most cases, imaging provides a precise diagnosis, with the CT demonstrating bone destruction in one or both temporal bones. Magnetic resonance imaging is able to clarify the differential diagnosis between cholesterol granuloma and petrous bone cholesteatomas or genuine tumors such as schwannoma, angioma, chondroma, etc. (Table 13.2).
Indications for Surgery
Although cholesterol granulomas of the middle and external ear are unanimously regarded as indications for surgical removal, there is controversy over the difficulty of reaching cholesterol granuloma in the petrous apex. Reliable, permanent drainage of cholesterol cysts in this region can be achieved by opening them via a sublabyrinthine or transsphenoidal approach (Gacek 1975, 1980, Gherini et al. 1985, Giddings et al. 1991). However, recurrences must be expected in a substantial percentage of cases, due to the inevitable tendency of nonepithelialized tunnels to become obliterated by scar formation. Permanent recovery can only be achieved if the granuloma is halted after initial decompression, with cleaning of the crystals and blood. Due to the small number of observations that have been made, it is not possible to offer any statistics regarding the prognosis with drainage operations.
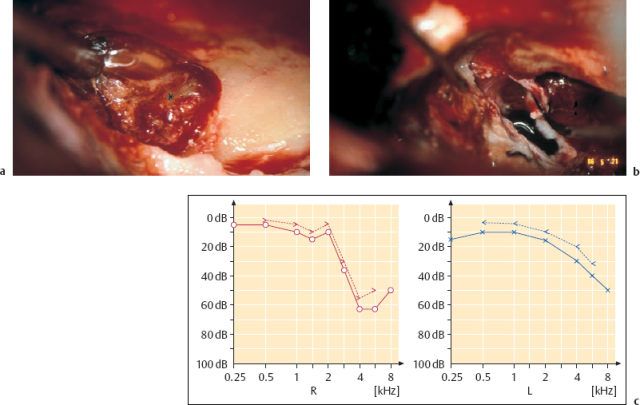
Fig. 13.9 Eradication of the cholesterol granulomas via the enlarged middle fossa approach (same patient as in Fig. 13.8). a Dissection of the right cholesterol cyst (*) from the cerebellar dura in front of the internal auditory canal. b Exenteration of the left petrous apex below the cochlea. c The postoperative audiogram shows that hearing was preserved at the preoperative level.
Complete or near-total ablation of the granuloma cyst appears to be the safer option. Surgical removal should be carried out whenever it appears possible to expose the lesion without endangering vital structures such as the cranial nerves, inner ear, and internal carotid artery. This attitude is supported in the current literature (Lorenz et al. 1996). In six of our own cases of cholesterol granuloma involving the anterior petrous bone, it was always possible to remove the lesions completely. The technique of choice was the middle fossa approach to the petrous apex (Fig. 13.9). Coronal CTs are valuable for providing precise delineation of the extent of a cholesterol granuloma and showing the height of the apical lesion. If it extends beneath the level of the eustachian tube, it may be necessary to carry out a second counter-operation, with inframeatal-infracochlear exposure of the inferior compartment. In our own small series, destructive procedures (with the translabyrinthine and transcochlear approaches) were not used, and bidirectional function-preserving interventions were carried out instead. Function was in fact preserved in all six patients, while nine of the 40 patients in the literature reviewed by Lorenz et al. (1996) underwent a destructive translabyrinthine approach. However, the latter approach may be justified, as it is easier to perform in patients with advanced disturbances of hearing and equilibrium (House and Brackmann 1982). We have treated a large number of middle ear cholesterol granulomas, not discussed here, using the ordinary endaural or transmastoid approaches.
Surgical Technique
Exposure of the floor of the middle cerebral fossa is described in detail in the section on acoustic neuromas below. For a lesion in the very tip of the pyramid, the skin incision and dissection of the temporalis muscle should be carried out 1–2 cm more rostrally, however. The frontal branch of the facial nerve must be avoided. The craniotomy must also be advanced slightly closer to the zygomatic arch, placing the middle meningeal artery in the center of the surgical field. After identification of the internal auditory canal and of the cochlea, bone removal follows the superior petrosal sinus to the apex, and uncovers the internal carotid artery. Usually, the cholesterol granuloma is reached just below the floor of the middle fossa. Using diamond drills to remove the roof of this gently, one will be able to expose the perimeter of the lesion without injuring the internal auditory canal. The sac can then be opened and emptied by suctioning out its semifluid contents. Irrigation with saline under moderate pressure helps with hemostasis. Hemorrhage from the snus or apical bone marrow can be stopped by applying Surgicel. With tender, blunt dissection, the granuloma membrane can now be lifted in continuity from the adjacent dura and from the bony walls of the cavity. The ball-tipped dissector, Zaufal’s hook, or a round knife are appropriate tools for stripping it off the cleavage plane. Removal of the membrane from deep extensions can become difficult. If small residual areas are resistant, leaving them in place is not problematic, since they are free of epithelium. They can be cauterized for definitive destruction. It is even more difficult when there is avulsion of the cholesterol granuloma from the internal auditory canal, which has occurred twice in my experience. Blunt dissection from the caudal (!) meatal dura was possible only under the guidance of an angled-view endoscope. Bilateral granulomas of the apex were removed in two sessions (Fig. 13.9).
Giant Cell Granuloma
Giant cell tumor (osteoclastoma) consists of a well vascularized network of spindle-shaped fibrocytes and ovoid fibroblasts, with islands of multinuclear giant cells. Microhemorrhage is typical, producing deposits of hemosiderin—which is why the term “brown tumors” is used for this lesion. The ample dispersion of osteoclasts has also led to the term “osteoclastoma.” The transition from a grade I benign lesion, via the grade II borderline type, to a grade III malignant lesion indicates genuine tumor growth, destroying the temporal bone. The pathogenesis is not uniform; hyperparathyroidism is the cause of some of these lesions. The neoplasia may grow considerably without becoming symptomatic. Aural pain and headache are among the presenting symptoms, but ocular dysfunction (diplopia, deficits of the visual field) and facial paralysis are signs of local impairment that lead to neurological examination. Kattner et al. (1998) recently summed up relevant data from the scarce literature on giant cell tumors of the sphenoid bone. They emphasize that radiographic evaluation is not capable of identifying the lesion. One feature that may be typical is compression of larger vessels or of the internal auditory canal, rather than encasement of them.
Surgical removal is appropriate for the unilocular form, but widespread multilocular lesions have been described. Incomplete exenteration leads to early recurrence. The approach that is suitable for the affected area must be selected in relation to the individual location. A supra-auricular or preauricular route (Fig. 13.10) is appropriate for access to the external auditory canal and mandibular joint, while the transtemporal middle fossa approach (Fig. 13.11) provides good exposure of anterior apex lesions. Radiotherapy has not proved successful (Bertoni et al. 1992), and can induce sarcoma (Freyschmidt and Ostertag 1988). However, long remissions have also been described.
Histiocytosis X
Histiocytosis X (Hand-Schüller-Christian disease, Letterer-Siwe disease, eosinophilic granuloma). This is a systemic disease that may have a milder course, or a severe and even fatal one. It produces polyostotic bone defects with sharp borders in the skull and tympanic bone. Bone necrosis is marked by clusters of histiocytes, and by various patterns of intermittent eosinophilic cell granulomas. Involvement of the external auditory meatus and of the middle ear leads to symptoms resembling those of otitis media, with otorrhea and hearing loss. Destruction of the meatal walls and of the mandibular joint causes pain and disturbances of mastication. The diagnosis is obtained by radiography and biopsy. The round to oval form, limited size, and sharp margins of the radiolucent areas can lead to a differential diagnostic suspicion of metastases. Treatment is conservative, including radiotherapy, but otologic surgery may be indicated for removal of obstructing granulomas and for drainage of the middle ear. Reconstructive measures follow the rules of tympanomeatoplasty.
Fibrous Dysplasia of Bone and Paget’s Disease
Fibrous dysplasia of the temporal bone (Jaffé-Lichtenstein disease) is very rare. It is more frequently located in the midface and anterior skull base. Its cause is not known, and the histopathological findings are characterized by irregular osteoclastic bone destruction. The bone defects are filled with connective tissue and with trabecular, immature bone formation. The affected areas of the calvaria and skull base are thickened, and invade the adjacent lamellar bone without sharp borders. Similar tumor-like changes of the temporal bone can be caused by Paget’s disease (osteitis deformans). An alternating process of resorption and apposition of bone, with multinucleated osteoclasts and chains of osteoblasts, is typical (Friedman 1974). Ossifying fibroma and osteopetrosis (Albers-Schönberg disease) also belong to this group of fibrous lesions of the bone, which can be distinguished from each other histologically and radiologically but have similar manifestations in the region of the ear.
The lesions are histopathologically benign, but they become clinically significant if they lead to compression of the external auditory meatus or middle ear structures, or of the labyrinth, internal auditory meatus, or facial nerve. Apart from the external protrusion of the temporal region that may be visible, the presenting symptoms therefore include deep aural pressure or pain, inner ear hearing loss, dizziness, and tinnitus, as well as facial paralysis and even disturbance of deglutition. A CT examination can delineate the process and identify the nature of the lesion (Fig. 13.12). A biopsy taken with a chisel from the mastoid can be helpful in obtaining a specific diagnosis. No treatment of the cause is available, and complete excision is possible only in the initial stages. However, most cases are not diagnosed before there is extensive proliferation. There are monostotic and polyostotic forms. A wait-and-scan policy is justified. With progressive palsies or deafness, however, decompression operations with unroofing of the fallopian canal or of the internal auditory meatus via the transmastoid or middle fossa approaches may be indicated.
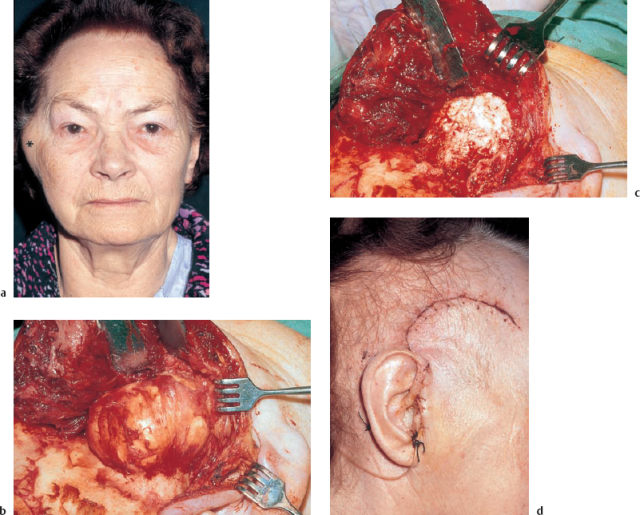
Fig. 13.11 Giant cell granuloma of the right petrous bone. a The tumor-like lesion (*), with a reddish-brownish color and a soft consistency, is exposed via the middle fossa approach in front of the superior semicircular canal, over the attic. b The clean tegmen epitympani is checked from above.
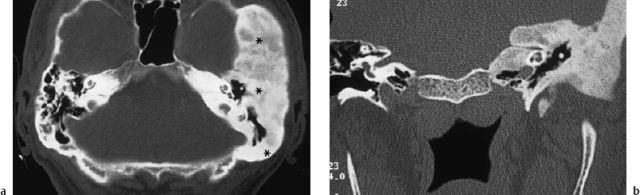
Fig. 13.12 Diffuse space-occupying tumor-like lesions of the temporal bone. a Fibrous dysplasia of bone (Jaffé-Lichtenstein disease) on an axial CT. There is massive encasement of the pyramid (*) by fibrous dysplasia of the temporal bone. The floor of the middle cranial fossa is enlarged, and the pneumatization has been replaced by cloudy, thickened bone (in a 58-year-old man who presented with tinnitus and progressive sensorineural hearing loss). b Ossifying fibroma (repeated histopathological diagnosis) on a coronal CT. There is compression of the internal auditory canal (in a 44-year-old man suffering from progressive hearing loss, tinnitus, and vertigo). Both ossifying fibroma and the fibrous dysplasia shown in Fig. 13.12a slowly compress the middle and inner ear structures, internal auditory canal, and related cranial nerves.
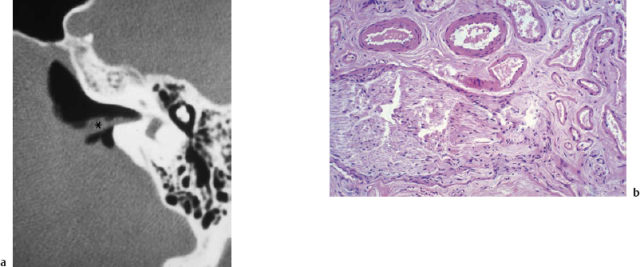
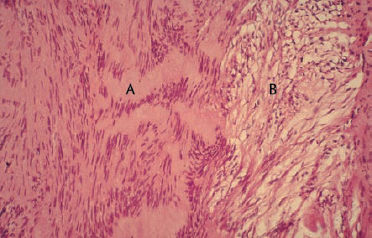
Fig. 13.13 Angiodysplastic granuloma of the temporal bone. a A tumor-like lesion (*) at the internal auditory canal, simulating a small acoustic neuroma (air cisternography). b The histopathological image (hematoxylineosin, original magnification 250 ×).
Vascular Lesions
Various types of angiodysplasia may become spaceoccupying, and can consequently compromise functional structures in the middle ear or internal auditory canal The histopathological appearance resembles that of cavernous hemangioma, and the lesions are usually monolocular. However, other entities such as arteriovenous shunts or fistulas can also grow with multilocular and confluent formations. While the former are benign in their behavior, the latter are very destructive, and have a fatal prognosis with cardiac insufficiency. Aneurysms can also occur in the internal auditory meatus, as well as vascular deformities and dislocations such as a persistent stapedial artery, an internal carotid artery located in the tympanic cavity, or a high-reaching jugular megabulb.
Depending on their size and location, tumor-like vascular lesions can lead to severe impairment. Angiodysplastic granulomas and aneurysms in the internal auditory canal produce the same symptoms as a small acoustic neuroma, and also have a very similar configuration on MRI (Fig. 13.13). In a series of 112 cases in which surgery was carried out with a presumed diagnosis of intrameatal schwannomas smaller than 5 mm in diameter, 8% turned out to be angiodysplasias or well-vascularized arachnoid connective tissue. Since all of these lesions were located near the meatal fundus, the differential diagnosis when this type of “minineuroma” is suspected must include vascular lesions and facial nerve schwannoma in the supralabyrinthine portion. An aneurysm (Fig. 13.14) in the internal auditory canal or a jugular megabulb (Fig. 13.15) can also lead to progressive sensorineural hearing loss, giddiness, and intractable tinnitus. Imaging always has to be extended to the contralateral side, since hypoplasia or even aplasia of the contralateral sigmoid sinus is not infrequent when there is a megabulb on one side. Recognition of such lesions has a significant impact on the surgical planning. Intentional or unintentional occlusion of the impinging vessel has to be avoided in these conditions.
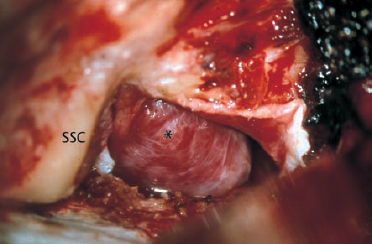
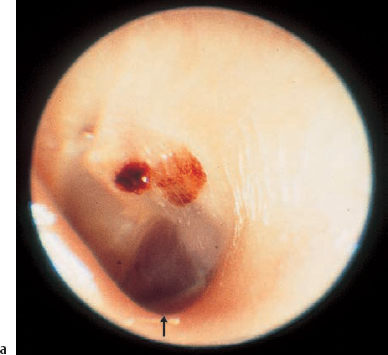
Fig. 13.15 Jugular megabulb causing cochlear hearing loss and tinnitus. a Otoscopy shows a dark blue shadow behind the drum membrane (arrow).
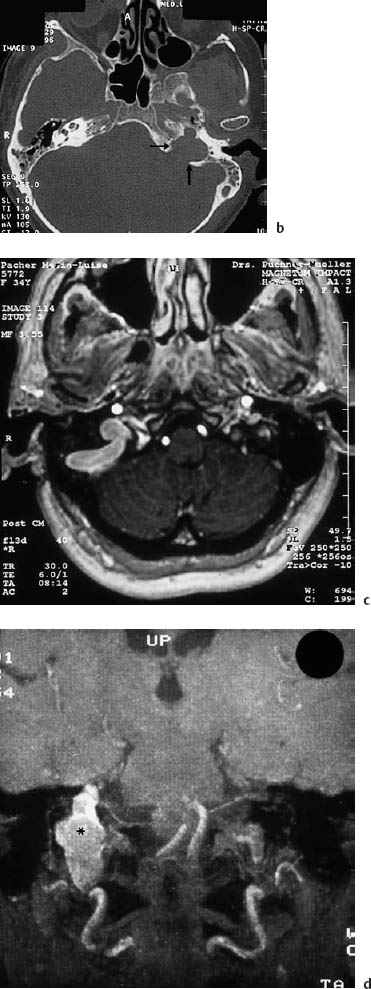
Fig. 13.15b The axial CT shows the space-occupying bulb (arrow) at the level of the labyrinth, and an underdeveloped contralateral sigmoid sinus. c, d A unilateral jugular megabulb (*) visualized with magnetic resonance angiography on the axial projection (c) and coronal projection (d).
As with vascular hyperplasia, an endolymphatic megaduct may also be found during imaging procedures when the temporal bone is being examined due to progressive sensorineural hearing loss and giddiness (Fig. 13.16). The pathological mechanism underlying the insufficiency of inner ear functions is not known here, however. Surgical intervention would appear to involve a risk of further sensorineural hearing loss, and it does not lead to any benefit in terms of improved hearing (Welling et al. 1999).
The indications for surgery have to be selected individually. Progressive hearing loss, vertigo, severe tinnitus, or facial palsy are reasons for considering a palliative intervention. Intratympanic lesions can be approached endaurally. A vascular granuloma can be destroyed using bipolar cautery or by excision in the same way as a hemangioma. The CO2 laser is valuable if the vessels have a diameter of less than 1 mm. A persistent stapedial artery can be closed or narrowed using a sling of connective tissue, which can be expected to shrink and srangulate the vessel after a few weeks. A jugular bulb that extends up to the round niche and causes noise can be mobilized and pushed downward. A piece of Silastic sponge enveloped by fascia is then anchored in the hypotympanum as a separating cushion.
Vascular lesions at the geniculate ganglion and within the internal auditory canal are best exposed using the middle fossa approach (Gjuric et al. 1999). Depending on the nature of the process, excision or displacement of the space-occupying process will decompress the affected nerves, and unroofing the internal auditory canal itself produces substantial symptomatic relief.
 Facial Nerve Schwannoma
Facial Nerve Schwannoma
Clinical Entity
Schwannomas of the facial nerve (neurinoma, neoplasia of the Schwann cells), first described by Schmidt (1931), are rare tumors. Conley and Janecka (1974) found 76 cases in a review of the literature up to 1972. There are now many reports describing manifestations between the internal auditory canal and the parotid gland, as well as bilateral growth (O’Donoghue et al. 1989, Fenton et al. 1999). Fisch and Rüttner (1977) postulated that the tumors grow in continuity, although some nodular expansion may give the impression of a multifocal lesion. Our own observations in 19 cases confirm this (Fig. 13.17). The histopathology corresponds to the typical appearance of other schwannomas, with Antoni type A or type B textures. There is no particular growth pattern in patients with von Recklinghausen’s disease (neurofibromatosis types 1 or 2).
The presenting symptoms are often untypical, and correspond to the site of the lesion. Facial palsy is fairly uncommon. Gerhardt et al. (1985) found only three schwannomas in 1128 cases of facial paralysis. In our series, hearing loss, giddiness, and tinnitus were predominant (Table 13.6), with locations in the middle ear and supralabyrinthine area often involving the ossicular chain or inner ear. Conductive deafness when the tympanic membrane has a normal appearance is therefore a reason for including facial nerve schwannoma (Fig. 13.18) in the differential diagnosis of otosclerosis. In the series reported by Nelson and House (1982), 17 lesions in 23 facial nerve neuromas had sites within the labyrinth, internal auditory canal, or cerebellopontine angle. The clinical course in these cases therefore most often resembled that of acoustic neuromas. When the patient history and the symptoms described give rise to a suspicion of facial nerve schwannoma, the following examinations are therefore required: electrophysiological testing, testing of lacrimation and taste sensation, CT of the temporal bone (Fig. 13.19), and ultrasound B-scanning of the parotid gland. Deficits in facial motor function were not always present in our group of patients; a pathological Schirmer test and hypogeusia were found exceptionally.
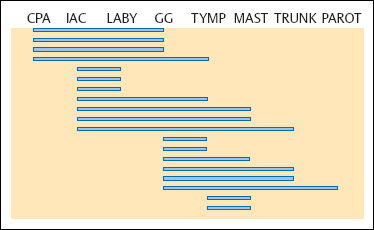
Fig. 13.17 Growth patterns in 19 facial nerve schwannomas (in our own series) located between the cerebellopontine angle and the parotid gland. The geniculate ganglion was involved in the majority of cases.
CPA Cerebellopontine angle
IAC Internal auditory canal
LABY Labyrinth
GG Geniculate ganglion
TYMP Tympanic membrane
MAST Mastoid
TRUNK Inframastoid portion
PAROT Parotid gland
| Sensorineural hearing loss | 16 | |
| Total deafness | 3 | |
| Facial nerve palsy | 8 | |
| No other symptoms | 2 | |
| + SNHL | 4 | |
| + SNHL + vestibular symptoms | 1 | |
| + Conductive hearing loss | 1 | |
| Vestibular symptoms | 4 | |
| Conductive hearing loss | 2 | |
| Tinnitus | 2 | |
| Parotid gland tumor | 1 | |
| SNHL: sensorineural hearing loss. |
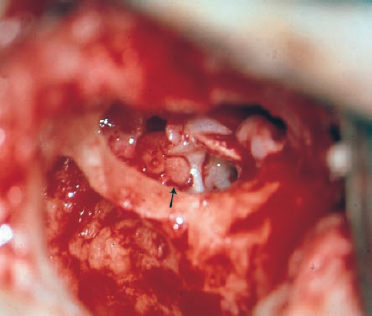
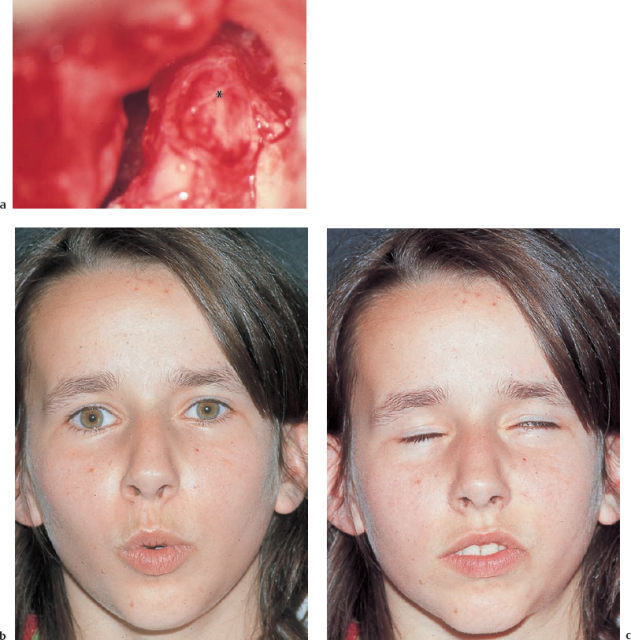
Fig. 13.18 A small intratympanic facial nerve schwannoma (arrow), exposed over the oval niche via an endaural tympanomeatoplasty (right ear). The tumor is visible in front of the bony bridge, after removal of the incus, the lenticular process of which was preserved to provide acoustic contact with the drumhead. Resection of the nerve with immediate interposition of a free graft from the greater auricular nerve led to a House–Brackmann grade of III.
As 68% of the cases of facial nerve schwannoma identified in our series had not been recognized as such before surgery, the intraoperative findings were surprising in some cases. Unexpected confrontation with a nerve tumor creates particular problems, since the consent form signed by the patient may not cover this eventuality, and biopsy procedures may lead to a palsy that was not previously present.
Indications for Surgery
Basically, complete surgical ablation of the tumor is indicated, implying resection of the nerve within healthy margins and subsequent nerve grafting. This indication may become a relative one if the symptoms are having little impact on the quality of life in elderly patients, or when patients are going through a critical career phase in their profession. If careful functional and imaging check-ups are carried out regularly, it may occasionally be a prudent decision to postpone postsurgical facial paralysis. The tumor is benign, and we have never observed malignant degeneration except in one case of neurofibromatosis type 1.
Surgical Technique
Infrapetrosal tumors in the parotid gland are best exposed using the typical bayonet-like subauricular skin incision, with dissection of the nerve trunk at its exit from the stylomastoid foramen. From here, the facial nerve branches can be followed to the schwannoma, as in parotidectomies. Harvesting a free graft from the great auricular nerve is very easy with this access route. Schwannomas of the tympanic and mastoid portions are excised via the endaural approach, with immediate restoration of normal anatomy, or using a transcortical, transmastoid access route if the lesion has a larger sublabyrinthine extension. In all schwannomas that extend into the intralabyrinthine portion and are located preoperatively using CT (MRI has recently also proved valuable here), a middle fossa approach is indicated (pp. 230–241), either alone or in combination with endaural exposure or with the transmastoid route. In ears with total deafness, the translabyrinthine access route to the geniculate ganglion and internal auditory canal may have the advantage of a smaller intervention. I prefer transtemporal exposure from above, as it provides a better overview of the petrous apex and allows easier reconstruction of the tympanic cavity using an additional endaural approach (Fig. 13.20).
When there is an eccentric growth pattern (Fig. 13.21a, b), partial resection of the facial nerve may be feasible by shaving off the tumor together with the involved fascicles from the underlying healthy fibers. We were able to use this technique in eight of 19 cases (Wigand and Wolf 1998). A free nerve graft was interposed into the partial defect (Fig. 13.22). If more than 50% of the nerve fibers were invaded, however, thorough excision of the involved nerve portion was carried out, with subsequent interposition of a full nerve graft. The oncological and functional results (Tables 13.7, 13.8) were satisfactory, with no recurrences being observed.
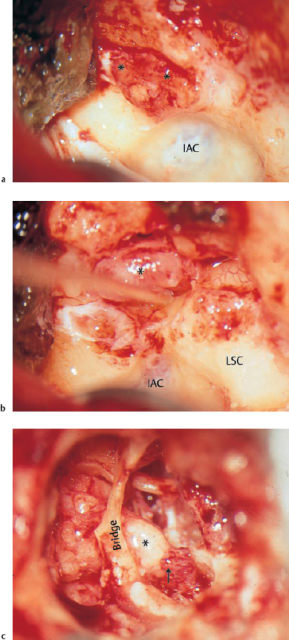
Fig. 13.20 Combined transtemporal and endaural approach for restitutional management of an advanced supralabyrinthine facial nerve schwannoma (right ear, same patient as in Fig. 13.19b). a Middle fossa exposure of the exophytic lesion (**) over the geniculate ganglion. b Blunt lifting of the tumor (*) with the geniculate ganglion. Extension of the nerve lesion into the tympanic portion. The greater petrosal nerve and the internal auditory canal were free of tumor. c Endaural exposure of the tympanic cavity, with interposition of a sural nerve graft (*) down to the facial recess. Synthesis of the nerve (arrow) with a coating of fibrin tissue adhesive behind the pyramidal process. The bony bridge was preserved to allow a normal tympanomeatal reconstruction.
IAC Internal auditory canal
LSC Lateral semicircular canal
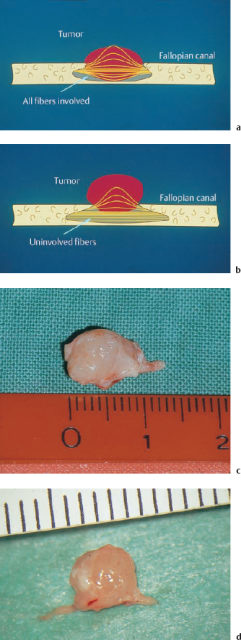
Fig. 13.21 Growth patterns of schwannomas. a Concentric growth. b Eccentric growth. This type of growth leaves options for partial nerve resection over a considerable time period. c, d Surgical specimens of these two growth patterns in acoustic neuromas completely excised via the enlarged middle fossa approach.
| No cases of malignant degeneration |
| No cases of multiple growth |
| No cases of recurrent tumor growth |
| House-Brackmann grade | ||||||
| I | II | III | IV | V | VI | |
| After partial resection of the facial nerve | 3 | 1 | 4 | – | – | – |
| After total nerve excision with graft interposition | – | – | 4 | 2 | – | 2 |
 Acoustic Neurinoma, Vestibular Nerve Schwannoma
Acoustic Neurinoma, Vestibular Nerve Schwannoma
Clinical Entity
The reported incidence of acoustic neurinomas has been increasing dramatically in recent years, probably because it is more easily detectable using magnetic resonance imaging. Before the era of modern imaging, an otology service would be likely to diagnose only a few cases per year using sophisticated audiological tests or vestibulometry, with subsequent radiography. Nowadays, however, MRI examinations are quickly requested by many otologists when there is unclear vertigo or progressive unilateral sensorineural hearing loss. Small and very small tumors are consequently diagnosed early, and 50–80 patients per year may be referred for treatment at a single otorhinolaryngology service. Tos et al. (1997,1999) have estimated the current incidence of acoustic neurinoma in Denmark at 12.4 per million (before 1983 the figure was 7.8 per million). This figure contrasts with the data given by Stewart et al. (1975), in which a 0.9% rate of acoustic neurinomas was found in a series of 893 unselected temporal bone autopsies. Selesnicketal. (1999) did not find a single lesion incidentally in 161 patients undergoing MRI examinations.
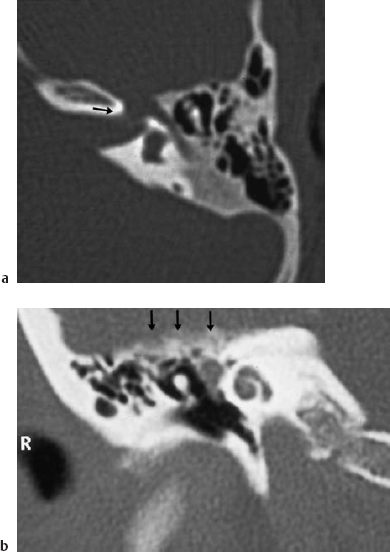
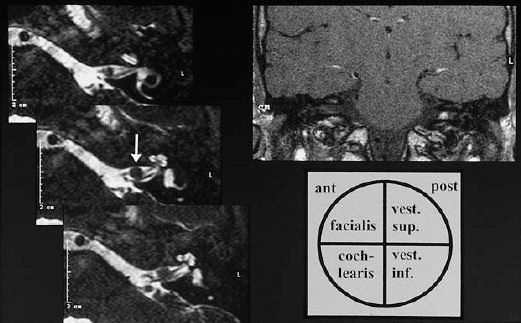
Henschen (1910, 1915) pointed out that the tumor always originates from the vestibular nerve (mostly from its inferior branch), but never from the cochlear nerve, and that it starts growing from the meatal fundus. With experience in more than 800 operations in our department, I can confirm this rule, which implies that all acoustic neurinomas start growing within the internal auditory canal, and only extend into the cerebellopontine angle secondarily. “Medial schwannomas,” growing from the cerebellopontine angle with an appendix extending into the internal auditory canal, do not exist. Support for this statement is also provided by the configuration of small and large acoustic neurinomas on MRI. Whether they are small, medium-sized or large, almost all tumors give a positive signal within the internal auditory canal (Fig. 13.23a–c), indicating that their growth starts lateral to the opening of the internal acoustic meatus (porus acusticus internus). Even if a contrast-enhanced MRI suggests an apparently “medial” tumor, with no extension into the internal auditory canal (Fig. 13.23d), surgery will demonstrate that it has the typical growth deep within the meatus (Fig. 13.23e).
Intralabyrinthine schwannomas, originating from the terminal fibers of the eighth nerve, are very rare (Fitzgerald et al. 1999). They can be found within the vestibule or the cochlea, and they have to be distinguished from the usual acoustic neurinomas penetrating the meatal fundus into the inner ear (Wigand and Haid 1976, Wigand 1981). According to these observations, and for practical reasons, our own size categories are differentiated pragmatically into A (intrameatal), B (with extension into the cerebellopontine angle, but not involving the brain stem), C (with broad contact to the brain stem), and D (with a diameter of more than 3 cm within the cerebellopontine angle; giant tumors). Intralabyrinthine extension is classified as L (Table 13.9).
Acoustic neurilemmomas, usually incorrectly referred to as neuromas, are histologically benign, and basically feature two well-known growth patterns (Antoni types A and B), both of which can exist within the same tumor (Fig. 13.24). Malignant degeneration is notobservedin unilateral acousticneuromas, norinbi-lateral neurinomas in neurofibromatosis type 2. In neurofibromatosis type 1, however, a neurosarcoma can develop in one or more of the ubiquitous nodes. The speed of growth of acoustic neurinomas—an important parameter in relation to the indication for surgery—seems to be variable. There is still a lack of adequate statistical evidence. Various series have reported observations in patients who did not undergo surgery, showing rapid expansion of the neurinomas in some and stagnation of growth in others. An average growth rate of 1–2 mm per year has been found by most authors, and our own observations have shown similar values (Triebswetter 1999).
Acoustic neurinomas occur with equal frequency in both sexes. The peak incidence is in middle age (Fig. 13.25), but the lesions also occur in children. It is interesting that the different tumor sizes are equally represented in all age groups (Table 13.10). Small tumors (size A) seem to grow less rapidly than larger ones (Fig. 13.26).
The pathogenesis of unilateral acoustic neurinomas is still unclear. The preferential location of such lesions in a specific branch of the eighth cranial nerve suggests some type of tissue abnormality. In the eighth nerve, the demarcation line between the neuroglia and the neurilemma is thought to lie more distally than in others, namely within the internal auditory canal (Nager 1969). Molecular genetic studies have identified a tumor suppressor protein, Merlin (moesin, ezrin, radixin-like protein), encoded by the NF2 gene, which is pathologically absent in bilateral neurofibromatosis type 2. This genetic abnormality is very seldom found in unilateral acoustic neurinomas, but Bikhazi et al. (1998) have described hereditary characteristics in families with unilateral vestibular schwannomas.
| A | Only intrameatal | ≤11 mm |
| B | Extension into CPA, no contact to brain stem | ≈3—;15mm* |
| C | Broad contact with brain stem | ≈ 16–30 mm* |
| D | Giant tumors | > 30mm* |
| L | Intralabyrinthine growth |
CPA: cerebellopontine angle.
*Largest diameter, within cerebellopontine angle.
| % of age groups | ||||
| A | B | C | ||
| 10–30y | 6.6 | 16.7 | 43.3 | 40.0 |
| 31–60y | 74.3 | 16.6 | 45.0 | 38.5 |
| >60y | 19.1 | 11.5 | 48.3 | 40.2 |
| Total | 15.6 | 45.5 | 38.9 | |
A, B, and C refer to the neuroma sizes given in Table 13.9.
Symptoms
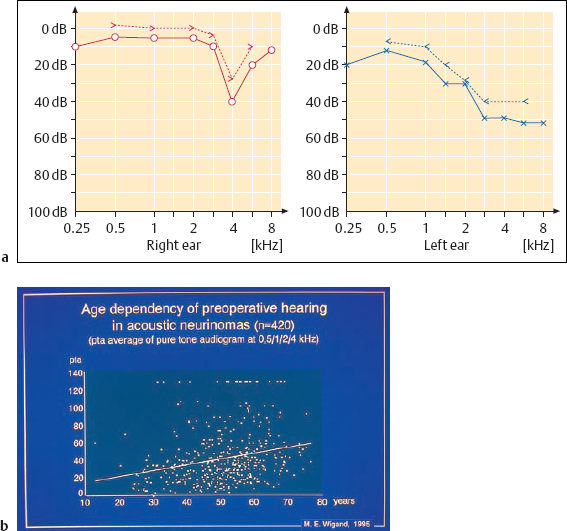
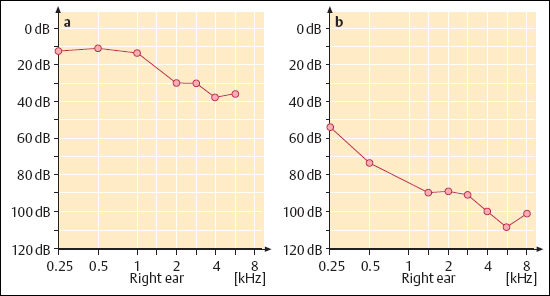
The symptoms are characterized by a triad of vertigo, deafness, and tinnitus. The individual complaints vary considerably. Although it originates in the vestibular nerve, the tumor often induces only discrete periods of giddiness and disequilibrium initially. The slow compression and destruction of the vestibular nerves can apparently be compensated for by the vestibulo-optokinetic system. With later impression into the cerebellum, however, severe ataxia may become predominant. The presenting symptoms in the majority of cases are unilateral progressive deafness and tinnitus. The typical pattern of the audiogram shows a moderate high-tone perception deficit (Fig. 13.27). In a group of 56 unoperated ears in which regular audiometric check-ups were carried out for a considerable period, the calculated annual average pure tone audiometry (PTA) hearing loss amounted to 10.8 dB. About 25% of our patients presented with single or repeated attacks of sudden hearing loss, and they were often treated conservatively for extended periods. Accompanying tinnitus is frequent, but is not always present. It is marked by high-pitched narrowband noises, or sometimes has a pure tone quality with low to marked intensity. Many patients report deep aural pressure when asked about this symptom. The range and prominence of the symptoms do not correspond to the size of the tumors.
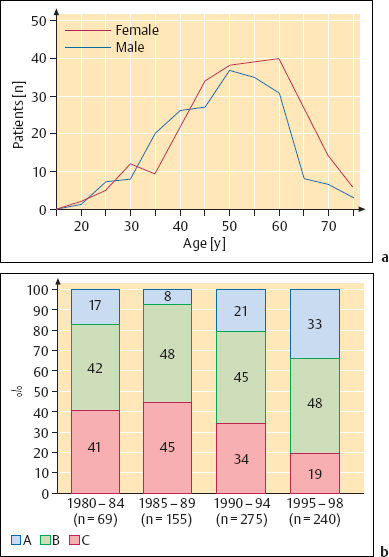
Fig. 13.25 Survey of our own series of patients with acoustic neurinomas, who underwent surgery via the extended middle fossa approach. a Distribution of sex and age in our own series of 420 cases of vestibular nerve schwannomas. b The distribution of tumor sizes A–C (in 739 unilateral lesions) shows a shift toward smaller tumors over the years. For definitions of sizes A–C, see Table 13.9 (p. 223).
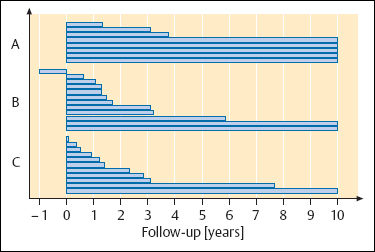
Fig. 13.26 Growth rates in 30 acoustic neurinomas, with regular MRI follow-up examinations for 10 years, showing doubling of the calculated volume. For definitions of sizes A–C, see Table 13.9 (p. 223).
Facial numbness, pain, impaired swallowing, and ocular dysfunction are rare. These indicate involvement of the trigeminal nerve or other cranial nerves, or even increased intracranial pressure. I have seen a woman with cerebrospinal fluid rhinorrhea of unknown cause, in whom the CT revealed a large, otherwise asymptomatic acoustic neurinoma.
Diagnosis
The required examinations are: pure tone audiometry, speech audiometry, early evoked brain stem responses, vestibular testing, and contrast-enhanced MRI. The audiogram generally shows unilateral high-frequency sensorineural hearing loss, which may fluctuate or even recover after conservative treatment in patients in whom sudden hearing loss is the presenting symptom. The majority of patients develop a slowly progressive threshold shift. In 550 candidates referred for surgery, 484 (88%) had a preoperative pure tone loss, averaging 500–4000 Hz, of 60 dB or less. Speech audiometry is not indicative. Discrimination that is disproportionately bad in comparison with the tone audiogram (considered to be a typical finding) was not confirmed in our series. Other forms of supraliminal auditory testing—e.g., for positive recruitment—have become less important due to high failure rates in comparison with imaging techniques. Auditory brain response (ABR) audiometry has proved valuable for detecting retrocochlear lesions, and in most countries it is used as an otological search test for selecting candidates for more expensive imaging modalities. Pathological latencies at peaks II–V, particularly an increase in the interpeak latency I–V to more than 0.2 ms above the normal range (4.04 ± 0.34 ms), and an interaural difference in the peak V latency of more than 0.4 ms, are highly suspicious and require further diagnostic procedures. Don et al. (1997) trace small lesions with special band-noise.
On the other hand, vestibular examination is indispensable. Thermic hyporeflexia and discrete positional nystagmus (Fig. 13.28), provoked by refined positioning tests, are valuable signs for early detection of acoustic neurinomas (Haid 1981). With the increasing incidence of referred small intracanalicular lesions, the numbers of patients with normal hearing and a normal caloric response have increased. On the other hand, severe inner ear depressions have also been encountered in ears with tumors less than 10 mm in diameter.
The contemporary standard method of imaging acoustic neurinomas, particularly for identifying small intrameatal tumors, is MRI with gadolinium contrast enhancement (meglumine gadopentetate, Magnevist) (Fig. 13.29). Contrast is absolutely necessary, since the tumor may remain invisible in noncontrast images (Fig. 13.29a). This technique allows detection of early tumor growth, with lesion diameters as small as 1–2 mm. It should be noted, however, that suspicious signals of less than 5 mm in diameter can also correspond to nontumorous lesions, such as vascularized soft tissue or arachnoid cysts (Cidlinsky et al. 1990).
Another experience is that a physical measurement in situ often gives slightly larger diameters than those calculated on the preoperative image. Cystic parts of a schwannoma, in particular, can easily be overlooked. For more precise identification of nerves and vessels and their relation to the lesion in the internal auditory canal, the turbospin sequences in which the T2-weighted phase is reformatted provide excellent clarity in the negative contours. Sophisticated three-dimensional turbospin sequences provide an even more sculptural impression of the inner ear structures and the internal auditory canal, with a fast imaging technique that is not stressful for the patient (Fig. 13.30). If MRI diagnosis is not easily available, the older technique of CT with appropriate contrast media is adequate. It also provides important information on destructive changes in the bone and labyrinth. As long as 20 years ago, using air cisternography with CT, we were able to demonstrate clearly the smallest intrameatal tumors and vascular malformations such as arterial loops (Rettinger et al. 1981). This technique has in the meantime been replaced by noninvasive MRI. The size of an acoustic neuroma is measured on the MRI to assess its extension within the internal auditory canal, but when it has expanded into the cerebellopontine angle, only the extrameatal extension is given in millimeters. Wolf et al. (1996) recently reported a volumetric technique for calculating the size of the tumors from the MRI.
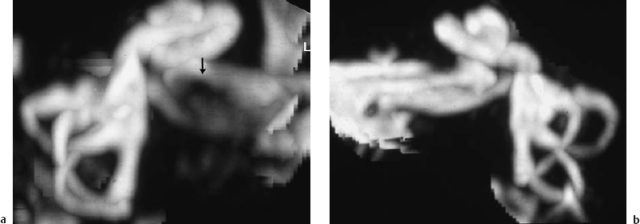
Fig. 13.30 Imaging of a small acoustic neurinoma with three-dimensional reformatting of T2-weighted magnetic resonance images (maximum intensity projection). a The affected side shows an incomplete defect in the cerebrospinal fluid contents of the internal auditory canal (arrow). b The facial and cochleovestibular nerve bundles can be identified on the healthy contralateral side.
Differential Diagnosis
If the typical growth pattern, with an intrameatal cylindrical shadow and an extrameatal bud is visible (Fig. 13.23), there is no doubt about the nature of the lesion. Facial nerve schwannomas within the internal auditory canal may imitate this pattern. Bigelow et al. (1998) reviewed 67 lipomas of the internal auditory canal reported in the literature and 17 of their own cases, showing that good differentiation is possible with MRI. Meningiomas can occur in the internal auditory canal, but their appearance is generally different, with a broader basis along the surface of the temporal bone. The signs and symptoms of tumor-like lesions, granulomas, vascular malformations, etc., may also mimic those of acoustic neurinoma, but they are very rare in this location. Cystic schwannomas may be misinterpreted as congenital cholesteatomas (epidermoid in the posterior cerebral fossa). Contrast enhancement can differentiate them.
Indications for Surgery
The current literature shows a surprising diversity of opinion regarding the indications for surgery and the technique used, and also regarding the use of radiotherapy and the appropriateness of a wait-and-watch policy. The reason for this is probably the rate of complications after complete removal of tumors from the cerebellopontine angle, which is still considerable. In view of the slow average speed of tumor growth and the tendency for the growth of this benign lesion to stagnate, this conservative weighing up of the chances is understandable. Particularly when a small intrameatal lesion has been detected in an older individual (Fig. 13.31), a wait-and-scan attitude appears justified (Tos et al. 1997). The risk of missing good chances of an excellent functional result, however, has to be considered; Shelton and Hitselberger (1991) have pointed out this dilemma. On the basis of a series of 78 conservatively managed acoustic neuromas, Bance et al. (1999) also concluded that there is a significant risk of losing useful hearing even in lesions that are not growing. In an odd controversy over this sensitive subject, two editorials in Clinical Otolaryngology reached different conclusions within one year (Cox 1993, Ramsden and Moffat 1994).
Our own recommendations are based on the observation of about 600 patients, with an abnormally high percentage of small acoustic neuromas. With the increasing application of MRI techniques, the proportion of small tumors (size A) in our patients increased over the years, while large tumors (size C) became less frequent. Since convincing long-term results are not yet available with gamma-knife therapy (Pollock et al. 1998), and since the complication rate is higher than that with our surgical methods, radiotherapy does not yet appear to be a reasonable option except in disabled and inoperable patients. Schulder et al. (1999) emphasized that when stereotactic radiotherapy fails, an unusual degree of fibrosis, scarring, and adhesions makes preservation of the facial nerve impossible during salvage surgery. Delaying or refusing surgery may also be a fair option in patients with a small tumor who do not have a strong need for preservation of hearing in the ear concerned, since a loss of time will not substantially compromise the facial nerve or the brain stem. In patients with good or excellent hearing in the affected ear, however, postponing surgery for a few months may risk marked hearing loss or even sudden total hearing loss (Fig. 13.32). In our experience, the threat of acute deterioration of hearing is quite independent of the size of the tumor. As mentioned above, the average annual hearing loss in unoperated ears was 10.8 dB in our series. The counseling we offer is based on providing the patient with full information about the functional risks with small tumors (Wolf et al. 1995). The probability of lower complication rates after removal of a small lesion emphasizes the fact that early surgery is preferable. In larger tumors that have already become embedded in the brain stem, the decision to carry out complete ablation is clear, and no time should be lost.
The planning of treatment for neurofibromatosis 2 schwannomas is a different story (see pp. 257–261). With rare exceptions, complete bilateral ablation results in total deafness in both ears, often with severe facial paralysis. In a series of 27 patients, we found that partial ablation of the schwannoma, with preservation of a strip of tumor along the facial nerve led to short-term preservation of hearing and facial motor function in all cases; tumor growth was arrested in some patients, but there was delayed recurrence with subsequent total deafness after six years in others. In younger individuals going through important phases in their professional or private lives, incomplete or subtotal tumor resection with regular monitoring of auditory evoked potentials may be an appropriate policy (Wigand et al. 1998).
The arsenal of surgical techniques available is not small. Since the translabyrinthine and transcochlear approaches destroy the inner ear and make it impracticable to use a cochlear implant, we only use the translabyrinthine access route with larger tumors in totally deaf ears. There are only two techniques with a realistic chance of preserving hearing: the lateral suboccipital (retrosigmoid) approach, and the (extended) middle cranial fossa approach. With the lateral suboccipital approach, which is preferred by neurosurgeons and by some authors in neuro-otology, there is some difficulty in visualizing the lateral portion of the internal auditory canal, but House’s classic middle fossa approach is only appropriate for small schwannomas. The extended middle fossa approach (EMFA), however, allows the removal of tumors of up to 3 cm in diameter in the cerebellopontine angle. With resection of the labyrinth and cochlea, this type of exposure can be extended even further.
Surgical Techniques
Middle Cranial Fossa Approach and Enlarged Middle Fossa Approach
This section only describes the extended middle fossa approach (EMFA), of which the pure middle fossa approach can be regarded as a simpler part. This is my favorite method for complete removal of small intrameatal acoustic neuromas (size A) as well as type B lesions (intrameatal/extrameatal, without adhesion to the brain stem), and type C lesions (with broad adhesion to the brain stem) with diameters of up to 2.5 cm. Larger schwannomas are assessed using the translabyrinthine approach (if the ear is completely deaf) or the lateral suboccipital approach (if there is still acceptable hearing); the latter approach is used in collaboration with a neurosurgeon. At my department, the House technique (purely with opening of the internal auditory canal), is not used, since the enlarged version with exposure of the cerebellopontine angle has advantages even with intrameatal lesions. The positioning of the patient is described in Chapter 2; the method is illustrated below with procedures carried out in right ears.
Step 1: skin incision. Various methods are proposed in the literature for exposing of the squamous part of the temporal bone. I prefer to form a caudally based skin flap (Fig. 13.33) containing the main trunk of the superficial temporal artery. The caudal width of the flap allows wider retraction than a simple vertical incision. To avoid colliding with the parietal and frontal branches of the facial nerve, the anterior leg of the flap should lie within the area of hair-bearing skin. The local topographical relationships were studied in a series of wet specimens. Taking a connecting line between the lateral canthus and the tragohelicine incisure (Table 13.11), the temporofrontal branches, lying close together (Fig. 13.33b), cross this line at a distance of about 3 cm from the incisure (minimum 2.0 cm and maximum 4.0 cm).
I use a high-frequency electric needle for the skin incision, and hemostasis is accomplished by bipolar cautery. Larger branches of the superficial temporal artery are ligated, but clips can also be used.
Step 2: transection of the temporalis muscle. The temporalis muscle is transected with a Y-shaped incision using the electric needle (Fig. 13.33c). For broader exposure of the zygomatic root, it may be necessary to extend the anterior branch anteriorly into the infratemporal fossa. Profuse hemorrhage from a venous plexus can occur, and this has to be stopped using cautery.
Step 3: exposure and excision of a temporal bone flap. Using a semisharp dissector, the periosteum with its attached muscle is now retracted anteriorly and posteriorly. The exposed bone area should be centered above the zygomatic root. The bone flap, about 5 × 4 cm in size, which is now excised, should also have this comparatively anterior position, since it is only in this way that it will be possible to expose the internal auditory canal easily, without encountering difficulties due to the superior semicircular canal. For bone excision I prefer to use a rotating craniotome, which can be inserted between the dura and bone after initial drilling out of a hole (Fig. 13.34). The blunt shoe of the craniotome prevents dural laceration. If a craniotome is not available, the flap can also be drilled using a sharp burr (first) and a diamond (for the most internal bone layer) with a diameter of 1 mm. The dura adherent to the bone flap is then detached from the bone by bluntly advancing a curved dissector. The excised bone can now be pulled out. Bone wax is pressed into the fresh bone wounds, and strips of Surgicel are pushed under the margins to ensure a blood-free field.
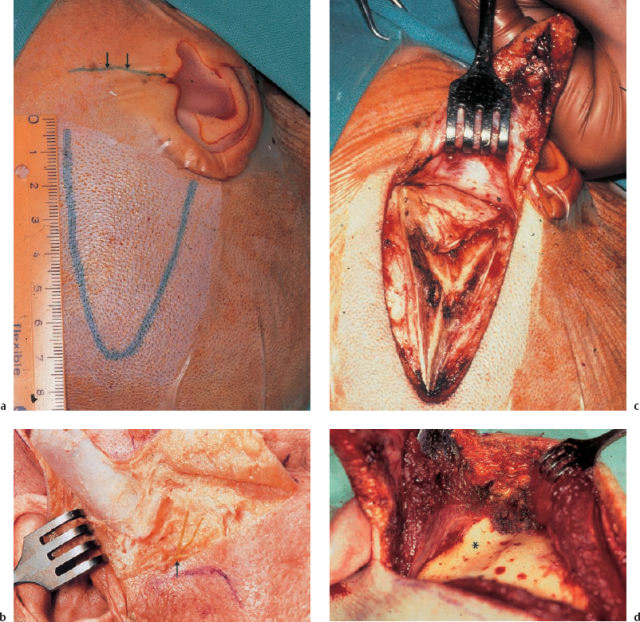
Fig. 13.33 Exposure of the temporal bone in the middle fossa approach. a Skin incision (right ear). The caudally based flap is centered over the zygomatic root (arrows). b The preauricular course of the temporal and frontal branches (arrow) of the facial nerve above the zygomatic arch (cadaver dissection, right ear). c The Y-shaped transection of the temporalis muscle. d Exposure of the zygomatic root (*) at the center of the exposed area (left ear).
|
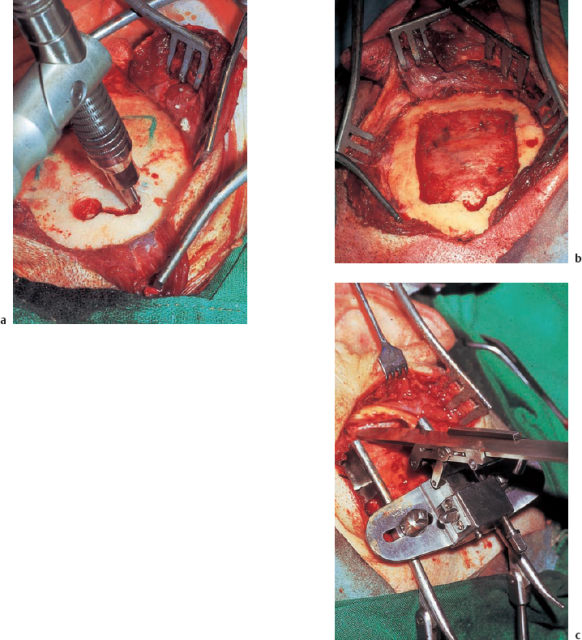
Step 4: extradural exposure of the petrous bone. After the bone flap has been lifted, the temporal dura can be bluntly detached from the skull base under direct visualization over 1–2 cm. Further extradural elevation of the temporal lobe will then require the use of a microscope and a self-retaining retractor. Different modifications are possible. I prefer Fisch’s very flexible model, which provides a stability and mobility that compensate for the disadvantage that the visual field is limited.
Variations of the temporal skull base may be encountered—e.g., broad or narrow openings into the pneumatizations, a deep or flat middle fossa, a strong or thin dural sheath, thin or thick bridging veins. Electrocautery will help arrest hemorrhage, and can also be used to allow the bulging dura to shrink, facilitating the subsequent blunt detachment of the dura from the anterior face of the temporal bone. The full length and breadth of the temporal bone extend between the sigmoid sinus and the petrous apex. Moderate to strong venous hemorrhage from bone and dura is unavoidable here for a moment, due to the rich vascularization. Cautery and packing with Surgicel (absorbable oxidized cellulose) will help. In addition, the middle meningeal artery can be cauterized and transected above the foramen spinae, which is then packed with Surgicel (Fig. 13.35). This resection, which I perform routinely in cases of larger tumors, considerably facilitates mobilization of the middle fossa dura.
Dissection of the dura also exposes both the greater and smaller petrosal nerves. Since these carry important autonomous nerve fibers, e.g. for lacrimation, care should be taken not to injure them. Traction can be avoided by sharp dissection of all the adherent dural fibers (Fig. 13.35b), and cautery has to be excluded in the vicinity of these.
Since the superior petrosal crest has to be resected in the EMFA, dissection of the petrous bone must always extend to the superior petrosal sinus. Resection of the superior petrosal sinus may be very helpful for clear exposure of larger tumors (size C). It is not difficult to identify the sinus from above and then carry out a tiny incision, packing the sinus gently with absorbable cellulose soaked in fibrin glue (Fig. 13.35c). I perform this interruption of the sinus twice—first anteriorly, and secondly close to the superior semicircular canal. The intermediate portion can then be carefully excised with the electric needle, providing ample space in the cerebellopontine angle
Step 5: supralabyrinthine bone resection for exposure of the internal auditory canal and cerebellopontine angle. The next phase consists of extended bone removal over, in front of, and behind the internal auditory meatus. This bone resection, which in fact represents a second penetration of the lateral skull base, is the essential component of the enlarged middle fossa approach. Orientation here can take advantage of two landmarks—the superior semicircular canal and the greater petrosal nerve. The plane of the superior semicircular canal is identified by cautiously flattening the arcuate eminence with a large diamond drill, until the gray line of the superior semicircular canal becomes visible (Fig. 13.36). This may lie very superficially or may be buried under deep, often well pneumatized, bone. The beginner may therefore be puzzled by small cells resembling a translucent semicircular canal. The position and direction of the axis of the gray line may also vary considerably. Some labyrinths lie more posteriorly or medially than expected; they are not always situated directly under the arcuate eminence. My advice is to mill down the bone very gently, with broad movements, and to note its consistency and color. The labyrinth is covered by very dense bone without any pneumatization, and its color is rather yellowish-green in comparison with the grayish-reddish color of the pneumatized bone (Fig. 13.36a).
After identifying the shadow of the superior semicircular canal, one can easily visualize the angle between its axis and the direction of the greater petrosal nerve. This is usually an angle of about 130°. It is easy to bisect this angle visually, and the bisection point will always indicate the axis of the internal auditory canal (Fig. 13.36b). This technique was also described by Garcia-Ibanez 1980.
Gentle caving in the direction of the internal auditory canal follows (Fig. 13.36c), with resection of the adjacent bone. Anteriorly, one proceeds medial to the cochlea into the pneumatization of the petrous apex, or into its bone marrow (Fig. 13.37).
Posteriorly, the posterior lip of the internal auditory canal is resected as far as the superior semicircular canal, and, if this lies laterally, around it to the wall of the sigmoid sinus. An opening with a very large diameter can be fraised into the petrous bone in this way. At the porus, the height of the internal auditory canal should be uncovered by at least 50%. At the fundus of the internal auditory canal, the canal can only be unroofed, due to the immediate vicinity of the cochlea and vestibulum (Fig. 13.37c). I use only diamond burrs of various sizes. The most delicate sites are the fundus, where the vertical crest and the fallopian canal have to be identified, and the anterior wall of the internal auditory canal, where the cochlea is only 1–2 mm distant (Fig. 13.38). If inadvertent injury of the cochlea or labyrinth occurs, the small fistula is immediately closed with a small piece of connective tissue, which is glued over it. No hearing loss is to be expected. Even partial labyrinthectomy need not necessarily result in deafness, as animal experiments have shown (Gjuric et al. 1992, Smouha and Inouye 1999). It may sometimes also become difficult to resect the bone from both sides of a stenotic porus, or to drill away the superior petrosal crest, which may be hidden by a tumor rising in the internal auditory canal The more precise and complete the bone resection has been, the easier and faster the tumor dissection along its outer surface will be.
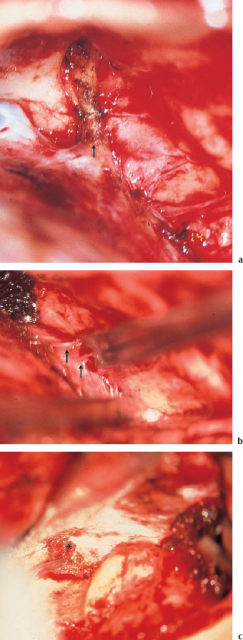
Fig. 13.35 Dissection of the petrous pyramid. a Isolation and coagulation of the middle meningeal artery (arrow) before it is transected and the foramen spinosum is closed with Oxycel. b Sharp transection of dural adhesions (arrows) over the greater petrosal nerve. c Closure of the superior petrosal sinus by packing its lumen with Oxycel (*) and fibrin glue.
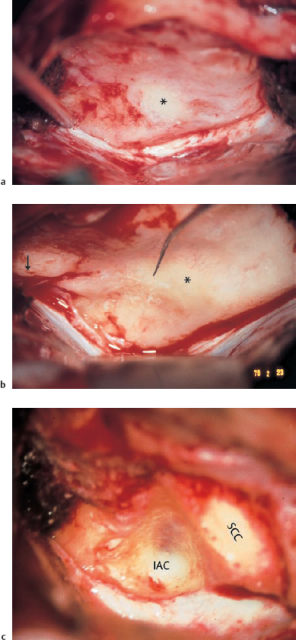
Step 6: exposure of the internal auditory canal and cerebellopontine angle. The next step exposes the internal auditory canal and the cerebellopontine angle in continuity from above. To do this, the meatal dura is incised along its axis. It is prudent to start at the fundus over the vestibular nerve compartment, and to continue medially to the porus. The first opening is made with a small sickle knife, and the axial opening is made using Fisch scissors (Fig. 13.39). A transverse incision of the cerebellar dura now follows, at a distance of about 2–3 mm parallel to the superior petrosal sinus. One should beware of the smaller vessels on the undersurface of the dura, and the scissors need to be inserted very superficially. The two incisions are then linked to form an inverted T shape by transecting the dural ring at the porus, together with an arachnoid fold, which is always present. Here and during the subsequent phases, it is often necessary to remove arachnoid vessels from the underlying tumor capsule. With gentle tearing of the arachnoid adhesions from the dura, an anterior and posterior dural flap can be developed, which can be fixed to the walls of the bone defect with a drop of fibrin glue. The exposed internal auditory canal and cerebellopontine angle are now framed by the cochlear bone complex anteriorly, the labyrinthine block posteriorly, and the superior border of the cerebellar dura inferiorly (Fig. 13.39b, c). The edge of the tentorium starts immediately posterior to and above the porus acusticus.
Step 7: mobilization of the facial nerve; resection of the superior vestibular nerve; tumor debulking. With complete distraction of both dural flaps down to the porus, the contents of the internal auditory canal become visible (Fig. 13.40). If unroofing of the canal was complete, the facial nerve (anteriorly) and the superior vestibular nerve (posteriorly) can immediately be identified easily on the upper surface of the tumor, due to their whitish color. Occasionally, the superior vestibular nerve is incorporated in the tumor; the facial nerve is never enwrapped by it, but it may be flattened and can even be split within the arachnoid sheath. The typical anterior location of the facial nerve may also be displaced by the tumor anteroinferiorly or dorsosuperiorly, making it difficult to identify. An electric stimulator should be at hand. A helpful technique is to tear the arachnoid sheath and apply saline at body temperature at the border between the two nerves, which is often marked by a small vessel accompanying the facial nerve in the axial direction. The border will then open, and the two nerves can be separated from each other using the ball-tipped dissector. In a typical operation, excision of the superior vestibular nerve close to the fundus now follows, exposing more of the superior tumor surface. This piece of nerve is usually tumor-free.
To mobilize the facial nerve from the tumor surface, I use the Zaufal 90° ball-tipped dissector, which is able to move safely in the correct cleavage plane and can always locate a vestibulofacial anastomosis (Fig. 13.40c), somewhere between the fundus and the porus. This has to be severed using the Fisch scissors. In the area of the porus, this type of anastomosis may be involved in the circular fibrous tissue, with tight adhesions to the tumor, which can mask its origin. Freeing the facial nerve is always difficult here, and often succeeds only after mobilization of the cerebellopontine angle portion of the nerve from medial to lateral, and after considerable reduction of the tumor volume by debulking.
Identification and separation of the facial nerve from the tumor is the most important task during this phase of the operation. It is greatly facilitated by consistent intracapsular debulking of the tumor. This is accomplished by incisions and morcellation, using round knives, forceps, bipolar cautery, or other instruments, depending on one’s own experience (Fig. 13.41). The Cavitron ultrasonic aspirator (CUSA) and laser are not applicable in small tumors. One should not try to isolate and remove a neuroma in one piece, except with a very small tumor with loose adhesions, because any traction can disturb the vascular supply to the cochlea and cochlear nerve. Meticulous debulking is really the secret of successfully preserving hearing and safely detaching adherent vessels. It allows the tumor capsule to be folded in without tension, making it possible to isolate and mobilize the facial and cochlear nerves precisely, as well as the vessels at the brain stem.
Step 8: cleaning of the fundus; dissection of the cochlear nerve. After marked reduction of the tumor volume within the internal auditory canal, it becomes possible to fold in the tumor capsule near the fundus. One can now palpate the entrance of the inferior vestibular nerve using the longer Zaufal hook, and gently tear its fibers away from the osseous channel. This zone is not always visible, despite meticulous uncovering of the meatal fundus. However, it is usually possible to assess complete ablation of the lateral tumor pole by checking the tumor-free fibers. This direct visualization of the fundus is one of the important features of the EMFA approach.
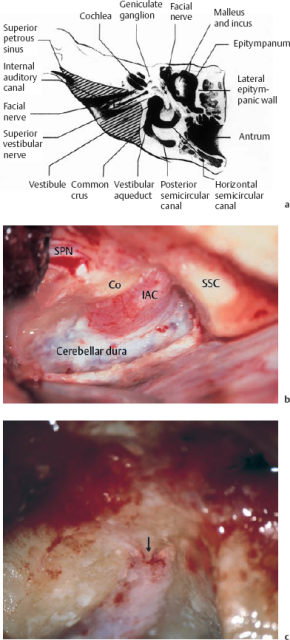
Fig. 13.37 Bone resection for the enlarged middle fossa approach. a Bone resection within the hatched areas (diagram reproduced with permission from Wigand et al. 1989). b Exposure of the meatal and cerebellar dura. c The vertical crest (Bill’s bar, arrow), separating the curved thin fallopian canal (left) from the channel of the superior vestibular nerve (right).
Co Cochlea
IAC Internal auditory canal
SPN Greater superficial petrosal nerve
SSC Superior semicircular canal
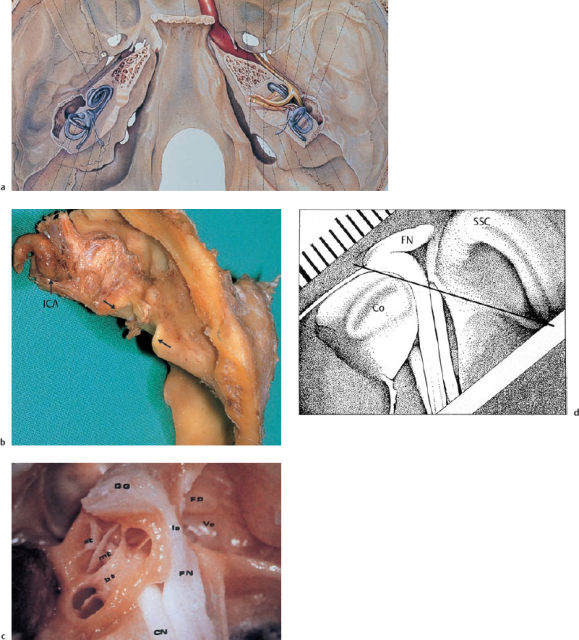
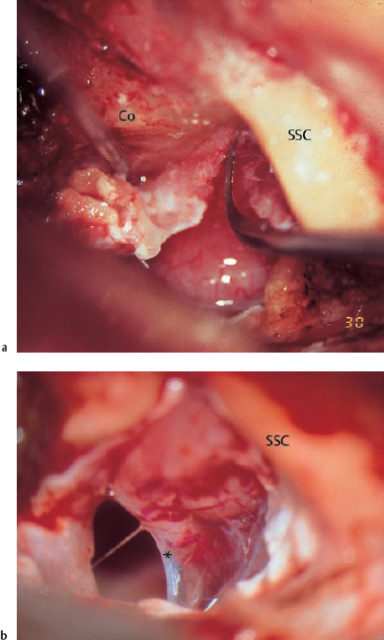
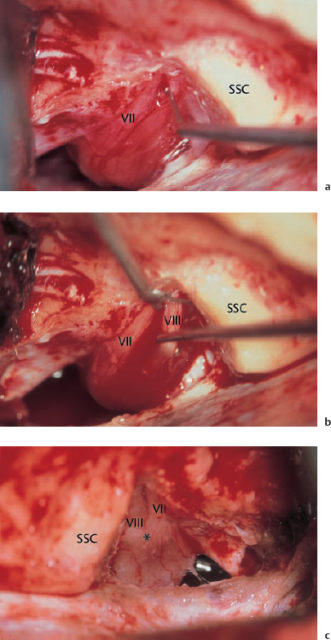
Fig. 13.40 Exposure of the tumor surface. a Mobilizing and separating the facial nerve in the internal auditory canal. b Transecting the superior vestibular nerve. c Identifying a thick intrameatal vestibulofacial anastomosis (*) (in another ear, in the left internal auditory canal).
VII Facial nerve
VIII Superior vestibular nerve
SSC Superior semicircular canal
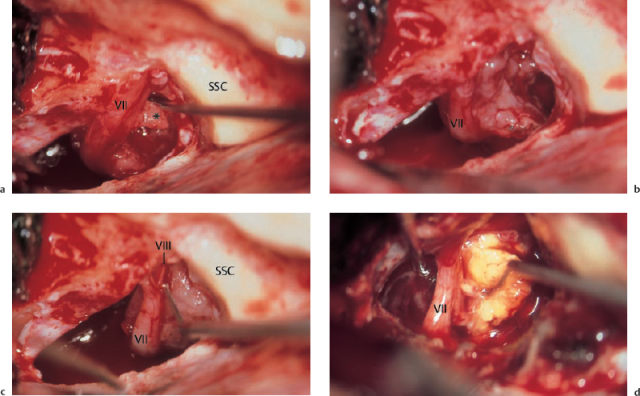
Subsequent avulsion of the shrunken tumor from the internal auditory canal should proceed, with continuous monitoring of auditory function. A depression of the evoked potentials is often noted during this phase. Manipulations are then immediately stopped at that point, and the dissection is continued at another place after a one-minute pause. Continuing the avulsion anteriorly, one will soon visualize the grayish-reddish bundle of the cochlear nerve, touching the tumor surface below the horizontal crest in the close vicinity of the inferior side of the facial nerve. Here, the cochlear nerve is a flat, broad bundle of fibers, representing the cribriform plate of the modiolus (Fig. 13.42). The aim is to separate the two nerves from the tumor, not to dissect them away from each other within the internal auditory canal! Both nerves are often supplied by the same branches of the labyrinthine artery, and any irritation of the accompanying vessels should therefore be avoided. This type of atraumatic isolation of the facial and cochlear nerves from the tumor may become extremely difficult if a tumor lobe is adherent to the lateral intrameatal portion of the facial nerve, with its artery.
After reaching the lateral pole and the anteroinferior surfaces of the tumor along their capsular plane, without any arachnoid tissue on it, further blunt dissection of the cochlear nerve is comparatively easy. Avoiding any traction from lateral to medial, one develops the cleavage plane down to the bifurcation of the cochleovestibular nerve using transverse (!) movements of the ball-tipped dissector. Whenever there is an impression of a tumor finger hiding in the cochlear nerve bundle, it is better to use a sharp hook to excise it, to ensure complete tumor removal. Complete isolation of the cochlear nerve down to the bifurcation can be achieved only after ablation of the tumor mass to almost zero. The two procedures have to alternate continuously.
Step 9: tumor removal in the cerebellopontine angle. After gross tumor removal from the internal auditory canal, with identification and separation of both the facial and the cochlear nerves, the surgeon concentrates on complete ablation of the extrameatal schwannoma. With a tumor size of 1.5–3.0 cm, the volume to be excised is larger than the opening to the cerebellopontine angle—i.e., the free space between the facial nerve and the superior semicircular canal. The dissection technique has to be adapted to this unique surgical field. Any blind manipulation or use of force have to be avoided, as they can cause life-threatening rupture of larger vessels. However, if one observes the principles of carefully mobilizing the tumor surfaces and gently pulling them into the surgical window of the petrous bone defect after continuous debulking, it is possible to control these large tumors with a high degree of security, and without greater risks than those inherent in all the other approaches.
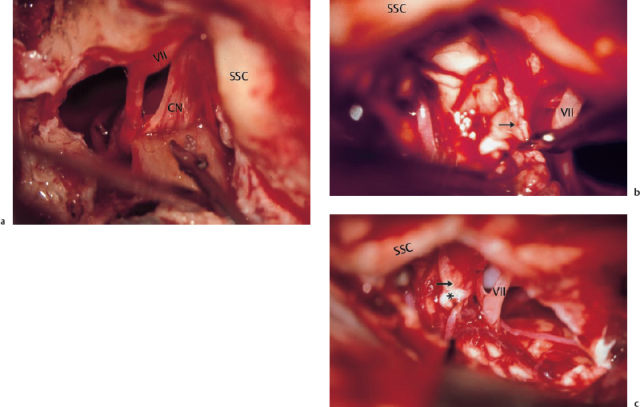
Fig. 13.42 Dissection of the cochlear nerve and final tumor excision. a The medial tumor pole is dissected from the flat-splayed cochlear nerve by spreading open scissors or using a ball-tipped probe in a transverse direction (right ear). b The proximal vestibular nerve stump is transected above its bifurcation (arrow) with the cochlear nerve (left ear). c The tumor-free bifurcation of the vestibulocochlear nerve (*) (left ear).
VII Facial nerve
CN Cochlear nerve
SSC Superior semicircular canal
Separating vessels from the tumor surface is greatly facilitated by consistent dissection of the arachnoid sheath away from the thin tumor capsule. It is easy for a beginner to misinterpret the sheath as a nice, stable surface. But difficulties arise as soon as one tries to dissect away an overlying vessel that is tightly adherent to the sheath. One should therefore identify the arachnoid web from the beginning of the dissection at the porus, and remove it consistently.
Debulking the tumor mass can be easy in soft, poorly vascularized schwannomas, but it may be tricky with hard tissue that has a rich vascular supply. Cystic formations are also difficult to handle if they have to be pulled into the surgical window from a laterocaudal position. If scissors, round knives, or forceps are not successful in debulking a larger, bleeding schwannoma, one can use bipolar coagulation for tissue destruction. Rinsing with saline will prevent tight adhesion of the burned tissue to the pincers, and will also protect the facial and cochlear nerves from heat trauma. Intermittent hemorrhage from intratumoral vessels that does not respond to bipolar cautery can be arrested by temporary compression with Gelfoam. Bleeding from innominate vessels in the tumor capsule can be arrested using bipolar cautery. One should always ensure that the surgical field is bloodless, and it is better to interrupt the operation for a minute or two to allow hemostasis than to continue the resection with uncontrolled hemorrhage. If an open vessel ever relapses in a position that cannot be reached with the bipolar cautery, one should try to cover it with a piece of Gelfoam. If it is a larger vessel, Gelfoam soaked in fibrin glue used for local tamponade will save the situation.
In larger tumors, the debulking procedure with subsequent resection of the tumor shell may be difficult in the caudal portions of the tumor. The resection of the superior petrosal sinus mentioned above may then become an essential aid. If it is unexpectedly not possible to visualize a dorsocaudal tumor lobe, resection of the labyrinthine block may help enlarge the surgical field. Immediate closure of the opened semicircular canals with fibrin glue and fascial tissue may even then be able to preserve hearing function.
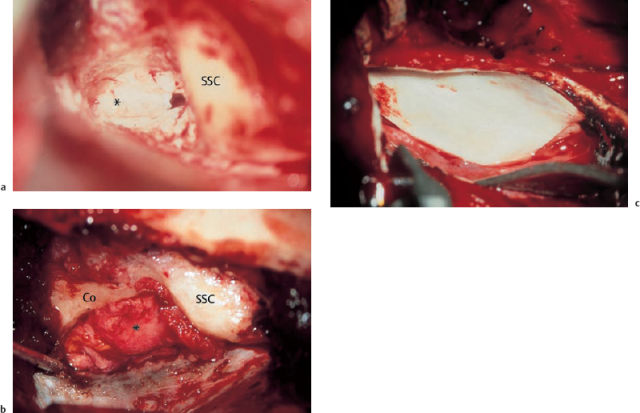
Fig. 13.43 Closure of the dural and pyramidal bone defect. a Replacement of the dural flaps (*) over the internal auditory canal and cerebellopontine angle. b Closure of the bone defect with free periosteal and fascial grafts (*). c Lining of the floor of the middle cranial fossa with homologous dura (Tutoplast).
Co Cochlea
SSC Superior semicircular canal
After approaching the most medial portions of the tumor at the brain stem, one should try to identify and mobilize the intermedius nerve together with the trunk of the facial nerve. Destruction of the nerve results in persistent dryness in the ipsilateral eye, but the gustatory deficits caused are often compensated for by the lingual nerve supply.
Dissection of the medial pole of larger schwannomas is another delicate step. While freeing this pole from the adjacent parts of the cerebellum or from the flocculus does not present any particular problems, skill and patience are required if it is fixed to the brain stem, as larger vessels may be tightly adherent. Arteries can be dissected fairly easily, but veins may rupture even with gentle movements. A particularly long pair of pincers is essential for bipolar coagulation, since the distance from the temporal squama to the brain stem may be as much as 16 cm. In about 1.5% of our cases, we left small tumor remnants on a vessel because removing them appeared to be too dangerous. Such fragments can be devitalized by bipolar coagulation. It was usually possible to identify the proximal stump of the vestibular nerve and transect it above its entrance into the cochleovestibular bifurcation. In other cases, dissection of the medial tumor pole was followed by spontaneous rupture of the last fibers. Ablation of the schwannoma, which is now complete, should be very carefully checked in all cases, and a search should be made under rinsing saline for any bleeding vessels that remain. Precise hemostasis using bipolar coagulation, or by gluing on small pieces of Gelfoam, is of the utmost importance.
Step 10: closure of the dural and petrous bone defects; prevention of extradural hematoma; reconstruction of the skull. Hemostasis of the cerebellopontine angle and internal auditory canal should be observed for more than five minutes before reconstruction of the skull base and definitive wound closure. I use this time span to harvest free grafts. Meanwhile, a large piece of Gelfoam is temporarily placed over the cerebellopontine angle to protect it from external hemorrhage. A large piece of muscle-periosteum is obtained from the superior mastoid area, and a piece of subcutaneous fascia can be excisedat the same place. Both of these grafts are harvested through the existing skin incision, without any further extension.
The opening in the petrous bone is closed in three or four layers. First, the dural flaps are replaced (Fig. 13.43) and glued together, which almost closes the dural defect. A second layer consists of muscle-periosteum, which is placed in the bone defect, overlapping the edges by a few millimeters. Careful filling out of the anterior, deeper part of the defect is necessary to avoid cerebrospinal fluid leakage via the opened pneumatization. A thinner layer consisting of fascia is then laid over the lateral parts of the surgical field for watertight closure of the middle ear cells. I like to place a fourth layer consisting of homologous dura (Tutoplast) to ensure definitive separation of the temporal dura from the petrous bone, which may prevent local invagination due to scar formation. Retraction of the temporal lobe is then released.
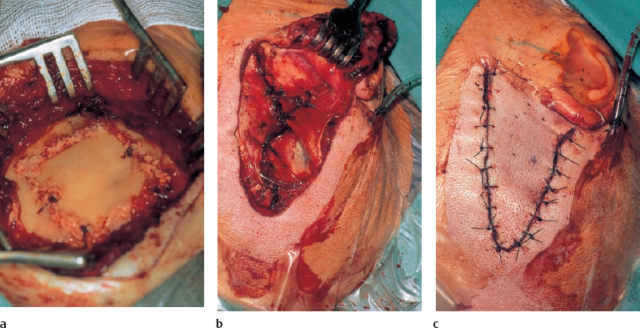
Fig. 13.44 Closure of the cranial wound. a Osteosynthesis of the bone flap by suturing and cementing with bone dust and fibrin glue. b Reconstruction of the temporalis muscle over a Redon drain. c Skin suturing.
It is not necessary to suture the temporal dura to the skull bone. Instead, we fill the empty spaces between the bone and dura using large pieces of Gelfoam soaked in saline and coated with fibrin glue. This is consistent enough to withstand possible slow venous hemorrhage, and will be resorbed during the time required for reexpansion of the retracted temporal lobe. Using this method, we have never observed a postoperative extradural hematoma. A large sheet of Gelfoam is also placed below the temporal bone flap that was stored in an antibiotic solution during the intracranial surgery, which is now fixed back in the window of the squamous part of the temporal bone using four Vicryl sutures, led through drilled holes (Fig. 13.44). The four clefts are additionally filled with bone paste with fibrin glue. This technique ensures fast osteosynthesis within a few weeks, as has been demonstrated by CT imaging.
Final wound closure consists of resuturing the temporalis muscle over a Redon drain applied to the zygomatic root, and double-layer skin suturing. It might be thought that this type of solid, definitive reconstruction of the skull base and calvaria over the surgical field in the cerebellopontine angle is questionable, in view of the possible need for an emergency revision procedure. However, we believe this type of immediate definitive reconstruction is justified, since in five hundred cases we have only had to reopen the cerebellopontine angle twice owing to a suspicion of intracranial hemorrhage, and we were able to reexpose it within about two minutes. However, it must be emphasized that after removal of an acoustic neuroma, the patient needs to be watched and monitored very carefully for 48 hours in an intensive-care unit very close to the operating room.
Postoperative course. After the usual EMFA operation, lasting four to six hours, the patient is extubated without delay, and observed for 48 hours in an intensive-care unit to monitor consciousness, vital functions, and pupillary reactions. If any alarming symptoms appear, such as severe headache, unconsciousness, or mydriasis, immediate reopening of the middle fossa is indicated (acute severe onset), or a CT scan is carried out (slow development). Both measures are sufficient for detection and immediate removal of an intracranial hematoma—the most dangerous early complication.
The urinary catheter is removed on the first day after surgery, and the patient can sit up and have a wash, with assistance. Vertigo is frequent, and can be treated by intravenous injections of 100 mg sulpiride; vomiting does not always occur.
Antibiotics with a high rate of cerebrospinal fluid diffusion are given for four to six days, or even longer if postoperative cerebrospial fluid leakage occurs. Perioperative corticoid administration is recommended. Lumbar drainage has only been required exceptionally in over 700 cases.
Translabyrinthine Approach
In the same period when House developed the middle fossa approach for removal of intrameatal acoustic neuromas (House 1961), he also worked out the translabyrinthine approach for lesions extending into the cerebellopontine angle (House 1964a, 1964b). In comparison with the neurosurgical suboccipital approaches, the translabyrinthine approach has the advantage of allowing direct broad exposure of the cerebellopontine angle without any compression of the cerebellum, and with excellent preservation of facial nerve function. However, it also involves the disadvantage of destroying the inner ear. Since many patients with mediumsized or larger acoustic neuromas still have useful or acceptable hearing, I rejected this technique very early on, and today only regard it as indicated when there are larger tumors in completely deaf ears. The approach is particularly suitable for intralabyrinthine schwannomas.
Step 1: postauricular mastoidectomy; sculpturing the fallopian canal, posterior semicircular canal, and sigmoid sinus. The posterior skin incision should be at a distance of 1–2 cm to the auricular fold. After mobilization of the skin on both sides, an anteriorly pedicled rectangular periosteal flap is circumcised, lifted, and retracted to the posterior circumference of the bony external ear canal. Henle’s spine should become visible. The exposed mastoid cortex is then resected using a large chisel or cutting burr, and the pneumatization around the antrum is removed from the aditus as far as the mastoid tip. The fallopian canal is then identified below the lateral semicircular canal, and is skeletonized using a diamond burr down to the stylomastoid foramen. The posterior semicircular canal is then identified in its typical location, lying in a perpendicular direction to the lateral semicircular canal and just behind it, with the two canals forming an inverted T shape. I prefer to expose the cerebellar dura in front of and behind the sigmoid sinus next, before starting the labyrinthectomy. A small, thin shell of bone can be left over the sigmoid sinus to protect its dural wall. When all the adjacent bone has been drilled away (with a diamond burr), this island of bone over the sinus serves as a protective shield when it becomes necessary to depress the sinus backward to obtain wider exposure of the cerebellopontine angle. Exposure of the cerebellar dura continues down to the jugular bulb and upward to the middle fossa dura (Fig. 13.45).
Step 2: labyrinthectomy and exposure of the internal auditory canal
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree