CHAPTER 169 Tests of Facial Nerve Function
Useful diagnostic tests add information to what is already known, influence the choice of therapy, and ultimately improve clinical outcomes.1 This chapter discusses several tests for evaluation of the facial nerve that can be helpful—as well as several that have not been shown to be helpful—in various clinical scenarios. Intraoperative monitoring of electrical activity of muscles innervated by the facial nerve is by far the most well-established, clinically useful means of assessing facial nerve function, other than physical examination. Outpatient electrical testing of facial nerve function makes sense in clinical settings in which surgical repair or decompression is under consideration, such as after temporal bone fracture or Bell’s palsy. Of note, however, most of the tests described in this chapter have very limited proven clinical usefulness.
Physical Examination
Attempts have been made to standardize measurement of facial function, using techniques as simple as measurements with hand-held calipers and as complex as digital photographic and videographic documentation.2,3 Whereas caliper methods have been shown to demonstrate a high intertest variability, digital photographic methods appear to be much more reliable.4
Several systems of clinical measurement of facial nerve function have been devised, but since the mid-1980s, the House-Brackmann system has been most widely used and has been endorsed by the American Academy of Otolaryngology–Head and Neck Surgery.5 In the House-Brackmann system, grade I is normal function, grade VI is complete absence of facial motor function, and grades II to V are intermediate (Table 169-1).
Table 169-1 House-Brackmann Facial Nerve Grading System
| Grade | Description | Characteristics |
|---|---|---|
| I | Normal | Normal facial function in all areas |
| II | Mild dysfunction | |
| III | Moderate dysfunction | Gross: Obvious but not disfiguring difference between two sides; noticeable but not severe synkinesis, contracture, or hemifacial spasm |
| IV | Moderately severe dysfunction | |
| V | Severe dysfunction | |
| VI | Total paralysis | No movement |
The House-Brackmann system is least ambiguous at its extremes and most prone to intertest variability at its intermediate grades II to V: Multiple regional descriptors of facial movement within each scale can overlap, leading to confusion in assigning the appropriate grade. Alternatively, major functional criteria can be used to create unambiguous categories of progression of deficit correlated with each grade (Fig. 169-1).
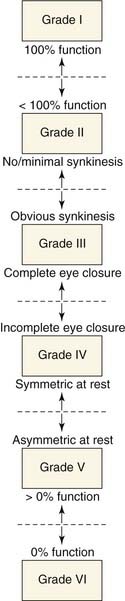
Figure 169-1. Schematic diagram of functional progression in assigning House-Brackmann grade to degree of facial paralysis.
Similarly, examiners do not always agree among themselves, even when using the same formal grading system. In two studies, each with more than 100 patients with various degrees of facial weakness, examiners unanimously agreed on the House-Brackmann grade as often as 80% (three examiners in one study) and as seldom as 33% (five examiners in the other study).6,7 Of course, the probability of disagreement should increase as more examiners are used; the probability of any two examiners disagreeing is as low as 13%8 (on the assumption that in all cases of three-examiner disagreement, two choose one grade and the third choose another grade) and as high as 40% (on the assumption of maximum dispersion of examiner judgments across House-Brackmann categories).9 It would be interesting to know how much of this variability is caused by individual examiners’ test-retest variability (i.e., the tendency of an examiner to assign different scores to the same patient on repeated examination) and how much is attributable to consistent differences among examiners (e.g., between “strict” and “lax” graders).
The wide acceptance of the House-Brackmann system has not prevented many others from proposing new systems. For example, Burres and Fisch10 described a method requiring multiple measurements of movement in different parts of the face; Croxson and colleagues6 showed the expected correlation between Burres-Fisch and House-Brackmann in 42 patients, but considerable variability of Burres-Fisch scores was observed for House-Brackmann grades III and IV. Murtry and coworkers9 proposed and tested the Nottingham system of subjective estimation of movement at each of several points on the face. This method correlated better with House-Brackmann than did Burres-Fisch, and it was simpler to perform. Newer systems include those devised by Ross and associates at Sunnybrook (Ontario, Canada)11 and Coulson and colleagues in Sydney, Australia.12 both of which place particular emphasis on the degree of synkinesis in facial movement. These systems demonstrate good overall test-intertest reliability and correlation with the House-Brackmann system, but subjective evaluation of degree of synkinesis is much more variable.12,13
Neely and associates14 showed the feasibility of computer-assisted image analysis for measuring facial movement. Other objective measurement systems have been proposed by Johnson15 and Jansen16 and their coworkers. These methods are promising, particularly for research and for the assessment of weakness affecting only a single region of the face (e.g., forehead, lower lip).
The House-Brackmann system applied in its strictest sense is well suited for evaluation of long-standing facial nerve dysfunction but not acute facial paralysis.17 Nonetheless, this system can be informally rethought for use in the acute setting, in terms of graduated degrees of facial weakness: grade I, normal or 100% function, grade II, mild weakness or 75% to 99% function; grade III, moderate weakness or 50% to 75% function; grade IV, moderately severe weakness or 25% to 50% function; grade V, severe weakness or 1% to 25% function; and grade VI, complete paralysis or zero function. The obvious disadvantage of this technique is in the reversion to a coarse, imprecise, unweighted method of scaling facial function. However, although not canonical, this method allows for continued use of a six-scale grading system to track facial nerve function through the entire clinical course, from acute onset of the paralysis to long-term outcome.
The best system would be reliable, easy to use, and correlate well with patient self-assessment. Kahn and coworkers18 developed a 15-item questionnaire for patient self-assessment of facial paralysis; questionnaire results in 76 patients correlated fairly well with both House-Brackmann and the Sunnybrook Facial Grading System.11 The American Academy of Otolaryngology–Head and Neck Surgery committee on facial nerve disorders has been in the process of developing a standardized grading system for facial nerve function, but this has not yet been formalized.19 For now, House-Brackmann still seems the most reasonable and widely used choice.
Topognostic Tests
Lacrimal Function
Schirmer’s test has the advantages of simplicity, speed, and economy: The physician places a folded strip of sterile filter paper into the conjunctival fornix of each eye and compares the rate of tear production of the two sides. Normally, the portion of the filter paper in contact with the conjunctiva acts as an irritant, stimulating an increased flow of tears, which are then wicked along the filter paper strip by capillary action. The length of the wetted portion of the strip after a fixed interval (usually 5 minutes) is measured and is proportional to the volume of tears produced. A defect in the afferent (the trigeminal nerve along the opthalmic division, or V1) or efferent (the facial nerve by way of the greater superficial petrosal nerve) limb of this reflex may cause a reduced flow. The reflex is consensual (i.e., a unilateral irritating stimulus in either eye causes tearing in both eyes, and a unilateral sensory deficit in either eye will reduce tearing bilaterally). However, unilateral corneal anesthesia reduces tearing asymmetrically, with a greater reduction on the anesthetized side; therefore, when a sensory deficit is present, presence of bilateral corneal anesthesia should be considered, and stimulation of lacrimation by other noxious stimuli (e.g., inhalation of ammonia) should be performed instead of the conventional Schirmer’s test.8
Schirmer’s test usually is considered positive if the affected side shows less than one-half the amount of lacrimation seen on the healthy side. This corresponds well to the normative data of Fisch,20 who found that 95% of healthy persons had relatively symmetric responses (the lesser response was less than 54% of the greater response). Fisch20 also pointed out that tearing often is reduced bilaterally in Bell’s palsy (perhaps because of subclinical involvement of other cranial nerves). Thus, both the symmetry of the response and its absolute magnitude are important; a total response (sum of the lengths of wetted filter paper for both eyes) of less than 25 mm is considered abnormal.
Fisch20 correlated the results of Schirmer’s test and ENoG (see later under “Electrodiagnostic Testing”) in patients with Bell’s palsy and herpes zoster oticus and found that all cases with 90% or greater degeneration by ENoG had an abnormal result on Schirmer’s test. However, the Schirmer’s test result did not indicate degeneration earlier than ENoG and was abnormal in several cases in which ENoG correctly predicted a good spontaneous recovery.
May21 added Schirmer’s test to the salivary flow test and MST (see later under “Electrodiagnostic Testing”) in a battery of prognostic tests. A decrease to 25% of normal in any of these was associated with a 90% chance of a poor recovery.
Stapedius Reflex
The reflex can be elicited by ipsilateral or contralateral acoustic stimulation or, in cases of bilateral severe hearing loss, by tactile or electrical stimulation. It is absent in 69% of cases of Bell’s palsy (in 84% when the paralysis is complete) at the time of presentation; the reflex recovers at about the same time as for clinically observed movements.22 The prognostic value of this test therefore seems limited.
Taste
Because the chorda tympani carries fibers subserving taste from the anterior two thirds of the tongue, many investigators have studied abnormalities of taste in Bell’s palsy. Psychophysical assessment can be performed with natural stimuli, such as filter paper disks impregnated with aqueous solutions of salt, sugar, citrate, or quinine, or with electrical stimulation of the tongue.23 The latter modality, termed electrogustometry (EGM), has the advantages of speed and ease of quantification. EGM involves bipolar or monopolar electrical stimulation of the tongue, with current delivery on the order of 4 µA (−6 dB) to 4 mA (34 dB). Threshold responses are denoted by the current level’s imparting a subjective sensation of one of the four cardinal tastes or of buzzing or tingling. In healthy persons, the two sides of the tongue have similar thresholds for electrical stimulation, rarely differing by greater than 25%.24 Older studies demonstrated little usefulness of taste testing, because results were abnormal in almost all patients who were in the acute phase of Bell’s palsy; accordingly, EGM could not be used to identify patients with a poor prognosis.25,26 A newer study suggests better predictive value for the test: Of a cohort of 50 patients with complete idiopathic facial paralysis, 18 demonstrated abnormal EGM responses (defined as no response or a threshold difference greater than 20 dB across sides), and 7 demonstrated no response on NET, all of whom were in the abnormal EGM responses group. Of these 7, 5 demonstrated incomplete recovery of facial nerve function on long-term follow-up.23 Taste function appears to recover before visible facial movement in some cases, so if the results of electrogustometry are normal in the second week or later, clinical recovery may be imminent.
Salivary Flow Test
Ekstrand27 stated that reduced salivary flow (less than 45% of flow on the healthy side after stimulation with 6% citric acid) correlates well with worse outcome in Bell’s palsy. Complete or incomplete recovery could be predicted with 89% accuracy. It is unclear whether these data, all collected within 10 days of onset of paralysis, provide an earlier or more reliable indication of prognosis than electrical or other tests.
May and Hawkins28 noted that salivary flow decreases sooner than do threshold changes in NET (see under “Electrodiagnostic Testing” later on) in idiopathic facial paralysis. These workers argued that a flow rate of 25% or less of that on the contralateral, unaffected side is an indication for surgery.
Salivary pH
At least one report29 showed that a submandibular salivary pH of 6.1 or less predicts incomplete recovery in cases of Bell’s palsy. Presumably only the duct on the affected side needs to be cannulated, because in this study, all of the control sides had pH levels of 6.4 or more. The overall accuracy of prediction was 91%. Unfortunately, the reported experience with salivary pH is very limited; it is unknown whether this test gives an earlier prognosis than other tests.
Imaging
Magnetic resonance imaging (MRI) with intravenous gadolinium contrast has revolutionized tumor detection in the cerebellopontine angle and temporal bone and is currently the study of choice when a facial nerve tumor is suspected (e.g., in a case of slowly progressive or long-standing weakness).30 However, enhancement also occurs in most cases of Bell’s palsy and herpes zoster opticus, usually in the perigeniculate portions of the nerve.31,32 This enhancement may persist for more than 1 year after clinical recovery; can be distinguished from neoplasm by its linear, unenlarged appearance; and has no apparent prognostic significance. Computed tomography (CT) is valuable for surgical planning in cholesteatomas and temporal bone trauma involving facial nerve paralysis but probably is less useful than MRI in the investigation of atypical idiopathic paralysis. Unfortunately, even MRI fails to detect a substantial number of malignant parotid tumors.33
MRI shows the greatest utility in predicting location and depth of parotid gland tumors, but even in this capacity it is no better than simple manual palpation alone. The MRI appearance of tumor indistinct from surrounding parotid parenchyma has a positive predictive value for malignancy of only 0.48; accordingly, fine needle aspiration biopsy continues to be the “gold standard” modality for preoperative evaluation of parotid masses.34
Pathophysiology
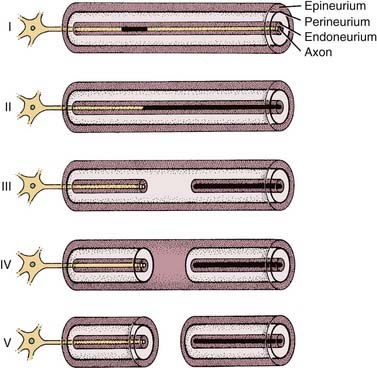
Sunderland35 provided a simple, five-category histopathologic classification of peripheral nerve injury based on a schematic framework proposed by Seddon36 (Fig. 169-2). Some investigators have since expanded this classification to include a sixth category involving mixed forms of injury proposed by MacKinnon and associates.37 This classification of nerve trunk injury is helpful in understanding and interpreting the results of electrical tests.
In classes II to VI, once wallerian degeneration has occurred, electrical stimulation of the nerve distal to the lesion will fail to produce a propagated action potential and muscle contraction. However, before axonal degeneration, the distal segment is still electrically stimulable. Wallerian degeneration begins immediately after injury but progresses slowly. Histopathologic degeneration of the distal segment becomes apparent approximately 1 week after insult and continues for the ensuing 1 to 2 months.38 In the case of the facial nerve, this delay in degeneration results in continued electrical stimulability of the distal segment for up to 3 to 5 days after injury.39 Thus, during these first days after an insult, electrodiagnostic testing of any form cannot distinguish between neurapraxic and neurodegenerative injuries.
Electrodiagnostic Testing
Nerve Excitability Test
The simplest and best-known test for facial nerve degeneration is the NET introduced by Laumans and Jonkees.40 The stimulating electrode is placed on the skin over the stylomastoid foramen or over one of the peripheral branches of the nerve, with a return electrode taped to the forearm. Beginning with the healthy side, electrical pulses, typically 0.3 msec in duration, are delivered at steadily increasing current levels until a facial twitch is noted. The lowest current eliciting a visible twitch is the threshold of excitation. Next, the process is repeated on the paralyzed side, and the difference in thresholds between the two sides is calculated.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree