Chapter 85 Strabismus surgery
Anatomy is important to strabismus surgery
Conjunctiva
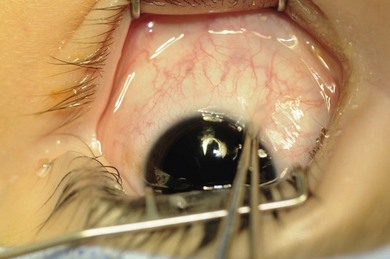
The conjunctiva is nearly transparent and well vascularized (Fig. 85.1). Adjacent to the limbus, it contributes to the rich anastamotic vascular network providing collateral flow to the anterior segment.1 After topical 2.5% phenylephrine, normal conjunctival vessels constrict; the underlying anterior ciliary vessels do not. The anterior ciliary vessels, their scleral penetration sites and anastamotic branches to neighboring rectus muscles can be seen. The insertion sites of the four rectus muscles can be visualized. The anterior termination of the extraconal fat pad is seen through the conjunctiva roughly 10 mm posterior to the limbus. These features help determine if there has been prior surgery and guide placement of the surgical incision.
Tenon’s capsule
Tenon’s capsule is a dense, translucent and nearly avascular fascial layer extending from the limbus to the optic nerve. Anteriorly, it is located just beneath the conjunctiva. The conjunctiva can easily be separated from the underlying Tenon’s capsule, beginning 1 mm posterior to the limbus. Three millimeters posterior to the limbus, Tenon’s capsule fuses with the underlying intermuscular septum, forming a single fascial layer.2 Posterior to this zone, Tenon’s capsule envelops the globe, forming a potential cavity (sub-Tenon’s space).
Tenon’s capsule separates the orbital fat from the globe. Anterior Tenon’s separates extraconal fat from the eye; posterior Tenon’s separates intraconal fat from the globe. Fibrous septae extend from the outer surface of Tenon’s capsule to the orbital periosteum.3 It is important to avoid violating Tenon’s capsule during surgery. Failure to do so permits fat to prolapse into sub-Tenon’s space. Scarring may result in fat adherence with restricted eye movement.
Muscle capsule and intermuscular septum
The rectus and oblique muscles and tendons are enveloped in connective tissue capsules. The capsules on the rectus muscles are well developed anterior to the site of the muscles’ penetration of Tenon’s capsule. With age, the capsules thin. Postoperative scarring is reduced if the muscle capsule is left intact in areas not involved with the procedure. Posterior to the penetration site in Tenon’s capsule, the muscle capsules become quite thin.4
The intermuscular septum is a thin, avascular layer of connective tissue that is continuous with the muscle capsules, extending from the border of each rectus muscle to the adjacent rectus muscles (Fig. 85.2). Anterior to the rectus muscles’ penetration of Tenon’s capsule, the intermuscular septum forms a thin, separate layer just beneath Tenon’s capsule, adjacent to the sclera. Posterior to the rectus muscles’ penetration, the intermuscular septum has been thought to separate intraconal from extraconal fat. However, the existence of this portion of the intermuscular septum has recently been questioned: it may be the anterior aspect of the connective tissue pulleys.5 The intermuscular septum is associated with the inferior oblique and forms a roughly 2 mm wide band, extending off the posterolateral margin of the muscle.
Blood supply
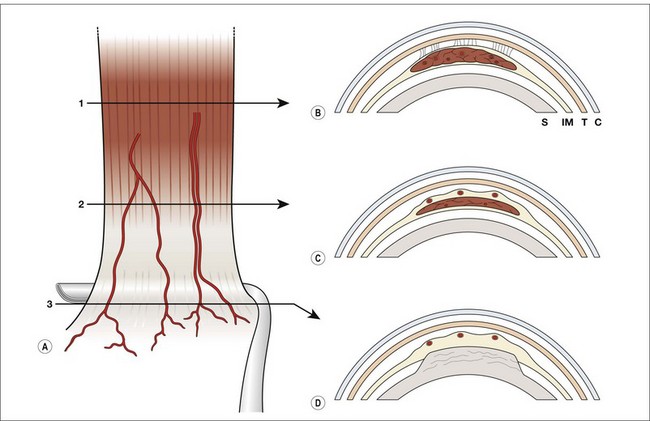
Each of the four rectus muscles carries anterior ciliary vessels that contribute to the blood supply of the anterior segment; the two oblique muscles do not. In the classic description, each rectus muscle has two anterior ciliary arteries, except the lateral, which has one.6 This diagram of the ocular circulation is reproduced repeatedly in ophthalmic literature6,7 and shows the anterior ciliary vessels traveling within the muscle and tendon. However, the vessels emerge from the substance of the muscle, posterior to the transition from muscle to tendon, and travel on the surface of the tendon8 (Figs 85.3 and 85.4). The number of anterior ciliary vessels is variable.9,10
The contribution of the anterior ciliary vessels to anterior segment blood flow has been studied by iris fluorescein angiography.11 Tenotomy of either of the vertical rectus muscles produces a significant delay in vascular filling in the corresponding segment of the iris.12 Surgery on the vertical rectus muscles impacts more on anterior segment blood flow than surgery on the horizontal rectus muscles and may influence the risk of anterior segment ischemia.
Pulleys of the orbit
Highly complex pulleys in the anterior orbit have been described. Each rectus muscle passes through an encircling ring of connective tissue near the equator of the globe, which attaches to the orbital wall. The pulleys are composed of collagen, bands of smooth muscle, and elastin.13,14 They have an intimate and active relationship with the extraocular muscles, which can include acting as the functional origin and limiting side-slipping.15,16,17 Orbital malformations, disease, trauma, and surgical procedures that affect the integrity of the orbital pulleys may be significant in ocular motility disorders.
Rectus muscles
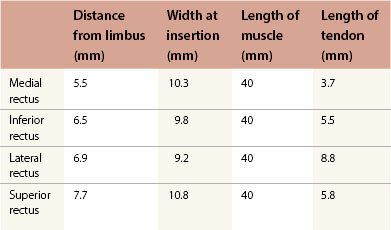
There are four striated rectus muscles arising from the annulus of Zinn in the apex of the orbit. Each is 40 mm in length and inserts on the sclera anterior to the equator of the globe. Following the “spiral of Tillaux,” the medial rectus inserts 5.5 mm from the limbus, the inferior rectus 6.5 mm, the lateral rectus 6.9 mm, and the superior rectus inserts 7.7 mm from the limbus (Table 85.1). The location of the insertions is actually somewhat variable.18 The insertions of the rectus muscles are curvilinear, particularly the vertical rectus muscles, in which the temporal border is further from the limbus than the nasal border.
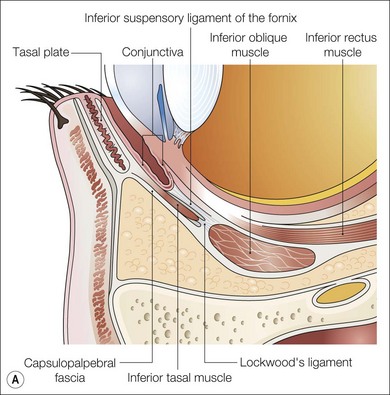
The inferior rectus muscle courses anteriorly, laterally, and inferiorly to insert on the sclera. The inferior rectus forms a 23° angle with the visual axis when the globe in the primary gaze position. It has fascial attachments to the inferior oblique muscle and the lower eyelid retractors (Fig. 85.5). If these attachments are not severed during recession or resection of the inferior rectus, eyelid fissure changes may occur. Connective tissue attachments between the superior rectus and the superior oblique may assist the surgeon in locating a ‘lost’ superior rectus muscle.
Superior oblique muscle
The superior oblique muscle arises from periosteum in the orbital apex above the annulus of Zinn and travels anteriorly along the superomedial orbital wall. It becomes a cord-like tendon, passing through the trochlea to run in a posterolateral direction at an angle of approximately 51° to 54° with the sagittal plane. The tendon passes under the superior rectus and fans out, inserting on the sclera in the superotemporal quadrant (Fig. 85.6). The anterior pole of its insertion is located close to the lateral border of the superior rectus, beginning 4 to 6 mm posterior to the superior rectus insertion. Its tendon is thin and broad, extending posteriorly for about 11 mm, but with considerable variability. The posterior end of the insertion lies 6−7 mm from the optic nerve. The superior temporal vortex vein can exit the sclera temporal to the superior oblique insertion, under the insertion or sometimes split the fibers of the insertion and pass through them.
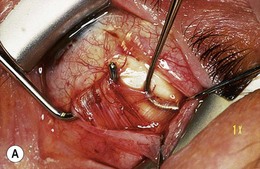
The superior oblique tendon can be abnormal.19 It may be redundant, lax, or misdirected. It can have an anomalous insertion nasal to the superior rectus or may insert into Tenon’s capsule and the trochlea, or be absent.20 Intraoperative “exaggerated”21,22 traction testing of the superior oblique can demonstrate laxity of the superior oblique tendon (Fig. 85.7).
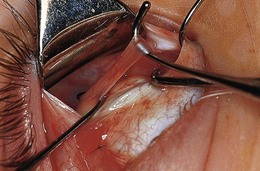
Fig. 85.7 Strabismus hooks beneath superior oblique tendon demonstrating tendon laxity.
(Patient of Dr R. Morris.)
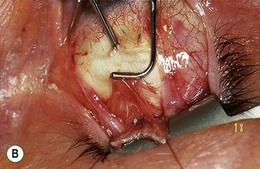
The nasal aspect of the superior oblique tendon has fascial relationships with the intermuscular septum, which envelops it to form a capsule. If the nasal intermuscular septum is thin, atrophic, or is damaged while performing a nasal tenotomy, the cut ends of the tendon may separate excessively, creating a superior oblique palsy. The ends of the tendon may be difficult to recover. There is a frenulum between the capsule on the underside of the superior rectus and superior surface of the superior oblique tendon. The frenulum must be lysed for significant procedures on either muscle (Fig. 85.8).
Inferior oblique muscle
The inferior oblique muscle originates from the maxillary bone adjacent to the lacrimal fossa. It passes posteriorly and laterally, forming approximately a 51° angle with a sagittal plane passing through the origin of the muscle. After passing beneath the inferior rectus, the inferior oblique travels upward inserting on the sclera, close to the macula.23 The tendon of the inferior oblique is 1−2 mm long, the shortest of the extraocular muscle tendons. The inferior oblique insertion is 8 to 12 mm posterior to the lateral rectus insertion, adjacent to its lower border. There is considerable variability in the location and configuration of the inferior oblique insertion.
The neurovascular bundle enters the inferior oblique muscle as it passes beneath the inferior rectus muscle. The neurovascular bundle may serve as the effective origin of the inferior oblique, particularly after anterior transposition of the muscle.24
Overaction of the inferior oblique is common, particularly in congenital esotropia, intermittent exotropia, and superior oblique palsy. Overaction of the inferior oblique is clinically graded on a scale of 1−4.25 Overaction of the inferior oblique must be distinguished from other causes of elevation of the globe in adduction. Overaction of the inferior oblique commonly coexists with dissociated vertical deviation.26
General principles of surgery
Anesthesia
Postoperative nausea and vomiting in strabismus patients receiving general anesthesia is common. Short acting inhalational anesthetics reduce, but do not eliminate, this problem.27,28,29 Dexamethasone and ondansetron decrease postoperative nausea and vomiting.30,31
Patient preparation
The patient should be positioned with the head level and the neck slightly extended (Fig. 85.9). The exposed skin is cleaned with aqueous povidone-iodine. One drop of 5% aqueous povidone-iodine is placed in the conjunctival cul-de-sac prior to surgery. The eyebrows are covered with drapes or an adhesive plastic strip (Fig. 85.10) and one is placed over the upper eyelid eyelashes. A separate speculum is used on each eye, when the procedure is bilateral.
Surgical instruments
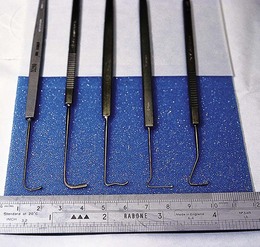
A selection of strabismus hooks is essential (Fig. 85.11). Hooks with tips slightly longer than the width of the rectus muscle insertion allow the tendon to be spread evenly on the hook, which facilitates dissection and suture placement (Fig. 85.12).
Traction testing
The exaggerated traction test of the superior oblique determines whether superior oblique laxity is present and may be helpful in Brown’s syndrome.32 It is performed by grasping the eye with two toothed forceps adjacent to the limbus. The eye is adducted while placing the eye in retropulsion, external rotation, and elevation. While maintaining this position, the eye is moved into abduction. A palpable “thump” is perceived with a normal superior oblique. An absent or reduced “thump” is evidence of superior oblique laxity. This test requires a considerable experience.
Incisions for strabismus surgery
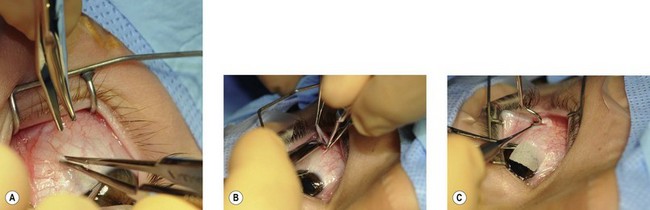
Limbal incision
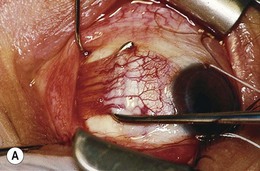
The limbal incision is a common approach for access to extraocular muscles (Fig. 85.13). The fusion of conjunctiva, Tenon’s capsule, and intermuscular septum a few millimeters from the limbus permits the three layers to be retracted together, providing direct access to the sclera. It provides an excellent view of the extraocular muscles.
Fornix or cul-de-sac incision
The fornix or cul-de-sac incision is also commonly employed. It reduces postoperative discomfort and places the surgical scar is beneath the eyelid (Fig. 85.14). It requires a skilled assistant, familiar with ocular anatomy.
Swan incision
The Swan incision is made directly over the rectus muscle insertion.33 It is less popular due to the interpalpebral location of the wound with scar formation and potential bleeding from the underlying anterior ciliary vessels. A modified Swan incision may be useful in reoperations.
Extraocular muscle surgery
Tissue adhesives
Tissue adhesives have been used in conjunctival closure during strabismus surgery37,38 but have not gained popularity in securing the muscles to the sclera.39–41
Rectus muscle surgery
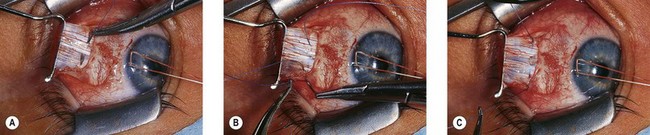
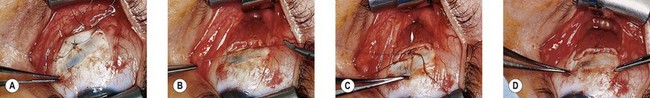
Proper exposure of the extraocular muscles is essential for successful strabismus surgery. Dissection of excess connective tissue at the suture line provides better exposure for precise suture placement and decreases the risk of slipped or lost muscles (Fig. 85.15). This requires excision of redundant muscle capsule and intermuscular septum. The muscle capsule and intermuscular septum can be lifted off the rectus muscle adjacent to the insertion and excised without disrupting the anterior ciliary vessels. Cauterizing anterior ciliary vessels prior to passing sutures or making incisions in the tendon or muscle reduces bleeding and decreases scarring.
Weakening procedures on the rectus muscles
Recessions
The preferred site for measuring a recession varies. Measurement from the limbus avoids the potential variability in the position of the muscle insertion.42 Those who measure from the insertion of the tendon feel the amount of recession is dependent upon the change in position relative to the original scleral attachment point, rather than the limbus. Measurement from the insertion should be determined prior to disinsertion; the elastic properties of the sclera allow the insertion to shift anteriorly following detachment of the muscle,43 especially with tight extraocular muscles.
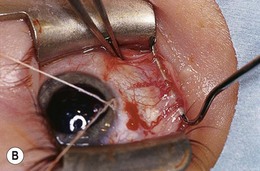
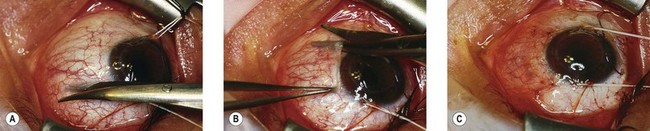
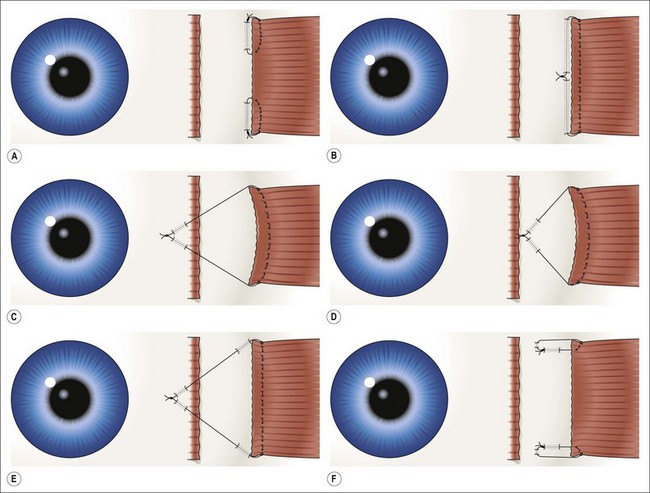
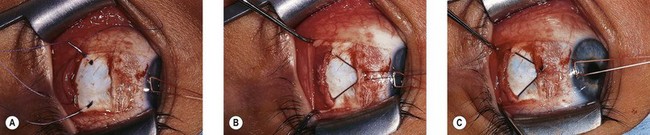
Direct fixation of the recessed muscle to the sclera is widely used (Fig. 85.16A,B). The scleral entry position of the needles is at the location of the desired recession and should be directly posterior to the original insertion site. Each pole should be spaced to maintain the width of the recessed muscle to avoid “central sag.” If sagging occurs, the center of the muscle can be sutured to the sclera (Fig. 85.17). Because the muscle is sutured directly to the sclera, it allows accurate supra- and infraplacement and “slanting” the recession, if desired, in “A” and “V” patterns.
Hang-back and hemi hang-back techniques are commonly employed in recessions (Fig. 85.16C,D).The hang-back technique suspends the muscle on a suture attached at or near the terminal poles of the original insertion or can be located near the mid-portion of the original insertion (see Fig 85.20). Hang-back recessions may reduce scleral perforations by placing the sutures in thicker sclera at the original insertion. However, with large recessions, the muscle may side-slip or creep anteriorly during or after surgery; this is reduced by placing shallow scleral “belt-loops” close to the recessed muscle or using an anchored hang-back suture (Figs 85.16E and 85.18).
In the hemi hang-back technique, the suture engages the sclera posterior to the original insertion site but anterior to the recessed muscle (Fig. 85.16D). Indications include: surgical exposure, thin sclera, scarring, the presence of an oblique muscle (particularly the superior oblique tendon), or a scleral buckle, glaucoma implant or other device on the sclera. Hemi hang-back sutures with large recessions may reduce the potential for side-slipping or muscle creep.
In a loop recession, a non-absorbable suture is placed through each pole of the muscle and sutured to the sclera behind the insertion (Fig. 85.16F).
Horizontal muscle recession
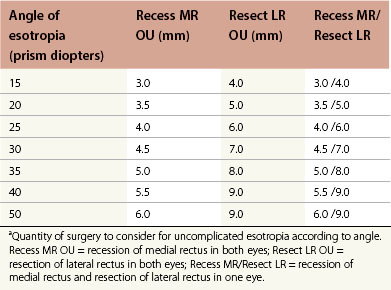
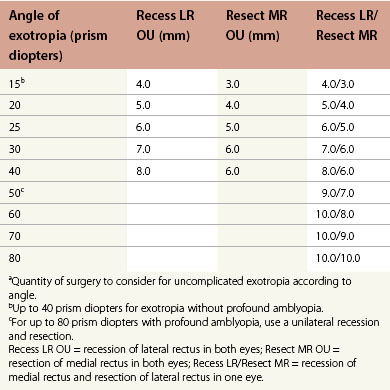
Dose-response tables for horizontal rectus recessions and resections are available. As a general rule, the medial rectus can be recessed a maximum of 7 mm without reducing adduction and the lateral rectus recessed up to 10 mm. Exceptions to these limits are common, particularly with restrictive and paretic strabismus, when much larger recessions are commonly employed. However, 7 mm bilateral medial rectus recessions may increase the risk of consecutive exotropia in children with congenital esotropia.44–46 The dose-response with large recessions is non-linear and less predictable. The guideline values listed in the dose-response tables are most useful in uncomplicated strabismus (Tables 85.2 and 85.3).
The quantity of surgery required to correct horizontal deviations is dependent upon a number of factors, especially the magnitude of the preoperative deviation. Axial length and refractive error are not usually significant.47 Recession of unoperated rectus muscles is more predictable than recession of previously operated muscles. Restrictive disorders, particularly thyroid orbitopathy, and recession of antagonist to a paretic muscle are associated with a reduced effect per millimeter of recession. Very large recessions combined with resections may result in limitation of movement.
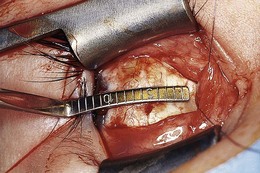
Recessions can be measured using a curved ruler or calipers. Curved rulers follow the circumference of the sclera, measuring the length of the arc (Fig. 85.19). Calipers measure the length of the chord: this disadvantage can be reduced by measuring the total recession as two small chord lengths, whose sum is equal to a large recession.
Inferior rectus muscle recession
There are no dose-response tables for vertical rectus recessions or resections. On average, 1 mm of inferior rectus recession will result in correction of 3 PD of vertical deviation in primary gaze and 5 PD in downgaze.48
Techniques to reduce lower eyelid retraction include:
1. Dissection and lysis of fascial attachments to the inferior rectus as far posteriorly as possible, including: intermuscular septum, check ligaments, and Lockwood’s ligament connections.
2. Suture the lower eyelid retractors to the inferior rectus.49 An adjustable suture can be placed on the capsulopalpebral head and passed through the inferior rectus insertion. Adjustment of the capsulopalpebral head is performed after the inferior rectus adjustment.50
3. Disinsertion of the lower eyelid retractors from the tarsal plate.51
Delayed overcorrection is a significant problem following inferior rectus recession. This is usually accompanied by limited depression in the operated eye with diplopia, particularly in downgaze. Delayed overcorrection is more likely to occur with thyroid orbitopathy treated with adjustable sutures. It occurs in 10−21% of cases.52,53,54
A number of mechanisms may contribute to “late slippage” of the inferior rectus.55,56,58 These include:
1. Thickened Tenon’s capsule under the inferior rectus muscle allowing the muscle to slip posteriorly after absorption of the sutures, forming a pseudo-tendon anterior to the insertion.
2. Inferior rectus attachments to Lockwood’s ligament and the lower eyelid retractors.
3. Hang-back adjustable sutures hold the recessed inferior rectus in place centrally. When the eye makes horizontal movements, the inferior rectus does not move fully with the eye; repeat side-slipping prevents firm reattachment of the muscle to the sclera.
4. Laterally directed traction of the inferior oblique may prevent adherence of the inferior rectus to the sclera.
5. Scarring around Lockwood’s ligament pulls the inferior rectus muscle anteriorly, creating slack in the anterior aspect of the muscle, weakening its depressor function.57
6. Following a large recession, the inferior rectus is not in apposition to the globe when the muscle is contracting and the eye is in downgaze.58 MRI studies demonstrate that an inferior rectus recession of 6.0 mm and a medial rectus recession of 7.5 mm in a normal eye leads to loss of inferior rectus apposition, with lower values in thyroid associated patients.59
7. Ipsilateral contraction of the superior rectus in thyroid orbitopathy leads to late overcorrection.
Poor or absent superior rectus function is a cause for undercorrection. “Double elevator” palsy and superior rectus palsy are likely to be undercorrected with inferior rectus recessions alone. Failure of superior rectus development in congenital fibrosis of the extraocular muscles causes disappointing results following inferior rectus recession.60,61 Retrobulbar and peribulbar block induced vertical strabismus can be complex and is often associated with paretic and restrictive components.62–67
Bilateral recessions of the inferior rectus muscles can create exotropia in downgaze, forming an “A” or “λ” pattern, especially in thyroid orbitopathy where large inferior rectus recessions have been performed. Nasal displacement of the inferior rectus muscles reduces the tendency for exotropia in downgaze. However, this may be associated with intorsion.63
Superior rectus muscle recession
There are also no dose response tables for superior rectus recessions or resections. Each millimeter of superior rectus recession generally corrects 3 to 4 PD of vertical deviation up to 6 mm of recession. Recessions of greater than 6 mm are avoided, except in dissociated vertical deviation (DVD), when hang-back recessions of up to 10 mm may be performed (Fig. 85.20).64,65 Superior rectus recessions beyond 4 mm require lysis of the fascial attachments between the underside of the superior rectus and the superior oblique tendon (see Fig. 85.8B). For very large recessions, it is important to dissect and cut fascial attachments and check ligaments on the superior rectus as far posteriorly as possible and identify the superior oblique tendon, both nasal and temporal to the superior rectus. The superior oblique insertion is in close proximity to the temporal border of the superior rectus. The thin fibers of the superior oblique near its insertion can be mistaken for intermuscular septum and inadvertently transected (Fig. 85.6).
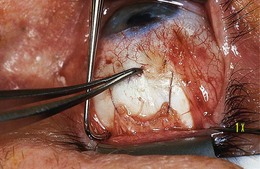
Fig. 85.20 Superior rectus hang-back recession: surgeon’s view from above.
(Patient of Dr R. Morris.)
Superior rectus recessions do not commonly cause upper eyelid retraction.66 Because of the potential problems associated with inferior rectus recessions, surgery for concomitant vertical deviations of up to 15 PD is often performed by superior rectus recession in the hypertropic eye.