CHAPTER 75 Tracheobronchial Endoscopy
Airway Anatomy and Nomenclature
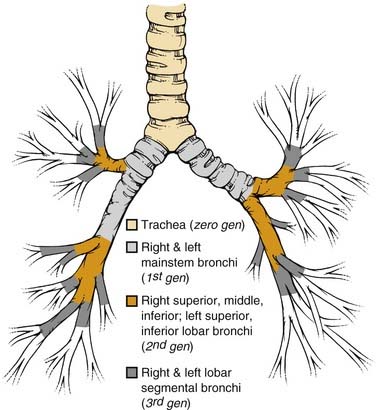
The basic pulmonary function of gas exchange occurs at the level of the alveolus, located in the distal acinus beyond respiratory bronchioles, but for the purpose of the bronchoscopist, it is more important to recognize the more proximal airway divisions. Distal to the trachea and the mainstem bronchi, the lobar bronchus defines the division of the lobes, and the segmental bronchi the pulmonary lobules (Fig. 75-1). There are thus three lobes in the right lung, with normally ten segmental lobules, although there may be some anatomic variants. In the left lung, the left upper lobar segments and the lingular subsegments come off the same left upper lobe bronchial division, which also explains why resection of the left upper lobe often requires inclusion of the lingular, and vice versa. Because of the anatomic divisions, the left lung is often divided into nine segmental lobules, although here again there are anomalies, and the bronchoscopist should be familiar with the variations. Because of the location of the heart, the right lung usually accounts for 55% to 60% of the total lung parenchyma, and the left lung the smaller remainder. This information becomes important in estimations of residual pulmonary function after a planned lobar resection or pneumonectomy.
An additional source of frequent confusion in the naming of the segmental and subsegmental bronchi has to do with the frequent but incorrect interchange of the terms generation and order of the bronchial segments. Standard nomenclature of the human airways denotes the trachea as the zero-generation airway, with each of the right and left mainstem bronchi being the first generation, lobar bronchi the second generation, lobar segmental bronchi the third generation, and so forth. Conversely, order of airway segment refers to the retrograde counting from the “lobular bronchiole,” which is the first airway segment with a diameter of less than 0.7 mm. Hence, more central airways of a larger dimension actually have a higher order.1,2 However, given the uneven segmentation and subsequent diminution of airway calibers, there is far less uniformity of division of airway in the different lobes, and lobules narrow down to 0.7 mm. Hence, the standard textbook description of the 0.7-mm lobular bronchiole located in the 10th- to 14th-generation bronchi does not match the bronchoscopic findings of airways navigable by a bronchoscope significantly wider than 0.7 mm beyond the number of branchings described previously.2–4 In summary, it is best to use the term generation in the description of sequential airway branching, starting from the trachea. Dr. Shigeto Ikeda,5 regarded as the founder of modern fiberoptic bronchoscopy, also developed a detailed system of naming segmental and subsegmental airways branchings, but that topic is beyond the scope of this chapter.
Indications and Preparations for Tracheobronchoscopy
Indications
Indications for tracheobronchoscopy include the evaluation of acute or chronic respiratory symptoms such as hemoptysis, nonresolving and worsening cough, and acute or worsening subacute dyspnea that may be accompanied by wheezing or stridor, pleurisy, chest pain, fever, or other symptoms suggestive of a pulmonary process (Box 75-1). Symptoms may be accompanied by radiographic abnormalities suggesting an endobronchial lesion, extrinsic compression of the airways by a mediastinal or lung mass, or presence of an infiltrate. Patients are often also referred for evaluation of asymptomatic lung or mediastinal masses and nonresolving parenchymal lung infiltrates. Pleural effusions usually lead to evaluation of the pleural fluid; however, effusions most often have an underlying parenchymal pulmonary cause, and a nonresolving effusion without an established cause after thoracentesis or thoracoscopy should prompt an examination for endobronchial obstruction. Bronchoscopy is helpful in assessing the placement of endotracheal tubes, especially in difficult intubation cases or when double-lumen tubes are required. A flexible fiberoptic bronchoscope (FOB) can be used to guide placement of an endotracheal (ET) tube in a difficult airway; in such cases, the ET tube is prepositioned and advanced over the bronchoscope. An FOB can also be used to guide percutaneous tracheostomy. Indications for tracheobronchoscopy in children include suspected foreign body, respiratory distress secondary to tracheomalacia or bronchomalacia, and evaluation of other congenital anomalies. Depending on the findings on radiography or during a diagnostic bronchoscopy, therapeutic interventions may be performed with an FOB to maintain airway patency and improve gas exchange.6,7
Box 75-1 Indications for Tracheobronchoscopy
Diagnostic Evaluation
Sedation and Airway Management
For patients undergoing bronchoscopy under conscious sedation, topical anesthetics are commonly used to reduce the amount of systemic sedative-narcotics needed to suppress cough, and also to diminish local discomfort in the nasopharynx during the passage of the bronchoscope. Lidocaine and its derivatives are most commonly used and applied topically as a gel to the nasal passage or as a spray to the posterior oropharynx. Solutions with concentrations between 1% and 4% are available, but the operator must pay attention to the total dosage applied because systemic lidocaine toxicities, including inadvertent deaths in healthy research subjects, have been reported when more than 500 to 1000 mg is delivered topically in a single session.8 Therefore, except for spraying to the posterior oropharynx, we limit the use of topical lidocaine injected via the operating channel of the bronchoscope onto the vocal cords and airway mucosa to less than 3 to 4 mg/kg total dosage (1% lidocaine has 10 mg of lidocaine per l mL solution). Cocaine is also a very effective topical anesthetic that has the advantage of vasoconstricting the nasal mucosa membrane; it is, however, not generally available for use in the average endoscopy suite.
Rigid Bronchoscopy
Before insertion of the rigid bronchoscope, either alone or with a telescope lens through it to provide a better distal view, the patient must be examined for neck stability and for any loose teeth or dentures. A shoulder roll may give room for added extension of the neck. With or without a tooth guard to protect the upper teeth, the operator’s left thumb is placed over the upper teeth and the index finger scissors open to lift up the lower teeth of the relaxed jaw. The bronchoscope with the bevel up is then directed midline and almost perpendicularly toward the hypopharynx until the uvula is passed. The operator thereafter slowly levels the bronchoscope angle toward the horizontal, seeking out the epiglottis while lifting the base of the tongue. When the vocal cords are clearly visualized, the bronchoscope is rotated 90 degrees such that the beveled edge can enter the trachea along the length of the vocal cord, thereby limiting trauma to the cords. Once the tip of the rigid bronchoscope is clearly in the trachea, it is rotated back to its starting position with the bevel up. The operator’s left hand and finger positions are maintained to protect the teeth, although it is also necessary to occasionally use the left hand to provide a better seal around the cuffless rigid bronchoscope at the level of the cricoid and thyroid cartilages when ventilation is applied. Because larger operative rigid bronchoscopes with diameters of 11 to 12 mm are now available for insertion of silicone stents, upper airway and vocal cord edema may be a more severe problem at the end of a prolonged procedure; hence careful examination of the cords and hypopharynx is important to avoid postprocedural stridor and upper airway obstruction. With the advent of flexible fiberoptic bronchoscopy, the number of pulmonary teaching programs routinely including rigid bronchoscopy in their training curriculum is falling. Therefore, even though rigid bronchoscopy should be a skill acquired by all bronchoscopists, generally only those being trained in otolaryngology–head and neck surgery, thoracic surgery, or interventional pulmonology generally receive adequate exposure to and practice with this technique.9
Flexible Fiberoptic Bronchoscopy
Range of Flexible Bronchoscopes and Special Features
Although there remains the tendency to subdivide bronchoscopes into “adult” and “pediatric” categories depending on their outer diameters, this distinction is arbitrary. Because bronchoscopists venture more peripherally with FOBs to sample focal lesions, and more interventional procedures via a flexible FOB are performed in the pediatric population, it makes more sense to describe the bronchoscopes and leave their judicious application to the bronchoscopist according to experience and the situational need.3,4
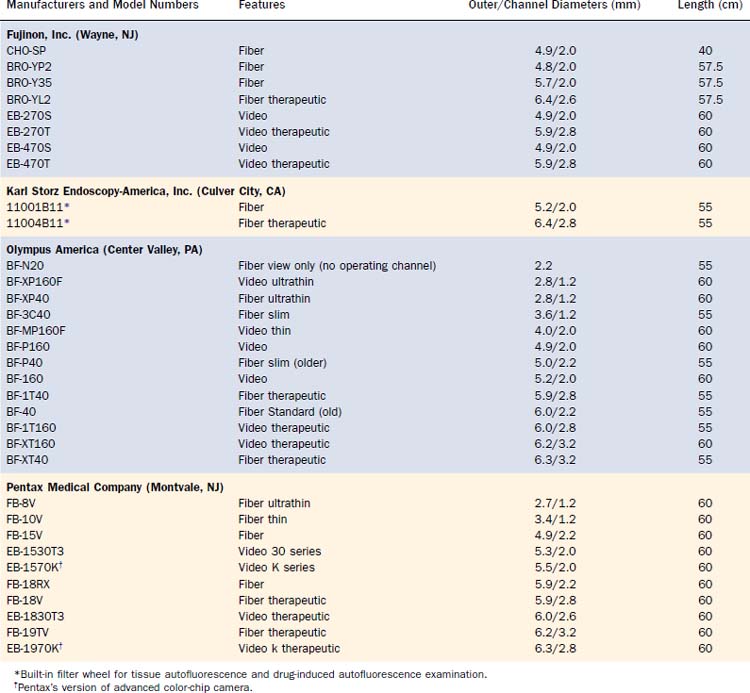
All FOBs have an illumination fiberoptic bundle and imaging fiberoptics or a camera. With the exception of the very few “ultrathin” bronchoscopes, there is also a channel for suction of secretions and blood, for the passage of topical medication and fluid for washing, and for the passage of various instruments for diagnostic retrieval of tissues or for therapeutic procedures (Table 75-1). The “average” diagnostic bronchoscope has an outer diameter of 5.0 to 5.5 mm and an operating channel of 2.0 to 2.2 mm. This caliber channel admits most cytology brushes, bronchial biopsy forceps, and transbronchial aspiration needles with sheathed outer diameters between 1.8 and 2.0 mm. Smaller bronchoscopes, in the range of 3.0 to 4.0 mm at the outer diameter and correspondingly smaller channels, are usually given a “P” designation (for pediatrics), but they can, of course, be used in the adult airways when narrowing is present because of benign strictures or malignant stenosis. Newer generations of “slim” video and fiberoptic bronchoscopes have a 2.0-mm operating channel with a 4.0-mm outer diameter. The one disadvantage of these bronchoscopes is the sacrifice of a smaller image area because of fewer optical bundles.
Larger “therapeutic” bronchoscopes (often designated with a “T” in the model number) can, of course, also be used for diagnostic purposes, but the larger outer diameter can cause greater discomfort and mucosal trauma in a conscious patient and can be harder to pass through an ET or tracheostomy tube, and thus can also impair gas exchange to a greater degree. Such therapeutic bronchoscopes have outer diameters between 6.0 and 6.3 mm, with operating channel lumens between 2.6 and 3.2 mm. Certain therapeutic instruments, including larger laser fibers, larger electrocautery forceps designed for gastrointestinal endoscopes, cryotherapy probes, and expandable balloons for bronchoplasty, require these larger diameters for their use through FOBs. Prototype bronchoscopes with a 9-mm outer diameter and a 5-mm operating channel have been made, the main application of which is to provide access for therapeutic instruments to airway segments that cannot normally be reached by rigid open-tube bronchoscopes and telescopes (Fig. 75-2).
At the other extreme are the ultrathin bronchoscopes, with outer diameters smaller than 3 mm. The Olympus production models fiber BF-XP40 and video BF-XP160F (Olympus America, Center Valley, PA) have outer diameters of 2.8 mm and operating channels of 1.2 mm. Special instruments (e.g., reusable cytology brush and forceps) of the proper caliber are available for tissue sampling (Fig. 75-3). Handling of these ultrathin bronchoscopes is often more challenging because their very floppy tips make steering more difficult. Suction via the narrow channels is also much more limited. Nevertheless, with practice and the use of small amounts of saline to flush open the distal airways in guiding the bronchoscope forward, the ultrathin bronchoscopes can traverse 12 to 16 generations of airways and, under fluoroscopic or CT guidance, are seen to approach the periphery of the lungs to sample focal lesions.3,4 The passage of a 2.8-mm instrument beyond the mid-teens division of the lobular bronchiole (measured at 0.7 mm in fixed tissue) calls into question some of the accepted measurements of the adult human airways. To permit the greater distance traversed, such ultrathin bronchoscopes usually have an operating length of 60 cm, 5 cm longer than the older bronchoscopes, although the current generation of videobronchoscopes are all built with a 60-cm working length.
New bronchoscopes manufactured today usually have a white ceramic insulated tip; this feature permits the safe use of electrocautery instruments with reduced risk of retrograde electric shock to the bronchoscopist and damage to the bronchoscope (see Figure 75-2).
Diagnostic Fiberoptic Bronchoscopy
General Approaches
Although there is usually one primary indication for bronchoscopy, an orderly and uniform approach should be taken for airway examination so that important pathology will not be missed because of impatience to attend to the primary focus. Starting at the upper trachea, mucosal integrity should be examined. Even when there are no gross endobronchial lesions, the presence of extrinsic tracheal deviation and compression due to paratracheal masses should be noted. TBNA can often successfully provide a tissue diagnosis of extrinsic lesions.10,11
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree