Fig. 5.1
Radial section schematic of spiral ganglion neurons in the cochlea. a The spiral ganglion is on left, showing several somas of type I spiral ganglion neurons (SGNs) all connected to one inner hair cell (IHC) in the organ of Corti on right, each via a single myelinated axon called a radial fiber. The radial fibers go through habenulas perforata (HP) to reach the neuropil of the inner spiral plexus (ISP) underneath IHCs in the organ of Corti. See boxed region enlarged in b. T, tunnel of Corti; BM, basilar membrane. One SGN is highlighted in black. SGN anatomical parts are labeled in italic font. b Synaptic transmission initiates a postsynaptic depolarization in the bouton and nonmyelinated fiber that triggers an action potential (AP) at the heminode near the HP (Sect. 5.4.4). An AP then propagates along the peripheral myelinated axon a via nodes of Ranvier to the soma in the spiral ganglion, then along the centrally projecting axon (not shown). c Each SGN receives excitatory synaptic input via one IHC presynaptic active zone, marked by a presynaptic ribbon (R), and surrounded by synaptic vesicles. (Modified from Rutherford et al., 2012). Spike encoding of neurotransmitter release timing by spiral ganglion neurons of the cochlea. The Journal of Neuroscience, 32(14), 4773–4789)
Starting from the periphery, in the organ of Corti, each SGN contacts a single synapse on one IHC via a single postsynaptic bouton (Fig. 5.1). The bouton is connected to the soma via the SGN’s short nonmyelinated segment and longer peripheral myelinated axon. Beyond the SGN soma in the spiral ganglion, the myelinated central axon projects to the brain stem (see Muniak et al., Chap. 6).
As the SGN fiber exits the organ of Corti into the osseous spiral lamina, the cable thickens and the myelin begins just beyond the habenula perforata (HP; Fig. 5.1a, b). APs are likely initiated there, relatively near the IHC–SGN synapses in the neuropil of the inner spiral plexus (ISP; Fig. 5.1b). In the neuropil environment of the ISP between bouton and HP, the SGN fiber is surrounded by other afferent fibers, presynaptic terminals of efferent fibers, and nonneuronal glia-like supporting cells. It is unclear whether this part of the SGN should be called a nonmyelinated axon or a dendrite. The great majority of SGN fibers do not branch (Liberman, 1980). Therefore, in general AP generation in each SGN depends on excitatory input to its postsynaptic bouton from a single presynaptic ribbon-type active zone (AZ) of one IHC (Fig. 5.1c).
5.1.3 The Inner Hair Cell
IHCs are the primary sensory receptors in the organ of Corti. They mediate mechanotransduction through the hair bundle comprised of stereocilia. Ionic current through the bundle drives the receptor potential continuously, depolarizing the IHC to modulate the opening of voltage-gated Ca2+ channels at synapses. IHCs release glutamate at rest and in response to sound, initiating the postsynaptic depolarization that generates spontaneous and evoked APs in SGNs. Each IHC excites multiple SGNs. For example, cochleae of mammals such as mice and rats have around 1000 IHCs and approximately 20,000 SGNs.
In murine species and other mammals as well, the number of SGNs per IHC varies tonotopically (Spoendlin, 1972; Bohne et al., 1982). As illustrated in Fig. 5.1, in the developed ears of cats and mice, for example, each SGN is excited by a single ribbon-type AZ (i.e., a single ribbon synapse on one IHC). Therefore, like the number of SGNs, the number of ribbon synapses per IHC varies tonotopically (Meyer et al., 2009). In general, there are fewer than 10 afferent synapses per IHC in the extreme cochlear base and apex, and 15–30 synapses per IHC in the mid-cochlea. Greater synaptic density correlates with greater hearing acuity for mid-cochlear frequencies. For example, the tonotopic location of peak innervation density corresponds to frequencies of peak behavioral sensitivity (Ehret, 1976).
The 1:1 connection between ribbon synapse and SGN means that each IHC AZ provides the sole excitatory input to its SGN. In this way, each IHC AZ has one private line of communication from ear to brain. The specific sound-response properties of these communication lines differ from each other, depending greatly on mechanisms inherent to the given IHC AZ and its paired SGN. Through these heterogeneous synaptic connections, information diverges from one IHC receptor potential to multiple SGNs with different response properties (Sect. 5.5).
5.1.4 The IHC Ribbon-Type Active Zone
The presynaptic AZ of each afferent synapse is occupied by a synaptic ribbon, a vesicle-tethering presynaptic electron-dense structural hallmark of the IHC–SGN synapse (Smith & Sjöstrand, 1961). Synaptic ribbons, found in cell types that release transmitter in response to graded stimulus-evoked receptor potentials, are composed predominantly of Ribeye (Schmitz et al., 2000; Khimich et al., 2005), a protein with both an enzymatic function (Schwarz et al., 2011) and an aggregating property thought to bind the ribbon together (Magupalli et al., 2008). Each synaptic ribbon has tens of vesicles tethered to it, a fraction of which are also tethered to the plasma membrane of the AZ (Frank et al., 2010). Structurally and molecularly, synaptic ribbons seem to be exocytosis nanomachines (Lenzi & von Gersdorff, 2001; Rutherford & Pangršič, 2012). One hypothesis is that ribbons inexhaustibly support high rates of transmitter release at continuously active sensory synapses by promoting the association of Ca2+ channels with fusion-competent vesicles. However, the complete functions of synaptic ribbons in IHC–SGN sensory encoding remain incompletely understood.
Ribbon-type AZs are large relative to AZs in the brain. Relative to each other, the ribbon-type AZs of hair cells exhibit marked heterogeneity in size. Putatively, differences in AZ size and protein content significantly influence AZ function (Sect. 5.5). In the absence of sound, different SGNs fire APs at mean rates that range from fewer than 1 to greater than 100 APs per second (s−1). These so-called “spontaneous” APs are not generated cell-endogenously. Rather, both spontaneous and sound-evoked APs require the endocochlear potential, IHC depolarization, voltage-gated Ca2+ influx, and glutamate release from the IHC AZ onto its paired SGN bouton (Sewell, 1984; Glowatzki & Fuchs, 2002; Robertson & Paki, 2002).
5.1.5 Voltage-Gated Ca2+ Channels Controlling Exocytosis
Continuous, graded receptor potentials arise from mechanoelectrical and voltage-gated conductances (Corey & Hudspeth, 1979; Roberts et al., 1990). These changes in hair cell transmembrane potential modulate the temporal pattern of synaptic voltage-gated Ca2+ channel activity. Gating of Ca2+ channels modulates synaptic transmission by triggering exocytosis of glutamate from synaptic vesicles. In the cochlea, the receptor potential of each IHC is sampled over time by several SGNs, separately, based on the details of Ca2+ channel activity at each presynaptic AZ. Therefore the synaptic transfer function can differ among synapses stimulated by the same IHC receptor potential.
Hair cells are said to release neurotransmitter continuously because their synapses are never truly at rest. From IHC AZs, glutamate is released in an ongoing temporal sequence of discrete quanta. These packets of glutamate arrive onto the SGN bouton at rates that increase with the level of depolarization of the IHC receptor potential, which changes in response to sound. In IHCs, the voltage-gated Ca2+ channels controlling exocytosis are not prone to use-dependent inactivation. Thus, IHC depolarization produces an increase in Ca2+ channel activity at each ribbon synapse that is sustained for the duration of the depolarization. The opening of just one voltage-gated Ca2+ channel may be sufficient to trigger exocytosis of glutamate onto the SGN postsynaptic bouton (Sect. 5.3).
5.1.6 Abbreviations Used in This Chapter
AMPAR
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor
AP
Action potential
AZ
Active zone
BAPTA
1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid, “fast” Ca2+ chelator
[Ca2+]
Ca2+ concentration
C2
Ca2+-binding protein domain
CaBP
Ca2+ binding protein
CaV1.3
Voltage-gated Ca2+ channel, L-type, pore-forming α-1D subunit
CaVß
Auxiliary ß-subunit of voltage-gated Ca2+ channel
CaVα2δ
Auxiliary α2δ-subunit of voltage-gated Ca2+ channel
CDI
Ca2+-dependent inactivation
CtBP2
C-terminal binding protein 2
EGTA
Ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid, “slow” Ca2+ chelator
EPSC/P
Excitatory postsynaptic current/potential
ex vivo
Experiments in acutely explanted organs
GluA
Glutamate receptor subunit type, comprising AMPARs
HCN
Hyperpolarization-activated, cyclic nucleotide-gated nonspecificationic current (I h)
IHC
Inner hair cell
KV
Voltage-gated K+ channel
m
Apparent Ca2+ cooperativity of exocytosis; from a power function fit to the relationship between exocytosis and Ca2+ influx
NaV
Voltage-gated Na+ channel
PSD
Postsynaptic density
px
Postnatal day x
RRP
Readily releasable pool of vesicles
SGN
Type I spiral ganglion neuron, also called auditory nerve fiber, cochlear nerve fiber, or auditory nerve single-unit
SNARE
Soluble NSF attachment protein receptors, including SNAP, syntaxin, and synaptobrevin proteins
SR
Spontaneous AP rate of a SGN (in the absence of sound)
5.2 Synaptogenesis of IHC and Type I Spiral Ganglion Neuron
5.2.1 Development from Pattern Generator to Sound Receiver
Before the onset of sensory function, IHCs drive patterned APs in the auditory nerve that seem to be required for normal wiring of the auditory brain (Walsh & McGee, 1987; Clause et al., 2014). These presensory APs in SGNs are driven by synaptic transmission, evoked by Ca2+ spikes in immature IHCs (Fig. 5.2a–c). Calcium spikes are regenerative potentials, similar to Na+ APs in neurons. Spike-driven exocytosis in immature IHCs (Kros et al., 1998; Beutner & Moser, 2001) is mediated predominantly by CaV1.3 Ca2+ channels (Brandt et al., 2003; Marcotti et al., 2003). Although mature IHCs do not spike, they use the same type of voltage-gated Ca2+ channels to mediate hearing.
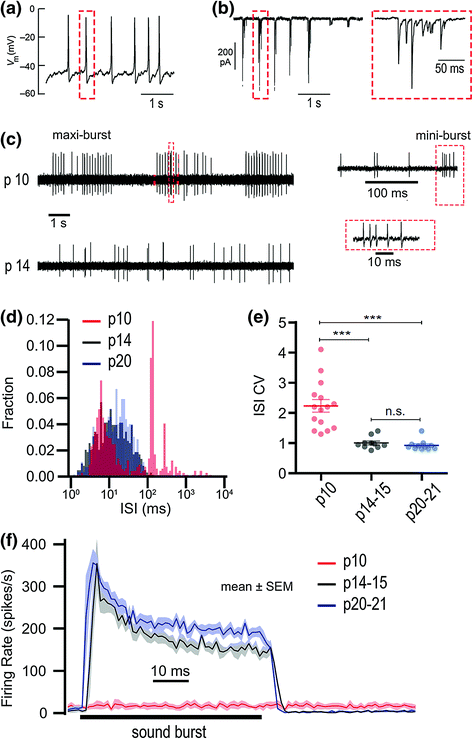

Fig. 5.2
Functional maturation from pattern generator to stimulus transducer. a Patch-clamp recording of a semiperiodic sequence of Ca2+ spikes in a developing IHC. b Left, bursts of EPSCs in a patch-clamp recording of a developing SGN, due to presynaptic Ca2+ spikes in the IHC. Right, one burst is enlarged. Each Ca2+ spike and EPSC burst lasts for approximately 100 ms and consists of several events of exocytosis. c Upper, in vivo SGN spontaneous AP train at p10 showing semiperiodic discharge. Each SGN AP mini-burst (red dashed boxes in c) is evoked by a burst of EPSCs (red dashed box in b) triggered by a Ca2+ spike in the presynaptic IHC (red dashed box in a). The timing between each mini-burst in a maxi-burst (the interburst interval) corresponds to the interspike interval in the immature IHC a. The periods between maxi-bursts represent durations over which the IHC is not spiking. Lower, SGN spontaneous AP train at p14 is relatively irregularly timed. d Interspike interval histograms for SGN spontaneous AP trains at p10, p14, and p20. The distribution of intervals changes from bimodal to unimodal between p10 and p14, and then remains relatively unchanged by p20. e Interspike interval coefficients of variation (CV: variance/mean) for individual SGN recordings (filled circles) and their means (horizontal bars) are significantly less by p14 because of the absence of long intervals that made the bimodal distribution at p10 d. f Mean instantaneous AP rates for repetitions of 50 ms sound bursts (horizontal bar) at the three developmental stages. After the onset of hearing (after p14) the SGNs exhibit an onset response that adapts and approaches a steady-state spike rate of ~200 s−1. (Modified from Wong et al., 2013. Concurrent maturation of inner hair cell synaptic Ca2+ influx and auditory nerve spontaneous activity around hearing onset in mice. Journal of Neuroscience, 33(26), 10661–10666)
By the onset of hearing, at approximately postnatal day 14 (p14) in mice and rats, reduction in number of CaV1.3 channels (Brandt et al., 2003) and upregulation of K+ channels disable regenerative Ca2+ spikes in IHCs. For example, the large-conductance Ca2+– and voltage-activated K+ channels (BK channels) carry a hyperpolarizing conductance that ensures a nonspiking, graded response of the mature IHC transmembrane potential (Kros et al., 1998; Oliver et al., 2006). Another developmentally upregulated K+ channel, KV7.4 (KCNQ4,), defective in human deafness DFNA2 (Kubisch et al., 1999), is partially active when IHCs are at rest, and contributes to setting the IHC resting membrane potential (Oliver et al., 2003). Many aspects of IHC development depend on thyroid hormone signaling (Rüsch et al., 2001; Sendin et al., 2007).
Presensory spiking in IHCs generates bursts of APs in SGNs at p10 in vivo (Fig. 5.2c). These bursts are replaced by mature-looking AP trains in SGNs around p14 (Wong et al., 2013). Experiments in organ of Corti explants have investigated what underlies the temporal pattern of Ca2+ spikes in IHCs, but the mechanism is still under debate. Release of ATP onto IHCs from cells in the developmentally transient Kölliker’s organ may be important for hair cell excitation (Tritsch et al., 2007; Tritsch & Bergles, 2010), although patterned activity proceeded in the presence of inhibitors of ionotropic purinoceptors (Sendin et al., 2014). Alternatively, patterned electrical activity may be intrinsic to the IHC but modulated by ATP release (Johnson et al., 2011).
There is general agreement that presensory activity is likely regulated by inhibition of IHCs via the efferent synapses of olivocochlear neurons. Inhibitory cholinergic transmission could periodically interrupt the IHC depolarization resulting from resting mechanotransduction (Walsh & Romand, 1992; Glowatzki & Fuchs, 2000; Sendin et al., 2014), similar to efferent inhibition of mature outer hair cells (Géléoc & Holt, 2003). However, olivocochlear neurons have somas in the brain, and it is unclear how intrinsic activity in their axons is altered in the excised organ of Corti. Whatever the mechanism, dramatic changes in SGN AP trains between p10 and p14 (Fig. 5.2c–f) are concurrent with IHC synaptic maturation.
5.2.2 Anatomical and Physiological Synaptic Maturation
In mice, SGN fibers reach cells in the differentiating organ of Corti already at birth. The numbers of fibers and synapses in the organ of Corti appear to increase in number during the first postnatal week (Lenoir et al., 1980; Shnerson et al., 1981). Then, presynaptic ribbons and postsynaptic densities (PSDs) decrease in number (Huang et al., 2007, 2012). By p21, IHC–SGN synapses are predominantly mature (Sobkowicz et al., 1982; Grant et al., 2010). This section highlights some structural and functional aspects of synaptic maturation and discusses underlying molecular-anatomical mechanisms.
In the first postnatal week, the IHC Ca2+ current and exocytosis increase as they approach their peak sizes. Then, during the second postnatal week, they decline differently as the efficiency with which Ca2+ influx triggers exocytosis increases. The number of CaV1.3 channels decreases but the smaller Ca2+ current of mature IHCs causes comparably large amounts of exocytosis (Beutner & Moser, 2001; Brandt et al., 2005; Zampini et al., 2010). Immunofluorescence microscopy in fixed tissue (Fig. 5.3a–c) as well as Ca2+ imaging in live tissue revealed that overall CaV1.3 immunoreactivity declined while it accumulated synaptically and the Ca2+ influx increased specifically at the ribbon synapses (Wong et al., 2013, 2014). Thus, maturation involved reduction of extrasynaptic Ca2+ channels not directly coupled to synaptic vesicle exocytosis. Unlike immature IHCs, Ca2+ influx in mature IHCs is largely confined to AZs.
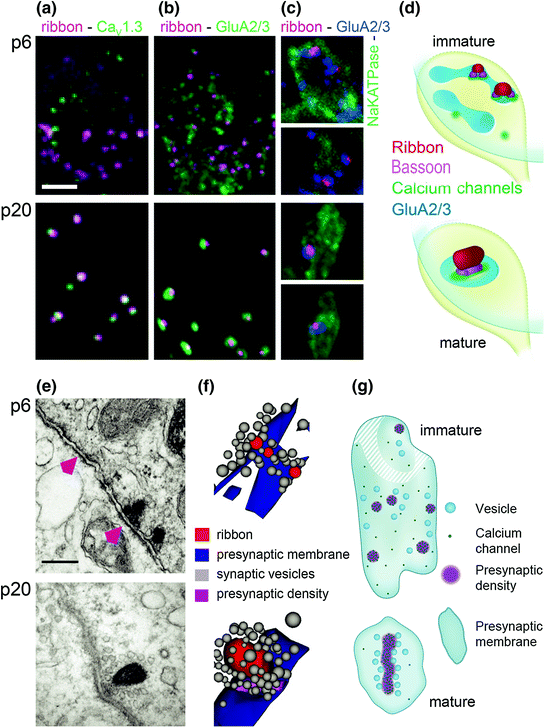

Fig. 5.3
Structural maturation of IHC-SGN ribbon synapses. a Ribbons (anti-CtBP2, magenta) and voltage-gated Ca2+ channels (anti-CaV1.3, green) in one IHC at p6 (immature, upper) and one IHC at p20 (mature, lower). b Similar to a but with AMPA-type glutamate receptors on the green channel (anti-GluA2/3). Between p6 and p20, presynaptic voltage-gated Ca2+ channels and postsynaptic glutamate receptors become restricted to ribbons at IHC–SGN connections. c Ribbons (magenta), GluA2/3 (blue), and NaKATPase (green, labeling SGN boutons) demonstrate refinement of molecular anatomy within the synaptic regions defined by each bouton contact. d Schematic of changes in a–c illustrates the development of 1:1 connectivity between ribbons and SGNs between p6 and p20. e Electron micrographs of IHC–SGN synaptic contacts. At p6, some postsynaptic densities are juxtaposed to presynaptic ribbons while others are not (magenta arrowheads). By p20, almost all synapses have a single, larger ribbon. f Three-dimensional AZ reconstructions of a few small immature ribbons (p6, upper) and one large mature ribbon (p20, lower) anchored to the presynaptic membrane and surrounded by vesicles. g Schematic of IHC–SGN synapses shows a more ordered arrangement of voltage-gated Ca2+ channels and synaptic vesicles upon maturity. (Modified from Wong et al., 2014. Developmental refinement of hair cell synapses tightens the coupling of Ca2+ influx to exocytosis. The EMBO Journal, 33(3), 247–264; Copyright Wiley-VCH Verlag GmbH & Co. KGaA. Reproduced with permission)
Individual IHC–SGN synapses at p6 displayed several small appositions of AZs and PSDs, only some of them occupied by a presynaptic ribbon. These groups of appositions encircled the perimeter of the bouton contact (Wong et al., 2014). Only after the onset of hearing was a single juxtaposed AZ–PSD complex found per SGN bouton (Fig. 5.3).
As AZs and PSDs decreased in number they increased in size, as shown via electron microscopy (Fig. 5.3e, f) and corroborated with confocal immunohistochemistry using antibodies against CaV1.3 Ca2+ channels, GluA2/3 glutamate receptors, and the ribbon protein CtBP2 (Fig. 5.3a–c; Wong et al., 2014). The ratio of ribbons to glutamate receptor puncta increased to nearly 1 by p20, indicating that ribbonless AZs disappeared and the 1:1 connection between ribbons and PSDs prevailed for each SGN.
Ribbons are synaptically anchored via the presynaptic protein bassoon (Khimich et al., 2005). In keeping with the notion that Ca2+ channels cluster underneath ribbons in the presynaptic density (schematized in Fig. 5.3d, g), bassoon and CaV1.3 immunofluorescence closely aligned in elongated stripes at p19 when measured with two-color stimulated emission depletion (STED) microscopy (Wong et al., 2014; Rutherford, 2015). In contrast, before the onset of hearing synaptic CaV1.3 channels formed only smaller spot-like clusters.
Two candidate mechanisms for this anatomical refinement are (1) merging—small AZs or PSDs of a synaptic contact coalesce via interactions of scaffold molecules possibly involving transsynaptic regulation and (2) pruning—small AZs and PSDs are selectively eliminated via protein degradation. Bassoon and the similar protein piccolo each inhibit ubiquitin ligase activity (Waites et al., 2013). Their greater abundance might protect the largest of the initially formed AZs.
These structural refinements are accompanied by developmental changes in synaptic function and changes in molecular composition. At p0, rodent IHCs show relatively little Ca2+ current or exocytosis. As mentioned previously in this section, this is followed by an increase during week 1, then a decrease in Ca2+ current but relatively little decrease in exocytosis during week 2. This increase in efficiency of exocytosis is at least partially due to the positioning of more CaV1.3 channels at AZs and fewer CaV1.3 channels away from AZs (Fig. 5.3a). Moreover, during the first postnatal week exocytosis seems to employ a different molecular program than later in development. For example, otoferlin, essential for exocytosis in mature IHCs, seems dispensable for presynaptic function at this early stage while the neuronal Ca2+ sensor of exocytosis synaptotagmin 2 is temporarily expressed (Beurg et al., 2010; Reisinger et al., 2011). In addition to the increase in efficiency of exocytosis, a change is also observed in the apparent Ca2+ dependence of exocytosis when manipulating the Ca2+ current by changing the number of open channels (Johnson et al., 2005; Wong et al., 2014).
Two mechanisms have been proposed to contribute to changes in the Ca2+ efficiency and apparent Ca2+ dependence of exocytosis in IHCs around the onset of hearing: (1) the intrinsic Ca2+ dependence of exocytosis changes due to a switch in synaptic protein type and/or (2) tightening of the spatial coupling between Ca2+ channels and vesicles at the AZ. A developmental upregulation of synaptotagmin IV has been proposed to underlie the increase in Ca2+ efficiency and the linearization of the apparent Ca2+ dependence of IHC exocytosis around the onset of hearing (Johnson et al., 2010), which might support hypothesis 1.
The intrinsic Ca2+ dependence of exocytosis in mouse IHCs was compared before and after the onset of hearing by measuring the Ca2+-dependent rate constant of the fast component of exocytosis, elicited by step changes of [Ca2+] in response to intracellular Ca2+ uncaging (Wong et al., 2014). The intrinsic Ca2+ dependence was found to be similar, which does not support hypothesis 1. In contrast, when changing the Ca2+ current by manipulating the number of open channels, a developmental difference was found in the apparent Ca2+ dependence (or cooperativity) of exocytosis. The apparent Ca2+ cooperativity of exocytosis was supralinear before hearing onset but near linear in mature IHCs, suggesting a transition from Ca2+ micro-domain control of exocytosis before the onset of hearing to Ca2+ nano-domain control of exocytosis after the onset of hearing. Development of Ca2+ nanodomain control of exocytosis upon maturation implies tightening of the spatial coupling between Ca2+ influx and exocytosis, which supports hypothesis 2. Indeed, the topography of membrane-proximal vesicles, assumed to form the readily releasable pool, is more ordered around presynaptic densities after the onset of hearing (Fig. 5.3g). For more on the subjects of intrinsic and apparent Ca2+ cooperativity as well as Ca2+ microdomain and nanodomain control of exocytosis, see Sect. 5.3.3.
5.3 Presynaptic Mechanisms Encoding Sound
5.3.1 Presynaptic Ca2+ Influx
Unlike typical L-type Ca2+ currents known in other systems to be activated by high-voltage (e.g., in cardiomyocytes of the heart), the L-type Ca2+ currents in hair cells of the inner ear activate at relatively hyperpolarized potentials, exhibit fast activation, and undergo slow and mild inactivation (Fuchs et al., 1990; Roberts et al., 1990; Spassova et al., 2001). In mouse cochlea, the pore-forming alpha subunit is CaV1.3 (Platzer et al., 2000; Brandt et al., 2003; Dou et al., 2004). Without Ca2+ influx through this channel, IHC synaptic exocytosis is abolished (Moser & Beutner, 2000; Brandt et al., 2003) and there is profound deafness in rodents and humans (Zhang et al., 1999; Platzer et al., 2000; Baig et al., 2011).
Hair cells are thusly similar to retinal photoreceptors and bipolar neurons, which also employ L-type Ca2+ channels, have synaptic ribbons, and transduce graded receptor potentials for controlling transmitter release (Barnes & Hille, 1989; Heidelberger & Matthews, 1992; Tachibana et al., 1993). They are different from conventional central nervous system (CNS) synapses that use N- and P/Q-type Ca2+ channels for transmitter release. The number of channels depends on species, developmental stage, and AZ number which varies by tonotopic location but, on average, the number of Ca2+ channels per mature mouse IHC is approximately 1700, with the majority being synaptic (Brandt et al., 2005; Frank et al., 2010; Wong et al., 2014). Evidence from various technical approaches agrees that each AZ of a mature auditory hair cell has on average approximately 100 Ca2+ channels in the frog (Roberts et al., 1990; Issa & Hudspeth, 1996; Rodriguez-Contreras & Yamoah, 2001), turtle (Tucker & Fettiplace, 1995), and mouse (Brandt et al., 2005; Zampini et al., 2013).
IHC CaV1.3 currents have little Ca2+-dependent inactivation (CDI) and activate at relatively negative potentials (Koschak et al., 2001), likely due to the IHC-specific molecular composition of the CaV1.3 Ca2+ channel complex and specific intracellular modulators of its activity. CaVß2 was identified to be the predominant ß-subunit of IHCs that co-regulates channel inactivation and enables sufficient numbers of Ca2+ channels to accumulate at the AZ (Neef et al., 2009). The CaVα2δ subunit(s) involved in the IHC Ca2+ channel remain to be identified. Calmodulin, an obligate mediator of CDI (Lee et al., 2000), is expressed in IHCs, where it regulates CDI of CaV1.3 channels (Grant & Fuchs, 2008). However, calmodulin-mediated CDI of CaV1.3 channels is antagonized by Ca2+ binding proteins (CaBPs), several of which are expressed in IHCs (Yang et al., 2006; Cui et al., 2007). In humans, mutation in the gene coding for CaBP2 results in hearing impairment DFNB93 (Schrauwen et al., 2012).
The list of putative regulators of the IHC CaV1.3 Ca2+ channel complex is steadily growing and includes bassoon, Rab3-interacting molecule (RIM), RIM-binding protein (Hibino et al., 2002), harmonin, and otoferlin. Of the two described mechanisms of interaction between RIM and Ca2+ channels, via RIM–PDZ binding to the proline-rich PDZ interacting motif in the C-terminus of CaVα or via RIM C-terminal C2 domain binding to CaVß, the CaV1.3 Ca2+ channel complex seems to employ only the C2 domain-CaVß binding (Gebhart et al., 2010; Kaeser et al., 2011). Harmonin, a scaffold protein mutated in Usher 1C syndrome (Verpy et al., 2000), is an important organizer of the mechanotransduction machinery in the hair bundle. Harmonin also interacts with CaV1.3 via binding of its second PDZ domain to the proline-rich PDZ interacting motif in the CaV1.3 C-terminus (Gregory et al., 2011). In this interaction harmonin imposes an inhibition on CaV1.3 gating that is relieved by depolarization, thereby contributing to voltage-dependent facilitation of CaV1.3. In addition, harmonin appears to facilitate ubiquitination and proteasomal degradation of CaV1.3, potentially co-regulating the abundance of Ca2+ channels at the presynaptic AZ (Gregory et al., 2011). Finally, proper number and morphology of CaV1.3 Ca2+ channel clustering have been attributed to the presynaptic scaffold protein bassoon and/or its associated supramolecular ribbon nanomachine (Frank et al., 2010; Jing et al., 2013). IHCs from mice lacking function of bassoon protein had fewer ribbons and less Ca2+ channel immunofluorescence at AZs (Fig. 5.4a). Reduction of Ca2+ channel immunoreactivity was greatest at the ribbonless AZs. Because the remaining ribbons were more loosely anchored to the AZ than wild-type ribbons (Fig. 5.4b, c), the extent to which functional deficits were due to lack of bassoon alone versus disruption of the entire ribbon complex is unclear.
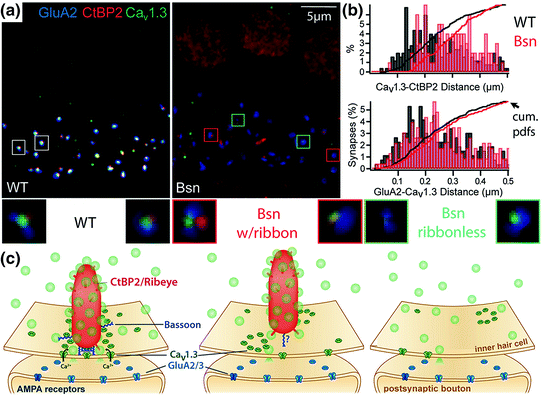

Fig. 5.4
Bassoon anchors the ribbon to the active zone, organizing Ca2+ channels and vesicles. a AMPA receptors (anti-GluA2, blue), ribbons (anti-CtBP2, red), and Ca2+ channels (anti- CaV1.3, green) in mature IHCs of wild-type mice (left, WT) or bassoon-deficient mice (right, Bsn). In Bsn IHCs, a minority of ribbons remained and all AZs appeared to have fewer Ca2+ channels. Small boxes are centered on individual AZs enlarged below for WT synapses (white boxes, left), ribbon-occupied Bsn synapses (red boxes, center), and ribbonless Bsn synapses (aqua boxes, right). b Analysis per AZ: without bassoon (red, Bsn) the distances between CtBP2 and CaV1.3 puncta (upper) are greater than in wild-type (black, WT). The distance between GluA2 and CaV1.3 puncta (lower) was relatively unaffected. Vertical bars are frequency histograms and lines are cumulative probability density functions (cum. pdfs). c Schematic of protein localizations at AZs of WT (left), ribbon-occupied Bsn (middle), and Bsn ribbonless IHCs (right). (Modified from Jing et al., 2013. Disruption of the presynaptic cytomatrix protein bassoon degrades ribbon anchorage, multiquantal release, and sound encoding at the hair cell afferent synapse. Journal of Neuroscience, 33(10), 4456–4467)
Presynaptic Ca2+ influx has been imaged in living hair cells with confocal microscopy in excised inner ear endorgans. On strong depolarization, spatially confined Ca2+ signals rapidly rise and decay with two time constants (Issa & Hudspeth, 1996; Frank et al., 2009), dependent on cytosolic diffusion of free and buffered Ca2+ (Roberts, 1993). Among IHC AZs, a marked heterogeneity of Ca2+ signal amplitude and voltage of half-maximal activation was observed (Frank et al., 2009). This presynaptic heterogeneity may enable the IHC to decompose sound amongst SGNs having different sensitivities, to encode the entire audible range of sound pressures at any characteristic frequency. For more about synaptic heterogeneity, see Sect. 5.5.
5.3.2 Presynaptic Transmitter Release
The ensuing Ca2+ signal drives rapid exocytosis of the readily releasable pool (RRP) of synaptic vesicles at the AZ, which releases glutamate onto the postsynaptic SGN bouton (Sect. 5.4). The IHC AZ has a molecular composition and structure that enables temporally precise release at high rates over long periods of time, as required for normal hearing (Moser et al., 2006; Matthews & Fuchs, 2010; Rutherford & Pangršič, 2012). The synaptic ribbon tethers synaptic vesicles to its ellipsoid-like surface. Moreover, two rows of vesicles align with the presynaptic membrane density at the base of the ribbon (Frank et al., 2010), some tethered to the plasma membrane. Because of their number and their preferential loss during stimulation, these vesicles are often considered to be the ultrastructural substrate of a finite RRP measured physiologically (Moser & Beutner, 2000; Lenzi et al., 2002). The vesicles immediately surrounding and near ribbons are thought to refill the RRP. Vesicle density can differ between high- and low-frequency hair cells, which may be an important tonotopic specialization (Schnee et al., 2005). After fusion with the plasma membrane, vesicles are regenerated via endocytosis in the perisynaptic space (Neef et al., 2014).
Sound-response properties of single auditory nerve units have been measured with extracellular electrophysiological recording of APs from the central axon of single SGNs in vivo (Kiang, 1965; Taberner & Liberman, 2005). The 1:1 connectivity between IHC AZ and SGN makes these recordings extremely valuable for understanding sound encoding at the IHC afferent synapse but also, more generally for neuroscience, because there is probably no other synaptic connection for which an in vivo readout of a single AZ exists. Computational models have used the acoustic signal as input and the APs of individual SGNs as measured output to describe cochlear filter properties mathematically (Weiss, 1966; Meddis, 2006).
To measure exocytosis of synaptic vesicles, the patch-clamp technique was applied to hair cells in inner ear explants (Parsons et al., 1994). Patch-clamp measurements of presynaptic plasma membrane capacitance allow one to monitor exocytosis and endocytosis because fusion and fission of synaptic vesicle membrane with plasma membrane cause increases and decreases, respectively, in surface area that are proportional to capacitance. Applied to the whole cell, measurements of capacitance changes report the summed activity of all synapses. On average, each AZ in a mouse IHC has RRP of about one dozen vesicles that undergoes exocytosis with a time constant of about 10 ms and is replenished with fast and slow time constants of about 140 ms and 3 s (Moser & Beutner, 2000). For single AZ measurements of exocytosis and synaptic transmission with the patch-clamp technique applied to SGN boutons, see Sect. 5.4.2.
Insights into the molecular composition of transmitter release have been provided along three main avenues of investigation: (1) candidate gene approaches driven by knowledge of conventional synapses (e.g., Safieddine & Wenthold, 1999; Nouvian et al., 2011), (2) genetics of human deafness (e.g., Yasunaga et al., 1999; Ruel et al., 2008), and (3) proteomics (Uthaiah & Hudspeth, 2010; Kantardzhieva et al., 2012; Duncker et al., 2013). The synaptic ribbon is composed primarily of the protein Ribeye (Schmitz et al., 2000), a splice variant of the transcriptional co-repressor CtBP2 that has lysophosphatidylacyl-transferase activity (Schwarz et al., 2011). The presence of ribeye at AZs seem to promote endocytic vesicle regeneration, vesicle tethering/docking/priming, and Ca2+-channel clustering in hair cells (Frank et al., 2010; Sheets et al., 2011; Jing et al., 2013; Khimich et al., 2005).
Some components of the presynaptic AZ machinery seem not to be conserved between conventional neuronal synapses and ribbon-type synapses of IHCs, specifically the proteins that mediate Ca2+ sensing and lipid membrane fusion. Otoferlin, a multi-C2-domain ferlin protein specifically expressed in inner ear hair cells is defective in human deafness DFNB9 (Yasunaga et al., 1999) and is currently the best candidate for a vesicular Ca2+ sensor. Exocytosis was nearly abolished in otoferlin-deficient IHCs despite the presence of synaptic vesicles at the AZ (Roux et al., 2006). A definitive conclusion on otoferlin as a Ca2+ sensor of fusion will require mutagenesis of Ca2+ binding sites, biochemical characterization of altered Ca2+ binding, and physiological assessment of the Ca2+ dependence of IHC exocytosis with the mutant otoferlin. In addition to its putative role as Ca2+ sensor, otoferlin seems to facilitate vesicle replenishment (Pangršič et al., 2010).
The core membrane fusion machinery is thought to be conserved at all synapses. In neurons it consists of the soluble NSF attachment protein receptors (SNAREs) synaptobrevin 1 or 2, SNAP25, and syntaxin 1. However, experiments that used neurotoxins and genetic mutations to disable SNARE proteins indicated that IHC exocytosis may operate without neuronal SNARE proteins (Nouvian et al., 2011). Interestingly, otoferlin has been shown to interact with neuronal SNAREs (Roux et al., 2006; Ramakrishnan et al., 2009) but hair cells seem to lack SNARE regulators such as synaptotagmins (Beurg et al., 2010; Reisinger et al., 2011) and complexins (Strenzke et al., 2009; Uthaiah & Hudspeth, 2010). Investigations into the fusion machinery of IHCs are ongoing.
5.3.3 Stimulus–Secretion Coupling
There is an intimate functional relationship and perhaps even direct molecular binding between release-ready vesicles and Ca2+ channels in a proximity of 10–30 nm. From the perspective of the Ca2+-sensing protein on a given release-ready synaptic vesicle, it seems that only one or very few CaV1.3 channels dominate the local [Ca2+] (Brandt et al., 2005; Goutman & Glowatzki, 2007; Graydon et al., 2011). In other words, Ca2+ control of exocytosis appears to operate in nanodomains. Alternatively, vesicle fusion at a given AZ may be controlled by a Ca2+ microdomain (Johnson et al., 2008, 2010; Heil & Neubauer, 2010), in which many Ca2+ channels contribute to the local [Ca2+] signal acting on individual vesicles.
To test the nanodomain versus microdomain hypotheses, the relative number of CaV1.3 channels contributing to exocytosis can be experimentally tested by studying the incremental dependence of RRP exocytosis on Ca2+ influx. The apparent Ca2+ cooperativity m is obtained by fitting a power function to the relationship between exocytosis and transmembrane Ca2+ charge (Q Ca): exocytosis = A(Q Ca) m , where A is the amplitude of the exocytic response and the exponent m is the apparent cooperativity. Different data points are obtained by manipulating the Ca2+ influx, either by changing the number of open channels or by changing the charge through each channel, while depolarizing the IHC for a brief duration to probe the RRP. If m is smaller when manipulating Ca2+ influx by changing the number of open Ca2+ channels than it is when changing the current through a given channel, then Ca2+ nanodomain control of exocytosis is suggested. If m is close to unity then the dependence of RRP exocytosis on Ca2+ influx is near linear. This implies little or no cooperativity of Ca2+ in its coupling to vesicle fusion and suggests nanodomain stimulus-secretion coupling. In the extreme interpretation of nanodomain, one vesicle undergoes exocytosis for each opening of a Ca2+ channel because a sufficient [Ca2+] is reached to saturate the sensor. Ca2+ from further channels would be insufficient. On the other hand, if comparable estimates of m are obtained for these two types of manipulation of Ca2+ influx (changing the number of open Ca2+ channels versus changing the current through a given channel), then m should be similar to the intrinsic biochemical Ca2+ cooperativity of IHC exocytosis (m = 4; Beutner et al., 2001). This would suggest Ca2+ microdomain control (Augustine et al., 1991). In a Ca2+ microdomain control of exocytosis, many channels must open with overlapping effects before [Ca2+] is high enough to evoke fusion.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


