CHAPTER 167 Surgery for Vestibular Disorders
Procedures for Benign Paroxysmal Positional Vertigo
Benign paroxysmal positional vertigo (BPPV) most often affects the posterior semicircular canal. Variations of this condition affecting any of the three semicircular canals have now been recognized, as discussed in Chapters 164 and 165. BPPV seems to be caused by displaced otoconial debris within the membranous labyrinth. The original pathogenic hypothesis proposed that otoconial debris became lodged within the ampulla of the posterior semicircular canal (cupulolithiasis), rendering it sensitive to gravity. In the early 1990s, evidence accumulated to support the concept of otoconia floating freely in the membranous canal (canalithiasis).1–3 These particles within the semicircular canal are purported to cause a transient flow of endolymph after a change in head position, producing a deviation of the cupula and triggering a change in the neural firing rate in the vestibular nerve. The brain perceives this change as rotation in the plane of the semicircular canal, with resulting vertigo and nystagmus. This theory is largely confirmed by the favorable results of particle repositioning maneuvers and the posterior canal occlusion procedure. It also better satisfies the conceptual framework for the biomechanics of BPPV originally suggested in the 1970s by McCabe and Ryu.4 Although cupulolithiasis may certainly result in vestibular symptoms, this entity occurs much less frequently.
Before surgical procedures for BPPV are considered, it should be recognized that this condition has a high rate of spontaneous resolution and also is highly responsive to management by physical therapy interventions. Surgery is rarely required and should be reserved for refractory or multiply-recurrent cases. Nonsurgical management options5 include traditional habituation exercises, Brandt-Daroff exercises, the liberatory maneuver of Semont, and particle repositioning techniques popularized by Epley and Parnes. A detailed discussion of these management options is provided in Chapters 163 and 165.
Singular Neurectomy
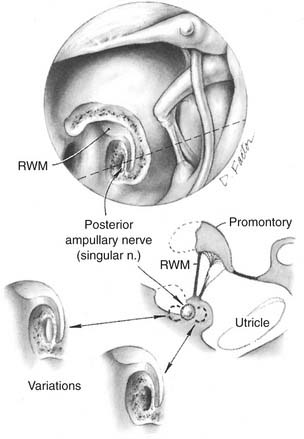
Gacek and Gacek6 have advocated the singular neurectomy procedure for BPPV. This procedure involves selective transection of the branch of the inferior vestibular nerve innervating the posterior canal ampulla. The nerve is interrupted within the singular canal, which travels from the posterior portion of the internal auditory canal to the posterior canal ampulla (Fig. 167-1). This procedure is a highly rational choice for management of intractable posterior canal BPPV, because it is safer and more specific than a complete section of the vestibular nerve. However, this procedure is demanding technically and should not be undertaken unless the surgeon has mastered the approach in the temporal bone dissection laboratory. Gacek has performed 74% of the reported cases in the world literature, with preservation of hearing in 97% of the operated patients; other investigators report poor hearing results with this procedure.7
For this procedure, a tympanomeatal flap is elevated using a transcanal approach, and the bony lip of the round window niche is removed to visualize the round window membrane itself. The surgeon approaches the singular canal by drilling at a site inferior to the round window. Ideally, the nerve is transected just before its point of entry into the ampulla. The ampulla itself and the vestibule are at risk for injury during the procedure, and injury of either structure may result in severe vertigo and sensorineural hearing loss. One study suggested that the nerve measures approximately 5 mm in length and is best accessed by drilling approximately 0.7 mm posteroinferiorly to the posteromedial margin of the round window membrane after saucerization of the niche.8 This study also found that whereas the average depth of the nerve was 1.5 mm, the posterior canal ampulla, the cochlear aqueduct, and the inferior cochlear vein were all within 2 mm of the preferred drilling point, which highlights the dangers of this approach. Another study showed that the nerve was always at least 0.7 mm from the edge of the exposed round window membrane, with an average depth of 1.3 mm.9 Transtympanic access was believed to be straightforward in only 20% of specimens and impossible in 28%. This procedure has largely been supplanted by the technically simpler semicircular canal occlusion procedure.
Posterior Semicircular Canal Occlusion
Parnes and McClure2 introduced the concept of surgical occlusion of the posterior semicircular canal for the relief of BPPV. Conceptually, the canal occlusion procedure is based on the assumption that otoconial debris, originally from the macula of the utricle, settles into the endolymphatic space of the posterior semicircular canal and produces the signs and symptoms of BPPV.
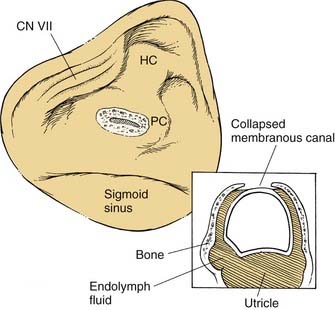
The surgery is conducted to obstruct the bony lumen of the dome of the posterior semicircular canal without disruption of the membranous labyrinth (see Fig. 167-2). After a complete mastoidectomy is performed, the horizontal semicircular canal is identified. The dome of the posterior semicircular canal is then identified between the horizontal canal and the posterior fossa dural plate. The lumen of the posterior canal is approached with a diamond drill until it is visible as a dark line through a thin layer of residual bone. The lumen is opened by removing the remaining thin bone over the canal lumen using delicate instrumentation, with care taken to avoid damaging the membranous canal or suctioning fluid from the perilymphatic space. The perilymph is gently removed by placing a wick of absorbent material near the labyrinthotomy site, causing the membranous semicircular canal to collapse against the deep wall of the bony canal lumen. The canal may then be occluded by placing small plugs of periosteum into the canal, one toward the ampulla and one toward the common crus. The labyrinthotomy is repaired by filling the lumen with wet bone dust harvested during the mastoidectomy. This material can be secured in position by placing bone wax or a piece of connective tissue over the dome of the canal.
The patient may have mild to moderate unsteadiness after the procedure but usually is fit for discharge from the hospital within 24 to 48 hours. A small risk of reactive labyrinthitis and sensorineural hearing loss is associated with the need to open the bony labyrinth. Approximately 15% of patients have protracted postoperative dysequilibrium, but vertigo control rates approach 100%, and hearing loss is uncommon.10 Dizziness Handicap Inventory scores are reduced significantly, and patient satisfaction is 85%.11
Surgery for Superior Semicircular Canal Dehiscence
Minor12 first described the syndrome of superior semicircular canal dehiscence (SSCD) in 2000. Further detailed characterization and recommendations for treatment have evolved since that ground-breaking report. Morphometric evidence indicates that SSCD may be associated with generalized reduction in the thickness of the squamous portion of the temporal bone,13 which would predispose affected patients to middle fossa superior semicircular canal defects and explain the frequent association with natural dehiscences of the tegmen tympani and mastoideum. Aural pressure and unusual sensitivity to bone-conducted sounds are common complaints. Patients frequently report autophony, so this condition must be distinguished from the patulous eustachian tube. Vestibular symptoms typically are transient and are induced by loud sounds or any activity that leads to a transient increase in intracranial pressure. Some patients also report chronic dysequilibrium. Eye movements that are characteristic of superior semicircular canal stimulation may be seen in response to applied pressure stimuli—specifically, a torsional downward deviation of the eye. Vestibular evoked myogenic potential (VEMP) thresholds are reduced and amplitudes elevated in the setting of open dehiscence. Auditory findings include an apparent conductive or mixed hearing loss with a significant low-frequency conductive component. The apparent conductive hearing loss is believed to be caused by shunting of air-conducted acoustic energy away from the basilar membrance within the cochlea toward the dehiscence, which is functioning as a third mobile window.14 It is essential to distinguish this condition audiometrically from otosclerosis by requesting testing for supranormal thresholds (often better than 0 dB) on bone conduction audiometry, and for the presence of acoustic reflexes.15
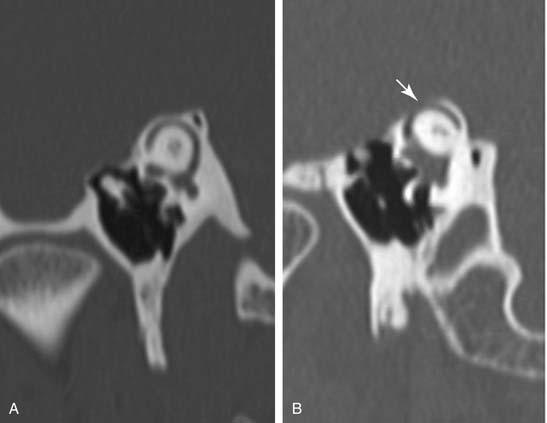
In cases in which clinical symptoms and test results suggest this diagnosis, high-resolution computed tomography (CT) of the temporal bone is indicated in an effort to visualize a dehiscence of the canal at the dural interface in the floor of the middle cranial fossa. These studies should be performed in a parasagittal plane orthogonal to the superior canal (Fig. 167-3). Generally, the dehiscence is repaired through a middle cranial fossa craniotomy, and evidence is mounting that disrupting the canal by obstructive plugging is superior to a simple repair (“resurfacing”) of the defect.16 Plugging of the SSCD reliably relieves vertigo symptoms and reduces the gain of the angular vestibulo-ocular reflex (VOR) by an average of 44% for the superior canal, along with a modest 10% reduction for the ipislateral posterior canal.17 The procedure will resolve most of the auditory symptoms, but results are less predictable than those for vestibular symptoms. Care should be taken to avoid suctioning perilymph from the dehiscence during surgery; plugging may be accomplished with tissue plugs supported by bone chips or bone paté. In carefully selected patients, it may be possible to repair the dehiscence using a transmastoid approach, obviating the need for a craniotomy.18
Repair of Perilymphatic Fistula
Perilymphatic fistula is a controversial diagnosis that centers on the phenomenon of perilymph leaking from the inner ear into the middle ear cleft through the oval window, round window, or other abnormally patent fissures in the bony labyrinth.19 A perilymph fistula may develop after stapedectomy surgery, penetrating middle ear trauma, head trauma, or barotrauma.20 Uncertainty regarding the clinical criteria for the diagnosis and the inability to document the presence of a microfistula at surgery contribute to the problematic nature of this diagnosis.21 Most authors agree that acute perilymph fistulas generally heal spontaneously, so a 2- to 3-day period of bedrest initially is appropriate. With suspected fistula after penetrating trauma, early exploration is indicated to preserve residual hearing, unless complete hearing loss is already present at diagnosis. In suspected chronic cases, a trial of vestibular rehabilitation before middle ear exploration may be appropriate if the diagnosis is uncertain, particularly if the hearing is stable.
Some investigators have attempted to use β-2 transferrin in fluids collected from the middle ear to support a clinical diagnosis of perilymph fistula, but results have been conflicting using this protein as a marker for perilymph.22,23 Unpublished data from my laboratory suggest that assays for β-2 transferrin used for identification of cerebrospinal fluid leak are not sensitive enough to detect this protein in only 1 to 2 µL of perilymph. On the other hand, exquisitely sensitive assays will detect trace amounts that are present in serum, which could lead to false-positive results. Accordingly, use of this technique in an effort to confirm this diagnosis is not recommended.
An exploratory tympanotomy for perilymph fistula should be undertaken with use of local anesthesia and mild sedation whenever possible, so that the patient may be enlisted to perform the Valsalva maneuver to help identify perilymph leaks. After a tympanomeatal flap is elevated, bone is removed from the posterior superior wall of the bony external auditory canal until the oval window is completely visible. Likewise, it may be wise to remove any mucosal folds and the bony overhang of the round window niche to better visualize the round window membrane. It is rare to see a dramatic defect in the oval or round window in routine perilymph fistula explorations. Fluid may be suctioned from the niche to see if it reaccumulates, although this phenomenon is common and does not verify that a perilymph fistula is present. Poe and Bottrill24 have added credence to the suggestion that pooling of clear fluid in the oval or round window is due to transudation or to dependent accumulation of local anesthetic solutions. They performed transtympanic endoscopy of the middle ear before raising a tympanomeatal flap, and documented that fluid was not evident in the windows until after injection of the local anesthetic solution. In fact, endoscopic visualization of the middle ear could supplant formal tympanotomy for diagnosis of this disorder. The ear also should be inspected for abnormally patent fissures in the bony labyrinth, especially anterior to the oval window and inferior to the round window. Any suspicious defect should be repaired by removing surrounding mucosa and patching with connective tissue, secured by packing. My own practice is to routinely patch the round window niche in explorations with negative findings, but a more conservative approach is indicated in the oval window region to avoid the risk of an iatrogenic conductive hearing loss.
Specific Operations for Meniere’s Disease
A less invasive alternate treatment for Meniere’s disease, intratympanic gentamicin injection, has become an accepted alternative to ablative surgery.25–27 Uncontrolled reports of success with intratympanic corticosteroids are accumulating, but higher-quality studies of efficacy in this disorder are needed.28 Therapy with a novel mechanical device, the Meniett device (Medtronic Xomed, Jacksonville, Florida), has demonstrated a significant reduction of episodic vertigo among surgical candidates in a placebo-controlled randomized clinical trial.29 This therapy involves insertion of a tympanostomy tube in the affected ear, with subsequent application of intermittent pulsatile overpressure to the inner ear in 5-minute treatment cycles three times daily. Sustained remissions were typical, and only 24% elected to proceed to surgical treatment within 2 years.30
Cochleosacculotomy
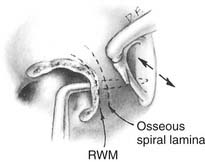
The cochlear endolymphatic shunt procedure, usually referred to as cochleosacculotomy, is designed to create a permanent communication to equilibrate endolymphatic and perilymphatic pressures in the hydropic ear.31 The goal is to create a small permanent fistula in the osseous spiral lamina within the basal turn of the cochlea. This is performed by elevating a tympanomeatal flap and exposing the round window niche. A 3-mm right-angle pick is inserted through the round window membrane and directed toward the oval window niche, hugging the lateral wall of the inner ear (Fig. 167-4), and fracturing the osseous spiral lamina. As might be expected, the operation has been associated with a 25% incidence of high-frequency sensorineural hearing loss and a 10% incidence of profound deafness.32 Despite reports of 70% success with long-term relief of vertigo, the operation has enjoyed only limited application. In one series of elderly patients with Meniere’s disease, vertigo control was documented in almost every patient, but hearing was almost universally worse.33 The study authors recommended that this operation be used as an alternative to labyrinthectomy in elderly patients with preexisting severe hearing loss.
Endolymphatic Sac Surgery
Although the specific physiologic defect causing Meniere’s disease is unclear, many investigators report a beneficial effect in uncontrolled studies of surgical intervention directed toward the endolymphatic sac in patients with Meniere’s disease. The first operative procedure on the endolymphatic sac was performed by Portmann.34 Many modifications have been proposed, including bony decompression of the sac by Shambaugh,35 placement of an endolymphatic-subarachnoid shunt by House,36 and an endolymphatic-mastoid shunt by Shea.37 Complications of endolymphatic sac procedures include hearing loss, cerebrospinal fluid leak, and facial nerve paralysis, although they are infrequent.
The efficacy of endolymphatic sac procedures has been widely questioned and debated. Some investigators suggest that the operation is ill-conceived and that the reported results may be biased or reflect a nonspecific effect of the operative undertaking or the natural history of this spontaneously remitting condition.38,39 Evidence of a nonspecific effect of anesthesia on Meniere’s disease has been provided by Gates,40 who documented in a prospective observational trial that administration of a potent neuroleptic analgesic (Innovar) provided lasting relief of vertigo for most patients in whom medical management had failed to produce improvement. Advocates of sac surgery argue that a 50% to 70% success rate improves on the anticipated natural history in this patient population, making an endolymphatic sac procedure preferable to immediate use of an ablative procedure.41 One uncontrolled observational study suggested that vertigo and vestibular disability could be largely corrected in 90% of patients evaluated 2 years after wide bony decompression of the sac and sigmoid sinus.42 An important randomized clinical trial performed in Denmark during the 1970s compared endolymphatic sac decompression with a “sham” cortical mastoidectomy operation. The patients and those evaluating the outcomes were blinded to group assignment. Multiple subsequent reports were published, culminating with a 9-year follow-up report.43 Both surgical groups demonstrated improvements that were not statistically different, and the investigators concluded that clinical improvement with decreased vertigo was attributable to a “placebo effect.” The conclusions of this study have been widely debated, with proponents of sac surgery arguing that the sample size was too small to detect clinically important differences and pointing out that both groups did improve, thus arguing in favor of the operation even if the effect was nonspecific. Welling and Nagaraja44 reanalyzed the data from this study by more sophisticated modern statistical methods. They demonstrated from the original data set that, even with the small sample size, the sac surgery group showed statistically significant reductions in postoperative vertigo, nausea and vomiting, and tinnitus compared with the mastoidectomy control group. Unfortunately, because the study did not include a nonsurgical control group, the potentially important influence of disease natural history on the reported efficacy could not be assessed. The Danish group performed another small controlled study comparing endolymphatic sac decompression and insertion of a tympanostomy tube, demonstrating that both groups had similar levels of improvement.45 Again, a nonsurgical control group was not used.
One difficulty in performing vertigo outcome studies has been a lack of validated metrics for measuring treatment outcome. A previously validated medical quality-of-life instrument, the SF-36, has been demonstrated to correlate well with the 1995 disease reporting classification44 proposed by the American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS).46 The study showed that preoperative patients scored well below population norms, and that those achieving AAO-HNS class A or B outcomes (vertigo relief) normalized their scores. Alternative metrics have been proposed for future prospective studies that are more specific to Meniere’s disease.47,48
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree