Surgical Management of Intraocular Tumors
Surgical Management of Intraocular TumorsSurgical Management of Intraocular Tumors
Surgical management of intraocular tumors has been mentioned in several parts of this book, but specific steps have not been illustrated. Techniques illustrated here include fine needle aspiration biopsy (FNAB), application of radioactive plaque, and surgical tumor removal by iridectomy, partial lamellar sclerouvectomy, enucleation, and orbital exenteration (1, 2, 3, 4, 5, 6, 7, 8, 9, 10). Laser photocoagulation and cryotherapy are discussed elsewhere (1,2) and are illustrated earlier in this book as they apply to specific lesions,
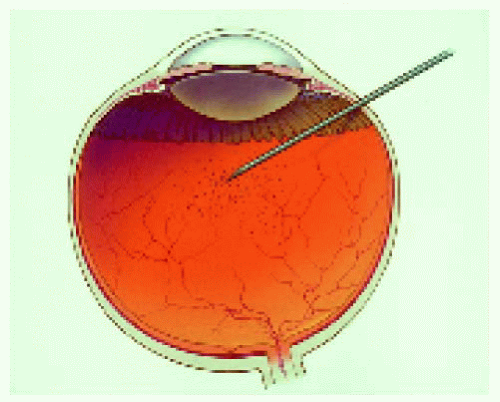
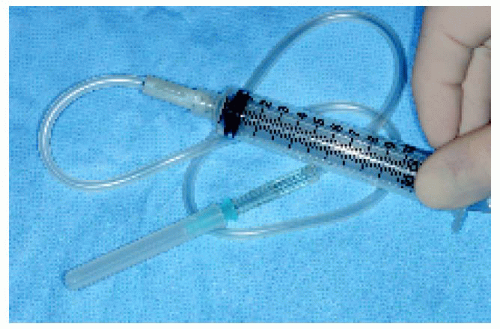
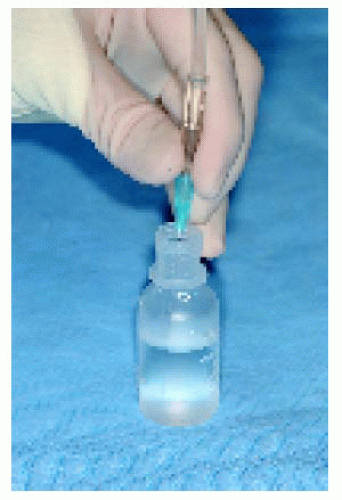
Intraocular FNAB is a method of diagnosing selected lesions that defy an accurate diagnosis using less invasive diagnostic modalities (3). It is a rather difficult procedure that requires the cooperation of an experienced ocular oncologist and cytopathologist (3). It should be reserved for selected cases in which an accurate diagnosis will influence the therapeutic choice. It is performed by passing a fine needle into the suspicious tissue and obtaining cells for special preparation and cytologic analysis. In the case of an iris lesion, the needle is passed through the corneoscleral limbus directly into the tumor, using the surgical microscope for better visualization. For posteriorly located lesions, the needle is passed through the pars plana and vitreous and into the tumor, using either indirect ophthalmoscopy or the surgical microscope for guidance. Although false results sometimes occur, it is an accurate diagnostic procedure in most cases (3).
Selected iris tumors can be removed by partial iridectomy. Selected ciliary body or choroidal tumors can be removed by techniques of partial lamellar sclerouvectomy. It is used mainly for melanoma, leiomyoma, and tumors or the pigmented or nonpigmented ciliary epithelium (4,5). Enucleation is indicated for advanced malignant tumors such as retinoblastoma and uveal melanoma that cannot be successfully managed by other methods (2,6,7). A gentle technique should be employed in all cases, and the method may vary depending on whether the tumor is a melanoma (2) or a retinoblastoma (6). Orbital exenteration, particularly the eyelid-sparing technique, is reserved for some advanced tumors, particularly uveal melanomas with large degrees of extraocular extension (7,8).
The techniques employed are subsequently illustrated and discussed in more detail in the references cited.
Selected References
1. Shields JA, Shields CL. Diagnostic approaches to intraocular tumors. In: Shields JA, Shields CL, eds. Intraocular Tumors. A Text and Atlas. Philadelphia: WB Saunders; 1992:11-23.
2. Shields JA, Shields CL. General principles of management. In: Shields JA, Shields CL, eds. Intraocular Tumors. A Text and Atlas. Philadelphia: WB Saunders; 1992:25-43.
3. Shields JA, Shields CL, Ehya H, et al. Fine needle aspiration biopsy of suspected intraocular tumors. The 1992 Urwick Lecture. Ophthalmology 1993;100:1677-1684.
4. Shields JA, Shields CL. Surgical approach to lamellar sclerouvectomy for posterior uveal melanomas. The 1986 Schoenberg Lecture. Ophthalmic Surg 1988;19:774-780.
5. Shields JA, Shields CL, Shah P, et al. Partial lamellar sclerouvectomy for ciliary body and choroidal tumors. Ophthalmology 1991;98:971-983.
6. Shields JA, Shields CL, De Potter P. Enucleation technique for children with retinoblastoma. J Pediatr Ophthalmol Strabismus 1992;29:213-215.
7. Shields JA, Marr BP, Shields CL. Enucleation for uveal melanoma. In: Albert DM, Jakobiec FA, Azar DT, et al., eds. Principles and Practice of Ophthalmology, 3rd ed. Philadelphia: WB Saunders, 2007. In press.
8. Shields JA, Shields CL, Suvarnamani C, et al. Orbital exenteration with eyelid sparing: indications, technique and results. Ophthalmic Surg 1991;22: 292-297.
9. Shields JA, Shields CL. Massive orbital extension of posterior uveal melanoma. J Ophthalmic Plast Reconstr Surg 1991;7:238-251.
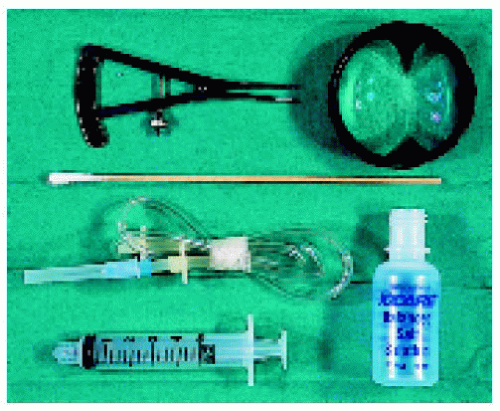
▪ Fine Needle Aspiration Biopsy: Instrumentation and Technique
The instrumentation, techniques, limitations, complications, and results for FNAB of suspected intraocular tumors and inflammations are reported in more detail in the references cited and are only illustrated briefly here.
1. Shields JA, Shields CL, Ehya H, et al. Fine needle aspiration biopsy of suspected intraocular tumors. The 1992 Urwick Lecture. Ophthalmology 1993;100:1677-1684.
2. Shields CL, Manquez ME, Mashayekhi A, et al. Fine needle aspiration biopsy of iris tumors in 100 consecutive cases. Technique and complications. Ophthalmology 2006;113:2080-2086.
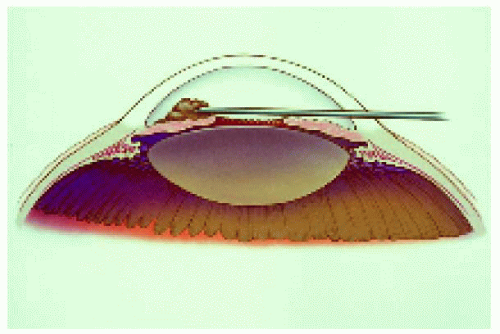
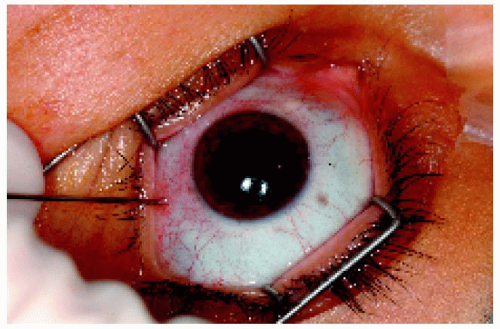
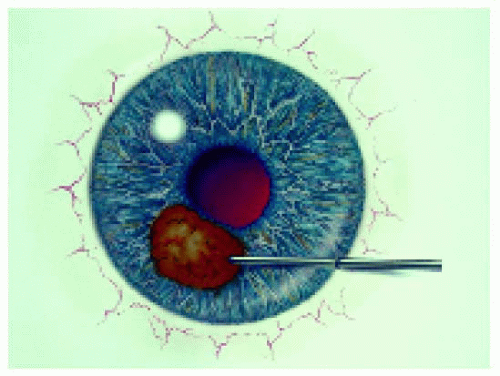 Figure 25.2. Translimbal, transaqueous approach for iris lesions, front view. The surgical microscope is generally used for this technique. |
▪ Fine Needle Aspiration Biopsy of Anterior Segment Lesions
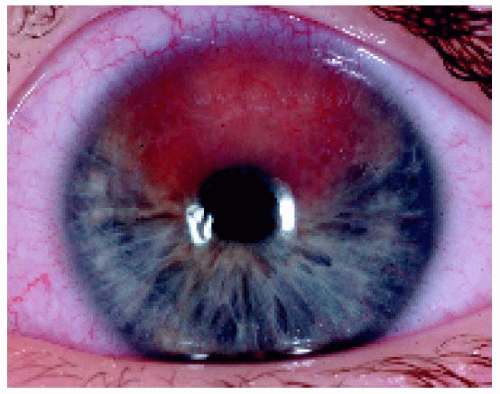
Examples are shown of metastasis lung cancer to the iris and a primary iris melanoma.
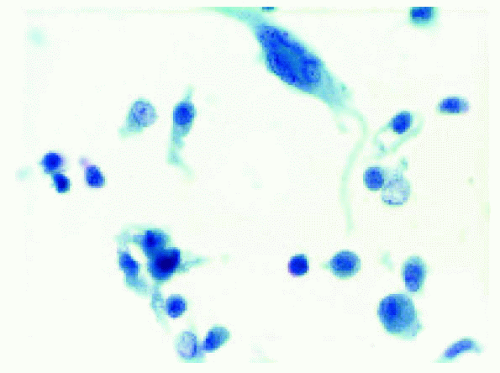 Figure 25.8. Cytopathology, showing histiocytes and a giant cell, findings compatible with juvenile xanthogranuloma. |
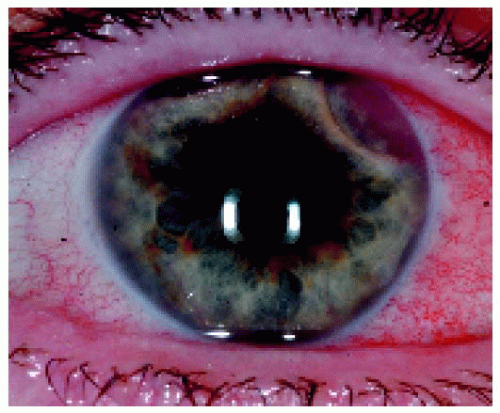 Figure 25.9. Two peripheral iris masses in a 63-year-old man, one superotemporally and one superonasally. |
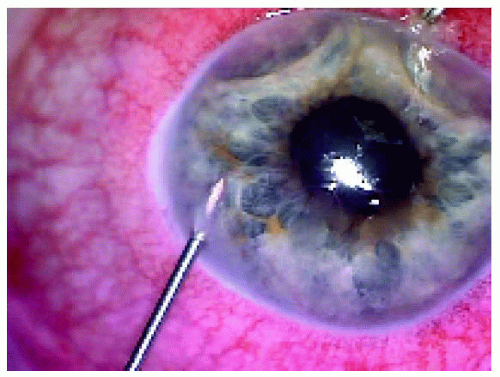
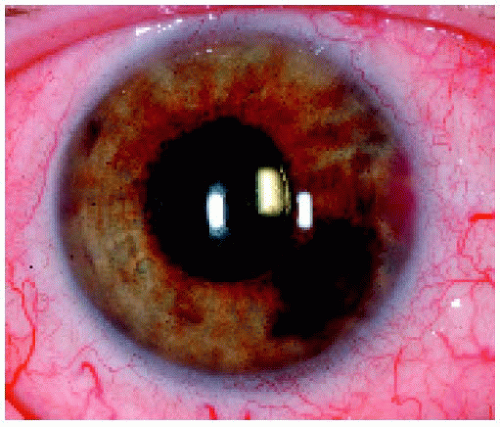 Figure 25.11. Pigmented iris lesion with tumor seeding on the iris surface in a 58-year-old man. Fine needle aspiration biopsy was performed. |
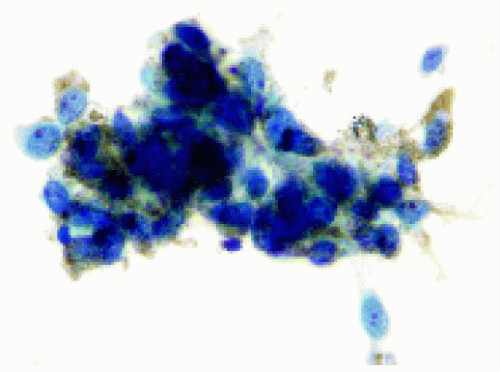 Figure 25.12. Cytopathology of fine needle aspiration biopsy on the lesion shown in Figure 25.11, revealing pigmented spindle and epithelioid melanoma cells. (Papanicolaou x200.) A radioactive plaque was applied. |
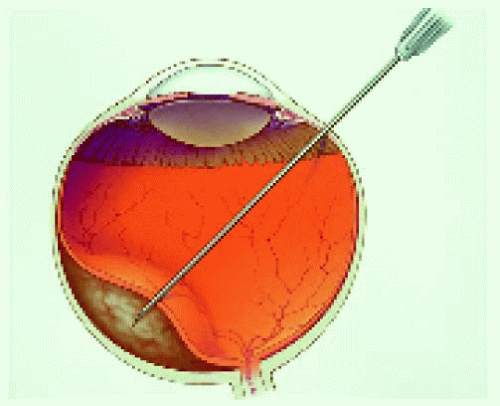
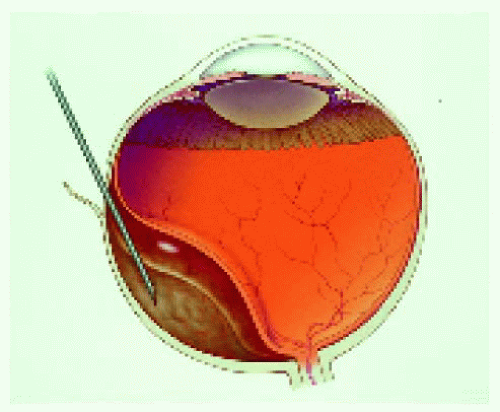
▪ Fine Needle Aspiration Biopsy of Posterior Segment Lesions
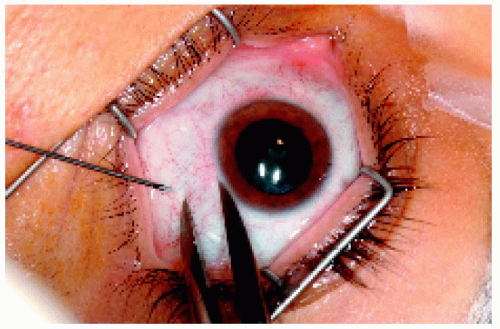 Figure 25.15. Calipers set at 4 mm to ensure entrance through the pars plana by the 25-gauge needle. |
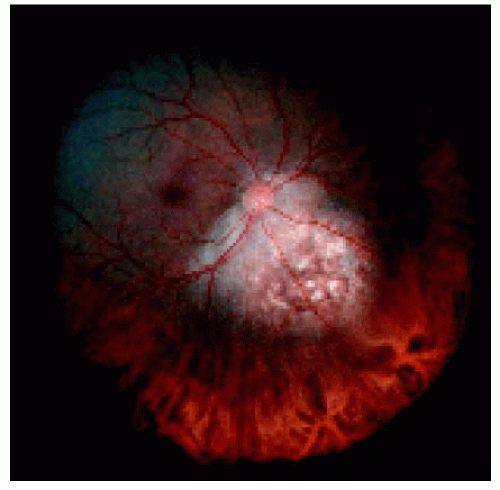
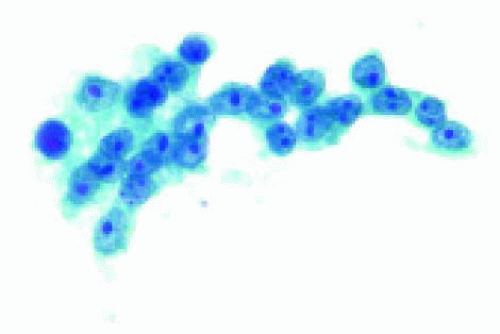 Figure 25.18. Cytopathology of an aspiration of the lesion shown in Figure 25.13, demonstrating cells compatible with choroidal metastasis from breast cancer. |
▪ Plaque Radiotherapy for Intraocular Tumors: Plaque Design and Application
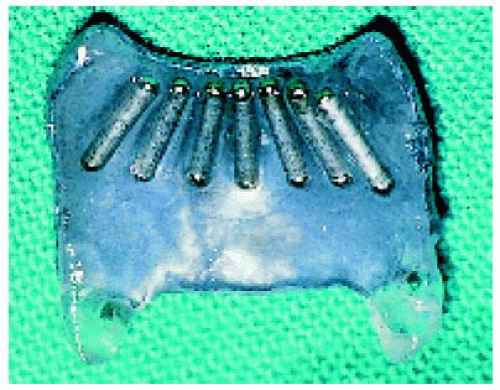
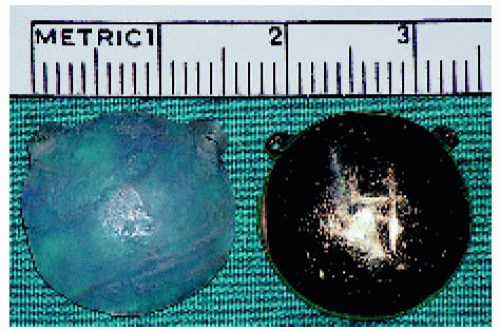 Figure 25.19. Standard round, 15-mm iodine-125 plaque. The dummy plaque is to the left and the shielded side of the active plaque is to the right. |
 Figure 25.20. Opposite side of the active shielded plaque, showing the iodine-125 seeds. The side shown here is directed toward the sclera at the time of plaque application. |
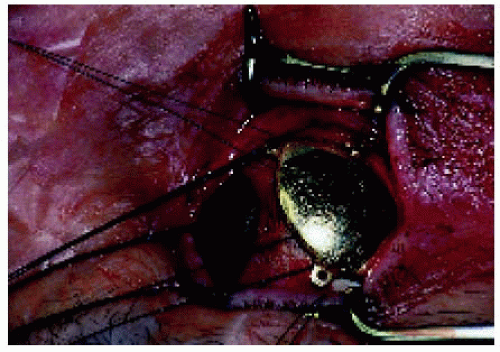
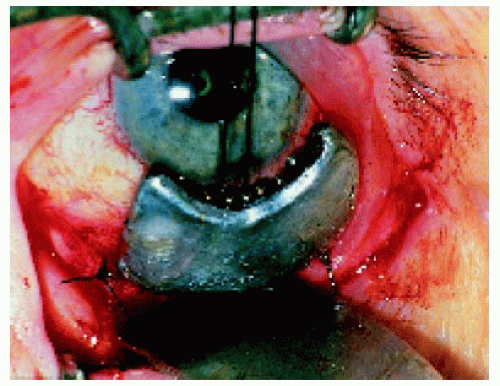 Figure 25.24. The plaque shown in Figure 25.23 being positioned at the time of surgery. |
▪ Application Technique of the Radioactive Plaque
Brachytherapy, using a radioactive plaque sutured to the sclera over the base of the intraocular tumors, is used most often for selected malignant tumors such as uveal melanoma, retinoblastoma, and uveal metastasis. It can be used under special circumstances for circumscribed choroidal hemangioma, retinal vascular tumors, and perhaps other intraocular lesions. The use of radioactive plaques for these tumors is illustrated under the specific chapters in this book.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree