Fig. 4.1
The peripheral innervation patterns and classical morphologies of spiral ganglion neurons are illustrative of their respective functional specializations. a Image of the vestibular and auditory bony structures within the inner ear. (Adapted from Brödel & Malone, 1946.) b Illustration of the anatomical arrangements within the organ of Corti with many type I spiral ganglion fibers innervating a single inner hair cell and individual type II fibers innervating multiple outer hair cells. (Adapted from Held, 1926. Die Cochlea der Säuger und der Vögel, ihre Entwicklung und ihr Bau. In Handbuch der Physiologie.) c–f The classically bipolar and pseudomonopolar spiral ganglion neuronal cell bodies described in vivo are also observed in tissue culture. c Bipolar primary afferent neurons compose the spiral ganglion. (From Gray & Lewis, 1918.) d Pseudomonopolar profiles are highlighted in a longitudinal section through a spiral ganglion of a chick embryo on the 12th day of incubation using the reduced silver nitrate method. The figure illustrates the full range of morphologies, although the relative percentages are not representative. (From Santiago Ramón y Cajal 1995. Histology of the nervous system, Vol 1., Figure 33. Oxford University Press.) e, f Examples of bipolar e and pseudomonopolar f neurons in vitro isolated from their peripheral and central synaptic targets and co-labeled with anti-MAP2 antibody (green) and anti-neuron-specific β-III tubulin antibody (red). (Adapted from Chen et al., 2011, cover illustration) and F. L. Smith and R. L. Davis (in preparation), respectively
A consideration of the role played by type I spiral ganglion neurons in afferent signal transmission would be incomplete without some discussion of the extensive descending efferent control that is exerted at the neuronal postsynaptic membrane. Neurotransmitters released from the lateral efferent system include acetylcholine (ACh), γ-aminobutryic acid (GABA), calcitonin gene–related peptide (CGRP), dopamine, serotonin, adenosine triphosphate (ATP), and opioids such as dynorphin and enkephalin (Dulon et al., 2006; Ciuman, 2010). This list includes both excitatory (ACh, CGRP, dynorphin) and inhibitory (GABA and dopamine) transmitter types (Simmons et al., 2011). Further, combinations of transmitters can reside within the same presynaptic terminals (e.g., ACh and dynorphin), thus complicating an already intricate microchemical environment (Altschuler et al., 1985; Safieddine & Eybalin, 1992). This indicates that the receptor-generated synaptic signal is exquisitely controlled within the first moments of synaptic transmission, even before the action potential is generated at the spike initiation zone. This complex organization at the source of electrical signal generation, together with its innervation pattern, suggests that the type I primary afferent is specialized to precisely shape a high-resolution neural signal that is a subcomponent of the total output of a single IHC.
The type II spiral ganglion neurons are a second, enigmatic class of primary afferents that compose the remaining 5 % of the ganglion. Although little is known about their in vivo responses to sound stimuli, their peripheral synaptic connections, like the type I afferents, also illuminate aspects of their potential role in coding. As described in Chaps. 1 by Dabdoub and Fritzsch and 2 by Goodrich, type II afferents innervate multiple outer hair cells (OHCs; Fig. 4.1b; Spoendlin, 1973; Perkins & Morest, 1975). This classically convergent pathway is designed for signal integration rather than resolution, and has a proposed role in the sensory determinations of threshold detection and pain perception (Spoendlin, 1973; Brown, 1994), two functions that, although divergent in their sensory input, similarly require the specialization of signal integration. What ultimately distinguishes one function from the other is the detection characteristics of the sensory receptors themselves. In this regard, one might expect that the network of OHCs is highly sensitive to sound stimuli due to cellular specializations such as the transduction channels at the tips of the stereocilia, indicating a role in threshold detection. Conversely, because synaptic input from satellite cells may contribute to the synaptic potential generated onto apical type II spiral ganglion neurons (Burgess et al., 1997), it is possible that input is also gathered from high-intensity events, perhaps indicating a role in pain perception. It is possible that both views may be correct with regard to type II neurons, acting either individually or as an overall population and being capable of integrating an array of input with widely divergent intensities, potentially contributing to a broad detection range with low resolution. Thus, investigators have found that whether type II neurons receive, process, and transmit low threshold input, high-intensity stimuli, or both, their endogenous membrane properties reflect the underlying requirement to mediate signal integration across a wide range of sensory input, including prolonged time constants (Reid et al., 2004; Weisz et al., 2014).
A description of the role of type II afferents in sensory processing is also incomplete without a consideration of efferent innervation. In this case, the efferent innervation originates from the medial superior olivary (MSO) complex and is delivered directly to the OHCs (Guinan, 2011). However, what is unique about efferent cochlear controls at the level of the OHCs is that type II neurons themselves exert a postulated feedback control on OHCs through reciprocal synapses (Thiers et al., 2008). Thus, type II afferents may serve as a local efferent regulator, in addition to their role as integrators of multiple sensory receptor inputs.
Beyond the spiral ganglion neuronal classes and their innervation patterns, a concerted effort has been made in recent years to characterize the intrinsic electrophysiological features of these cells. This goal was made possible by in vitro systems that allow access to whole-cell and single-channel patch-clamp recordings and provide a stable environment for manipulating experimental conditions. Although it is necessary to separate the neurons from their peripheral and central targets to determine unequivocally their endogenous characteristics, it is critical that many of their overall features remain unchanged. Evaluations have shown that at the most basic level, spiral ganglion neurons retain their distinctive features. As shown (Fig. 4.1c–f), their bipolar and pseudomonopolar soma shapes are evident, and they elaborate long processes that, in many culture conditions, reiterate their simple in vivo branching patterns (Whitlon et al., 2006). Further, intermediate filaments that distinguish type I from type II spiral ganglion neurons in vivo (Hafidi, 1998; see also Muniak et al., Chap. 6) are retained in vitro (Mou et al., 1998), which makes it possible to separate the firing features of type I from type II neurons even when isolated from their peripheral targets. Moreover, immunocytochemical studies have shown that many voltage-gated ion channels characterized for these neurons in vitro are also detected in postnatal and adult spiral ganglion tissue sections. Thus, although there inevitably will be differences found between the electrophysiologically relevant phenotype of isolated spiral ganglion neurons in vitro and those still connected to their synaptic partners in vivo, these studies allow a more comprehensive view of the capabilities of the neurons on their own so that the role of the spiral ganglion can be assessed as a component part of the auditory neural pathway.
4.2.2 An Unusual Neuron
The functional challenges carried out by the convergent and divergent pathways formed by the primary auditory afferents are met by a class of neurons with an unusual morphological configuration. Compared to the entirety of the nervous system, the type I spiral ganglion neurons that compose the VIIIth cranial nerve are unique because of their bipolar and pseudomonopolar configurations, in which the somata are part of the conduction pathway (Fig. 4.1). In many species, the type I spiral ganglion somata, interposed in the conduction pathway, possesses a unique form of myelin, termed loose myelin (Rosenbluth, 1962), whereas in humans the interposing somata are unmyelinated (Nadol, 1988). Thus, the configuration of this class of primary afferents is unlike that of a typical central neuron in which electrical input is received in the dendrites and integrated at the soma before generating an action potential at the spike initiation zone. Instead, in the spiral ganglion, the action potential is initiated in the type I cells proximal to the sensory receptors and transmitted along the axonal segment, which is interrupted by a large expanse of soma membrane (Hossain et al., 2005). Without specific compensatory electrophysiological specializations, one might expect that this configuration would create an impedance mismatch that leads to action potential failure (Despres et al., 1994), much like that observed at asymmetric axonal branch points (Luscher and Shiner 1990; Debanne et al., 2011). However, the bipolar spiral ganglion somata have multiple morphological specializations, such as the close proximity of surrounding nodes and differential diameter of their central versus peripheral initial processes (Liberman & Oliver, 1984; Spoendlin & Schrott, 1989), which presumably counteract “branch failures” that, if unchecked, would ultimately impede action potential conduction into the CNS.
Why would a high-precision system evolve to interpose the soma in the conduction pathway only to necessarily overcome its presence? One might speculate that these elaborate morphological specializations are designed to filter or modify, rather than block, signal transmission. The electrophysiological significance of the soma is compounded by the fact that spiral ganglion somata sizes are graded along the cochlear contour. Although heterogeneous, neurons in the apex are significantly smaller than those in the base (Nadol et al., 1990). Yet, rather than the soma area differences being rigidly graded, one observes small but systematic size increases from the apex through the mid-basal region, and then the soma area abruptly increases in the most extreme end of the base (Echteler & Nofsinger, 2000). One study that addressed the electrophysiological consequences of this size disparity concluded that soma area is related to action potential filtering (Robertson, 1976). An additional possibility is that the soma size regulates conduction time through the soma (Lawson & Waddell, 1991), which is also dependent on input resistance, the length constant, and membrane area (Johnston et al., 1995). Whether these somatic specializations contribute to filtering, transmission delays, or other aspects of shaping the endogenous electrical profile, the system design appears to support specialized processes separately for low–mid- versus high-frequency regions.
The unique soma placement and presence of loose myelin initially suggest that the soma, itself, is an internodal axonal structure that serves exclusively to transmit signals. However, there is evidence that the soma is electrogenic and therefore potentially capable of integrating signals as well as conducting them (Robertson, 1976). In support of this latter view, microtubule-associated protein 2 (MAP2), a marker of dendrites and somatic integrating regions, was shown to be present in spiral ganglion neuron somata (Chen et al., 2011) in both bipolar (Fig. 4.1e, green/yellow) and pseudomonopolar (Fig. 4.1f, green/yellow) cell types. This staining extends into the processes that emanate from the soma, yet was not observed along their axons (Fig. 4.1e, f red). Thus, despite their axonal location, spiral ganglion neuronal somata possess a dendritic marker, suggesting a distinct functionality that is separated from the postsynaptic dendritic region by a length of myelinated axon. Further, a related protein, CASPR, known to flank the nodes of Ranvier and found within the spike initiation zone (Peles et al., 1997), has also been localized to the somatic region (Hossain et al., 2005), along with multiple types of voltage-gated ion currents (Rusznak & Szucs, 2009). Thus, it appears that spiral ganglion neurons are unusual, not because they lack a soma that can potentially integrate and shape electrical signals, but because this region is morphologically discontinuous from the postsynaptic membrane and primary spike initiation zone.
Further evidence that spiral ganglion somata may contribute to shaping the signals that they ultimately transmit comes from the now well-established presence of somatic voltage-gated ion channels located beneath the loose myelin. A first step toward determining whether resident voltage-gated ion channels affect electrogenicity was performed on neurons from the goldfish (Carassius auratus) saccular nerve, which are bipolar myelinated neurons (Fig. 4.2a, b) that convey sound (Furshpan & Furukawa, 1962). Mechanical microdissection of the myelin (Fig. 4.2c, d) permitted single-channel patch-clamp recordings from the underlying somatic membrane (Davis, 1996). Recordings indeed showed multiple classes of voltage-gated ion channels (Fig. 4.2e–h). Four separate K+ channel types were identified based on their distinctive kinetics, conductances, and inactivation profiles (Fig. 4.2e–g), one of which was the large-conductance, voltage- and calcium-activated potassium (BK) channel (Fig. 4.2g, h). These experiments revealed that voltage-gated channels were functional in the cell bodies of primary auditory afferents, despite their location beneath myelin, and thus highlighted the electrogenic capacity of these neurons. Further, these studies show that the complexity of the electrophysiological profile of the soma membrane goes well beyond the simple combination of nodal Na+ channels and paranodal delayed rectifier K+ channels (Rasband & Trimmer, 2001).
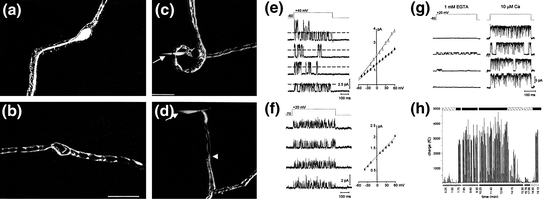

Fig. 4.2
Identification of K+ channels in acutely demyelinated primary auditory neurons. a Bipolar goldfish saccular neuron encased in myelin labeled intracellularly with Lucifer Yellow. b The myelin covering was labeled independently by breaking through the Schwann cell membrane surrounding a bipolar goldfish saccular neuron. c The initial stage of mechanical demyelination. A glass micropipette was used to penetrate the myelin sheath without affecting the neuronal membrane. The arrow indicates the pipette position after the myelin above the soma membrane had been partially removed. d The pipette (arrow) was subsequently threaded between the remaining myelin and neuronal membrane and then lifted to remove the soma from the myelin covering. Arrowhead indicates the edge of the myelin. e–h Evidence for multiple types of somatic voltage-gated K+ channels obtained from patch-clamp recordings of the internodal membrane. e Overlapping openings of different amplitudes indicate the presence of two distinct K+ channel types. Right, current-to-voltage relationships for the small (filled diamond) and large (open triangle) channel openings. The conductances were 18 and 30 pS, respectively, for the small and large conductance channels. f Similar to e but this channel type had much briefer openings. I–V graph to the right indicates a conductance of 14 pS and a reversal potential of −81 mV for this channel. g An inside-out single-channel recording revealed the presence of a large-conductance K+ channel that was activated following elevation of external Ca2+ (compare left side sweeps with the right when 10 μM Ca2+ was added). h Plot of charge (fC) calculated for each sweep over the course of a recording of a BK channel. Below the x-axis (time, min) are the voltage protocols used (hatched bars, −60 mV holding potential, stepped to 0 mV; black bars, −40 mV holding potential, 0 mV step potential; white bars, −60 mV holding potential, +10 mV step potential). Represented above the graph are durations of EGTA application (hatched bar) and 10 μM Ca2+ (black bars). Note the increase in charge with the addition of Ca2+. (Adapted from Figs. 1–3 of Davis, 1996)
4.3 The Basic Firing Patterns
The unusual morphological configuration of the spiral ganglion neuron, in which an electrogenic cell soma lies directly in the signal conduction pathway, makes it imperative to characterize the firing properties of isolated neurons to understand better their contribution to coding. The first intracellular recordings of this type were made from adult spiral ganglion neurons (Santos-Sacchi, 1993). Voltage-clamp traces revealed a transient inward Na+ current, which could be blocked by tetrodotoxin (TTX), followed by an outward current with properties that typified a classic delayed rectifier (Fig. 4.3a1; Santos-Sacchi, 1993). Although current-clamp traces showed an atypical gradation in amplitude, a fully formed action potential was evident at the highest depolarization (Fig. 4.3a2). This basic electrophysiological profile is consistent with standard single-unit recordings in vivo, in which post-stimulus time histograms revealed rapid and dynamic firing patterns in response to pure tone stimuli (Kiang, 1965). These findings prompted the examination of responses to prolonged stimuli to determine whether primary auditory afferents are capable of displaying the more varied properties expected of a sophisticated sensory system that could also potentially account for the nonstandard behavior of some reported single-unit recordings (Kiang, 1990).
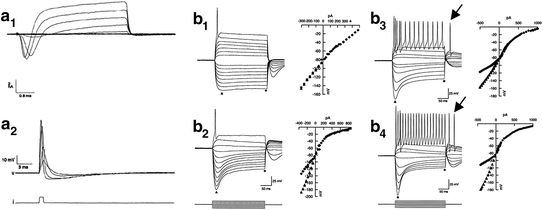

Fig. 4.3
Somatic electrophysiological recordings of spiral ganglion neurons in culture confirm electrogenicity and display a wide range of firing features. a 1 Whole-cell voltage-clamp traces demonstrate the presence of an inward Na+ current and an outward K+ current from an isolated, adult rat spiral ganglion neuron in culture. a 2 Current-clamp recording from the cell shown in the top panel. Increasing amounts of current injection elicit a fast depolarization followed by a rapid repolarization and an after-hyperpolarization that are consistent with an action potential. (From Fig. 2 of Santos-Sacchi, 1993.) b Whole-cell patch-clamp recordings and voltage-current (V–I) relationships for rapidly accommodating (RA) and slowly accommodating (SA) neurons. b 1 Voltage responses of an RA neuron. Depolarizing current injection resulted in a single action potential. Hyperpolarizing current injection (downward-going traces) produced a nearly ohmic response. Together, they result in a nearly linear V–I relationship (right). Current injection is shown below b 2. b 2 Another RA neuron but the voltage sag produced by hyperpolarizing current injection resulted in a nonlinear V–I relationship. The voltage sag is observed as the difference in magnitude between the peak (filled triangle) and plateau (filled diamond). Note the difference in voltage sag magnitude between b 1 and b 2. b 3 and b 4 were similarly analyzed SA neurons. Note the difference in the maximum number of action potentials fired between b 3 and b 4. In response to hyperpolarizing current injection, both neurons fired rebound action potentials (shown by arrows). (From Figs. 7 and 8 of Mo & Davis, 1997b)
Experiments that evaluated murine postnatal spiral ganglion neurons using longer pulse durations expanded the view of the electrophysiological complexity of these cells (Fig. 4.3b1–b4). Some neurons showed a rapid accommodation (RA) profile in which action potentials ceased to fire during a prolonged depolarizing current injection (Fig. 4.3b1, b2; Mo & Davis, 1997b). The majority of the neurons within this class of cells fired only a single action potential even at the highest levels of current injection. The other neurons within this class, while accommodating during the depolarizing stimulus, typically fired fewer than eight action potentials just following the onset of the stimulus (Mo and Davis, 1997b).
Slowly accommodating (SA) neurons, which fired action potentials throughout a prolonged depolarizing current injection, were also detected (Fig. 4.3b3, b4; Mo & Davis, 1997b). Examination of responses to a series of current injections revealed that the maximal number of action potentials a cell was capable of firing varied from neuron to neuron. This variation was largely the result of differences in the maximum firing rate (minimum interspike interval). For example, the two cells illustrated in Fig. 4.3b3, b4, although firing the maximum number of action potentials do so at different rates. Coincident with these depolarization-evoked responses, a distinctive voltage response was also noted in these neurons to hyperpolarizing constant current injections. Some neurons displayed essentially linear responses (Fig. 4.3b1), whereas most others showed a hyperpolarizing sag indicative of the hyperpolarization-activated (I h) cationic current (Fig. 4.3b2–b4; Chen, 1997; Liu & Davis, 2007; Kim & Holt, 2013). As seen from the examples, the magnitude of the hyperpolarizing sag does not appear to correlate with the degree of accommodation, revealing an additional point of electrophysiological diversity (Fig. 4.3b1–b4).
4.4 Voltage-Gated Ion Channels: The Fundamental Building Blocks
The levels of accommodation from mammalian cells, whether categorized as rapidly accommodating (RA) or slowly accommodating (SA), require a greater ensemble of voltage-gated ion channel types than the classic transient Na+ and delayed rectifier K+ channels described by Hodgkin and Huxley (Hodgkin & Huxley, 1952; Hodgkin et al., 1952). Using pharmacological, molecular, genetic, and immunocytochemical approaches, evidence for an abundance of voltage-gated channel types has indeed been found in spiral ganglion neurons.
4.4.1 Ionic Currents Present in Spiral Ganglion Neurons
Initial studies pairing electrophysiology with pharmacological blockers paved the way for identifying the basic underlying voltage-dependent ion channels that shape the membrane properties in spiral ganglion neurons. Multiple voltage-gated K+ channels, through their varied time course and voltage dependence, have a profound regulatory effect on neuronal firing patterns (Hille, 2001). Recordings from spiral ganglion neuron somata using tetraethylammonium (TEA) revealed a delayed rectifier current while experiments with 4-aminopyridine (4-AP) provided evidence for a transient inactivating current (Fig. 4.4a–d; Garcia-Diaz, 1999; Szabo et al., 2002). Further, α-dendrotoxin and dendrotoxin-K, specific blockers of delayed rectifier subtypes KV1.1, KV1.2 and KV1.6, were effective in revealing both low-voltage- and high-voltage-activated K+ currents that contributed to the overall whole-cell outward current (Mo et al., 2002). In addition, application of linopirdine, a blocker of the KV7 family (KCNQ) of channels, identified these currents in dissociated cultures of mouse spiral ganglion (Lv et al., 2010). These observations were further refined to specifically include KV7.4, which was genetically deleted in mice (Beisel et al., 2005).
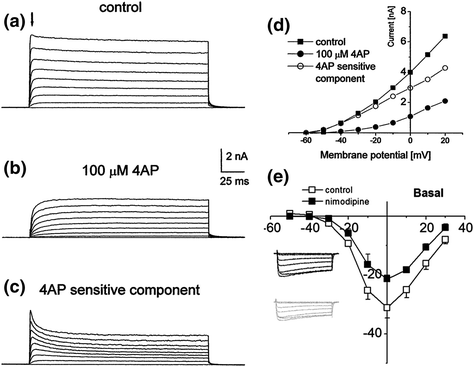

Fig. 4.4
The sensitivity of spiral ganglion neurons to pharmacological blockers. a–d Cultured guinea pig neurons were voltage clamped at −60 mV and stepped to 20 mV in 10-mV increments. a Outward currents prior to 4-AP application and b after 100 µM 4-AP application. c Subtracted traces (b from a) reveals the 4-AP-sensitive currents. d I–V plots of control (filled square), 4-AP (filled circle), and subtracted component (open circle). The scale bar for sweeps is shown between a and b. The measurements for the I–V plots were taken from the peak current (downward arrow in a). (From Fig. 4 of Szabo et al., 2002.) e Sensitivity of basal neurons to nimodipine. I–V plot of whole-cell voltage-clamp recordings from 3-month-old mouse spiral ganglion neurons in culture demonstrates the presence of L-type Ca2+ channels. The amplitude of the control (open squares) was clearly reduced by nimodipine (filled squares). Inset shows the effect of nimodipine before (dark traces) and after (gray traces). (Adapted from Fig. 3 of Lv et al., 2012)
Although the investigations summarized in the preceding text provide evidence for the diversity of the membrane K+ currents, other studies have identified additional current types. For example, of the 10 voltage-gated calcium channel (VGCC) α-subunits identified to date (Catterall et al., 2005), 8 are expressed in the ganglion and 7 were found to be localized to the neurons in differing intra- and intercellular distributions in postnatal and adult animals (Lopez et al., 2003; Chen et al., 2011). Electrophysiological analysis indicates that L-type calcium channels predominate; however, evidence exists for T-, P/Q-, N-, and R-type calcium channels actively contributing to the endogenous membrane properties (Fig. 4.4e; Yamaguchi and Ohmori 1990; Szabo et al., 2002; Lv et al., 2012). Another current type, underlying the aforementioned hyperpolarizing sag, is the cationic I h current, identified in spiral ganglion neurons using the blockers Cs+ and ZD-7288, polymerase chain reaction of of HCN1-4, and genetic deletions of HCN1 and -2 (Kim & Holt, 2013; Liu et al., 2014a). Interestingly, the voltage dependence of activation showed unusually broad heterogeneity under the control of cAMP (Banks et al., 1993; Mo and Davis, 1997a), supporting the variations in hyperpolarizing sag first noted in current-clamp recordings (Fig. 4.3b).
Together, several somatic whole-cell patch-clamp studies have identified a range of voltage-gated currents from the classic rapidly inactivating Na+ current to multiple types of K+ and Ca2+ currents to I h currents. These findings are important because they reveal a functional complexity beyond that expected for the axonally localized neuronal cell body. The abundance and diversity of ion channel types found in neurons will likely increase with additional studies.
4.4.2 Currents that Contribute to the Complex Firing Patterns
To understand better the functional impact of the aforementioned currents on the firing patterns of spiral ganglion neurons, pharmacological blockers were applied during whole-cell current-clamp recordings to evaluate changes in the voltage responses. In this way, the altered firing patterns caused by a pharmacological blocker can be compared to control conditions to determine how a specific current type contributes to the overall neuronal firing.
A pharmacological blocker of BK currents, charybdotoxin, altered the firing patterns of spiral ganglion neurons (Fig. 4.5a), thus confirming the presence of this large conductance voltage- and calcium-activated K+ channel in mammalian, as well as goldfish, primary afferents (Davis, 1996; Adamson et al., 2002b). These experiments support the idea that BK currents exert multiple effects. At voltages close to action potential threshold, BK currents reduced the onset time course, whereas at suprathreshold levels, BK currents increased accommodation.
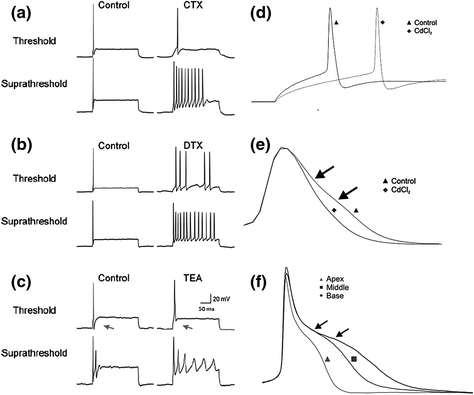

Fig. 4.5
Effects of K+ and Ca2+ channel blockers illustrate the diversity of spiral ganglion neuron firing properties. a Charybdotoxin (CTX), a BK channel blocker, was most effective at suprathreshold stimulation levels as shown by the increase in the number of action potentials. b Dendrotoxin (DTX), a KV1 channel blocker, was effective at both threshold and suprathreshold levels. c TEA application blocked the after-hyperpolarization at threshold (denoted with arrows) and also changed the firing at suprathreshold levels by increasing the number and duration of the action potentials. d, e Effect of Cd2+ on action potential latency at threshold d and action potential duration e. f TEA application revealed tonotopic differences in action potential duration. (a–c from Fig. 6 of Adamson et al., (2002b) and d–f adapted from Figs. 2 and 4 of Chen et al., 2011)
The KV1 blocker α-dendrotoxin also had a profound effect on the endogenous firing patterns of spiral ganglion neurons. Neurons exposed to the toxin showed predominantly SA responses, depolarized resting membrane potentials (RMPs), and lower thresholds, resulting in greater overall excitability (Fig. 4.5b; Mo et al., 2002; Liu et al., 2014b). Conversely, cells exposed to the broad-spectrum blocker TEA showed very little alteration of threshold or RMP, but instead slowed action potential repolarization (Fig. 4.5c). This effect on repolarization has implications not only for individual action potentials but also for prolonging the interspike intervals of neurons that fire more than once. These examples demonstrate that K+ channel types, which are responsible for hyperpolarizing the membrane, do so at distinct voltages, and therefore can affect firing patterns in very different ways.
In addition to effects of K+ currents, depolarization contributed by VGCCs can also alter firing patterns, and, similar to K+ channels, the voltage dependence of a particular Ca2+ channel can affect the mechanism of action. Thus, blocking Ca2+ currents with the broad-spectrum blocker cadmium produced effects at both threshold and suprathreshold voltages (Chen et al., 2011). Cadmium application eliminated a depolarization near threshold to increase response latency (Fig. 4.5d) and abolished a subtle plateau-like potential on the falling phase of the action potential that resulted in a spike with faster repolarization (Fig. 4.5e, arrows). The Ca2+ effect on action potential duration was clearly revealed with the application of TEA. Following an initial rapid repolarization was a pronounced plateau that substantially prolonged the action potential duration (Fig. 4.5f, arrows), which is a hallmark of a Ca2+ spike (Mason & Leng, 1984; Sundgren-Andersson & Johansson, 1998). Thus, despite the rapidity with which these neurons are capable of firing action potentials, buried beneath the powerful repolarizing currents carried by delayed rectifiers and BK channels is a capacity for prolonged depolarization that could have significant effects on signal transmission and, ultimately, neurotransmitter release (Borst & Sakmann, 1999; Yang & Wang, 2006).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


