and Armando G. Correa3, 4
(1)
Department of Otorhinolaryngology—Head and Neck Surgery, University of Maryland School of Medicine, 16 S Eutaw St Ste 500, Baltimore, MD 21201, USA
(2)
Department of Pediatric Otolaryngology, University of Maryland School of Medicine, 16 S Eutaw St Ste 500, Baltimore, MD 21201, USA
(3)
Department of Pediatrics, Baylor College of Medicine, 6701 Fannin St; Suite 1540, Houston, TX 77030, USA
(4)
Texas Children’s Hospital, Houston, TX, USA
Keywords
TracheitisCroupStridorAirway obstructionLaryngotracheitisParainfluenzaSteroidsEpinephrineIntubationMist therapyIntroduction
A plethora of epidemics , changes in immunization practices and endotracheal intubation have resulted in a better understanding of the pathogenesis of tracheal infections. Historic descriptions first appeared for croup; with the name derived from Anglo-Saxon root kropan referring to a child with a barking cough. This was first described in print as early as 1854 [1]. Much deliberation ensued concerning the treatment of this condition, with O’Dwyer being the first physician credited with treatment of acute croup either by insertion of a modified endotracheal tube [2] or a tracheostomy [3] in separate instances.
The first reported case of tracheitis was published in 1823 by Pierre Blaud [4]. An increase in incidence was observed during each influenza A virus pandemic—H1N1 during the great Spanish flu (1918), Asian flu caused by H2N2 (1957), Hong Kong flu resulting from H3N2 (1968), and more recently the pandemic H1N1 of 2009. Autopsies performed during the 2009 pandemic showed tracheal denudation, maceration, de-epithelialization and other pathologic changes consistent with tracheitis [5]. Bacterial tracheal infections still maintain a low level of presence in infants and children presenting with symptoms of airway obstruction, requiring ICU admission and potentially endotracheal intubation.
Different diagnostic terms have been used for conditions that affect the larynx and trachea. Although it is useful to distinguish between supraglottic and subglottic laryngitis, this distinction is often difficult when the child is first seen. Laryngotracheitis or croup syndrome is a useful preliminary descriptive diagnosis until more definitive information is available [6]. “Croup syndrome ” also has been used to emphasize the variety of possible causes and location of laryngotracheal disease. In this chapter, “croup” is used to refer to subglottic laryngitis or laryngotracheitis, presumably viral. “Epiglottitis ” is an imprecise term often used in place of the better “supraglottitis ” as the epiglottis may be minimally involved in some cases in which most of the swelling is in the aryepiglottic folds. Preferred terms for tracheal infections are (with the usual terms in parentheses): croup, supraglottitis (epiglottitis), and suppurative tracheitis, laryngotracheitis, laryngotracheobronchitis, or laryngotracheobronchopneumonitis (bacterial tracheitis), depending on the extent of the bacterial superinfection [6].
Epidemiology
Tracheal infections have a significantly lower incidence compared to infections of the upper respiratory tract, with 1–5 % of all children requiring outpatient evaluation for viral croup within the first 3 years of life. Croup also requires hospital admission in about 1.3–5.6 % of all children evaluated for the same in emergency settings [7, 8].
Viral croup has the highest incidence in the second year of life and is virtually non-existent in the first 3 months. The incidence is slightly higher in male children (odds ratio = 1.43), and is highest in late fall and early winter [7]. A time-series analysis performed from a large number of children admitted with a principal diagnosis of croup in Ontario suggested a strong component of seasonality, with a biennial mid-autumn peak and annual summer trough [9]. Of interest, the overall number of hospitalizations has continued to decrease in the last 20 years, given the improvement in diagnosis and treatment. Marx et al. [10] from the Centers for Disease Control (CDC) studied the overall burden of croup and showed that the mean annual number of croup hospitalizations is about 41,000 (range, 27,000–62,000/year) in the U.S. Ninety-one percent of hospitalizations occur among children <5 years of age. The authors also reported that minor peaks in croup hospitalizations occurred each year in February, and major peaks occurred in October of odd-numbered years, which coincides with peak parainfluenza type 1 activity .
Supraglottitis , in contrast, has no seasonal peak. This disease, almost always caused by Haemophilus influenzae type b and accompanied by bacteremia, has been virtually eradicated by widespread immunization during infancy. While the peak age frequency for croup is 1–3 years, supraglottitis occurs in older children, with a peak between 3 and 6 years. Suppurative tracheobronchitis also tends to be a disease of preschool and school-age children [6].
The reported incidence for bacterial tracheitis in the literature is about 0.1/100,000 [11]. This estimate was based on the combined experience of four pediatric intensive care units. The incidence of tracheal infections caused by other pathogens such as fungal or mycobacterial origin is exceedingly low. Over the last two decades, the availability of nebulized epinephrine as well as injectable corticosteroids have changed the landscape of serious, life-threatening tracheal infections, with the re-emergence of bacterial tracheitis. Currently, bacterial tracheitis has three times the risk of respiratory failure associated with it than epiglottitis and viral croup combined [4].
Etiology
Acute laryngotracheitis, considered to be the most common cause for croup, is almost exclusively caused by viral organisms. Both bacteria and viruses may be responsible for infections with collateral components, such as laryngotracheobronchitis, and the more general laryngotracheobronchopneumonitis. In 1958, the first evidence for association between croup and two newly isolated myxoviruses, parainfluenza virus types 1 and 2, resulted in separation of two categories of cases—mild, requiring only outpatient follow up, and severe, requiring hospitalization [12]. Parainfluenza is a RNA paramyxovirus that actively replicates in respiratory epithelial cells and is comprised of four major serotypes. Parainfluenza type 3, more commonly associated with bronchiolitis or bronchopneumonia, can also produce severe croup in an endemic pattern, while type 4 is rarely seen. Parainfluenza 1 and 2, account for >65 % of all causes of croup. A large series studied 6165 instances of lower respiratory tract infections (LRIs) wherein approximately 75 % of all isolates were identified as parainfluenza viruses. Of these, parainfluenza type 1 accounted for about 60 %. Conversely, the propensity of the various organisms to produce symptoms of croup reached 60 % for both parainfluenza 1 and 2. For parainfluenza type 3, the number dropped to about 30 %, whereas all the other microorganisms accounted for about 5–15 %. Thus, parainfluenza viruses were the most common cause for all age groups; whereas respiratory syncytial virus (RSV) caused croup in infants and the influenza viruses and M. pneumoniae were significant causes of croup only in children older than 5 years of age. Summertime croup may be due to enteroviruses, adenovirus, or parainfluenza type 3.
Among other important viral pathogens causing tracheal infections, RSV was studied in isolates from sentinel practices in England and Wales from 1975 to 1990, during which an increase in mortality, by as much as 60–80 %, was observed in comparison with parainfluenza and influenza viruses [13]. Prematurity is associated with an increased risk for mortality, with factors such as a decrease in gestational age, increased perinatal oxygen requirements and discharge within 3 months of the RSV season increasing the likelihood for hospitalization [14]. Among the rare viral causes, measles, by virtue of immunosuppression, leads to a bacterial superinfection that results in a condition termed measles-associated bacterial tracheitis (MABT) , which carries an increased risk for need of artificial airway and intensive care admission [15].
Bacterial tracheitis is much less common when compared to that of viral origin. Previous reports have shown that the most consistent organism is S. aureus , followed by S. pneumoniae and M. catarrhalis [4]. Due to the universal immunization against H. influenzae type b , the incidence has dropped significantly. Similarly, immunization against C. diphtheriae has restricted the incidence of diphtheritic tracheitis to unimmunized children only. Reports of this are largely limited in modern literature, compared to the beginning of the century when tracheostomy was a routine practice to circumvent acute airway obstruction due to formation of pseudomembranes [16].
Fayon et al. [17] studied independent risk factors for development of bacterial tracheitis in a large series of children admitted to the PICU (n = 955), and found that the incidence of bacterial nosocomial tracheitis in that population was about 1.8 %. The pathogens isolated in this series were in agreement with other studies of bacterial tracheitis, comprising Staphylococcus aureus and gram-negative bacteria, and sometimes, mixed flora. In this population, tracheitis was attributed to young age, with small-sized airways in which thick secretions and mucosal inflammation being blamed for impairment of air flow and increased stasis. Head trauma, neuromuscular blockade and mechanical ventilation were independent variables that increased the risk of infection, but the last two risk factors may be physiologically collinear, given that most patients who were administered neuromuscular blockade were intubated, and vice versa.
Given the evolution of design features of modern day endotracheal tubes as well as enhanced monitoring of cuff pressures, reports of laryngotracheitis induced by indwelling endotracheal tubes have largely been limited to historic data [18]. Modern endotracheal tubes use materials that intrinsically inhibit or are coated with substances such as micronized silver to reduce bacterial growth by providing less scaffolding for colonization [19, 20].
Infectious agents such as Mycobacterium tuberculosis and fungi have been previously reported to have caused isolated instances of tracheal infection with a picture of long-term respiratory failure requiring a tracheostomy during the course of treatment that may be prolonged [21]. Chronic aspiration as well as gastroesophageal reflux (GERD) may accelerate laryngotracheal injury facilitating the development of tracheitis in those children.
Clinical Presentation
Despite the narrow spectrum of pathogens isolated from the plethora of tracheal infections described, these can have varied presentations, and typically differ in the outpatient vis-à-vis inpatient setting.
Acute viral croup manifests in the form of a viral prodrome characterized by clear rhinorrhea, low-grade fever, sore throat (in older children) and cough [22]. This usually lasts about 12–72 h, and typically progresses to hoarseness and the pathognomonic croupy cough that has a bark-like character. Rarely, febrile convulsions can occur. Nighttime symptoms are usually worse which frequently prompts the parents to seek care in the emergency room.
The typical course of progression goes through stridor that is inspiratory in nature but may also be associated with an expiratory component that results from unique features of the larynx in young children [23]. The infant larynx is narrowest in the subglottic segment, and inflammation of this area results in a fixed obstruction that leads to expiratory stridor. Children presenting multiple times with acute viral croup may have a masked presentation of subglottic stenosis wherein the already narrowed subglottic larynx is further reduced in diameter due to croup-related inflammation [24]. Hoarseness results from edema of the true vocal cords, often reducing their mobility. Wheezing is infrequently present. With increased severity, suprasternal and intercostal retractions may be present, and tachycardia and tachypnea are relatively common. It is important to note that reduction in the intensity of stridor in a sick-appearing child may be sign of impending respiratory failure as airflow may be reduced to the point where stridor may not be present.
When an infectious cause is not present in croup, the clinical course is abbreviated, with the noticeable absence of the viral prodrome. This condition is frequently referred as “spasmodic croup ”, although episodic croup is a more appropriate term as it is typically triggered by an allergic etiology and often recurs [25]. Pediatric angioedema shares features with croup, but is often associated with facial or neck swelling that is acute in onset [26]. Rarely, an undetected foreign body may masquerade as acute croup. Although a viral prodrome may be absent, an unsuspecting physician may be drawn into an acute airway emergency due to commonality of symptoms [27].
The severity of viral croup may be assessed using one of the many scoring systems available. The most well-known of these, the Westley Croup Score [28], utilizes key clinical signs including chest retractions, stridor, cyanosis, level of consciousness and air entry to obtain a composite score that is predictive of the need for intubation. As croup is primarily a clinical diagnosis, the utility of the Westley Score, as with other stratification systems, such as the Alberta Clinical Practice Guideline Working Group [29], may be limited to use in a research scenario. For example, using the Westley Score , Johnson et al. [30] showed that ≥ 85 % of children present with symptoms of generally mild croup, and less than 1 % were diagnosed with severe croup.
Peltola et al. [31] studied the clinical courses of croup caused by parainfluenza and influenza viruses to highlight the differences in morbidity caused by the different viral strains in hospitalized children. In general, there were no significant differences in the patterns of clinical features due to infections with the three parainfluenza subtypes, except that parainfluenza 3 was associated with wheezing. However, children with croup due to microbiologically-confirmed influenza virus tended to be hospitalized for longer (4 days vs. 2 days for parainfluenza). In addition, the rates of readmission were higher for influenza due to the relapsing course of respiratory distress during the few days following discharge. The requirements for corticosteroids as well as supplemental oxygen also tended to be higher for those caused by influenza virus, emphasizing its enhanced virulence.
Notwithstanding the generally predictable course of viral croup, it is important to differentiate it from other acute disorders of the pediatric airway. Rapidity in progression to high fever, odynophagia, anxiety and relative aphonia should always alert the practitioner to supraglottitis (epiglottitis), which is a rare occurrence following introduction of universal immunization against Hemophilus influenzae type b . Posturing in the upright position and extension of the neck with drooling in an anxious-appearing child mandates the need to secure the airway in a controlled setting such as the operating room. Care should be taken to not agitate the child for that may precipitate respiratory collapse. If there is history to suggest incomplete immunization, laryngeal diphtheria should be considered. This condition tends to have a slower progression, and has historically been associated with the presence of exudative membranes in the oropharynx.
Bacterial tracheitis typically is a secondary infection following a primary viral respiratory infection due to a cascade of events resulting from tracheal mucosal injury, impaired phagocytic function and cytopathic effects of the viral infection. This condition usually is recognized after reasonable efforts to treat viral croup have failed. Children with bacterial tracheitis are acutely ill, with symptoms to suggest dehydration and organ failure in the presence of other host factors such as immunodeficiency. Often seen in an inpatient setting in children admitted to the ICU with respiratory failure, bacterial tracheitis can have a variable presentation in the absence of pathognomonic clinical signs [32].
The cardinal initial signs of bacterial tracheitis include cough, stridor and a rapidly changing course of illness that progresses to respiratory failure quickly. Children affected are usually older (>5 years). Other symptoms on admission may include choking episodes, dyspnea, dysphagia, neck pain, dysphonia and agitation. In one study by Bernstein et al. [33], children younger than 5 years of age were twice as likely to be intubated, compared to older children. The same study compared changing figures for mortality—prior to the 1940s, the mortality rate for bacterial tracheitis approached 40 %, but with advances in mechanical ventilation and airway management, that figure has dropped to 0–20 % in more recent series.
Diagnostic Tests
From the majority of studies, it is clear that the diagnosis of acute viral croup is chiefly based on clinical examination and does not necessitate laboratory testing. That said, if there is suspicion for a concurrent lower respiratory tract infection, white blood cell count with differential, as well as routine postero-anterior/lateral chest and neck radiographs may be indicated. In viral croup, the white count is often at the high end of normal, and may be higher in approximately 50 % of hospitalized children [34]. Administration of corticosteroids may cause leukocyte demargination, which can lead to spuriously elevated counts during the course of treatment.
Plain film radiography often is utilized to evaluate laryngotracheal edema in croup, but has inconsistent results. The typical picture is that of narrowing of laryngeal air column in the subglottic segment, approximately for ~5–10 mm below the level of the vocal cords, resulting from mucosal edema [35]. This has been historically referred to as the steeple sign (Fig. 12.1), but is observed only in ~50 % of instances [23]. This, coupled with reduced sensitivity for differentiating between viral croup, epiglottitis and bacterial tracheitis undermines the usefulness of routine radiographs for diagnosis. However, some investigators such as Mills et al. [36], have reported sensitivity and specificity of >90 % respectively. The best practice in these circumstances is to consider radiographs in those children in whom the clinical presentation is atypical and whose respiratory status is stable enough to undergo positioning prior to obtaining the films [23].
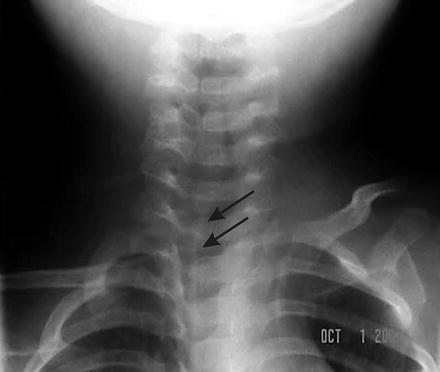

Fig. 12.1
Antero-posterior view of a plain film radiograph showing a long segment of subglottic and tracheal inflammation in a child with croup
Alveolar gas exchange is usually not affected by viral croup, unless there is concurrent presence of laryngotracheobronchitis, asthma or pulmonary insufficiency [23]. Thus, pulse oximetry and respiratory rate have been shown to have poor correlation with clinical status or hypoxia due to artifacts [37]. Evidently, the uncompromised standard is clinical observation with pulse oximetry as a useful adjunct in instances wherein the lower airway is also affected.
In cases where operative control of the airway is required, telescopic tracheobronchoscopy , aided by the ventilating bronchoscope provides the gold standard for assessment of the airway in severe croup, or when alternate pathology, such as supraglottitis, is suspected. In the ambulatory setting, children who present with recurrent croup should be examined for concurrent abnormalities. Chun et al. [38] evaluated 30 children who were previously diagnosed with recurrent episodes of croup. A third of these children were found to have synchronous lesions such as subglottic stenosis, edema and cysts. In the same study, abnormal rigid endoscopic findings were more likely to be seen in children under the age of 3 years, highlighting the need for a higher index of suspicion and lower threshold for performing airway endoscopy in this age group.
Microbiologic investigations to determine etiology are increasingly being performed due to the availability of molecular and standard virologic methods. These tests are usually not recommended for diagnosis in mild cases of croup, but may be warranted in children hospitalized and/or requiring mechanical ventilation. Real-time polymerase chain reaction (RT-PCR) and viral cultures are also indicated with atypical courses of the infection, as described by reports of novel pathogenic strains for viral croup, e.g. coronavirus NL63 detected in samples isolated from Europe [39]. An improved panel based on an RT-PCR assay has been developed for influenza A and B viruses, RSV and parainfluenza 1, 2, 3 and 4. According to one study, the application of PCR increases the sensitivity of respiratory viral diagnosis, with results being made available within 6 h, thus increasing clinical relevance [40]. With claimed sensitivity of ~80 % and specificity approaching 100 %, several authors have increasingly validated their cost-effectiveness [41]. As mentioned earlier, the routine use of these tests in mild croup is unsubstantiated.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


