Purpose
To report results of aflibercept therapy in eyes with neovascular age-related macular degeneration previously treated with bevacizumab, ranibizumab, or both.
Design
Retrospective, interventional, noncomparative, consecutive case series.
Methods
Ninety-six eyes from 85 patients with neovascular age-related macular degeneration who previously had received bevacizumab, ranibizumab, or both were treated with aflibercept monthly for 3 months followed by a fourth injection within 2 months. Outcomes were determined 4 ± 1 months after the first aflibercept dose and included: proportion of patients gaining or losing 2 lines or more of best-corrected visual acuity, proportion remaining within a gain or loss of 1 line, mean change in logarithm of the minimal angle of resolution visual acuity, mean change in central foveal thickness, mean change in macular cube volume, and qualitative anatomic response as assessed by spectral-domain optical coherence tomography.
Results
At baseline, 82 (85%) eyes had signs of active exudation despite a mean of 17 previous anti–vascular endothelial growth factor injections. At final visit, 82 (85%) remained stable within a gain or loss of 1 line, 7 (7%) gained 2 lines or more, and 7 (7%) lost 2 lines or more of best-corrected visual acuity. Mean logarithm of the minimal angle of resolution visual acuity showed minimal change 0.02 (range, −0.46 to 0.70; P = .14). Mean central foveal thickness decreased −18 μm (range, −242 to 198 μm; P = .06). Mean macular volume decreased −0.27 mm 3 (95% confidence interval, −0.4 to −0.1 mm 3 ; P = .004). On qualitative analysis, 4 (5%) eyes had complete resolution of exudative fluid, 40 (49%) showed partial resolution, 26 (32%) remained unchanged, and 12 (14%) showed worsened exudative fluid.
Conclusions
Aflibercept seems to be an effective alternative for neovascular age-related macular degeneration patients previously treated with bevacizumab, ranibizumab, or both at 4 months of follow-up. Most treated eyes demonstrated stable visual acuity and anatomic improvements by spectral-domain optical coherence tomography.
Age-related macular degeneration (AMD) is a leading cause of blindness for people older than 65 years in the United States. In the neovascular (wet or exudative) form of AMD, choroidal neovascular membranes disrupt the normal architecture of the choriocapillaris, Bruch membrane, and retinal pigment epithelium layer leading to retinal edema, submacular hemorrhage, as well as debilitating atrophy and scarring.
The use of intravitreal anti–vascular endothelial growth factor (VEGF) therapy currently is the standard of care for neovascular AMD. The 2 most commonly used agents, bevacizumab and ranibizumab, reduce exudative fluid and have been shown to improve best-corrected visual acuity (BCVA) in eyes with neovascular AMD compared with controls.
The VEGF Trap-Eye: Investigation of Efficacy and Safety in Wet AMD 1 and 2 studies led to the United States Food and Drug Administration approval of aflibercept (Eylea [VEGF Trap-Eye]; Regeneron, Tarrytown, New York, USA, and Bayer HealthCare, Berlin, Germany) for the treatment of neovascular AMD in November 2011. These pivotal randomized, multicenter, double-masked, active-controlled studies showed that intravitreal injections of 2 mg every 4 weeks or 2 mg every 8 weeks after a loading dose of 3 monthly injections demonstrated BCVA and anatomic outcomes at years 1 and 2 that were comparable with those after monthly ranibizumab injections.
Aflibercept is a recombinant soluble decoy receptor fusion protein consisting of the binding domains of VEGF receptors 1 and 2 fused to the fragment crystallizable portion of human immunoglobulin G-1 (IgG-1). This protein binds VEGF-A, VEGF-B, and placental growth factor, which inhibits the binding and activation of VEGF receptors.
Although aflibercept has shown efficacy as primary therapy for treatment-naïve neovascular AMD, most patients in the clinical setting previously were treated with other VEGF inhibitors, including bevacizumab, ranibizumab, or both. We report the short-term efficacy of aflibercept for neovascular AMD in the setting of chronic VEGF blockade in this manuscript.
Methods
Approval for this study of VEGF inhibitors for neovascular AMD was obtained from the Emory University Institutional Review Board before the research was conducted. Patient information was de-identified to remain compliant with the Health Insurance Portability and Accountability Act. This was a retrospective, interventional, noncomparative, consecutive series of neovascular AMD patients who were treated previously with ranibizumab, bevacizumab, or both and transitioned to aflibercept between February 1, 2012, and May 21, 2012. Indications for transition to aflibercept included persistent, recurrent, or worsening exudative fluid or hemorrhage on examination or spectral-domain optical coherence tomography (SD OCT). Patients also were transitioned if they had intolerance to previous bevacizumab or ranibizumab injections. As a general rule, before switching to aflibercept, eyes were treated aggressively with injections of bevacizumab or ranibizumab every 4 to 5 weeks as long as signs of exudation were present. Eyes that received at least 2 aflibercept injections and had follow-up at 4 months (±1 month) were included in the study. Patients with follow-up intervals outside of this specified time frame or those who reverted back to another anti-VEGF agent were excluded.
Each eye received intravitreal injections of aflibercept at the recommended dose of 2 mg (0.05 mL) every 4 weeks for the first 3 months, followed by a fourth intravitreal injection 1 to 2 months later. Variations in this protocol occurred based on the discretion of the treating physician or if patient follow-up precluded monthly injections.
Office-based Snellen visual acuity (VA) measurements using best spectacle-corrected or pinhole VAs were obtained on the day of aflibercept initiation, at subsequent follow-up visits, and at the patient’s final follow-up appointment. The study’s primary outcomes were the mean change in logarithm of the minimal angle of resolution (logMAR) VA and the percentage of patients with improved, stable, or worsened Snellen VA at 4 ± 1 months after starting aflibercept injections. Improvement and worsening were defined as gaining or losing 2 lines or more of Snellen VA. Stabilization was defined as remaining within a 1-line gain or loss from baseline Snellen VA. The number of eyes with baseline and final VA of 20/40 or better as well as the number of aflibercept injections were quantified.
Additional outcomes included the mean change in central foveal thickness, macular cube volume, as well as qualitative changes of intraretinal fluid (IRF), subretinal fluid (SRF), pigment epithelial detachments (PEDs), subretinal hemorrhage (SRH), and overall response to therapy on Cirrus SD OCT (Carl Zeiss Meditec, Inc, Dublin, California, USA). Changes in the degree of fluid by SD OCT from respective baseline images were classified as complete resolution, partial resolution, unchanged, or worse. Complete resolution was defined as resolution of all IRF, SRF, PEDs, and SRH. Any adverse events such as endophthalmitis, retinal detachment, sustained increase in intraocular pressure requiring topical ocular hypertensive medications, uveitis, and thromboembolic events (nonfatal stroke, nonfatal myocardial infarction, or vascular death, including deaths of unknown cause) also were recorded. Statistical analyses were performed using Microsoft Excel (Microsoft Corporation, Redmond, Washington, USA), and a 2-tailed paired t test was applied to compare means of continuous variables.
Results
Demographics and Clinical Characteristics
One hundred twenty-three eyes of 107 patients were transitioned from bevacizumab, ranibizumab, or both to aflibercept between February 1, 2012, and May 21, 2012. A total of 27 eyes were excluded from the study for the following reasons. Four eyes received only a single aflibercept injection. Fourteen eyes had follow-up outside the designated 4 ± 1-month window. Nine eyes reverted back to bevacizumab or ranibizumab. Of these, 6 were the result of subjective declines in vision that were not supported by objective BCVA testing and 2 were related to expense. A final patient experienced a drop in Snellen acuity from 20/50 to 20/400 (remained at 20/40 by potential acuity meter) 1 month after his second aflibercept injection, so the third injection was withheld. When he returned 2 weeks later with BCVA of 20/70, he was reverted back to ranibizumab injections. Ultimately, 96 eyes of 85 patients, 34 males and 51 females, were included in the study. Of these, 77 eyes (80%) had central foveal thickness and 67 eyes (70%) had macular cube volume values documented at both baseline and final examinations.
The average patient age was 79 years (range, 62 to 91). For the entire study population, the mean number of any previous anti-VEGF injections was 17 (range, 1 to 60). Thirty eyes (31%) previously had been treated exclusively with bevacizumab (mean, 14 injections; range, 1 to 53 injections), 43 eyes (45%) had been treated exclusively with ranibizumab (mean, 19 injections; range, 1 to 49 injections), and 23 eyes (24%) had been treated with both intravitreal agents (bevacizumab: mean, 13 injections; range, 1 to 32 injections; and ranibizumab: mean, 8 injections; range, 1 to 36 injections). At the time of transition to aflibercept, 82 (85%) eyes had evidence of active exudation in the form of SRF, IRF, PED, or SRH. Mean follow-up for study eyes was 114 days (range, 90 to 133 days). The mean number of aflibercept injections from initiation of therapy to last follow-up appointment, excluding the injection at the last visit, was 2.6 (range, 2 to 4). No patients received fewer than 2 injections before the last follow-up appointment ( Table 1 ).
| Characteristic | Value (Range) |
|---|---|
| No. of eyes | 96 |
| No. of patients | 85 |
| Gender | |
| Male | 34 |
| Female | 51 |
| Mean age (y) | 79 (62 to 91) |
| Interval between last agent and aflibercept (days) | 49 (18 to 355) |
| Mean follow-up after initiation of aflibercept (days) | 114 (90 to 133) |
| Mean no. of aflibercept injections before last follow-up visit | 2.6 (2 to 4) |
| No. of eyes with exudative fluid, hemorrhage, or both at first aflibercept injection | 82 |
| Mean no. of prior anti-VEGF injections for all study eyes | 17 (1 to 60) |
| Bevacizumab-only group (no. of eyes) | 30 |
| Mean no. of bevacizumab injections | 14 (1 to 53) |
| Ranibizumab-only group (no. of eyes) | 43 |
| Mean no. of ranibizumab injections | 19 (1 to 49) |
| Bevacizumab and ranibizumab group (no. of eyes) | 23 |
| Mean no. of bevacizumab injections | 13 (1 to 32) |
| Mean no. of ranibizumab injections | 8 (1 to 36) |
| Other prior treatment methods (no. of eyes) | |
| Photodynamic therapy | 5 |
| Pegaptanib injections | 1 |
| Other ocular conditions (no. of eyes) | |
| Epiretinal membrane | 4 |
| Glaucoma | 5 |
| Idiopathic polypoidal choroidal vasculopathy | 1 |
| Central serous chorioretinopathy | 2 |
| Presumed ocular histoplasmosis syndrome | 1 |
Visual Outcomes With Aflibercept After Prior Therapies
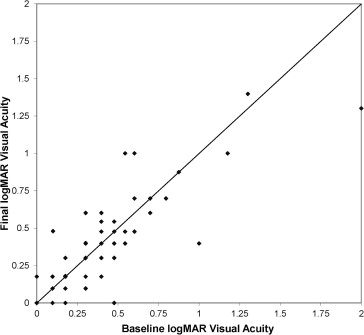
The average VA at the time of first treatment with aflibercept ranged from 20/40 to 20/50 in the groups receiving prior bevacizumab or ranibizumab monotherapy and was 20/60 for the group who received both prior bevacizumab and ranibizumab. For the group as a whole, the mean baseline VA was 20/50, and of these 96 eyes, 7 (7%) gained 2 lines or more, 82 (85%) remained stable within a 1-line gain or loss, and 7 (7%) lost 2 lines or more of Snellen VA. The mean gain in logMAR VA was 0.02 logMAR (range, −0.46 to 0.70 logMAR; P = .14; Figure 1 ). Thirty-eight (83%) of 46 eyes with 20/40 vision or better at baseline maintained this level of acuity and 8 eyes improved to this range by the end of the study.
Of the 30 eyes previously treated exclusively with bevacizumab, 1 (3%) gained 2 lines or more, 27 (90%) remained stable within a 1-line gain or loss, and 2 (7%) lost 2 lines or more of Snellen VA. The mean change in logMAR acuity for this group was −0.03 logMAR (range, −0.38 to 0.12 logMAR; P = .26). Fourteen (78%) of 18 eyes with 20/40 vision or better at baseline maintained this level of acuity by the end of the study.
Of the 43 eyes previously treated exclusively with ranibizumab, 3 (7%) gained 2 lines or more, 36 (84%) stabilized within a 1-line gain or loss, and 4 (9%) worsened by 2 lines or more of Snellen VA. The mean change in logMAR VA for this group was −0.004 logMAR (range, −0.46 to 0.47 logMAR; P = .8). Twenty-two (85%) of 26 eyes with 20/40 vision or better at baseline maintained this level of acuity and 3 eyes improved to this range by the end of the study.
In the 23 eyes whose prior treatment consisted of both bevacizumab and ranibizumab, 3 (13%) improved by 2 lines or more, 18 (78%) remained stable within a 1-line gain or loss, and 2 (9%) worsened by 2 lines or more of Snellen VA. There was a statistically significant improvement in mean logMAR VA of 0.13 logMAR (95% confidence interval [CI], 0.06 to 0.20 logMAR; P = .003). All 3 eyes with 20/40 vision or better at baseline maintained this acuity and 3 eyes improved to this level at last follow-up.
Anatomic Outcomes With Aflibercept After Prior Therapies
Central foveal thickness
The mean central foveal thickness of 77 eyes at the time of first treatment with aflibercept was 276 μm (range, 130 to 559 μm) for the entire study population, 278 μm (range, 130 to 459 μm) in the bevacizumab-only group, 258 μm (range, 165 to 348 μm) in the ranibizumab-only group, and 299 μm (range, 181 to 559 μm) for the group who previously received both agents. The mean change in central foveal thickness at the end of follow-up for the study population was −18 μm (range, −242 to 198 μm; P = .06; Figure 2 ), −20.4 μm (range, −168 to 198 μm; P = .26) for the bevacizumab-only group, −11.2 μm (range, −109 to 54 μm; P = .07) in the ranibizumab-only group, and −28.9 μm (range, −242 to 123 μm; P = .2) for eyes that received both agents.
Macular cube volume
The mean standard macular cube volume of 67 eyes at the time of first treatment with aflibercept was 9.8 mm 3 (range, 7.7 to 13.80 mm 3 ) for all patients, 9.8 mm 3 (range, 8.2 to 13.3 mm 3 ) in the bevacizumab-only group, 9.7 mm 3 (range, 7.8 to 12.2 mm 3 ) for the ranibizumab-only group, and 9.8 mm 3 (range, 7.7 to 13.8 mm 3 ) for patients who received bevacizumab and ranibizumab. The mean change in macular cube volume at the end of follow-up was −0.27 mm 3 (95% CI, −0.4 to −0.1 mm 3 ; P = .004) overall ( Figure 3 ), −0.35 mm 3 (95% CI, −0.53 to −0.11 mm 3 ; P = .007) for bevacizumab-only eyes, −0.02 mm 3 (range, −1.5 to 0.7 mm 3 ; P = .12) for ranibizumab-only eyes, and −0.53 mm 3 (95% CI, −1.02 to −0.04 mm 3 ; P = .02) for eyes that previously received both bevacizumab and ranibizumab.




