Patient Selection
The selection of the appropriate skull base approach depends on the type of lesion and location of the disease, as well as the hearing status and medical status of the patient. Preoperative hearing status should be determined with a comprehensive audiogram because some skull base approaches result in profound deafness in the operated ear. Temporal bone abscesses and cholesterol granulomas may only require the creation of a drainage pathway, whereas cholesteatoma and neoplasms often require more extensive access to attempt a complete excision. Complete eradication of the disease process may not always be possible. The surgeon in select circumstances may elect to leave cholesteatoma behind when resection could result in facial paralysis, vascular injury, or profound hearing loss. Periodic staged procedures may be required to evacuate accumulated debris in these cases.
42.3 Presentation and Differential Diagnosis
Chronic ear disease usually presents with hearing loss and otorrhea that can be treated with standard tympanoplasty with or without mastoidectomy. Symptoms such as otalgia, retro-orbital pain, vertigo, diplopia, facial paralysis, lower cranial nerve deficits, or profound hearing loss suggest more extensive disease. Petrous apicitis may present with Gradenigo’s triad of retro-orbital pain, abducens nerve palsy, and chronic otorrhea.1,2 Profound sensorineural hearing loss, with or without vertigo, implies direct involvement of the labyrinth. Hoarseness or dysphagia suggests involvement of the jugular foramen. Appropriate use of computed tomography (CT) and magnetic resonance imaging (MRI) will confirm the extent of the disease and allow for surgical planning.3
The differential diagnosis for COM is extensive (▶ Table 42.1). Cholesteatoma is common and often presents with a posterior-superior retraction pocket and debris collecting under the scutum. A chronically draining perforation that is unresponsive to ototopical antibiotics should raise suspicion for a more serious underlying problem, such as a cholesteatoma, petrositis, or neoplasm. If granulation tissue is identified that is not associated with a definite cholesteatoma and not responsive to ototopical antibiotic/steroid treatment, a biopsy is warranted to evaluate for a neoplasm. Deep otalgia should raise suspicion of a more serious problem, such as petrositis or a neoplasm.
Inflammatory disorders | Neoplasms |
Chronic suppurative otitis media | Squamous cell carcinoma |
Cholesteatoma | Endolymphatic sac tumor |
Apical petrositis | Middle ear adenoma |
Sigmoid sinus thrombosis | Lymphoma |
Epidural abscess | Glomus tumor |
Otitis externa | Langerhans’s cell histiocytosis |
Malignant otitis externa | Chondrosarcoma |
Wegener’s granulomatosis | Osteosarcoma |
Eosinophilic otitis media | Rhabdomyosarcoma |
Tuberculous otitis media | Adenocarcinoma |
Cholesterol granuloma | Adenoid cystic carcinoma |
Syphilis | Metastasis |
42.4 Petrous Apex Drainage Pathways
The petrous apex is the pyramid-shaped medial aspect of the temporal bone that is bounded laterally by the otic capsule, tensor tympani muscle, and petrous carotid artery. Most individuals have a marrow-filled or sclerotic petrous apex, with only a small minority having aeration. A number of pre-existing pathways to the petrous apex can be used to access disease. Selection of a surgical approach for apical disease is facilitated by dividing the petrous apex into anterior and posterior segments using a parallel line through the internal auditory canal (▶ Fig. 42.1).4
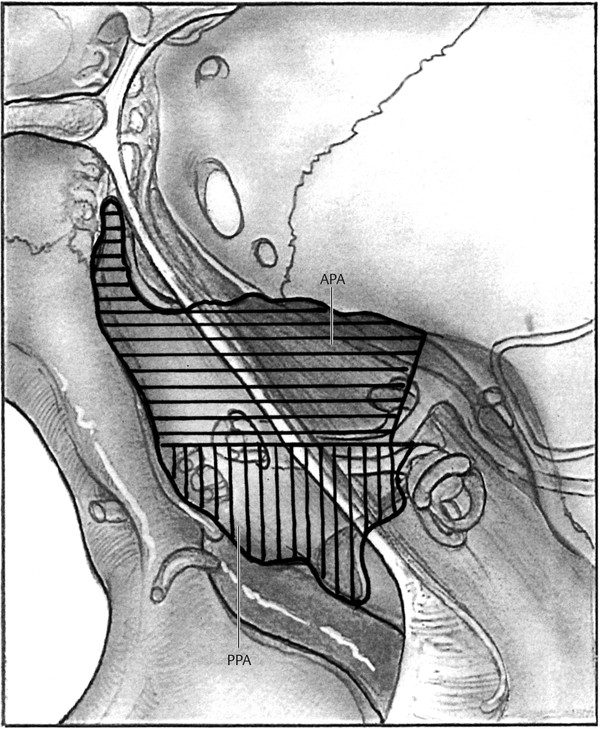
Fig. 42.1 Petrous apex. The petrous apex can be divided into an anterior (APA) and posterior (PPA) compartment by the internal auditory canal.
Three pathways (sinodural angle, subarcuate, infralabyrinthine) to the posterior petrous apex are accessible via a standard postauricular, complete mastoidectomy approach (▶ Fig. 42.2). The sinodural angle and subarcuate tracts provide an avenue for drainage of the superior aspect of the posterior petrous apex. The sinodural angle tract is opened after skeletonizing the posterior and superior semicircular canals, transverse sigmoid sinus junction, and the posterior aspect of the mastoid tegmen. The petrous ridge and superior petrosal sinus are followed into the posterior petrous apex. The subarcuate approach requires skeletonization of the semicircular canals and mastoid tegmen. An opening into the superior aspect of the posterior petrous apex is created by removing the bone through the arch of the superior semicircular canal. Bleeding from the subarcuate artery is typically encountered during this approach. A 2- or 3-mm bur is used to open the subarcuate tract, with care being taken not to enter the vestibule and semicircular canals.4 Lee used CT scans of 58 temporal bones in patients with aerated petrous apices to estimate the dimensions of the subarcuate tract, which was approximately 4.88 mm by 4.9 mm.5 Blue-lining the canals may provide additional reference for the surgeon to maximize the opening into the petrous apex via the subarcuate tract.4
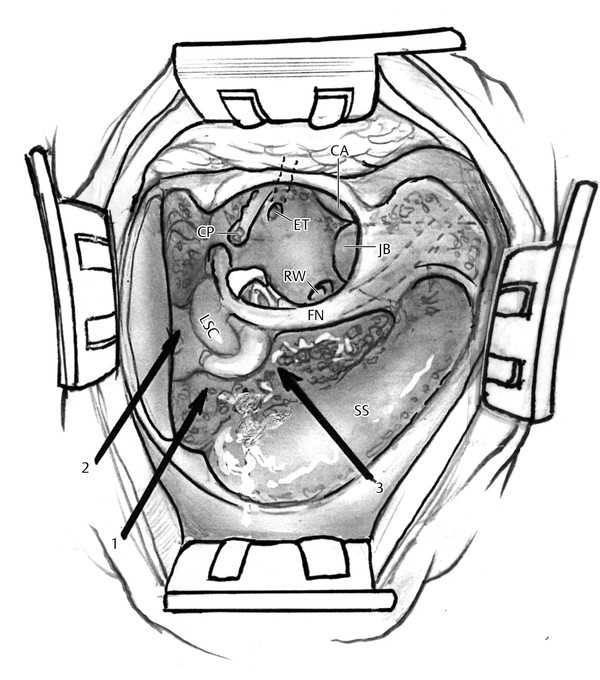
Fig. 42.2 Posterior tracts to petrous apex. Three pathways (1, sinodural angle; 2, subarcuate; 3, infralabyrinthine) to the posterior petrous apex are accessible via a standard postauricular, complete mastoidectomy approach. SS, sigmoid sinus; LSC, lateral semicircular canal; FN, facial nerve; RW, round window; ET, Eustachian tube, CA, carotid artery; CP, cochleariform process; JB, jugular bulb.
The transmastoid infralabyrinthine approach provides access to the inferior aspect of the posterior and, occasionally, the anterior petrous apex when the jugular bulb is low. The vertical facial nerve, sigmoid sinus, and the ampullated end of the posterior semicircular canal provide the boundaries for the infralabyrinthine approach. Excessive bleeding from the sigmoid sinus or jugular bulb, audiovestibular dysfunction from otic capsule violation, facial paresis, and cerebrospinal fluid (CSF) leak are all potential complications of this approach.4
The transtemporal (infracochlear, pericarotid, and complete apicectomy), transnasal, and extradural middle fossa approaches provide access to the anterior petrous apex (▶ Fig. 42.3). The infracochlear approach allows access to the inferior aspect of the anterior petrous apex. The inferior basal turn of the cochlea, vertical petrous carotid artery, and the anterior aspect of the jugular bulb are the boundaries of the infracochlear approach. The infracochlear approach necessitates removal of the inferior tympanic osseous annulus and a portion of the inferior canal wall. The pericarotid approach and complete apicectomy approach provide access to the superior aspect of the anterior petrous apex. The boundaries of the pericarotid approach are the horizontal petrous carotid artery, the ascending basal turn of the cochlea, and the middle fossa dura. Exposure and removal of the medial wall of the semicanal of the tensor tympani muscle is required to expose the anterior petrous apex via the pericarotid approach. A complete apicectomy is an extension of the pericarotid approach, described by Ramadier and Lempert, and requires removal of the anterior canal wall and mandibular condyle.4 The medial wall of the glenoid fossa is opened up to the horizontal segment of the petrous carotid artery. A less radical variation of the complete apicectomy that preserves the anterior canal wall and retracts the glenoid contents anteriorly can be utilized to provide wide access of the anterior petrous apex.4
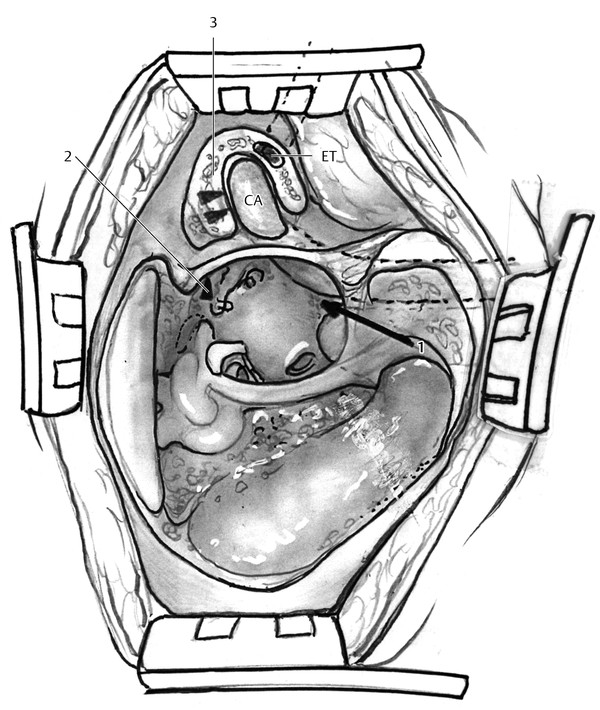
Fig. 42.3 Anterior tracts to petrous apex. The transtemporal (1, infracochlear; 2, supracochlear; 3, transglenoid) approaches provide access to the anterior petrous apex. The transnasal and extradural middle fossa approaches (not pictured) also can be used to approach the anterior petrous apex. CA, carotid artery; ET, Eustachian tube.
The petrous apex typically abuts the posterior lateral wall of the sphenoid sinus. An endoscopic transnasal sphenoidotomy in some cases will permit access to the anterior petrous apex.6 The extradural middle fossa approach provides wide access to the superior aspect of the anterior and posterior petrous apex. The greater superficial petrosal nerve, arcuate eminence, and the petrous ridge are the boundaries of the petrous apex as viewed from the middle fossa approach.7
42.5 Classification of Petrous Apex Cholesteatoma
42.5.1 Congenital
Congenital cholesteatoma is thought to arise from embryonic tissue from the abnormal development of the first brachial groove.8,9 Cholesteatoma has a linear growth pattern, unlike a true neoplasm. Congenital petrous apex cholesteatoma often presents in a delayed fashion as otoscopy is often normal even in symptomatic patients. Vestibular symptoms and sensorineural hearing loss, as opposed to conductive loss, occur secondary to erosion into the otic capsule or internal auditory canal. Patients typically present with slowly progressing facial paresis or long-standing paralysis secondary to internal auditory canal or Fallopian canal involvement. Diplopia or facial anesthesia may occur with medial cholesteatoma extension into Dorello’s canal or Meckel’s cave, respectively.10
42.5.2 Acquired
Neglected or obscured middle ear and mastoid cholesteatomas, on rare occasion, can spread to the petrous apex via the previously described preformed pathways or by erosion through the otic capsule.11 Inflammation and infection typically associated with chronic otitis media result in increased epithelial turnover, which leads to progressive cholesteatoma expansion and erosion of adjacent bone. Patients with a known history of prior cholesteatoma are at risk for apical extension regardless of the previous surgical approach (canal wall-up/down, tympanoplasty, etc.); however, canal wall-down mastoidectomy has a lower recurrence rate. Apical extension of cholesteatoma may still occur after canal wall-down mastoidectomy secondary to spread along preformed pathways from residual disease in the mastoid or the middle ear.9,11
Acquired petrous apex cholesteatoma is readily identified with CT but is sometimes not identified until the time of surgery. Imaging rarely alters management but is important when discussing potential outcomes and complications with patients prior to treatment. Preoperative imaging for cholesteatoma or chronic otitis media is indicated in patients with long-standing symptoms, sensorineural/mixed hearing loss, vestibular symptoms, facial nerve symptoms, facial anesthesia, otalgia, or prior temporal bone surgery or trauma.3
42.5.3 Extent
Classifying the extent and location of a petrous cholesteatoma is important for surgical planning and for patient counseling regarding prognosis for hearing and facial nerve function. A number of classification systems have been reported for defining the location and extent of petrous apex cholesteatoma. The Moffat-Smith system uses the lesion location relative to the labyrinth.12 Supralabyrinthine, infralabyrinthine, and massive labyrinthine disease refer to cholesteatoma extending superior, inferior, or through the otic capsule, respectively, without extension into the petrous apex. Extension of the cholesteatoma into the petrous apex in each of the above three categories adds the additional designation of “apical” (i.e., supralabyrinthine-apical). A cholesteatoma confined to the petrous apex without otic capsule invasion is designated as apical disease.12
42.6 Surgical Technique
42.6.1 Translabyrinthine Approach
The translabyrinthine approach allows wide access to perilabyrinthine air cell tracts, the petrous apex, the internal auditory canal, and the posterior fossa. The approach itself results in profound sensorineural hearing loss because the labyrinth is removed, so most candidates should have non-serviceable preoperative hearing. Severe postoperative vertigo and imbalance often occur, requiring hospitalization for a few days and vestibular rehabilitation.
The patient is placed in a supine position and facial nerve monitoring is utilized. The incision is performed 1–2 cm from the postauricular sulcus. The mastoid periosteum is divided along the linea temporalis and then toward the mastoid tip. The periosteum is then elevated until the external auditory canal is visualized. A cortical mastoidectomy is performed and the tegmen mastoideum, sigmoid sinus, and posterior fossa dura are identified. Dissection continues medially until the antrum, lateral semicircular canal, and short process of the incus are identified. The extent of the disease will dictate the extent and location of bone removal. If the cholesteatoma involves the perilabyrinthine air cells, the petrous apex, or the internal auditory canal, a labyrinthectomy may be performed.
The semicircular canals should be removed in a systematic fashion by first identifying the membranous labyrinth of the lateral semicircular canal. The lateral semicircular canal should be opened on the superior aspect of the canal to prevent injury to the second genu of the facial nerve. The superior canal is then identified near the ampullated end and followed posterior to the common crus. The posterior semicircular canal is then identified at the common crus and is opened toward the ampullated end. Removal of the inferior ampullated end of the posterior canal requires drilling medial to the second genu of the facial nerve. During a labyrinthectomy the facial nerve is at greatest risk along the medial portion of the distal tympanic segment. Once the labyrinth has been removed, dissection continues medially to the internal auditory canal or petrous apex. The jugular bulb can be at risk, especially if the jugular bulb is more superior than usual. If CSF is encountered, the Eustachian tube can be obliterated and the external auditory canal closed. All open air cell tracts should be closed with bone wax. If CSF is encountered, an abdominal fat graft should be used. If no CSF is encountered, postoperative imaging for recurrent disease may be easier to interpret without fat packing.
42.6.2 Variations of the Translabyrinthine Approach
Transotic Approach
The transotic approach is used to remove disease that involves the cochlea or anterior petrous apex. The facial nerve is not transposed, so dissection occurs on both the mastoid and middle ear side of the facial nerve. The posterior external auditory canal is removed to improve visualization of the middle ear and sinus tympani. The cochlea is removed, so profound sensorineural hearing loss is expected. If CSF is encountered, the Eustachian tube is obliterated and the external auditory canal is closed. This creates a blind cavity that can be followed by serial imaging to ensure cholesteatoma has not reformed.13
Trancochlear Approach
The transcochlear approach is similar to the transotic approach, with the differentiating step being the transposition of the facial nerve posteriorly. Transposing the facial nerve usually results in some degree of permanent facial paresis, so the patient should be counseled preoperatively. This approach is useful if disease is medial to the facial nerve and involves the jugular foramen. If possible, it is preferable to work on either side of the Fallopian canal rather than transposing the nerve to prevent chronic facial paresis. This technique has been referred to as the “Fallopian-bridge technique.”14,15,16 Profound sensorineural hearing loss occurs with the transcochlear approach, so appropriate preoperative counseling is essential.
Transcrusal Approach
A transcrusal approach is helpful in patients with serviceable hearing who have extension of disease medial to the semicircular canals. The superior and posterior semicircular canals can be removed by exposing the membranous labyrinth at each end of the canal and then occluding the canals with bone paté and bone wax. It is important to prevent occluding materials from entering the vestibule. The canals can then be removed, resulting in improved access to the petrous apex, perilabyrinthine air cell tracts, and clivus. Sensorineural hearing loss is possible because the labyrinth has been violated.17,18,19,20
Pearls and Pitfalls
Preoperative comprehensive audiometry should be obtained to establish hearing status because the approach will result in profound sensorineural hearing loss.
CSF fistula management should be considered, including obliteration of the Eustachian tube, closure of the external auditory canal, and use of abdominal fat graft.
If disease involves the cochlea or jugular foramen, a transotic or transcochlear approach should be considered.
The transcochlear approach requires transposition of the facial nerve and, likely, chronic facial paresis.
The transcrusal approach is useful to gain improved access to the petrous apex, with the potential for hearing preservation.
42.6.3 Middle Fossa Approach
The middle fossa approach provides varying degrees of exposure to the petrous apex, clivus, ventral brainstem, and anterior cerebellopontine angle. The standard and extended middle fossa approaches provide excellent exposure of the internal auditory canal from the porus acousticus to the fundus.21 The extended middle fossa approach entails greater exposure of the anterior and posterior petrous apex. The boundaries of the middle fossa approach are the lateral wall of Meckel’s cave and petrous ridge medially, the superior semicircular canal posterior laterally, and the greater superficial petrosal nerve anterior laterally.22 The Kawase approach, which requires down-fracturing the zygoma, provides access to the ventral pons by removing the anterior petrous apex down to the level of the horizontal petrous carotid artery.23,24
The primary disadvantage of the middle fossa approach is an increased risk of facial nerve injury because of its anterior-superior location in the internal auditory canal; however, several studies have demonstrated no long-term difference with other skull base approaches to the internal auditory canal.25,26 Temporal lobe injury from prolonged retraction or injury to the vein of Labbé is also an important consideration when using this approach.27 The middle fossa approach does not permit visualization of the inferior cerebellopontine angle. The middle fossa approach may be combined with the pre-sigmoid approaches to address lesions that extend into multiple intracranial compartments.28
A variety of skin incisions may be used for the middle fossa approach. Once the skin and subcutaneous tissues are elevated, the temporalis muscle is cut just proximal to its origin from the temporal line to provide a site of attachment during closure. The temporalis muscle is elevated anteriorly and inferiorly and is secured with retractors or hooks. The zygoma and osseous external auditory meatus provide landmarks for the craniotomy bone flap. A 4 by 4 cm craniotomy bone flap that is centered at the zygomatic root is elevated. The inferior edge of the craniotomy is lowered to the middle fossa floor to provide additional visualization. The dura is elevated off the floor of the middle fossa from posterior to anterior to prevent injury to the greater superficial petrosal and facial nerves. The dura is elevated from the petrous ridge posteriorly to the dura of Meckel’s cave anteriorly. The greater superficial petrosal nerve, arcuate eminence, petrous ridge, middle meningeal artery, and foramen ovale all serve as landmarks.7,29
A lateral-to-medial technique, described by Fisch, begins by blue-lining the superior semicircular canal and then identifying the internal auditory canal at a 60-degree angle.30 William House identified the fundus of the internal auditory canal by tracing the greater superficial petrosal nerve to the geniculate ganglion.31 Jackler and Gladstone described a medial-to-lateral technique that begins with drilling at the petrous ridge and identifying the internal auditory canal at the porous acousticus and following it laterally to the fundus.32 Once tumor removal is complete, air cells are waxed and muscle or fat is used to obliterate the defect.
Pearls and Pitfalls
The middle fossa approach provides wide access to superior aspects of both the anterior and posterior petrous apex. Inferior petrous apex disease is not as readily accessible with this approach.
The middle fossa approach is a hearing-preservation approach. Patients with anacusis may be better served with a presigmoid approach.
The bone flap should be centered at the zygomatic root.
Cauterization or clipping of the middle meningeal artery provides additional working space.
Identifying and placing a retractor on the petrous ridge is a mandatory step of the middle fossa approach.
Identifying the greater superficial petrosal nerve and blue-lining the superior semicircular canal provides consistent landmarks for identifying the internal auditory canal.
42.6.4 Combined Petrosal Approach
The combined approach provides exposure for lesions that extend in the middle and posterior fossa and typically uses a transtemporal approach (retrolabyrinthine, translabyrinthine, and transcochlear) in addition to a middle fossa craniotomy.33
A large postauricular C-shaped incision is typically utilized for the combined petrosal approach that allows for a middle fossa craniotomy as well as a transtemporal approach. Leaving the skin flap attached to most of the temporalis muscle and periosteum facilitates prevention of pseudomeningocele formation and incisional CSF leaks. A stair-stepped C-shaped incision parallel to the skin incision is made through the temporalis muscle and mastoid periosteum down to the mastoid tip. The composite skin periosteal flap is then elevated to the osseous external auditory meatus. Retractors are placed and a complete canal wall-up mastoidectomy is performed, with care being taken to minimize the opening of the aditus ad antrum. The tegmen and sigmoid sinus are identified along the labyrinth and vertical facial nerve. The facial recess is not opened to reduce the risk of CSF otorrhea. The sigmoid sinus and middle fossa dura are decompressed down to the semicircular canals, which are then skeletonized. Decompression of the retrosigmoid and lateral middle fossa dura allows for additional room for retraction.
A retrosigmoid combined middle fossa craniotomy is then performed, with care being taken around the transverse sinus. Bur holes on either side of the transverse sinus provide an additional measure of safety when elevating this bone flap. A horizontal middle fossa durotomy is created, and the vein of Labbé is identified. An incision is then made along the inferior temporal lobe dura down to the superior petrosal sinus. A vertical presigmoid dura incision just anterior to the sigmoid sinus is then made up to the level of the superior petrosal sinus. The superior petrosal sinus is then divided after placement of liga-clips or sutures. The tentorium is divided, with care taken not to injure the trochlear nerve, Dandy vein, superior cerebellar artery, and vein of Labbé.28 Prolonged retraction of the temporal lobe dura poses a risk of injury or thrombosis of the vein of Labbé, but intermittent release of the retractor or division of the sigmoid sinus may prevent this.27
The middle fossa retrosigmoid craniotomy bone flap is secured into place with titanium plates at the completion of tumor removal. Titanium mesh, bone cement, or methyl methacrylate can all be utilized to reconstruct the osseous defect created with the combine petrosal approach. The dura is partially re-approximated with sutures. All exposed air cells, including the zygomatic root, retrofacial, facial recess, and mastoid tip, are sealed with bone wax. A piece of bone from the middle fossa decompression is shaped to the approximate size of the opening in the aditus ad antrum. This bone chip is placed over the aditus ad antrum and secured into place with additional bone wax. The retrolabyrinthine cavity is filled with strips of abdominal fat, which are placed partially through the dural openings. The composite periosteal skin flap is then closed in a water-tight fashion in three layers. A pressure dressing is placed for three* days.28
Pearls and Pitfalls
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


