he goal of transconjunctival transciliary filtration (TCTCF) is to cause the posterior chamber fluid to drain under the subconjunctival space, without the need for dissection of any kind. An activated Fugo blade is directed through the conjunctiva, the tenon capsule, the sclera posterior to the scleral spur, and the pars plicata of the ciliary body to open the lateral wall of the posterior chamber. The track passes across the suprachoroidal space. The volume of the posterior chamber is merely one-fifth of the anterior chamber fluid. The inflow and outflow channels of aqueous are separated by the iris.
he goal of transconjunctival transciliary filtration (TCTCF) is to cause the posterior chamber fluid to drain under the subconjunctival space, without the need for dissection of any kind. An activated Fugo blade is directed through the conjunctiva, the tenon capsule, the sclera posterior to the scleral spur, and the pars plicata of the ciliary body to open the lateral wall of the posterior chamber. The track passes across the suprachoroidal space. The volume of the posterior chamber is merely one-fifth of the anterior chamber fluid. The inflow and outflow channels of aqueous are separated by the iris.
TCTCF is suitable for all patients in whom the posterior chamber anatomy is normal. This technique is eminently suitable in cases of phacomorphic glaucoma, either alone or immediately before lens surgery, acute congestive glaucoma, some cases of malignant glaucoma, and neovascular glaucoma. In eyes with scarred perilimbal conjunctiva resulting from earlier operations, you may find a healthy spot 1 or even 2 mm away from the limbus from where TCTCF can be performed.
TECHNIQUE
Anesthesia
Adequate local anesthesia includes a facial block and a choice between retrobulbar, peribulbar, and subconjunctival infiltration anesthesia. Mere surface anesthesia is not enough. After the injection, compress the eyeball for 5 minutes using a rubber ball to disperse the anesthetic agent in all directions. The patient will not feel pain during the procedure.
Surgical Technique
- Separate the eyelids with a speculum. Make sure that the speculum is not stretching the fornix conjunctiva too much, as this may prevent proper execution of important surgical steps. The conjunctiva above the upper limbus or wherever surgery is to be done should be loose and mobile.
- Visualize the posterior limit of the bluish surgical limbal area by pressing or stretching the conjunctiva. Mark it by pressing with the blunt side of a 1-mm diamond blade. Use the same blade to measure and mark 1 mm above the first mark. Now press the blunt side of a razor fragment on the sclera, parallel to the limbus. Maintain the pressure until a sufficient indentation (about 1 mm) is made on the conjunctiva and the sclera; the indentation acts as a guide when the conjunctiva is glided toward the limbus during the next step of surgery. A 1-mm diamond knife is the best measuring instrument. Performing TCTCF 1 mm behind the limbus site is not a hard-and-fast rule, as variations are possible; for example, a variation is to be expected when the corneal diameter is smaller or larger than normal.
- Use a blunt-edged nonmetallic tool to glide a fold of conjunctiva from the proximal to the limbal side, up to the marked indentation on the sclera. There, press the conjunctiva toward the sclera so that it cannot slip from underneath the pressing tool. An intentionally blunted diamond or sapphire knife is eminently suitable for this purpose, because it is nonmetallic.
- Use a straight 300-µm naked ablation tip to open the posterior chamber. The naked tip produces a scleral-ciliary track that is wider than the blade itself. This helps you to be aware to some extent of what is happening under the tip. The tip remains functional as long the posterior chamber fluid has not been reached.
- Place the naked 300-µm tip in direct proximity to the blunt diamond tool that controls the retracted conjunctiva. The Fugo blade is set at the highest energy level. The posterior chamber is approachable almost directly under the scleral indentation.
- From where the entry point has been chosen (1 mm from the limbus), direct the ablating tip at about 70 to 90 degrees.
- Activate the Fugo blade and insert it in small steps, taking it deeper with each step. After each little step, visualize the ablated pit for the depth.
- When the sclera is ablated, the black ciliary body appears. Ablate the ciliary body slowly, in many steps. One last step takes the blade into the posterior chamber, and the fluid begins to drain. The aqueous comes out, sometimes mixed with black-pigmented specks. The aqueous fills the scleral–uveal track and makes any further ablation by the naked tip ineffective. (The track can be made in one complete step that takes the tip to the posterior chamber; however, it is wise to take these steps slowly.) When fluid surrounds the naked Fugo blade tip, its ablating power decreases automatically. The crystalline lens is in no danger because it is, in practical terms, located far from the surgical site.
- Confirm the completion of transciliary filtration track formation by pushing air through it using a 24-gauge cannula. This can be achieved only if the conjunctiva retracting device is still in place after the aqueous has drained. Air flows into the anterior chamber after traveling under the iris and through the pupil.
- Rinse the posterior chamber with saline if blood appears during the procedure.
- If you use a 300-µm tip shielded with nonstick material, you will feel the resistance of the track wall as it is created. The ablation process is completely blocked from view as the tip fills the track. This not much of a problem, however, since the tip is repeatedly removed and the track observed as the track is being made.
- Allow the conjunctiva to retract.
- In selected cases, inject a drop of mitomycin 0.02% to 0.05% around the scleral opening with a thin cannula. The blebs resulting from filtration in these cases appear somewhat avascular. Mitomycin may help to prevent scarring in high-risk cases (Fig. 27.1).
- The conjunctival hole resulting from the procedure is situated about 2 to 3 mm away from the scleral opening. It is secured with a suture or with fibrin glue. It can also be left alone to close naturally in a day or two.
- Use a blunt-edged nonmetallic tool to glide a fold of conjunctiva from the proximal to the limbal side, up to the marked indentation on the sclera. There, press the conjunctiva toward the sclera so that it cannot slip from underneath the pressing tool. An intentionally blunted diamond or sapphire knife is eminently suitable for this purpose, because it is nonmetallic.
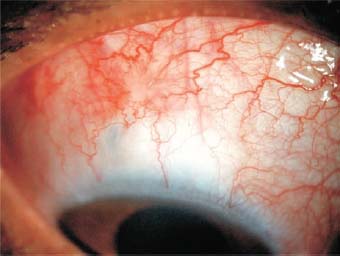
Figure 27.1. Mitomycin use causes avascularity in the area where it was applied during surgery.
In all cases with a shallow anterior chamber, the depth is seen to increase right after the posterior chamber begins to drain. In neovascular glaucoma cases, blood may begin to ooze from the angle of the anterior chamber. Nothing needs to be done; chances are that the blood in the anterior chamber will not compromise the ciliary body opening because the blood will collect in the lower part of the anterior chamber, while the filtering track is superior and behind the iris.
Postoperative Management
Worn off, remove the eye patch. Instruct the patient to instill topical steroid-antiobiotic drops six to eight times during the day for 3 weeks. The patient should gradually decrease the drops over the next month. Use an antibiotic–steroid ointment at bedtime.
Examine the eye for the depth of the anterior chamber and the condition of the filtration area and the surrounding bleb. Examine the eye every week for a month. Beyond that, follow-up is determined by the individual needs of the patient.
The filtration area is prone to all the normal reactions of the subconjunctival area, the tenon capsule, and the scleral and ciliary body tissues surrounding the filtration track. However, the TCTCF technique causes very little tissue reaction.
CASE STUDIES
The utility of TCTCF is illustrated by the following diverse examples.
Case 1: Congestive Glaucoma
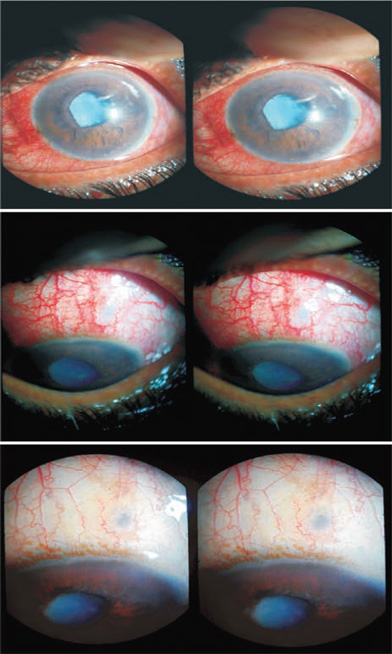
A 52-year-old patient had acute congestive glaucoma. The intra-ocular pressure (IOP) was above 50 mm Hg. The anterior chamber was very shallow and the cornea was edematous. The IOP could not be reduced with local or systemic medication. Surgery was recommended and performed as described below (Fig. 27.2).
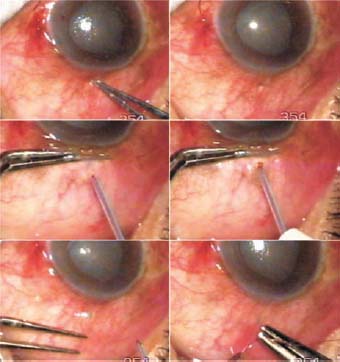
Figure 27.2. TCTCF in a case of acute congestive glaucoma (Case 1). A scleral depression is made with the tip of a forceps. The conjunctiva is pulled down beyond the depression using a plain forceps. A TCTCF track is made with a 300-µm tip. A bleb arises. The conjunctival hole is closed with a suture.
The tip of a forceps was pressed at the point of intended entry through the sclera. The pressure created a pit in the sclera that was visible through the conjunctiva. The conjunctiva was then slid down with a plane forceps. A 300- µm Fugo blade tip shielded with nonstick material was used to effect TCTCF. During the process of making the filtration track, the tip was not allowed to touch the metallic forceps. Once the fluid drainage was started, the surgery was complete. The conjunctival hole was closed with a suture. Postoperative recovery was uneventful (Fig. 27.3).
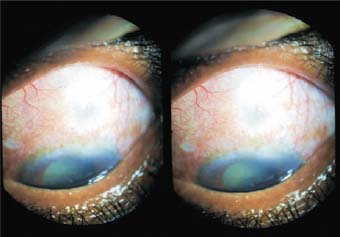
Figure 27.3. The same patient (Case 1) 15 days after surgery. The bleb is well formed and the anterior chamber is now normal.
Cases 2 and 3: Phacomorphic Glaucoma
In case 2, a 65-year-old patient had had acute phacomorphic glaucoma for 1 week. The IOP was above 50 mm Hg. The patient’s pain was a major concern, as was the restoration of vision. Two possible avenues existed for treatment: either one-stage surgery for glaucoma and cataract, or control of the IOP as the first stage. We chose the latter. TCTCF surgery was performed as usual. The operation was considered satisfactory, as fluid mixed with some pigment drained out. The movement of the fluid was confirmed by putting a drop of trypan blue on the conjunctival hole (Fig. 27.4). One suture was applied to it. The recovery was uneventful (Fig. 27.5).
In a very similar case (Case 3), photographs taken before surgery and 3 days and 40 days after surgery show profound deepening of the anterior chamber. This could not happen without considerable shrinkage in the volume of the intumescent cataract (Fig. 27.6).
This case brings up the question of the relationship between phacomorphic glaucoma and intumescence of the cataract. The attack of glaucoma no doubt results from angle closure. As the IOP rises, more aqueous enters the cataractous lens by hydrostatic pressure and increases its volume. This further increases the severity of obstruction, and thus begins a vicious circle. When the IOP falls, the fluid moves out of the cataract and the anterior chamber depth increases.
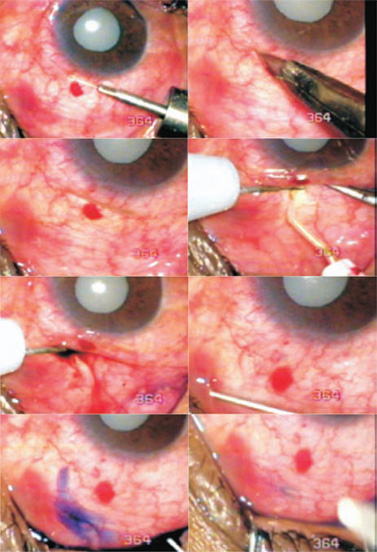
Figure 27.4. A case of phacomorphic glaucoma (Case 2). Successful TCTCF brings out some pigment and blood. Trypan blue dropped in the conjunctival opening flows away with the aqueous stream. The opening is closed with a suture.
Case 4: Phthisis Bulbi and Staphylomas
A 20-year-old female patient had phthisis bulbi in one eye and extensive ciliary staphylomas, a deep anterior chamber, occlusion pupillae, and an IOP of 32 mm Hg in the second eye. Ultrasounic B mode (UBM) showed a voluminous space behind the iris produced as a result of thinning and stretching of the ciliary body. The sclera was nearly nonexistent in the staphyloma areas. Surprisingly, the patient had good all-around light projection. Any kind of standard filtration procedure was unthinkable. The entire anatomy of the anterior segment was disturbed, which made operations like trabeculectomy, nonperforating filtration, viscocanalostomy, and glaucoma valve procedures nearly impossible to perform. Over the preceding 2 years, the patient’s search for control of her IOP was fruitless because the pressure could not be controlled with medication and surgery was never suggested.
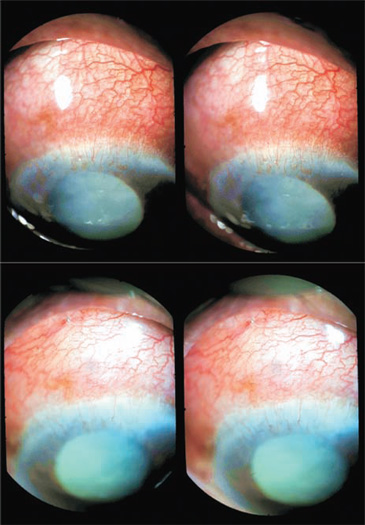
Figure 27.5. The same patient (Case 2), before and 3 hours after surgery, in three-dimensional imaging. The depth of the anterior chamber has considerably improved.
Judging from our earlier experience with three similar cases, we believed that TCTCF could resolve the patient’s glaucoma, and in fact the surgery was very simple. The freely mobile conjunctiva was glided down over the staphyloma at the 12 o’clock position. A 300- µm Fugo blade tip was used. The first light touch crossed the conjunctiva; the second light touched ablated the uveal tissue, and that released the aqueous fluid. The conjunctiva was slid back to its original position and a suture was applied to the conjunctival opening. The surgery produced a delicate extensive bleb over the surface of the sclera. The chances for the survival of this bleb are high, since the surgery was atraumatic and there were no tenon capsule or sclera to react to surgery and close the track (Fig. 27.7).
Figure 27.6. Phacomorphic glaucoma (Case 3) before surgery and 4 days and 40 days after surgery. The depth of the anterior chamber has increased. The congestion is gone.
Blebs after Transconjunctival Transciliary Filtration
The most striking thing about the blebs in the first 1 or 2 days after surgery is the remarkable similarity of their appearance. Most cases look alike in the first few days. The blebs are well formed and spread out diffusely. The most raised area of the conjunctiva lies over the scleral end of the filtration track. From this central point, the aqueous fluid drains diffusely. The appearance changes over time. In the coming days, the bleb becomes flatter as the filtration volume settles down according to the facility/resistance from the subconjunctival tissues, including the lymphatics (Fig. 27.8). They might flatten out yet remain effective (Fig. 27.9). The flattening appears quite natural since no obstruction from tissue reaction is formed and fluid is carried away by the lymphatics. It is not surprising to see a barely visible but fully functional bleb in an eye that had an IOP above 50 mm Hg before surgery.
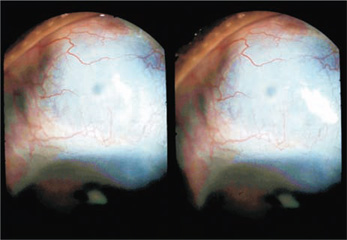
Figure 27.7. TCTCF in a case of ciliary staphyloma (Case 4), 1 day after surgery. A totally transparent conjunctiva (visible in three dimensions) is lifted by the outward flow of the aqueous.
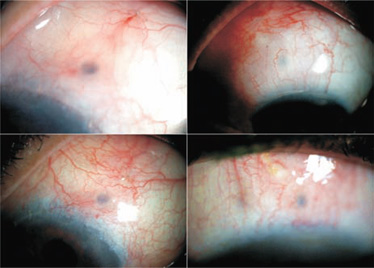
Figure 27.8. Examples of blebs that are less than 1 week old. The blebs are shallow and extensive.
Postoperative Management
The dressing is removed as soon as the effect of local anesthesia has worn off. The usual finding is a normal anterior chamber and a raised extensive bleb. The patient goes home with steroid antibiotic drops to be instilled seven to eight times a day and an antibiotic– steroid ointment for bedtime.
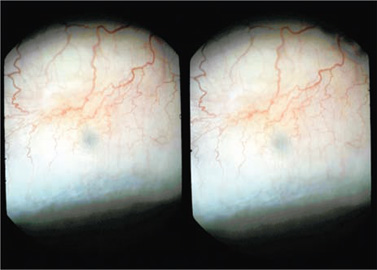
Figure 27.9. A TCTCF bleb, 2 months after surgery, in three dimensions. The bleb is barely raised from the surface, but is functional. The conjunctiva show a normal transparent gossamerlike appearance. The IOP is 15 mm Hg, down from 51 mm Hg before surgery.
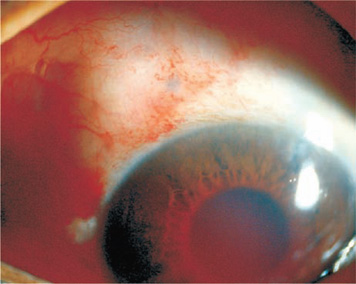
Figure 27.10. A postoperative blood ooze that originated either from the area of filtration or from the angle of the anterior chamber.
An occasional adverse finding is a blood ooze (Fig. 27.10). This is of no great concern, however, as the blood settles and absorbs spontaneously and does not interfere with the filtration track, which is far removed from the site, on the back of the iris.
Summary
Transconjunctival transciliary filtration has some merit in the treatment of glaucoma. A conjunctival flap is unnecessary, as is interference with the tenon capsule and sclera. The route of tissue ablation to reach the posterior chamber is straight and does not damage adjacent tissues. This technique then becomes the minimally traumatic way to perform major filtration surgery. The key is to enter the sclera at the right point and to move in the right direction toward the posterior chamber. If suitable precautions are taken, the crystalline lens is in no danger. The Fugo blade tip becomes inactivated when it reaches the fluid in the posterior chamber.
In aphakic and pseudophakic pupillary block, TCTCF is very effective. UBM findings provide useful information for better placement and direction of the Fugo blade ablating tip. Repeat operation is easy and may be performed at the original site or at an adjacent site. Going through the tenon capsule as in TCTCF minimizes the chances of a tenon cyst formation.
In the future, a highly flexible miniature fiberoptic light and camera may be mounted on the Fugo blade tip to provide a high-quality high-magnification view of the ablation pit. Also, an electronic echo function may be incorporated in the ablating tip to show the precise shortest path in which the tip should be moved to cross the ciliary tissue to reach the fluid chamber.
Suggested Reading
Garg A, Fine H, Pallikaris IG, Chang DF, Tsuneoka H, Bovet JJ, eds. Innovative techniques in ophthalmology. New Delhi, India: Jaypee, 2006:414.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree