16 Multiple symptoms are commonly experienced, including decreased vision (a decline in central visual acuity affecting distance and near vision), metamorphopsia (image distortion, the most sensitive symptom for macular elevation) (Table 16–1), micropsia (objects appear smaller than normal), dyschromatopsia (colors appear faded), and loss of contrast sensitivity. These symptoms can be unilateral or bilateral and are often asymptomatic when the pathology is unilateral with normal vision in the unaffected eye or when the fovea is uninvolved. A systematic approach to the evaluation of these symptoms is outlined in Figure 16–1. Determining the morphology of the macula is crucial in determining the cause of metamorphopsia. A thorough fundus examination with biomicroscopy and utilization of a fundus contact lens is necessary to appreciate the subtle changes that may be present. The most obvious sign of a thickened or elevated macular retina is often the loss of the foveal light reflex. This occurs when the normal concavity of the fovea is disrupted; however, changes can occur in the macula that are extrafoveal and therefore do not affect the foveal light reflex. Five areas can be involved in producing an elevated or thickened macula. These include, from outer to inner layers, suprachoroidal space, choroid, subretinal pigment epithelial (RPE) space, subretinal space, and intraretinal space. Most of the changes at these levels are caused by abnormal accumulation of fluid, blood, or other substances, like lipofuscin. Elevation of the choroid from suprachoroidal hemorrhage or thickening of the choroid due to choroidal effusions can secondarily elevate the macular retina and produce metamorphopsia (Fig. 16–2). Common causes of choroidal effusion include ocular hypotony, uveitis, and scleritis. The sub-RPE area is another location where pathology can occur and produce elevation of the macula. A change in this location will result in a pigment epithelial detachment (PED). These elevated areas will often appear as a small, rounded, yellowish gray elevation of RPE underneath an otherwise normal retina. A serous PED results from a collection of fluid beneath the RPE; whereas an ingrowth of choroidal neovascularization is categorized as a fibrovascular PED. Transillumination of the lesion with a slit beam will often differentiate a serous and fibrovascular PED. Other forms of PEDs develop from bleeding or large drusen formation beneath the RPE, termed hemorrhagic or drusenoid PEDs, respectively (Fig. 16–2B,C). A fluorescein angiogram is instrumental in differentiating between the various types of PEDs. These lesions most commonly are caused by age-related macular degeneration or other causes of choroidal neovascularization and can be present with central serous chorioretinopathy. The subretinal location is the next potential space internal to the RPE. Again, the most common substances that accumulate in this area are fluid and blood. Other materials, like lipid and lipofuscin, also collect in this position. Choroidal neovascularization and macroaneurysms are common causes of subretinal fluid, hemorrhage, and lipid (Fig. 16–2A). The presence of lipofuscin in the subretinal space produces a yellowish color that is seen with Best’s disease and pseudovitelliform macular dystrophy. Occasionally, the appearance of a pseudohypopyon can develop with layering of the subretinal fluid and lipofuscin1 (Fig. 16–3). Central serous retinopathy is characterized by leakage from an incompetent RPE layer and can produce localized neurosensory detachments. Fluorescein angiography (FA) is helpful in differentiating between the various causes of a localized macular neurosensory detachment of the retina. The specific characteristics are discussed later in this chapter.
Thickened or Elevated Macular Retina
Thickened or Elevated Macular Retina
What Are the Symptoms of a Thickened or Elevated Macular Retina?
How Do You Differentiate the Causes of Metamorphopsia?
| Decreased vision |
| Effects both near and distance acuity |
| Metamorphopsia |
| Image distortion |
| Evaluate with an Amsler grid |
| Micropsia |
| Image minification |
| Results from increased separation of photoreceptors |
| Dyschromatopsia |
| Poor color vision |
| Colors appear desaturated |
| Decreased contrast sensitivity |
The ability to determine whether the disturbance of the macular retina is secondary to elevation of more outer layers or thickening of the retina can be difficult. A ring of light reflex that replaces the normal foveal light reflex may outline the involved retina. Further examination of this abnormal retina with a slit beam positioned at an oblique angle of incidence will highlight a neurosensory detachment from the underlying RPE with an increased separation of the light reflexes between the surfaces of the RPE and retina. In addition, a localized detachment of the macular retina will appear more diaphanous due to the transparency of the retina than when elevated RPE is also present.
A thickening of the macular retina can be present with or without a neurosensory retinal detachment from the RPE. The most common causes of retinal thickening are vascular abnormalities that lead to macular edema from accumulation of intraretinal fluid. Therefore, the presence of microaneurysms or telangiectasias within the retina can be helpful in determining the cause. These most commonly are associated with diabetic retinopathy and venous occlusive disease. Intraretinal cysts also can develop from fluid accumulation in the foveal area. A characteristic petal configuration of cysts can form as a result of the architecture of the outer plexiform layer, that is, the Henle layer, within the perifoveal area. This results in cystoid macular edema, which is most often due to diabetic macular edema, the Irvine–Gass syndrome2,3 or other inflammatory conditions. Large foveal cysts often have the appearance of a macular hole and are considered a pseudohole. Referral to a retinal specialist is often necessary for differentiation with the assistance of optical coherence tomography (OCT) or FA.
Macular Retinal Detachment
Central Serous Chorioretinopathy and Its Common Risk Factors
Central serous chorioretinopathy (CSC) is most commonly identified in young males between the ages of 30 and 50 years. There is a male preponderance, with a ratio of approximately 9:l.4–6 The incidence in women, however, doubles between the second and third decades of life7 and in persons over the age of 50 years, the male-to-female ratio is 2.6:1.0.8 There may be a racial predisposition for whites, Hispanics, and Asians; the incidence in blacks is very low.9,10 Stress and other psychological factors also have been linked with a causal relationship to this disorder.11,12
What Are Clinical Signs Indicative of Central Serous Chorioretinopathy?
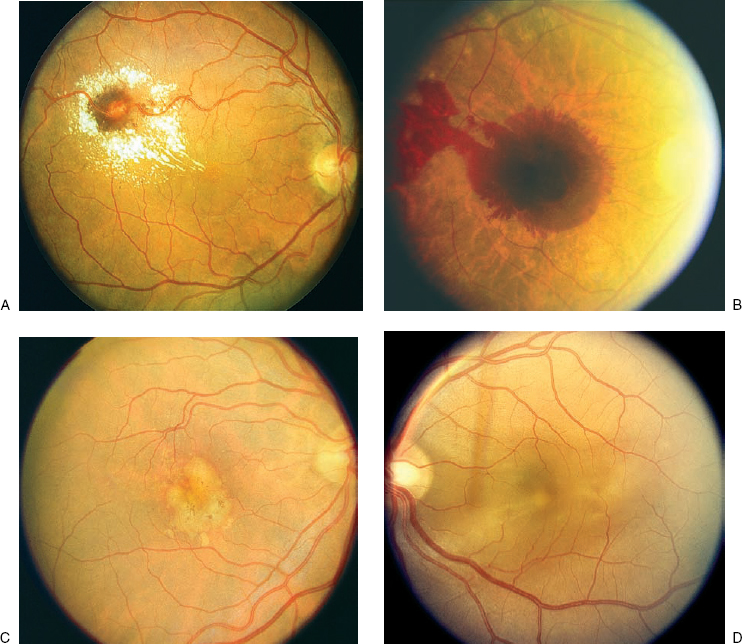
A focal area of neurosensory retinal detachment is often present in the macula (Fig. 16–4). The small area of elevation appears as a well-defined blister filled with transparent fluid. When the fovea is involved, the foveal light reflex is not present. The area of neurosensory elevation can be associated with an underlying PED, or a PED can exist without separation of the overlying retina. These features are best appreciated by using a fundus contact lens with the slit beam at an oblique angle to highlight any increased separation between the surfaces of the pigment epithelium and retina. Occasionally, the outer surface of the retina contains whitish yellow fibrin precipitates. Other manifestations in the RPE can range from subtle pigmentary changes with areas of pigment atrophy and pigment clumping. Perivascular pigment clumping in the form of bone spicules has been termed pseudoretinitis pigmentosa-like atypical CSC presentation.13 Severe or chronic leakage can produce RPE atrophic tracts that extend inferiorly and cause localized exudative retinal detachments.14,15
What Causes Central Serous Chorioretinopathy?
Although the exact cause of CSC is not known, dysfunction at the level of the RPE or choroid is commonly implicated. The normal RPE functions both as a barrier between the retina and choroid16 and as a pump that moves fluid in a retinochoroidal direction.17 Therefore, several investigators have suggested that defective RPE can lead to hyperpermeability and secondary accumulation of subretinal fluid.17,18 Others have described widespread choroidal abnormalities seen with indocyanine green (ICG) green angiography and conclude that hyperpermeability of the choroid leads to abnormalities of the RPE.8,19 The trigger that ultimately causes CSC may be a combination of environmental, genetic, and behavioral factors12 (Table 16–2).
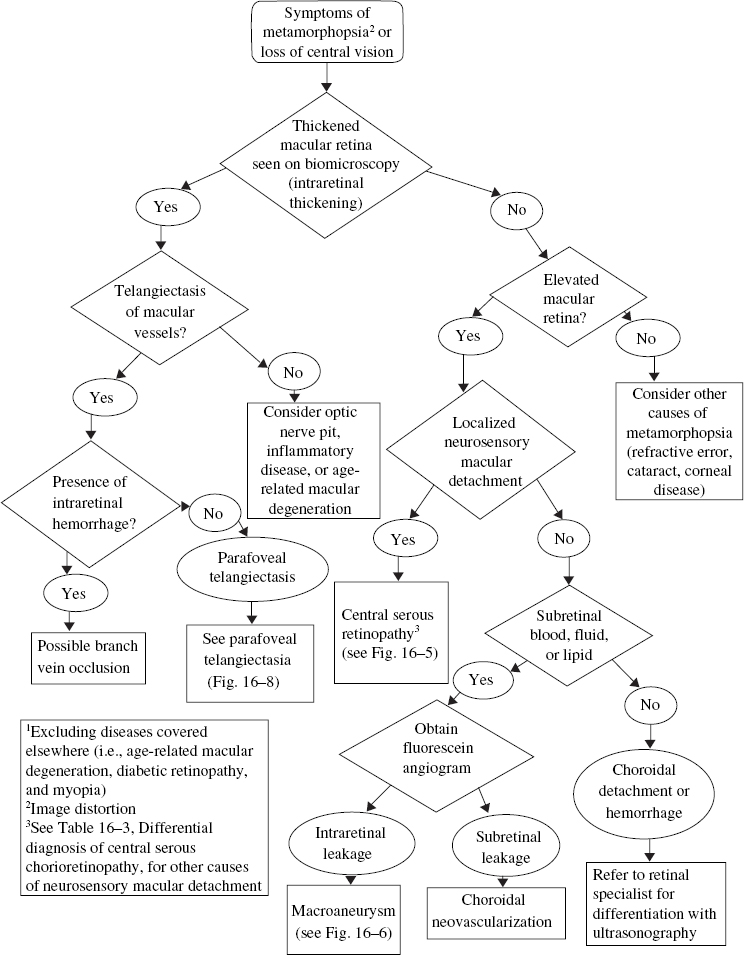
FIGURE 16–1. Evaluation of metamorphopsia secondary to elevated or thickened macular retina.
What Are the Findings of Central Serous Chorioretinopathy with Angiography?
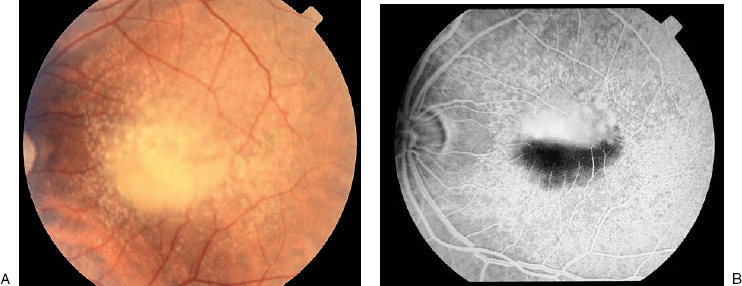
The early descriptions of CSC using FA revealed an area of leakage at the level of the RPE that accumulates within the neurosensory detachment20,21 (Fig. 16–4). In about 95% of cases, there is usually one focal point of leakage, but up to seven leakage points have been documented.5,22 Rarely, a diffuse area of leakage is present instead of a focal point of leakage. The classic smokestack phenomenon23 of leakage is present in only a minority of cases, about 724 to 20%.5,6 In the typical smokestack presentation, the fluorescein dye rises from a leakage point to the upper limit of the neurosensory detachment and then spreads laterally.
Although the blister of neurosensory detachment often involves the fovea, only about 10% of cases have the leakage point within the foveal area. Most cases contain the leakage source within 1 mm of the fovea, with the superonasal quadrant the most frequent location.22 In prolonged cases of CSC, dependent neurosensory detachments may develop that extend inferiorly. These “gutter” formations are often seen on FA as tracts of atrophic RPE with mottled hyperfluorescence.
The use of ICG angiography has provided evidence that supports the theory of diffuse choroidal or RPE dysfunction leading to CSC. With ICG, multiple areas of early and midphase hyperfluorescence are apparent throughout the fundus; these areas fade in the late phase. Often these abnormal spots are not associated with areas of leakage on FA and can be present in normal fellow eyes lacking neurosensory retinal detachments.19,25,26
What Is the Prognosis of Central Serous Chorioretinopathy?
Central serous chorioretinopathy is usually a self-limited disease with reports of 100% recovery without treatment.5,7 Patients usually recover within 3 to 4 months with visual acuities better than 20/40 in all cases, most achieving 20/20. Recurrences are reported in almost 50% of cases in these series, however, with half of the recurrences present in the first year but some up to 10 years after initial presentation.7
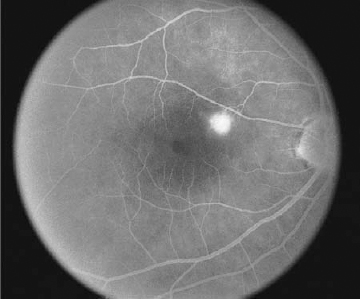
FIGURE 16–4. Central serous chorioretinopathy. Late-phase fluorescein angiogram with leakage from retinal pigment epithelium defect.
Fewer than 5% of patients have secondary complications from CSC that lead to a poor visual outcome. These can include peripheral atrophic tracts,14 choroidal neovascularization,27 macular RPE atrophy,28 and cystoid macular edema.
What Is the Treatment for Central Serous Chorioretinopathy?
Almost all cases of CSC will resolve spontaneously without the need for treatment. In cases with chronic leakage from a persistent source, however, photocoagulation can hasten the resolution of the leakage and reduce the recurrence rate, but it does not improve final visual acuity.29
Most patients should be observed for spontaneous resolution for 6 months (Fig. 16–5). FA at the initial visit is helpful in identifying the leakage source, and if the same source is the cause of persistent leakage after 6 months, laser photocoagulation to that area may be considered. When a patient has an occupational need for prompt recovery of visual acuity, or the lesion is greater than 500 μ from the fovea, earlier treatment is feasible.
| RPE, retinal pigment epithelium. |
| Serous detachment of the macular retina |
| Serous retinal pigment epithelial detachment |
| Hyperpigmented and atrophic RPE changes |
| Subretinal precipitates |
| Inferior, atrophic RPE tracts (gutter) |
| Dependent exudative retinal detachment |
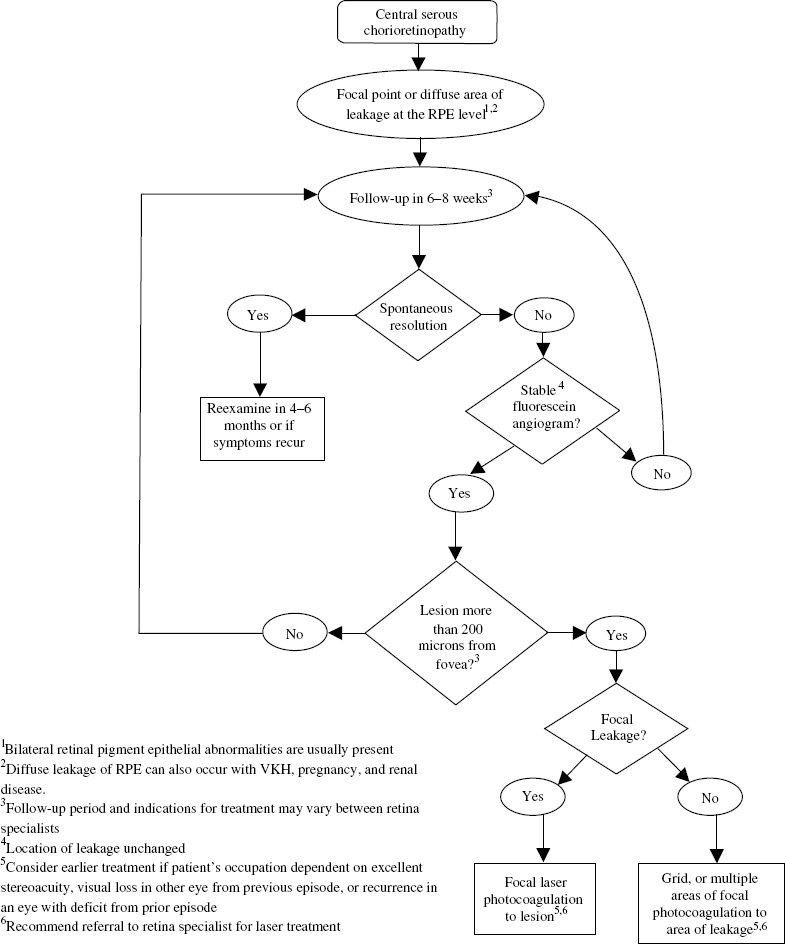
FIGURE 16–5. Management of central serous chorioretinopathy.
When treatment is recommended, a recent FA is required to determine the location of the source of leakage. Green, yellow, or red lasers are effective, but the krypton red is recommended for treatment within the foveal avascular zone or papillomacular bundle.30 A light gray reaction with a 200-μ spot over the area of leakage is often sufficient. A low intensity delivered over a moderate duration (0.2 to 0.3 seconds) with slow titration of power to reach the desired endpoint is recommended. In areas of diffuse leakage, multiple burns may be necessary to cover the entire lesion.
Multiple complications can arise from laser treatment, including choroidal neovascularization,31 gradual enlargement of the laser scar into the fovea, and accidental photocoagulation of the fovea.
What Is the Differential Diagnosis for Central Serous Chorioretinopathy?
Any disorder that causes a serous neurosensory detachment of the macula can be confused with CSC. Diseases associated with choroidal neovascularization like age-related macular degeneration, pathologic myopia, and ocular histoplasmosis syndrome must be considered. Posterior scleritis, multifocal choroiditis, Harada disease, and Eales’ disease are inflammatory conditions that can be associated with elevation of the macula. Other disorders include primary ocular or metastatic tumors, lymphoma, uveal effusion syndrome, optic nerve pits, or retinoschisis (Table 16–3).
| Disorders with choroidal neovascularization |
| Age-related macular degeneration |
| Pathologic myopia |
| Ocular histoplasmosis syndrome |
| Inflammatory disorders |
| Posterior scleritis |
| Multifocal choroiditis |
| Harada disease |
| Eales’ disease |
| Ocular tumors |
| Metastatic tumors |
| Lymphoma |
| Posterior uveal effusion syndrome |
| Optic-nerve pit |
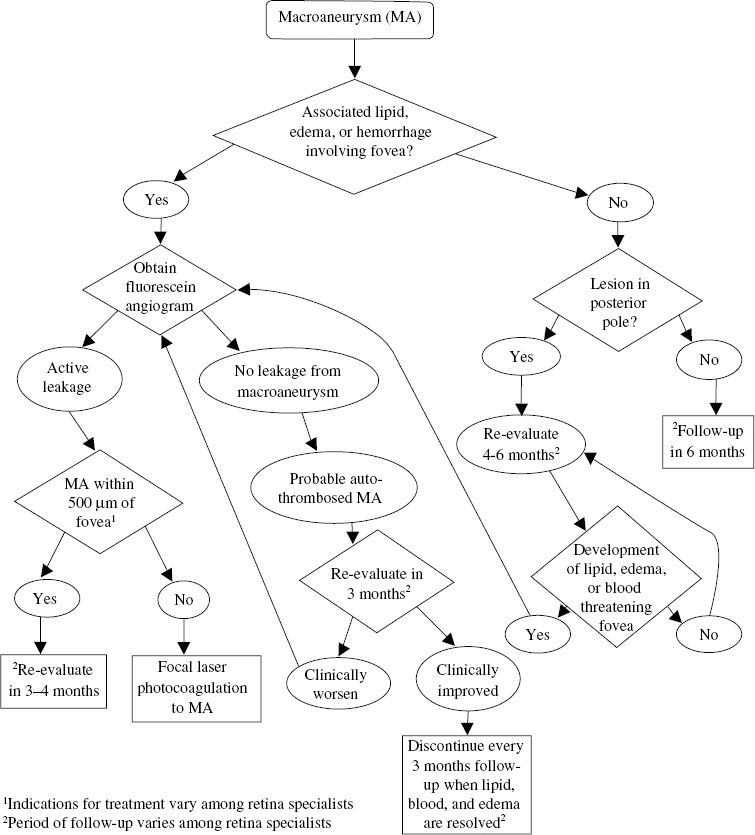
Macroaneurysm
A macroaneurysm is an acquired degeneration of a retinal artery causing a round or fusiform dilation that most often occurs on one of the four major branch retinal arteries. Macroaneurysms occur within the first three orders of retinal arterial branching. A macroaneurysm is most commonly present on the superotemporal arcade and often is associated with an arterial bifurcation or arteriovenous crossing32 (Fig. 16–2A).
What Causes Macroaneurysms?
Macroaneurysms are most commonly observed in the sixth and seventh decades of life,33 which signifies that aging is a predisposing factor. There is also a large predisposition for macroaneurysms to occur in females, and they are commonly associated with hypertension, arteriosclerosis, elevated serum lipids, and lipoprotein abnormalities.33–35 Histopathologic studies of macroaneurysms reveal linear breaks in the arterial wall surrounded by a laminated fibrin-platelet clot, hemorrhage, exudates, lipid-laden macrophages, hemosiderin, and a fibroglial reaction.32,36
How Does a Macroaneurysm Cause Loss of Vision?
Frequently, macroaneurysms are asymptomatic and diagnosed on routine funduscopic examination. When a patient does notice visual changes related to a macroaneurysm, the symptoms may be a gradual decrease or sudden visual loss that correlates with the location of the macroaneurysm and the presence of macular edema, hemorrhage, or both. Macroaneurysms are susceptible to chronic exudation, which can lead to macular edema with a secondary circinate retinopathy from deposition of lipid exudates. In addition, persistent leakage can cause a shallow, serous neurosensory retinal detachment (Fig. 16–2A). This results from the amount of exudation exceeding the clearing capacity of the retina and gravitating toward the fovea. The symptoms therefore depend on the proximity of the macroaneurysm to the macula and the extent that the secondary complications extend into the macular region. Thus, a decline in vision related to leakage from a macroaneurysm is usually mild to moderate with a gradual and chronic onset, but severe vision loss can result from chronic deposition of lipid in the fovea.
The other frequent cause of visual loss related to macroaneurysms is hemorrhage. Bleeding from a macroaneurysm usually results in acute, severe loss of vision. These hemorrhages can be subretinal, intraretinal, or preretinal. Often simultaneous bleeding into all three potential spaces results in an “hourglass” configuration. Extensive accumulation in the preretinal space can result in a significant vitreous hemorrhage.
Acquired retinal arterial macroaneurysms have been studied according to their morphologic characteristics and classified into two groups.37,38 Group I macroaneurysms are predisposed to developing hemorrhage from acute aneurysmal decompensation. These macroaneurysms are usually located on larger arterioles closer to the optic nerve and have a larger average diameter. Group II macroaneurysms are more susceptible to chronic exudation that often involves the macula. These exudative macroaneurysms tend to be located within 3 mm of the fovea and are smaller in diameter.
What Are the Characteristics of a Macroaneurysm on Fluorescein Angiography?
A macroaneurysm may cause partial, complete, or no obstruction of the associated arteriole on FA.39 There is usually complete filling of the macroaneurysm in the early phase, but partial filling occurs when an intraluminal thrombosis is present. Also, associated lipid or hemorrhage can block the hyperfluorescence of the aneurysm. Other secondary microvascular abnormalities may develop near the macroaneurysm, including attenuation of the proximal and distal arteriole, areas of capillary dilation or nonperfusion, microaneurysms, and intraarterial collaterals. Leakage often occurs from the macroaneurysm and occasionally from the associated microvascular abnormalities.32 In some cases, a Z-shaped kink has been described at the site of the microaneurysm following closure.40 The use of ICG angiography can help to identify a macroaneurysm when there is significant hemorrhage.41
How Is a Macroaneurysm Treated?
Although most macroaneurysms will resolve spontaneously with preservation of good vision,33 treatment has been recommended in cases where the vision is compromised (Fig. 16–6) by chronic exudation or hemorrhage involving the fovea39,40 (Fig. 16–2A). Different techniques for treatment have used laser photocoagulation indirectly to the microvascular abnormalities surrounding the macroaneurysm and directly to the macroaneurysm. These treatments are performed with xenon-arc, argon-green, and krypton-yellow lasers.35,40,42 The beneficial absorption of the krypton-yellow laser by hemoglobin and oxyhemoglobin has popularized the direct treatment of macroaneurysms.43–45 The natural history of these lesions is usually benign, however, and treatment with laser photocoagulation is controversial,46 especially because complications of the direct laser photocoagulation can include bleeding from rupture of the macroaneurysm, development of a distal branch arteriole occlusion,44 and visual loss secondary to photocoagulation from treatment of macroaneurysms close to the fovea.
When laser photocoagulation is indicated, a slow burn with a large spot size is used. Careful consideration needs to be taken with macroaneurysms that are within 500 μ of the fovea because the laser treatment may be more harmful than the natural history. An argon-green or krypton-yellow laser with a 500-μ spot size and duration of 0.5 seconds is used to treat the microaneurysm directly with enough power to whiten the lesion.32 The patient should be monitored every 3 months for persistent or recurrent exudation or hemorrhage or until these substances resolve.
In cases where significant subretinal, preretinal, or vitreous hemorrhage is limiting the vision, other treatment modalities may be indicated. Submacular surgery for removal of subretinal blood may provide a better outcome than allowing the hemorrhage to resolve on its own.47 When preretinal bleeding is trapped under the internal limiting membrane or posterior hyaloid, the neodymium:yttrium–aluminum–garnet (Nd:YAG) laser has been used to puncture the membrane and allow the hemorrhage to escape into the vitreous.48 Finally, non-clearing vitreous hemorrhages of unknown cause may be due to an underlying macroaneurysm, and a pars plana vitrectomy for removal of the blood can be both diagnostic and therapeutic.49
Choroidal Detachment (Uveal Effusion)
Choroidal detachment is synonymous with several clinical terms like uveal effusion or ciliochoroidal effusion. In these conditions, detachment and effusion have the same meaning. These terms are anatomic descriptions of a pathologic condition that has several etiologies. An effusion describes an abnormal accumulation of serous fluid in the choroid or outer layer of the ciliary body, which more accurately describes the clinical situation than a detachment. Occasionally, the fluid collects in the suprachoroidal space and causes disruption of the fine connective tissue fibers in this layer, leading to an actual detachment of the choroid.
What Is the Clinical Appearance of a Choroidal Effusion?
A typical choroidal effusion appears as a brownish orange, lobular elevation with smooth, convex surfaces. These effusions have a solid appearance and do not undulate with eye movements, like a rhegmatogenous retinal detachment; however, a serous retinal detachment can develop from a choroidal effusion. Transillumination of the globe is helpful in differentiating a serous collection from hemorrhage or a solid tumor. These elevations can occur anywhere in the posterior segment, but they are more commonly present at the pars plana or peripheral choroid and often allow visualization of the ora serrata without scleral depression. The anterior predisposition results from the connective tissue fibers in the suprachoriodal space being tangentially oriented and longer anteriorly compared with the shorter, perpendicular connections posteriorly. A characteristic four-lobed configuration occurs in large effusions as a result of the choroid–scleral attachments at the vortex vein ampullae.50