Dibb WL, 1991 [11]
Clark WB, 1997 [12]
Jones, RN 1997 [13]
Drehobl M, 2008 [14]
Number of patients
226
23
433
627
Gram–positive organisms
Staphylococcus aureus
34.1 %
30.4 %
11.8 %
10.8 %
Gram–negative organisms
Pseudomonas aeruginosa
22.1 %
60.9 %
48.7 %
62.2 %
Proteus mirabilis
3.5 %
8.7 %
5.3 %
–
Enterobacter species
4.0 %
4.3 %
3.7 %
4.0 %
Klebsiella species
5.3 %
–
2.5 %
4.9 %
Escherichia coli
4.9 %
–
–
2.1 %
Diagnosis
Acute otitis externa is a clinical diagnosis consisting of the rapid onset of signs and symptoms of ear canal inflammation [17]. Otalgia is the most common symptom. Patients can experience itching or fullness within the ear canal, hearing loss, or pain with chewing. The characteristic finding of exquisite tenderness with manipulation of the tragus or pinna is frequently observed in patients with diffuse canal swelling. On examination, one finds erythema and swelling of the EAC, often with purulent debris (Fig. 2.1). With severe disease, cellulitis of the pinna and regional lymphadenopathy can be present. Otoscopy with visualization of the tympanic membrane, while often difficult due to EAC swelling, should be performed in all patients as a nonintact tympanic membrane influences management decisions.
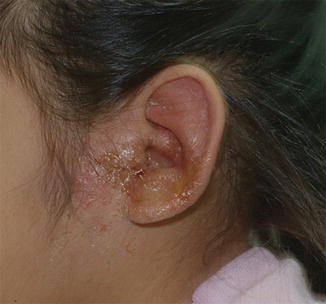

Fig. 2.1
Acute otitis externa . A severe case of acute otitis externa. There is profuse otorrhea with soft tissue swelling that extends beyond the canal, involving the pinna and the preauricular space (courtesy of Dr. Ellen Friedman)
Clinicians should be careful to differentiate AOE from other conditions that cause otalgia and otorrhea . Otitis media with perforation, frequently confused with AOE, will present with a middle ear effusion and less severe tenderness with tragus pressure. Fungal otitis externa (“otomycosis”) and chronic otitis media will have a more protracted duration of symptoms. When dermatologic conditions involve the ear canal, patients experience prolonged ear pain and less otorrhea than with AOE. Causes of ear pain unrelated to ear pathology include temporomandibular joint (TMJ) syndrome, tonsillitis or peritonsillar infections, and dental disorders.
Cultures of the ear canal are not necessary to diagnose AOE. For routine, uncomplicated AOE, cultures are unlikely to impact management decisions. However, for patients with severe disease or who have an inadequate response to treatment, cultures should be obtained.
Treatment
The management of uncomplicated AOE consists of topical antibiotics and pain control. In 2014, the American Academy of Otolaryngology—Head and Neck Surgery Foundation (AAO-HNSF) updated their evidence-based recommendations on the management of AOE [17].
Topical antibiotics are the cornerstone of treatment for uncomplicated AOE. Topical therapy allows for high concentrations of antimicrobials to be delivered to the site of infection while minimizing the risk of side effects [18]. In contrast, systemic antibiotics can promote selective pressure for resistant organisms, carry a greater risk of side effects, and achieve lower local drug concentrations [19]. The efficacy of topical therapy for AOE has been demonstrated in numerous randomized controlled trials [20–24]. Clinical cure rates up to 80 % can be achieved within 10 days of therapy with topical antibiotics [20]. Various preparations are approved for AOE, including antiseptics and antibiotics, with or without a corticosteroid (Table 2.2).
Table 2.2
Ototopical preparations for the treatment of acute otitis externa
Drug | Name | Bottle size (mL) | Cost | |
|---|---|---|---|---|
Trade | Generic | |||
Acetic acid 2.0 % solution | Acetic acid otic (generic) | 15.0 | – | $ |
Acetic acid 2.0 %, hydrocortisone 1.0 % | Acetasol HC (generic) | 10.0 | – | $ |
Neomycin, polymyxin B, hydrocortisone | Cortisporin otic (trade) | 10.0 | $$ | $ |
Ofloxacin 0.3 % | Floxin otic (trade) | 5.0 | $$ | $ |
Ciprofloxacin 0.2 %, hydrocortisone 1.0 % | Cipro HC (trade) | 10.0 | $$$ | – |
Ciprofloxacin 0.3 %, dexamethasone 0.1 % | Ciprodex (trade) | 7.5 | $$$ | – |
Antiseptic agents are used predominantly for the treatment of mild-moderate AOE or for the prevention of AOE in at-risk persons (i.e. swimmers). Acetic acid and alcohol are the most commonly utilized antiseptic agents, although other agents (boric acid, aluminum acetate, and silver nitrate) have also been used [17]. These agents work through various mechanisms, including acidifying the local environment, to make the ear canal less tolerable for bacteria. Due to their acidic nature, some patients experience pain and local irritation with administration [25]. While effective in most cases of AOE, a randomized controlled trial found acetic acid alone had a lower cure rate than either an acetic acid-steroid combination or an antibiotic-steroid combination at 2 and 3 week follow-up [22].
Topical antibiotics are the most commonly prescribed agents for AOE, and the most frequently prescribed antibiotic preparation is a combination of neomycin and polymyxin B with hydrocortisone [26, 27]. It is an inexpensive and effective preparation that has been available for over three decades. Polymyxin B is active against P. aeruginosa and other gram-negative organisms, while neomycin, an aminoglycoside, provides activity against S. aureus. Aminoglycoside ophthalmic preparations—tobramycin 0.3 % solution, tobramycin 0.3 % and dexamethasone 0.1 % suspension, and gentamicin 0.3 % solution—have also been used for the treatment of AOE. While not FDA-approved for this indication, several small trials have found that clinical cure rates for these agents are similar to other topical antibiotic preparations [23]. Several limitations, however, exist for preparations containing aminoglycosides—the dosing schedule, the risk of contact dermatitis, and the risk of ototoxicity in patients with nonintact tympanic membranes.
The fluoroquinolones ofloxacin and ciprofloxacin , were developed into topical formulations for ear infections in the late 1990s [25]. They have broad antimicrobial activity, including both gram-positive and gram-negative bacteria, and benefit from a convenient dosing regimen, an excellent safety profile, and the ability to be used in patients with nonintact tympanic membranes. Several studies have demonstrated that quinolone agents perform as well, or better, than non-quinolone preparations [20].
The clinical cure rates are similar between the various classes of ototopical agents used for AOE, according to a systematic review of randomized control trials [20]. In a multicenter, randomized clinical trial composed of 1072 adult and pediatric patients, ciprofloxacin-dexamethasone and neomycin-polymyxin B-hydrocortisone given for 7 days had similar cure rates after 3 days (14 % vs. 10 %), 8 days (75 % vs. 72 %), and 18 days (98 % vs. 97 %). The only significant difference was found in the time to cure, which was 0.6 days shorter for the ciprofloxacin-dexamethasone suspension [24].
Before selecting a topical agent for AOE, clinicians should determine whether the patient has a nonintact tympanic membrane due to tympanic membrane perforation or tympanostomy tube placement. In such patients, clinicians should avoid prescribing potential ototoxic topical agents. Animal models have demonstrated that application of ototoxic agents into the middle ear can damage the hair cells of the inner ear [28]. Products to avoid in patients with nonintact tympanic membranes include preparations with a low pH (most antiseptic agents), alcohol, or aminoglycosides [17]. Despite the concern for ototoxic injury, the risk from topical aminoglycosides in humans appears to be low. A review of 500 children who received an aminoglycoside containing preparation following tympanostomy tube placement revealed no cases of cochlear toxicity [29]. In a 2004 systematic review of the published literature, only 54 cases of inner ear toxicity attributed to gentamicin and 13 cases attributed to neomycin were identified [30]. Although the risk for ototoxicity may be low, given the availability of a safe and effective alternative, we recommend the use of a quinolone agent for all patients with AOE with a nonintact tympanic membrane.
Additional considerations when selecting a topical agent for AOE include the cost, dosing schedule, and side effects. The twice-daily dosing schedule for the quinolone agents is convenient and likely results in the greatest degree of adherence to therapy. In a single open-label trial, once-daily dosing of ofloxacin achieved good clinical outcomes [31]. In contrast, the neomycin-polymyxin B-hydrocortisone preparation generally requires that drops are administered four times daily. The risk of contact dermatitis appears to be greatest from the aminoglycoside-containing topical preparations, particularly neomycin [32]. While rare following a single course of treatment, these localized reactions can develop in patients when there is prolonged use.
Clinicians should promote the effective administration of ototopical medications through patient education and, if necessary, adjunctive drug delivery methods. Aural toilet or otologic debridement should be considered in patients with severe otorrhea or EAC swelling. The AAO-HNSF guidelines recommend that another person (i.e. the child’s parent or caregiver) administer drops with the patient lying down and the affected ear facing up. After instilling the drops in the ear canal, the patient should remain in this position for 3–5 min [17]. Debris in the EAC or severe swelling can interfere with the effective delivery of medication to the site of infection. For such patients, clinicians should consider performing aural toilet, otologic debridement, or inserting an ear wick. Additionally, as ear pain from AOE can be intense, the degree of pain should be assessed and a pain control strategy developed. Benzocaine-containing otic solutions are topical anesthetics that can provide pain temporary relief. These products, however, are currently not approved by the US Food and Drug Administration and may not be safe if used with a nonintact tympanic membrane. We recommend orally administered analgesics (i.e. acetaminophen, ibuprofen) for children with otalgia from otitis externa.
Systemic antibiotics, when combined with topical therapy, provide no additional benefit for uncomplicated AOE [17, 22]. However, systemic antibiotics should be considered for immunocompromised patients or those with severe disease extending beyond the pinna. Empiric antibiotic therapy should cover P. aeruginosa and S. aureus, while definitive therapy should be directed by culture results.
Prevention
Preventive measures should be considered for individuals with recurrent episodes of AOE, especially swimmers and individuals with predisposing dermatologic conditions. No randomized controlled trials have been performed to identify the most effective strategies. Potential preventive measures include wearing ear plugs, blow drying the ear following water exposure (using a low setting and keeping the hair dryer >12 in. from the ear), and using an antiseptic agent before and after water exposure to re-acidify the ear canal.
Necrotizing (Malignant) Otitis Externa
Necrotizing otitis externa (NOE) , also called malignant otitis externa , is a rare and severe invasive infection of the external auditory canal that involves the periauricular soft tissue and skull base. This disorder, which typically affects elderly individuals with diabetes mellitus, has also been reported in immunocompromised children, including children with HIV, malignancy, malnutrition, chemotherapy-induced neutropenia, aplastic anemia, and diabetes mellitus [33]. Pseudomonas aeruginosa is identified in >90 % of all cases of NOE in both adult and pediatric case series [33, 34]. Additional bacteria reported to cause NOE include S. aureus, Klebsiella species, and P. mirabilis. Rarely, fungal NOE has been reported, most often due to Aspergillus fumigatus [34].
Individuals present with exquisite otalgia and otorrhea , generally more severe than that found in uncomplicated otitis externa. While adult patients generally present with prolonged and progressive ear pain and otorrhea unresponsive to topical therapy, children tend to present more acutely. On examination, there is swelling of the pinna and periauricular soft tissue. Granulation tissue is frequently noted along the inferior portion of the EAC at the bony-cartilage junction (Fig. 2.2). Osteomyelitis of the skull base and cranial neuropathies develop with progression of the infection. Facial nerve palsy, which occurs at a higher rate in children than adults due to the proximity of the facial nerve to the ear canal, typically occurs early in the course of disease [33]. Additional complications, while rare, include meningitis, brain abscesses, and dural sinus thrombosis.
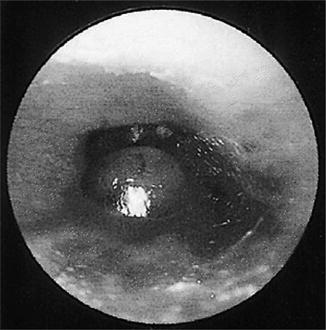

Fig. 2.2
Necrotizing otitis externa . Granulation tissue at the bony-cartilage junction (Reprinted with permission from [54]. Copyright Elsevier 2010)
Necrotizing otitis externa is a diagnosis based on clinical, laboratory, and radiographic findings. Laboratory findings reveal an elevated erythrocyte sedimentation rate or C-reactive protein. While non-specific, markedly elevated levels of these inflammatory markers may suggest invasive disease. Controversy exists regarding the optimal imaging modality for the diagnosis and follow-up of NOE [34]. Historically, radionuclide (technetium-99 or gallium-67) scans, with or without single photon emission computerized tomography (SPECT) scanning, were preferred to detect and monitor disease from NOE [34]. Recent literature, however, has shown that magnetic resonance imaging (MRI), which can identify soft tissue changes and intracranial complications, is effective in following the course of disease in patients with NOE [35]. In comparison to radionuclide scans, MRI is more readily accessible and avoids exposure to radiation, a particularly important point in the pediatric population. Computed tomography (CT) imaging is able to detect early bone erosion; thus it may be the preferred technique to distinguish NOE from cases of uncomplicated, yet severe, external otitis.
Treatment for necrotizing otitis externa consists of prolonged systemic antibiotic therapy. The empiric antibiotic regimen should have activity against P. aeruginosa, with definitive therapy directed by culture results. Antipseudomonal beta-lactam agents (piperacillin-tazobactam, ticarcillin-clavulanate, ceftazidime, cefepime), fluoroquinolones, and carbapenems are agents with activity against P. aeruginosa that effectively penetrate bone tissue. The fluoroquinolones are frequently used for NOE in adults due to their safe side effect profile, excellent bone penetration, and availability in an oral formulation [34]. While fluoroquinolones are generally avoided in children due to the risk of injury to developing bones or joints, their use in NOE is justified when effective alternative agents are not available [36]. Prolonged antibiotic therapy for 6–8 weeks is generally recommended. Surgical intervention is limited to debridement of granulation tissue or to obtain biopsies for microbiologic and histologic specimens when there is diagnostic uncertainty. We recommend combination therapy (an antipseudomonal cephalosporin or a fluoroquinolone, plus an aminoglycoside) for initial gram-negative coverage to increase the likelihood of susceptibility in the event there is a drug-resistant organism. Following culture and susceptibility results, definitive therapy can be completed with a single agent (an antipseudomonal cephalosporin or fluoroquinolone) for a minimum of 6 weeks.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


