CHAPTER 149 Sensorineural Hearing Loss in Adults
Clinical Evaluation of the Patient with Hearing Loss
Audiometric Testing
Only a brief discussion of audiometric testing is presented here. Conventional audiometric testing is discussed more thoroughly in Chapter 133, and electrophysiologic testing is addressed in Chapter 134.
Auditory brainstem response (ABR) testing is useful in evaluating the possibility of a retrocochlear etiology, and for establishing thresholds in difficult-to-test patients (young children or malingerers). In the past, the ABR was believed to be a highly sensitive test for the presence of a retrocochlear lesion. It is less commonly used in this role today because of reduced sensitivity in patients with small vestibular schwannomas—tumors that are readily detected with the high accuracy offered by magnetic resonance imaging (MRI).1–3 New modifications of the ABR may increase this study’s sensitivity.4
Electrocochleography differs from ABR testing in that the reference electrode is placed closer to the cochlea (on or close to the tympanic membrane or promontory). This allows measurement of the cochlear microphonic potential, the summating potential, and the auditory nerve action potential. Wave I of the ABR corresponds to the action potential of electrocochleography. Approximately two thirds of patients with classic Meniere’s disease have an elevated summating potential-to-action potential ratio. It is believed that such a finding suggests the presence of endolymphatic hydrops.5,6 Electrocochleography can also be useful in patients in whom wave I of the ABR is weak or not present because the location of the reference electrode inherently enlarges wave I.
Radiographic Testing
Radiographic imaging is warranted in selected patients with SNHL. MRI with gadolinium enhancement is currently the gold standard in evaluating potential retrocochlear hearing losses. The role of MRI versus ABR testing in this regard is controversial. It is clear, however, that MRI with gadolinium is much more sensitive than ABR for diagnosis of small lesions.1–3 Selective T2-weighted fast spin-echo MRI may be almost as sensitive as gadolinium-enhanced standard MRI and is less expensive.7 Computed tomography (CT) is useful in patients with suspected labyrinthine anomalies, such as large vestibular aqueduct syndrome or Mondini dysplasia. CT may also be useful in patients with suspected labyrinthine fistula or temporal bone fractures. High-resolution CT with reformatted images in the plane of, and perpendicular to, the semicircular canals is the study of choice for showing semicircular canal dehiscence syndrome.8
Etiology of Sensorineural Hearing Loss
Developmental and Hereditary Disorders
Hereditary Disorders of Adult Onset
The discussion of hereditary causes of hearing loss in this chapter is limited to the more common etiologies primarily presenting in adulthood. Hereditary factors frequently play a role in SNHL, and research in this area is expanding rapidly. For a complete categorization and review of these disorders, the reader is referred to an excellent and encyclopedic work by Toriello and colleagues.9
Nonsyndromic Hereditary Hearing Loss
Most hereditary SNHL is not associated with other hereditary abnormalities. Hereditary hearing loss without associated abnormalities is much more common than generally appreciated and frequently is overlooked. It is likely that genetic factors play a role in presbycusis and in susceptibility to noise-induced hearing loss (NIHL).10–13 Distinct patterns of hereditary hearing loss transmitted in autosomal-dominant, autosomal-recessive, and X-linked fashion have been well described. Recessive or dominant isolated SNHL can be progressive or static, and may be congenital, present first at birth, manifest in childhood, or manifest in adulthood. Approximately 90% of inherited SNHL is recessive.
Waardenburg’s Syndrome
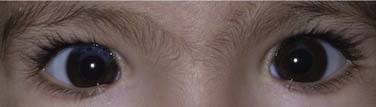
Waardenburg’s syndrome is transmitted in an autosomal-dominant fashion and consists of a constellation of findings, including (1) dystopia canthorum (lateral displacement of the medial canthi), (2) broad nasal root, (3) confluence of the medial portions of the eyebrows, (4) partial or total heterochromia iridis, (5) a white forelock, and (6) SNHL (Fig. 149-1). There is extreme variability in the expression of this disorder, and the hearing loss can vary from profound to none at all. The hearing loss can be unilateral or bilateral, and can be associated with vestibular abnormalities.
Alport’s Syndrome
Alport’s syndrome is characterized by interstitial nephritis, SNHL, and, much less commonly, ocular manifestations.14 This disease is unique because it is more common in women, but typically is much more severe in men. In the past, it has been thought to be transmitted in an autosomal-dominant fashion. It is now clear that there is significant genetic heterogeneity. Hearing loss is progressive and variable, usually beginning in the second decade of life. By age 20 to 40 years, 50% to 75% of men develop end-stage renal failure.
Inner Ear Anomalies
Many patterns of dysplasia of the inner ear have been described, and most have been associated with SNHL. These dysplastic patterns of development may be inherited, sporadic, or the result of chromosomal abnormalities. Commonly used descriptive terms for these dysplasias include Scheibe dysplasia (cochleosaccular dysplasia, involving membranous labyrinth only), Mondini dysplasia (dysplasia of bony and membranous labyrinth), and common cavity deformity (otocyst-like labyrinth with no cochlea or clear vestibular organs). Patterns of labyrinthine dysplasias form a spectrum with all manner of anomalies and patterns of hearing loss.15
Large Vestibular Aqueduct Syndrome
One form of inner ear dysplasia is unique because it has been associated with delayed onset of SNHL. An enlarged vestibular aqueduct is commonly seen in combination with other inner ear dysplasias, but more recently, it has been noted as an isolated finding in many ears. These patients may have any level of hearing from normal to a profound loss. Frequently, both ears are affected, and the losses are asymmetric. Fluctuation of hearing is common and usually affects one ear at a time; this may manifest as anacusis in one ear with fluctuation in the other. There is often evidence of a conductive component to the low-frequency portion of hearing loss. In patients who have been followed over time, a progressive stepwise loss has been noted in many.15 This syndrome has been found to be familial in some cases, and probably occurs much more commonly than generally appreciated.16 It is seen in isolation, as part of the Mondini malformation, and in patients with branchio-oto-renal syndrome17 and Pendred syndrome.18 It is well shown on high-resolution CT imaging of the temporal bone (Fig. 149-2).
Infectious Disorders
Labyrinthitis
An infectious or inflammatory process within the labyrinth can take two forms pathologically: serous (sometimes referred to as “toxic”) or suppurative. Serous labyrinthitis is defined as an abnormal process within the labyrinth, caused by the degradation of the tissue-fluid environment within the inner ear.19 It may be caused by bacterial toxins or contamination of perilymph with blood, products of tissue injury, or air at surgery. Bacterial toxins may enter the inner ear during the course of acute or chronic otitis media, presumably through either the oval or the round window membranes. Because both acute and chronic suppurative otitis media are common, and because SNHL associated with either condition is rare, these membranes seem to provide an excellent barrier preventing transmission of bacteria or their toxins to the inner ear. The principal abnormal finding in patients with serous labyrinthitis is endolymphatic hydrops, and the hearing loss and vestibular dysfunction associated with this state can be permanent or transient.
Commonly, a clinical diagnosis of labyrinthitis is made when patients present with sudden onset of SNHL and acute vertigo. The exact etiology in cases such as this is uncertain, but it is probably identical or similar to the etiology of sudden SNHL. The evidence tends to support the theory that this is most commonly caused by a viral labyrinthitis.20,21
Otitis Media
SNHL is rarely associated with acute otitis media. No study has shown a relationship between SNHL and frequency of acute otitis media.22 Patients with long-standing chronic otitis media commonly have a mixed hearing loss. Whether the sensorineural component of this loss is a result of the infectious process itself, or a result of other factors such as surgery or chronic use of ototoxic topical antibiotics, has been a long-standing controversy. When controlled for sensorineural losses associated with surgery, there seems to be no increase in SNHL in patients with chronic otitis media.23
Viral Infections
Cytomegalovirus infection is thought by some authors to be a common cause of congenital and progressive hearing loss in children.24–27 It has been proposed to be a cause of sudden SNHL in adults.28 Hearing loss associated with acquired immunodeficiency syndrome (AIDS) may represent reactivation of latent cytomegalovirus infections.29
Syphilis
Congenital or acquired syphilis has been well established as a cause of SNHL. Although hearing loss is not associated with primary acquired syphilis, its incidence has been estimated to be 80% in patients with symptomatic neurosyphilis, 29% in patients with asymptomatic neurosyphilis, 25% in patients with late latent syphilis, and 17% in patients with congenital syphilis.19 The mechanism of hearing loss in syphilis is either a meningolabyrinthitis as seen in neurosyphilis, or an osteitis of the temporal bone with secondary involvement of the labyrinth as seen in late congenital, late latent, or tertiary syphilis.30 Pathologically, a resorptive osteitis is seen in the temporal bone, with progressive endolymphatic hydrops noted within the labyrinth. Clinically, the presentation of syphilitic hearing loss often is indistinguishable from Meniere’s disease, with fluctuating hearing loss, tinnitus, aural fullness, and episodic vertigo. Hennebert’s sign (a positive fistula test without middle ear disease) and Tullio’s phenomenon (vertigo or nystagmus on exposure to high-intensity sound) have been strongly associated with otosyphilis.19 The generally recommended treatment consists of an antibiotic protocol adequate for neurosyphilis with the addition of systemic corticosteroids.31
Rocky Mountain Spotted Fever
Rocky Mountain spotted fever is a tickborne infection caused by Rickettsia rickettsii. Headache, fever, myalgias, and an expanding petechial rash follow the tick bite by approximately 1 week. The disease results in systemic vasculitis, resulting in encephalitis, nephritis, and hepatitis. Rapidly progressive SNHL has been associated with Rocky Mountain spotted fever and may be transient.32,33 Vasculitis involving the auditory system has been postulated to be the etiology of the hearing loss. Diagnosis is made primarily by clinical presentation and is confirmed by serologic titers. Treatment is with broad-spectrum antibiotics.
Lyme Disease
Lyme disease is a tickborne spirochetal illness caused by Borrelia burgdorferi. Although the most well-known otolaryngologic manifestation of the disease is facial paralysis, there is some evidence that the disease can be a cause of SNHL.34–37 Although its true significance remains unclear, Lyme disease should be considered a possible etiology of SNHL in endemic areas.
Pharmacologic Toxicity
Aminoglycosides
At least 96 different pharmacologic agents have potential ototoxic side effects.38,39 Among these, aminoglycoside antibiotics are perhaps the most common offending agents. This group of antibiotics includes streptomycin, dihydrostreptomycin, kanamycin, neomycin, amikacin, gentamicin, tobramycin, and netilmicin. Drugs that are ototoxic frequently are also nephrotoxic and vice versa (aminoglycosides, loop diuretics, potassium bromates, and nonsteroidal anti-inflammatory drugs [NSAIDs]). Alport’s syndrome, described earlier, is a hereditary disorder affecting the kidneys and the inner ear, and there are developmental disorders resulting in renal and inner ear abnormalities. The strong association between pathology of the renal and auditory systems has not been well explained.
Aminoglycosides target the hair cells and enter the hair cell in an energy-dependent process. The end result is death of the hair cell. The reader is referred to excellent reviews of the current understanding of the mechanism of aminoglycoside ototoxicity (see Chapter 154).40–42 The final common pathway of hair cell damage consists of the generation of reactive oxygen species. Different aminoglycosides have affinities for differing groups of hair cells, which result in different patterns of ototoxicity with different aminoglycosides. Kanamycin, tobramycin, amikacin, neomycin, and dihydrostreptomycin are more cochleotoxic than vestibulotoxic. Others, such as streptomycin and gentamicin, are more vestibulotoxic than cochleotoxic. The time course of the toxicity can vary.39 Neomycin toxicity is typically rapid and profound, whereas a significant delayed effect has been noted for systemically administered streptomycin, dihydrostreptomycin, tobramycin, amikacin, and netilmicin, and for gentamicin administered through the middle ear.43
The hearing loss may be unilateral or asymmetric and can progress during or after cessation of therapy. Some degree of reversibility of the hearing loss sometimes is noted weeks to months after treatment.39 Protective agents, including antioxidants, show promise for preventing or reducing aminoglycoside toxicity. More recently, the use of salicylates has been proposed.44 A placebo-controlled clinical trial in China has shown a beneficial effect from the use of aspirin during aminoglycoside administration.45
Ototopical Preparations
Topical preparations containing neomycin, gentamicin, and tobramycin have long been used directly in the ear for treatment of otitis externa and chronic otitis media. Placement of aminoglycosides within a healthy middle ear space frequently results in cochlear or vestibular ototoxicity as shown in experimental animals and in patients. This effect is now used to perform a titrated chemical labyrinthectomy for patients with Meniere’s disease.46,47 These same drugs have been used extensively over the years in countless ears with chronic otitis media with little to no apparent clinically significant effect on hearing or vestibular function.48,49 Reduced permeability of the inflamed round and oval window membranes and dilution of the toxic drugs by purulent fluids and increased absorption into the vascular system by the hyperemic mucosa probably account for this decreased toxicity in the presence of otitis media.
It is clear, however, that use of aminoglycosides in the middle ear of animals does cause significant and predictable cochlear and vestibular toxicity in animals.50 Based on this toxicity, and on the now widespread effective use of aminoglycosides to create a chemical labyrinthectomy, it is now generally regarded as unwise to use topical aminoglycoside antibiotics for treatment of otitis media. The American Academy of Otolaryngology–Head and Neck Surgery convened a consensus panel in 2004 that, after careful review of the literature, recommended against the use of aminoglycosides in topical form in the middle ear unless no alternative was available.51 Other ingredients of older ototopical preparations also have ototoxic potential (e.g., polymyxin B, propylene glycol, acetic acid, and antifungal agents).52,53 It seems prudent to use only agents specifically designed and approved for use in the middle ear for treatment of chronic otitis.
Loop Diuretics
The loop diuretics ethacrynic acid, bumetanide, and furosemide exert their diuretic effect by blocking sodium and water reabsorption in the proximal portion of the loop of Henle. The use of these drugs has been associated with a reversible SNHL and with the potentiation of aminoglycoside-induced hearing loss. The loss typically is bilateral and symmetric, and may be sudden in onset.54,55 These drugs seem to alter metabolism in the stria vascularis, resulting in alteration of endolymphatic ion concentration and endocochlear potential.56 Risk factors for loop diuretic–induced ototoxicity include (1) renal failure, (2) rapid infusion, and (3) concomitant aminoglycoside administration.125
Antimalarials
Quinine has long been known to be associated with the development of tinnitus, SNHL, and visual disturbances.57 The drug, derived from the bark of the cinchona tree, has a colorful history as an antipyretic. It was dispensed by quacks and in secret remedies in the 17th and 18th centuries. The syndrome of tinnitus, headache, nausea, and disturbed vision is termed cinchonism. Larger doses may cause a more severe form of the syndrome, which also includes gastrointestinal, CNS, cardiovascular, and dermatologic manifestations. Quinine is used as an adjunct in the treatment of malaria and nocturnal leg cramps.58 The ototoxic effect of quinine seems to be primarily on hearing and usually is transient. Permanent hearing loss may occur with large doses or in sensitive patients. Chloroquine and hydroxychloroquine (Plaquenil) are currently used antimalarial drugs structurally related to quinine. They have also been associated with ototoxicity and retinopathy. Ototoxicity with these drugs seems to be rare and possibly reversible.59,60
Salicylates
Aspirin and other salicylates are strongly associated with tinnitus and reversible SNHL. The hearing loss is dose-dependent and can be in the moderate-to-severe range. On discontinuation of the drug, hearing returns to normal within 72 hours.57 Tinnitus consistently occurs at a dose of 6 to 8 g/day of aspirin and at lower doses in some patients.61,62 Caloric responses also can be reduced by salicylates.63 The site of the ototoxic effect seems to be at the level of basic cochlear mechanics, as evidenced by SNHL, loss of otoacoustic emissions, reduced cochlear action potentials, and alteration of the “tips” of auditory nerve fiber tuning curves.64 These effects may be a result of alteration in turgidity and motility of outer hair cells.65
Nonsteroidal Anti-inflammatory Drugs
NSAIDs share many of the therapeutic actions and side effects of salicylates. Although there are isolated reports of hearing loss caused by naproxen,66 ketorolac,67 and piroxicam,68 ototoxicity resulting from use of NSAIDs generally is rare compared with salicylates.57,69 Similar to salicylates, animal models of NSAID ototoxicity show only reversible physiologic changes, without major morphologic changes.
Vancomycin
Vancomycin generally is believed to be ototoxic, but the available data are difficult to evaluate.70 In clinical reports of vancomycin ototoxicity, patients almost always also received loop diuretics or aminoglycosides. Vancomycin has been associated with ototoxicity when administered intravenously, but not when given orally. Auditory impairment has been reported to be permanent or transient and is extremely unusual if serum concentrations are less than 30 mg/mL. In animals, vancomycin ototoxicity does not occur, unless toxic levels are administered.71 Vancomycin is nephrotoxic and excreted by the kidneys. Renal failure can prolong vancomycin half-life and increase the likelihood of ototoxicity. Ototoxicity and nephrotoxicity are reputed to be less common with newer, more purified formulations of vancomycin.72 When given orally, or in appropriate intravenous doses, vancomycin ototoxicity seems to be very rare, but vancomycin may potentiate other ototoxic drugs.73,74 There is one reported case of severe, irreversible hearing loss associated with intrathecal administration of vancomycin.75
Erythromycin
Numerous case reports document SNHL associated with erythromycin administration.76 In almost all reports, the drug was given intravenously rather than orally. The hearing loss seems to be uncommon and, in most cases, recovers within 1 to 3 weeks after the drug is stopped. The risk of erythromycin ototoxicity seems to be greater in patients with renal or hepatic insufficiency. There have also been reports of ototoxicity associated with the newer macrolide antibiotic azithromycin.77 Limited histologic data suggest that the site of lesion in erythromycin toxicity is the stria vascularis.78
Cisplatin and Carboplatin
cis-Diamminedichloroplatinum (cisplatin) is a cell cycle–nonspecific cancer chemotherapeutic agent that produces dose-limiting SNHL and peripheral neuropathy, and a dose-related cumulative renal toxicity, hematologic toxicity, and gastrointestinal toxicity.79 The incidence of hearing loss varies in adults (25% to 86%) and children (84% to 100%). Children seem to be significantly more susceptible to ototoxicity.80 The hearing loss initially is worse at high frequencies, bilateral, and irreversible. It occasionally is accompanied by tinnitus or vertigo. The degree of hearing loss is dose-related, but there is considerable variability. Occasionally, severe hearing loss can occur after a single dose.81 If ultra-high-frequency hearing is tested, 100% of patients show a loss. Many factors influence this variability, including mode of drug administration, tumor site, age, renal function, diet, cranial irradiation, interaction with aminoglycosides and loop diuretics, preexisting hearing loss, cumulative dose, and total dose per treatment.79,82,83
Carboplatin is a cisplatin analogue with a similar spectrum of antineoplastic activity. Carboplatin is less nephrotoxic than cisplatin. Myelosuppression is the dose-limiting toxicity with carboplatin. It was initially thought that carboplatin was less ototoxic than cisplatin. More recent studies have shown higher rates of ototoxicity than initially appreciated, however. In a series of children, high-dose carboplatin therapy was associated with a very high rate of ototoxicity.84
Cisplatin and carboplatin ototoxicity seems to be due to the creation of reactive oxygen species within the inner ear that target the hair cells. Cisplatin results in greater outer than inner hair cell damage, whereas carboplatin seems to affect the inner hair cells preferentially.85
Nitrogen Mustards
Nitrogen mustards are antineoplastic agents that include mechlorethamine (Mustargen), chlorambucil (Leukeran), cyclophosphamide (Cytoxan), melphalan (Alkeran), and ifosfamide (Ifex). Of these drugs, only mechlorethamine has ototoxicity as a serious adverse effect, and it has limited usefulness today because of its severe toxic profile. Animal and human studies with mechlorethamine ototoxicity have revealed severe loss of outer hair cells.69 Other studies have shown shrinkage of the organ of Corti and loss of inner and outer hair cells.86
Vincristine and Vinblastine
The vinca alkaloids vincristine and, to a lesser extent, vinblastine are notable for their potent neurotoxicity. Peripheral neuropathy is common. Cranial neuropathies, ataxia, and hearing loss have been reported. Vincristine has been shown in animals to cause loss of hair cells and primary auditory neurons, whereas vinblastine has been shown to result in only hair cell loss.79
Eflornithine
Eflornithine (difluoromethylornithine) is a potent inhibitor of ornithine decarboxylase and is very effective in the treatment of trypanosomiasis. It also has proven useful in some patients with Pneumocystis carinii pneumonia, cryptosporidiosis, leishmaniasis, and malaria. It has shown potential as an antineoplastic agent. This drug has been reported to cause major and dose-related SNHL.87
Deferoxamine
Deferoxamine is an iron-chelating agent used in some patients with chronic iron overload or acute, severe iron intoxication. Auditory and visual neurotoxicity have been reported, particularly with larger doses in younger patients. SNHL is reversible in some patients when the dosage is reduced.88
Renal Disorders
Numerous genetic causes of SNHL are associated with renal abnormalities; Alport’s syndrome is the most well recognized.9 Acquired renal disorders have an unclear association with SNHL. Chronic renal failure, especially when managed with hemodialysis or renal transplantation, has been associated with progressive, fluctuating, or sudden SNHL. Oda and coworkers89 found that 15% of 290 hemodialysis and renal transplantation patients developed SNHL. The etiology of the SNHL is difficult to determine precisely and probably is multifactorial. In addition to the electrolyte and metabolic abnormalities caused by the renal failure and subsequent hemodialysis, these patients typically receive frequent doses of loop diuretics, aminoglycoside antibiotics, and vancomycin. Because of the altered pharmacodynamics of these drugs caused by renal failure, their ototoxic potential is increased.
Trauma
Noise-Induced Hearing Loss and Acoustic Trauma
The fact that excessive noise exposure could cause hearing loss was first recognized in the 18th century. In the early 20th century, NIHL was termed “boilermaker’s deafness.” Careful descriptions of the hearing loss sustained in industry would await development of the audiometer and were first published in the 1930s.90 NIHL is now recognized as one of the most common occupationally induced disabilities, and noise exposure is now regulated by the Occupational Health and Safety Administration.91
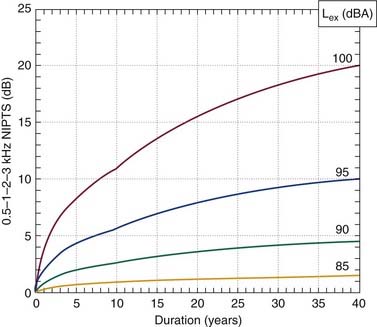
NIHL almost always results in a symmetric, bilateral hearing loss. It almost never results in a profound loss. Early in the course of NIHL, the loss usually is limited to 3 kHz, 4 kHz, and 6 kHz. The greatest loss usually occurs at 4 kHz. As the loss progresses, lower frequencies become involved, but the loss at 3 to 6 kHz is always far worse. The loss progresses most rapidly during the first 10 to 15 years of exposure and thereafter grows at a much-reduced rate. Figure 149-3 shows the 50% and 95% confidence limits for 20-year and 40-year exposures to 90 dBA (decibel, with “A”-weighting that fully weights 700-9000 Hz) sound. Figure 149-4 shows an example of the rate of growth of NIHL over time. The International Organization for Standardization has established standards for determining and quantifying occupational hearing loss and noise-induced hearing impairment.92
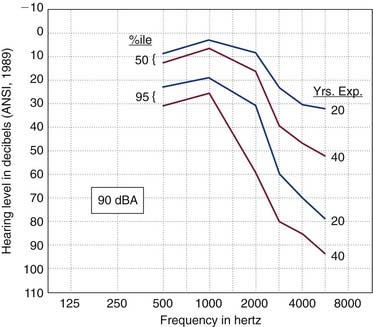
Figure 149-3. Predicted hearing thresholds (median and extreme values) after 20 years and 40 years of occupational noise exposure at 90 dBA.
(ISO-1999, from Dobie RA. Medical-Legal Evaluation of Hearing Loss. New York: Van Nostrand Reinhold; 1993.)
There are three exceptions. Conductive hearing losses are clearly protective for NIHL in the same way that earplugs or earmuffs would be. The lack of an acoustic reflex has been shown to predispose patients to NIHL (the protective effect of the acoustic reflex is primarily ≤2 kHz).19 Finally, patients with an unusually large PTS already should be considered to be more susceptible. TTS and PTS are commonly accompanied by tinnitus, and tinnitus after a noise exposure should be considered a warning sign.
Barotrauma and Perilymphatic Fistula
A perilymphatic fistula consists of a pathologic communication between the perilymphatic space of the inner ear and the middle ear. Perilymphatic fistulas can be congenital or acquired in origin. The fistulas can occur at either the round or the oval windows.93
Congenital defects can occur in the stapes footplate in patients with labyrinthine anomalies such as Mondini dysplasia.94,95 These fistulas can communicate with the subarachnoid space and result in cerebrospinal fluid leak and possible meningitis. Typically, these ears have a profound hearing loss. This phenomenon should be a consideration in the differential diagnosis of patients with recurrent meningitis.96–98
Acquired perilymphatic fistulas can result from barotrauma, direct or indirect trauma to the temporal bone, or a complication of stapedectomy surgery. A typical history attributed to perilymphatic fistula consists of the sudden development of SNHL and vertigo after a head injury, barotrauma, or heavy lifting or straining. The event is sometimes associated with an audible “pop.” Patients may have a positive Hennebert sign and positional nystagmus when the involved ear is placed in a dependent position.99 Some authors believe that perilymphatic fistulas develop spontaneously.100,101
Diagnosis is made by middle ear exploration. Visualization of fluid in the region of the oval or round windows is not definitive evidence of a fistula because serous fluid can ooze from the middle ear mucosa, or lidocaine from the local anesthetic can collect in the vicinity.102,103 Treatment consists of packing the area in question with tissue. Because of the lack of a definitive diagnostic test for the presence of a fistula, and because even surgical exploration does not reliably diagnose or exclude the possibility of a fistula, there is considerable controversy with regard to the management of this entity.93,100,104,105 In the 1980s and early 1990s, it was commonly believed that spontaneous perilymphatic fistulas were a common cause of otherwise unexplained hearing loss and vertigo. Many surgical fistula repairs were performed as a result of this belief. It has since become clear that spontaneous perilymphatic fistula is rare.106 No clear consensus exists regarding diagnosis or management.
Finally, labyrinthine fistula can result from erosion by cholesteatoma, or may develop spontaneously as in the superior semicircular canal dehiscence syndrome. The reader is referred to Chapters 140, 163, and 165 of this book for detailed descriptions of these entities.
Irradiation
The cochlea seems to be resistant to radiation injury at doses less than 45 Gy. At doses greater than 45 Gy, there is a clear, dose-dependent toxicity that manifests as hearing loss.107–109 Radiation also seems to cause a dose-dependent toxicity to the auditory nerve and brainstem.110 The latency period from radiation exposure to clinical hearing loss can be 12 months or more. One report notes that early-onset SNHL after radiation can recover to some degree.111 Fractionated irradiation has been used to a limited extent in the past to treat vestibular schwannoma. Whether it has had an effect on hearing in these patients is difficult to determine because of the limited data available.112 More recently, there has been much more extensive experience with stereotactic irradiation (radiosurgery) for vestibular schwannoma. This modality seems to be associated with substantial risk of SNHL, at least as high as with microsurgical removal.113
There is a relative paucity of published literature on the experimental effects of radiation on hearing. In a histologic study in chinchillas, sacrificed 2 years after fractionated irradiation of the cochlea, Bohne and coworkers114 showed a dose-dependent loss of inner and outer hair cells. Early studies of the effects of radiotherapy involving the cochlea on human hearing were flawed because lack of controls, inadequate follow-up, and the retrospective nature of the studies. Many well-designed studies now have shown that radiotherapy involving the cochlea causes SNHL in 50% of patients.107–109,115–117 The hearing loss occurs in a dose-dependent fashion, and seems to increase significantly at doses greater than 45 Gy. Advanced age, preexisting hearing loss, and adjuvant ototoxic chemotherapeutic agents are likely to amplify the effects of radiation. The hearing loss occurs with a latency of 0.5 to 2 years post-treatment and is probably progressive.111 This delayed onset, and the fact that many patients do not survive for sufficient follow-up, results in underestimation of the frequency and severity of this complication. Dose-dependent injury to the auditory nerve and brainstem also occurs; however, the frequency in the absence of other neurologic complications is difficult to determine.110
An understanding of the effects of radiation on hearing has become much more clinically relevant more recently because of the popularity of stereotactic radiotherapy for the treatment of vestibular schwannoma and other similar lesions. Although many reports suggest that the risk of hearing loss is minimal, these studies are limited by (1) their retrospective design, (2) lack of long-term follow-up, and (3) incomplete audiologic characterization. In addition, it is difficult to compare the studies that are available because of differences in tumor size, radiation dose, radiation field, technique of radiation administration, and follow-up. There is a wealth of literature on the treatment outcomes after stereotactic radiotherapy; however, almost all of them are limited by one of the above-mentioned shortcomings. More recent studies report hearing preservation rates (defined in different ways and with different follow-up periods) between 36% and 61%.118–121 The time course of hearing loss after stereotactic radiotherapy is poorly characterized; available data would suggest that the loss is progressive, and that early results may not predict later outcomes.
Neurologic Disorders
Multiple Sclerosis
SNHL develops in 4% to 10% of patients with multiple sclerosis.122,123 The hearing loss can be progressive or sudden in onset, and can be bilateral, unilateral, symmetric, or asymmetric.124–128 Frequently, the loss is sudden and unilateral, and can recover after days or weeks.125,129 Audiometrically, speech discrimination can be normal or reduced out of proportion to the increase of pure-tone thresholds. Abnormal patterns of acoustic reflexes can be seen in some patients.126,130,131 Abnormalities of the ABR frequently are seen and are a diagnostic criterion for multiple sclerosis. Patterns of abnormality vary and include latency prolongation of wave I or the later waves, absence or poor morphology of waveforms, and waveform abnormalities with increased stimulus presentation rate.124,128,130,132–134 MRI frequently is abnormal in multiple sclerosis and typically reveals periventricular white matter plaques on T2-weighted images. Plaques can be seen in the cochlear nucleus or inferior colliculus in patients with SNHL.135–138