Purpose
To assess baseline and follow-up characteristics of choroidal neovascularization (CNV) lesions in age-related macular degeneration in relation to the development of subfoveal subretinal fibrosis.
Design
Retrospective, observational case series.
Methods
settings and study population : One hundred ninety-seven treatment-naïve eyes in 197 patients with CNV in age-related macular degeneration without subfoveal fibrosis at first presentation who were treated with ranibizumab in a pro re nata regimen. main outcome measure : Subfoveal fibrosis at the conclusion follow-up of 24 months or fewer.
Results
The hazard ratio of any subfoveal fibrosis developing in eyes with predominantly classic CNV was 5.95 (95% confidence interval [CI], 3.25 to 10.90) compared with minimally classic and occult CNV, whereas the hazard ratio of fibrosis developing with foveal atrophy was 3.38 (95% CI, 1.47 to 7.81; mean follow-up, 1.80 years; 95% CI, 1.75 to 1.85 years). The hazard ratio of any fibrosis developing was 3.38 (95% CI, 1.10 to 10.38) in eyes with a baseline best-corrected visual acuity of 40 or worse using Early Treatment Diabetic Retinopathy Study letter scores, as compared with eyes with a baseline best-corrected visual acuity of 70 letters or more. An interval between diagnosis and treatment of 15 days or more was associated with a hazard ratio of any fibrosis developing of 2.24 (95% CI, 1.28 to 3.94) as compared with an interval of fewer than 15 days. Compared with eyes in which fibrosis did not develop, eyes in which prominent fibrosis or fibrosis developed with foveal atrophy lost 8.5 more Early Treatment Diabetic Retinopathy Study letters (95% CI, −1.0 to −15.9; P = .0242) and 10.3 more Early Treatment Diabetic Retinopathy Study letters (95% CI, −4.0 to −16.5; P = .0012), respectively.
Conclusions
The development of subfoveal fibrosis in neovascular age-related macular degeneration was associated with predominantly classic CNV and poorer visual acuity at first presentation, a longer interval between diagnosis and treatment, and approximately 2 lines of additional visual loss at the conclusion follow-up.
The introduction of intravitreal vascular endothelial growth factor (VEGF) A inhibitors has improved markedly the visual prognosis in neovascular age-related macular degeneration (AMD), but subretinal fibrosis and atrophy of the outer retina remain prominent pathologic elements in end-stage disease with severe visual loss. These limitations of current wet AMD management indicate that AMD will remain a prominent cause of visual impairment in people 50 years of age and older, despite a decreasing incidence of AMD-related legal blindness. In the Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular Age-Related Macular Degeneration (MARINA) study, atrophic scarring was related to poor visual outcomes after ranibizumab treatment. In this retrospective observational case series, we examined the impact of characteristics at first presentation on the development of subfoveal fibrosis in patients who underwent routine intravitreal ranibizumab therapy for neovascular AMD.
Methods
This retrospective, observational case series complied with the tenets of the Declaration of Helsinki. The Committees on Health Research Ethics for the Capital Region of Denmark waived institutional review board approval because the study was retrospective and did not involve biological material.
The study comprised patients of white ethnicity who began intravitreal ranibizumab therapy for choroidal neovascularization (CNV) in AMD at the Glostrup Hospital between February 1, 2007, and July 1, 2007. Final follow-up took place 2 years after the initiation of intravitreal ranibizumab therapy, which was the only therapeutic option used during the study period. The criteria for initiating ranibizumab therapy were active subfoveal CNV, best-corrected visual acuity (BCVA) of 20/400 or better, an ability to comply with treatment and follow-up examinations, an absence of extensive subfoveal fibrosis, and a greatest linear lesion dimension of no more than 5400 μm. The treatment protocol prescribed 3 initial 0.5-mg ranibizumab injections at intervals of 4 weeks followed by renewed clinical examination 4 weeks after the third injection, at which point flexible retreatment was provided. The first ranibizumab injection was administered at a later date from when the diagnosis of neovascular AMD was made. Follow-up examinations were scheduled monthly from 16 weeks after the first injection and included updating the history of symptoms, an assessment of BCVA, transfoveal optical coherence tomography (OCT), and fundus photography. Guideline criteria for ranibizumab retreatment were signs of CNV activity in the absence of criteria for terminating treatment. Signs of CNV activity were defined as an increase of fluid under the macula compared with any previous visit, an increase in macular thickness of 50 μm or more, persistent thickening and fluid in the macula despite ranibizumab injections, fresh hemorrhage, and growth of the CNV. After retreatment, which consisted of a variable number of injections, follow-up was rescheduled for 4 weeks after the last injection. Guideline criteria for the termination of treatment were persistent BCVA of 20/400 or worse and BCVA that was stable for 6 months or longer without treatment. Atrophy of the foveal photoreceptor layer, geographic atrophy of the foveal retinal pigment epithelium (RPE), and subfoveal fibrosis indicating the absence of a potential for visual acuity improvement were adjuvant criteria for terminating therapy and referring the patient back to his or her general ophthalmologist. Termination of therapy was not considered during the first year after the initiation of injections.
Clinical and demographic data were retrieved from patient records. BCVA was assessed using a Snellen chart at a 6-m distance and an Early Treatment Diabetic Retinopathy Study (ETDRS) chart at a 2-m distance. Data are converted from Snellen to ETDRS letters for analysis in a minority of cases (5%) in which the standard procedure had not been applied. Clinical examination included slit-lamp biomicroscopy and fluorescein angiography. All diagnostics were performed by ophthalmologists or physicians in ophthalmology training.
Inclusion criteria were that patients be 50 years of age or older, have a BCVA of 20/400 or better, be diagnosed with CNV secondary to AMD, have a greatest lesion extent no larger than 5400 μm, and have CNV comprising at least 50% of the lesion. Eyes were excluded from analysis if they previously had undergone photodynamic therapy, focal laser photocoagulation, or injection with ranibizumab, bevacizumab, or pegaptanib. Eyes with retinal angiomatous proliferations, predominantly hemorrhagic lesions, extrafoveal lesions, or subretinal fibrosis at baseline, and eyes in which RPE tears developed during follow-up were excluded from the analysis. Eyes with missing information about baseline BCVA and fluorescein angiogram results also were excluded. These criteria led to the exclusion of 72 patients: 6 (8%) because of RPE tears, 9 (13%) because of predominantly hemorrhagic lesions, 9 (13%) because of retinal angiomatous proliferations, 12 (17%) because of lesions larger than 5400 μm, 4 (5%) because of extrafoveal lesions, 3 (4%) because of subretinal fibrosis at first presentation, and 29 (40%) because of missing data at baseline. The type of lesion and lesion components at the time of the first diagnosis of neovascular AMD were determined independently by 2 masked graders. All photographs and OCT images at diagnosis and follow-up were reviewed twice by a masked investigator (S.B.B.). Intergrader reliability and intragrader reproducibility were 85% or better, depending on parameter (type, lesion size, stage of fibrosis, etc.). Adherence to criteria for initiating and resuming therapy was assessed by one of the authors (S.B.B.), who reviewed all data and found full compliance with departmental guidelines. Subfoveal fibrosis and retinal atrophy based on OCT and color fundus photographic readings were evaluated based on a visual evaluation of images and scans from 16 weeks after the first intravitreal injection and onward.
Baseline fundus photographs and fluorescein angiograms were 30- and 50-degree digital color and red-free greyscale photographs (ff450; Carl Zeiss-Meditech, Jena, Germany). Fundus photographic quality was graded as acceptable, questionable, not gradable, or missing. Lesion characteristics were measured from digital nonstereoscopic fundus photographs and fluorescein angiograms using standardized grading grids and assuming a disc area (DA) of 2.54 mm 2 (adapted from the Macular Photocoagulation Study and an extension of the Wisconsin age-related maculopathy grading system [University of Wisconsin Fundus Photographic Reading Center, 1998, unpublished]). Lesions were classified as type 1 for occult and minimally classic CNV lesions and type 2 lesions for classic or predominantly classic CNV lesions. Total lesion area was defined by the leakage area on early- to mid-phase angiograms (type 1 lesions) or early-phase angiograms (type 2 lesions) together with any area of contiguous thick hemorrhages, blocked fluorescence, subretinal fibrous tissue, or serous pigment epithelial detachment adjacent to the CNV lesion and likely to obscure the boundaries of the CNV. Temporal trends of fluorescein leakage during an angiography session were defined as no leakage, fading during or before the late phase, plateaus in mid and late phases, or increasing leakage throughout the session.
Subretinal fibrous tissue was diagnosed when whitish material under the retina that was not dehemoglobinized blood or hard exudate was seen. As in the Age-Related Eye Disease Study protocol, no formal attempt was made to distinguish between fibrin and fibrous tissue. Subretinal fibrin could be replaced by fibrous tissue, but once present, fibrous tissue should remain and be of stable or expanding character or should be shrinking somewhat with preserved lines of traction, outline, and so forth.
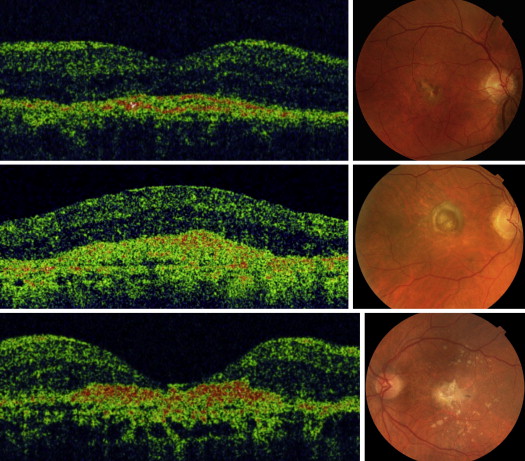
OCT was performed using a Topcon spectral-domain OCT/fundus camera with predefined scan options of 6 radial scans and 2 crosshair scans (Topcon, Tokyo, Japan). The appearance of subretinal fibrous tissue on OCT was assessed on the basis of standard photographs representing a 3-stage grading of OCT findings, which is an abbreviated and adjusted version of a previously published 5-stage classification. A basic assumption was that on OCT, subretinal fibrous tissue has the appearance of a continuous, highly reflective band between the neurosensory retina—or what is left of it—and the Bruch membrane, with the reservations that the Bruch membrane may not be visible if there is dense fibrous tissue in front of it and that it may be indiscernible if it is embedded in a fibrovascular CNV. Based on OCT, subfoveal fibrous tissue was categorized, with reference to standard photographs ( Figure 1 ), as minimal fibrosis with or without subretinal fluid (stage I), as prominent fibrosis with or without cystoid edema (stage II) and fibrosis with overlying neurosensory retinal atrophy (stage III). The diagnosis of subretinal fibrous tissue required concordance between color fundus photographs and OCT. Incident subfoveal fibrous tissue was the primary outcome parameter of the study, the principal end point being stage III fibrosis.
Data analysis and graphic illustrations were made using SAS software version 9.1.3 (SAS Institute, Cary, North Carolina, USA). A general linear model was used to assess the relationship between BCVA and subfoveal fibrous tissue after 24 months of variable ranibizumab treatment. A Cox proportional hazards model, taking length of follow-up into consideration and adjusting for age and gender, was used to find hazard ratios (relative risk ratios) for subfoveal fibrous tissue, depending on initial lesion characteristics. Cox regression analysis was performed with and without interaction terms. No significant association was found between baseline characteristics and developments of subretinal fibrous tissue if we excluded type 2 lesions from the Cox regression model and made a partial analysis within the type 1 lesion group. A likelihood ratio test was performed to show that the assumption of proportionality in the regression model was met. The exit date also was the last follow-up and could be registered until September 1, 2009, when the analysis was closed after 2 years of variable treatment with ranibizumab. Outcome analysis was executed using data from the date when fibrosis was first visible (event) or the exit date if no fibrosis had developed (survival) using Cox regression analysis. A Kaplan-Meier survival curve was made to illustrate fibrosis-free survival. The threshold of statistical significance was set at P ≤ .05 (2-sided).
Results
At study completion, follow-up data were available for 197 eyes from 197 patients with a mean period of observation of 1.80 years (95% confidence interval [CI], 1.75 to 1.85 years; Table 1 ). The mean interval between making the diagnosis of neovascular AMD and giving the first ranibizumab injection was 17.2 days (95% CI, 15.6 to 18.9 days). In the first year of treatment, the total number of intravitreal injections was 1015 in 197 eyes, that is, a mean of 5.15 injections per patient (95% CI, 4.94 to 5.37 injections). The total number of examinations between the first injection and 12 months was 977, that is, a mean of 4.96 per patient (95% CI, 4.84 to 5.08 examinations).
| Gender, no. (%), n = 197 | |
| Male | 73 (37.1) |
| Female | 124 (62.9) |
| Age (y) | |
| Mean (95% CI) | 78.7 (77.5 to 79.8) |
| Median (range) | 80.0 (53 to 97) |
| Visual acuity (ETDRS letters) | |
| Mean (95% CI) | 53.8 (51.7 to 56.0) |
| Median (range) | 58 (13 to 85) |
| Type of choroidal neovascularization, no. (%) | |
| Predominantly classic | 49 (24.9) |
| Minimally classic | 43 (21.8) |
| Occult | 105 (53.3) |
| Area of choroidal neovascularization (DA) | |
| Mean (95% CI) a | 2.5 (2.2 to 2.8) |
| Median (range) | 3.0 (0.125 to 12) |
| Area of total lesion (DA) | |
| Mean (95% CI) a | 3.3 (3.0 to 3.7) |
| Median (range) | 3.5 (0.125 to 12) |
| Total area of leakage from choroidal neovascularization (DA) | |
| Mean (95% CI) a | 3.3 (3.0 to 3.6) |
| Median (range) | 3.4 (0.5 to 12) |
| Central retinal thickness (μm) b | |
| Mean (95% CI) | 322 (306 to 338) |
| Median (range) | 300 (150 to 600) |
| Fellow eye characteristics, no. (%) c | |
| Nonneovascular AMD | 80 (41.2) |
| Advanced AMD with geographic atrophy | 12 (6.2) |
| Advanced neovascular AMD | 36 (18.6) |
| Advanced AMD with fibrovascular scarring | 64 (33.0) |
| Retinal pigment epithelial detachments | 0 (0) |
| Epiretinal fibrosis | 2 (0.01) |
a Means are calculated from log-transformed data.
b Available in 140 (71%) of 197 eyes.
On average, patients with type 2 CNV lesions read 9.6 ETDRS letters (95% CI, −4.8 to −14.4 ETDRS letters; P < .0001) fewer at baseline than patients with type 1 CNV lesions. The total lesion size and CNV size at baseline were 33% (95% CI, 14% to 47%; P = .0012) and 42% (95% CI, 25% to 56%; P < .0001) smaller, respectively, in type 2 lesions than in type 1 lesions. The total area of leakage was 32% (95% CI, 6% to 65%) greater in type 2 lesions than in type 1 lesions ( P = .0132). On initiation of treatment, fluorescein angiograms showed an increase in leakage intensity during subsequent phases of the angiogram in all type 2 lesions.
After 2 years, BCVA was 12.2 ETDRS letters (95% CI, −5.6 to −18.7 ETDRS letters; P = .0003) lower in eyes with type 2 lesions than in eyes with type 1 lesions, but eyes with type 2 lesions did not lose significantly more ETDRS letters (difference of −2.6 ETDRS letters; 95% CI, 3.0 to −8.1 ETDRS letters; P = .3512). Baseline age, interval from diagnosis to treatment, follow-up period, number of ranibizumab injections, and presence of thick bleeding were comparable for type 1 and type 2 lesions ( P > .05 for all analyses). Eyes with prominently fibrotic or fibroatrophic lesions (stages II and III) had lost 8.5 more ETDRS letters (95% CI, −1.0 to −15.9 ETDRS letters; P < .0242) and 10.3 more ETDRS letters (95% CI, −4.0 to −16.5 ETDRS letters; P = .0012), respectively, after 2 years than eyes in which fibrosis did not develop ( Figure 2 ). BCVA after 24 months was available for 176 (89%) of the 197 study eyes.
The mean area of subretinal fibrous tissue after 12 months was 0.9 DA (95% CI, 0.7 to 1.1 DA) and after 2 years, it had increased to 1.0 DA (95% CI, 0.8 to 1.0 DA). During the first 6 months of treatment with ranibizumab, subfoveal fibrous tissue had developed in 64 eyes (32%), mainly in the form of stage I fibrosis. After 12 months of treatment, subfoveal fibrous tissue was visible in 71 eyes (36%), of which 50 eyes had stage II and stage III fibrosis. During the 2-year period of analysis, subfoveal fibrosis developed in a total of 77 eyes (40%), of which most (43 eyes) proceeded to stage III fibrosis.
The hazard ratio for developing subfoveal fibrosis of any stage (stages I, II, and III) within 2 years was 5.95 in eyes with type 2 lesions relative to that in type 1 lesions ( Table 2 and Figure 3 ). In most cases, subfoveal fibrosis developed during the first 6 months after initiation of treatment ( Figure 3 ).
| Baseline Characteristics | Cases with Subretinal Fibrosis/Total (%) | No. of Follow-up Years | Multivariate Relative Risk (95% CI) a | P Value |
|---|---|---|---|---|
| Type of lesion | ||||
| Type 1 | 53/148 (36) | 270 | 1 | — |
| Type 2 | 41/49 (84) | 85 | 5.95 (3.25 to 10.90) | <.0001 |
| Area of CNV (DA) | ||||
| <1.5 | 18/41 (44) | 77 | 1 | — |
| 1.5 to 3.0 | 27/55 (49) | 99 | 2.36 (0.84 to 6.62) | .1028 |
| 3.0 to 5.0 | 24/47 (51) | 81 | 2.74 (0.92 to 8.17) | .0705 |
| 5.0 to 12.0 | 25/54 (46) | 97 | 4.49 (1.33 to 15.14) | .0155 |
| Trend test | .1173 | |||
| Visual acuity letters b | ||||
| ≥70 | 10/34 (29) | 65 | 1 | — |
| 60 to 70 | 15/50 (30) | 96 | 0.95 (0.31 to 2.87) | .9250 |
| 50 to 60 | 28/53 (53) | 95 | 1.93 (0.67 to 5.56) | .2220 |
| 40 to 50 | 20/31 (65) | 53 | 2.26 (0.77 to 6.61) | .1382 |
| 30 to 40 | 19/27 (79) | 45 | 3.38 (1.10 to 10.38) | .0336 |
| 20 to 30 | 12/12 (100) | 17 | 7.17 (2.17 to 23.70) | .0012 |
| Trend test | .0017 | |||
| Interval, diagnosis to first treatment (days) | ||||
| ≤14 | 47/100 (47) | 171 | 1 | — |
| >14 | 47/97 (48) | 184 | 2.24 (1.28 to 3.94) | .0050 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


