CHAPTER 201 Salivary Gland Disease in Children
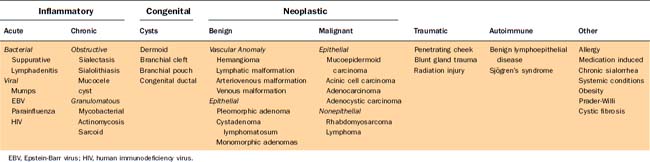
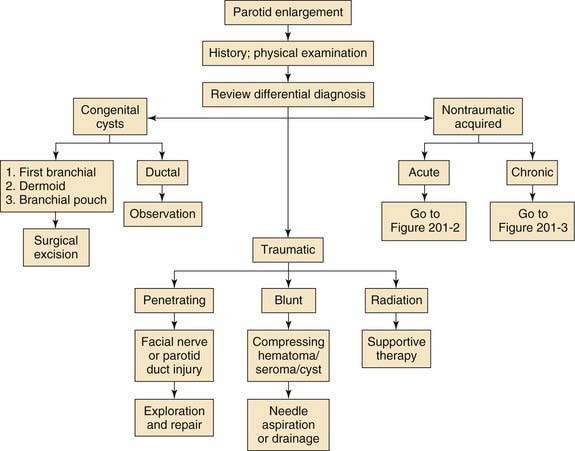
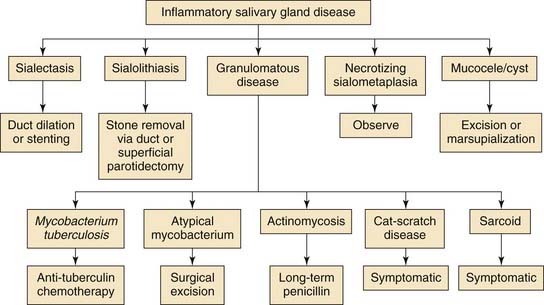
Salivary gland disease is an uncommon but manageable condition in the pediatric population. Inflammatory conditions of the salivary glands are more frequently encountered because salivary tumors account for only 1% of all childhood neoplasms. Infections can be either acute (bacterial and viral) or chronic (ductal obstruction, mycobacterial, and granulomatous disease) with the parotid being the most frequently affected gland. When a persistent salivary gland mass is present, possible neoplastic and congenital processes should be explored with an understanding that the risk of malignancy for salivary tumors is greater in young children. A complete differential diagnosis includes inflammatory, neoplastic, vascular, congenital, traumatic, and a myriad of less common diseases (Table 201-1). Clues gleaned from a thorough history and physical examination can help identify the disease in each pediatric patient. Ultrasound, computed tomography, magnetic resonance imaging, and fine-needle aspiration (FNA) have proven to be safe and valuable diagnostic tools for delineating inflammatory from benign and malignant salivary gland conditions (Figs. 201-1 to 201-3). Chronic sialorrhea (drooling) is a frequent and chronic problem, especially in neurologically impaired children, that can be addressed with both medical and surgical therapy.
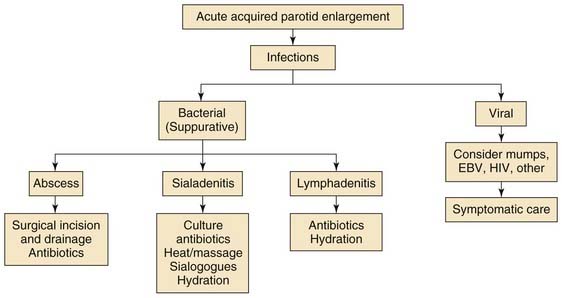
Figure 201-2. Acute acquired parotid enlargement. EBV, Epstein-Barr virus; HIV, human immunodeficiency virus.
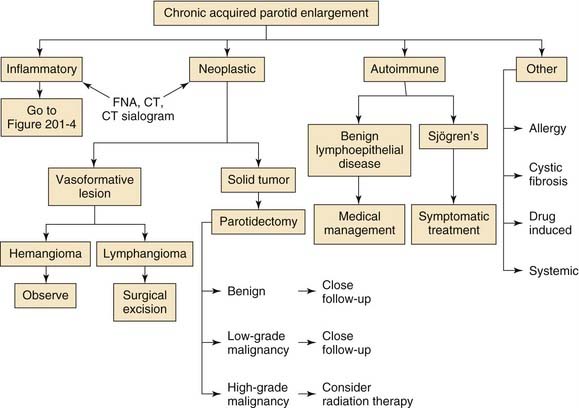
Figure 201-3. Chronic acquired parotid enlargement. CT, computed tomography; FNA, fine-needle aspiration.
Anatomy
Physiology of the salivary glands is thoroughly addressed in Chapter 84. Briefly, salivary flow is controlled by the superior salivatory nuclei (submandibular and sublingual glands) and inferior salivatory nuclei (parotid gland) via the autonomic nervous system. Only a small amount of saliva is secreted during the night because resting salivary flow increases during the day and peaks in the late afternoon. Resting salivary secretions are produced predominantly by the submandibular (60%) and sublingual (10%) glands with the submandibular glands creating the bulk of the fluid. The production of parotid secretions, which are serous in nature, is increased during eating. Saliva produced from all glands is critical for deglutition and acts as an infectious barrier for the mucosa and dentition by creating a surface coat of mucoid secretions, enzymes (e.g.. lysosyme), and immunoglobulins (IgA). In select children, excessive saliva and drooling (sialorrhea) can lead to aspiration, pulmonary compromise, mouth and skin maceration, and social isolation.1 These patients often require medical or surgical therapy, or both, to reduce salivary flow from the major glands.
History and Physical Examination
A thorough history is the most important tool in diagnosing salivary gland problems in children. The onset, duration, and severity of symptoms provide valuable clues to the pathologic nature of the problem and allow the practitioner to quickly delineate among neoplastic, congenital, and inflammatory processes. Congenital disorders such as hemangiomas and cysts will often become evident shortly after birth. Neoplastic processes are more frequently seen in children older than 10 years.2–4 In contrast, an inflammatory condition is the most common salivary pathology in toddlers and is marked by acute onset, brief duration, and painful swelling. Chronic conditions may be more difficult to differentiate by history alone, but gradual and painless growth at or near salivary tissue is suspicious for neoplasm until proven otherwise. Intermittent swelling associated with eating will signify salivary duct obstruction by stones or strictures, whereas recurrent and diffuse bilateral cheek swelling may indicate recurrent parotitis or chronic sialectasis.
The location and number of glands involved should be elicited in the history. Diffuse edema of the involved gland suggests inflammatory disease while focal and palpable lesions are the hallmark of neoplasm (Fig. 201-4). Bilateral involvement suggests systemic conditions. The character, quality, and quantity of the salivary secretions are also important. Serous secretions suggest a viral etiology, whereas malodorous purulent material indicates bacterial disease. The presence of fever, chills, or a recent upper respiratory infection should also be ascertained. An immunization history for mumps vaccine should be obtained. Questioning should elicit any history of trauma including injury from blunt or sharp objects, animal scratches, or recent dental procedures. The presence of congenital anomalies or cysts should be identified. Awareness of any underlying systemic diseases, which may include immune deficiency, allergy, or autoimmune disease, can be helpful.
Laboratory Studies
FNA is an easy and relatively safe technique for the diagnosis of palpable or deep-lying salivary masses. This can often be employed as an adjunct to diagnostic ultrasonography during the evaluation of pediatric salivary masses. With a well-trained cytopathologist, FNA can be an effective tool in diagnosing difficult lesions. In adults, up to 93% of benign and malignant lesions of the salivary glands can be differentiated.5 Unfortunately, FNA is not commonly employed in the pediatric population because sedation or general anesthesia is often required. Moreover, pediatric pathologists are not universally versed in identification of salivary tumors due to the rarity of neoplasms in this population. Nonetheless, the unique architecture of pleomorphic adenoma and Warthin’s tumor often makes it easy to diagnose them by FNA. Cytologic evaluation of sialadenitis shows polymorphonuclear leukocytes in a background of necrotic cellular debris. Cultures may also be obtained from aspirated contents at the time of FNA. Cystic lesions and poorly differentiated carcinomas are more difficult to assess by FNA and should undergo excisional biopsy for definitive answers.
In general, unusual and longstanding salivary masses should undergo FNA in tolerant pediatric patients before a surgical plan is developed. Surgical resection of discovered malignancies by FNA can be designed more appropriately. Misdiagnoses and nondiagnostic results are rare in FNA of pediatric head and neck masses. Regardless, most fine-needle aspirates lead to the diagnosis of a chronic inflammatory condition and can offset the use of open procedures in pediatric patients.6,7
Radiology
Radiographic imaging has played an increasing role in the diagnosis and perioperative decision making for salivary gland lesions. Improved radiographic instruments have allowed practitioners to delineate inflammatory and benign disease from malignant processes. Commonly employed techniques include plain film radiography, ultrasonography, sialography, radionucleotide scanning, computed tomography (CT), and magnetic resonance imaging (MRI). New MR technologies such as dynamic contrast-enhanced MRI, diffusion-weighted MRI, and MR spectrscopy have proven helpful in better differentiating benign from malignant salivary gland tumors.8
Sialography involves the injection of contrast medium into the salivary duct during radiographic imaging in order to outline the salivary ductal system. Similar to plain film radiographs, this technique is gradually being replaced by CT sialography, MRI, and salivary duct endoscopy.9,10 CT sialography can be valuable for detecting sialolithiasis, ductal dilatation, and strictures of the major salivary glands and has been used to evaluate obstructive disease, inflammatory lesions, penetrating trauma, and mass lesions. A filling defect and dilation of the main duct or intraglandular ducts will be seen when calculi are present. Chronic sialadenitis may show sialectasis or ductal ectasia. Salivary fistula or sialoceles can be picked up using this technique following trauma to a salivary gland.11 Regardless, in the face of acute sialadenitis, sialography should not be performed because this can exacerbate disease and injure the involved gland.
For complete evaluation of the nature and extent of salivary gland masses, cross-sectional imaging with CT or MRI is necessary. Contrast-enhanced CT is best fitted for evaluating inflammatory conditions such as abscesses, obstructing sialoadenitis, cystic disease, sialolithiasis, and acute inflammation. Imaging should be performed both before and after administration of intravenous contrast. CT is an excellent tool to study both intrinsic and extrinsic masses of the parotid gland, and the relationship of the mass to critical anatomic structures such as the facial nerve are well illustrated with coronal and sagittal reconstructions.8 The main benefits of CT scans in children are its speed and quietness relative to the slow, claustrophobic, and loud nature of MRI machines. Bony architecture is better defined by CT and can provide information on the integrity of adjacent bone in malignant processes.
However, in patients with suspected neoplastic disease, MRI provides the best information on the exact location and degree of disease penetration.11 In malignant processes, tumor infiltration of adjacent structures, soft tissue destruction, perineural spread, and meningeal penetration is best detected by MRI. This provides more information for operative planning. Standard axial T1- and T2-weighted MRI illustrate tumor margins and growth patterns. Coronal images, fat suppression, and gadolinium enhancement help identify perineural spread. Moreover, newer MR techniques have the ability to delineate benign from malignant processes with reported sensitivities and specificities above 90% using dynamic contrast or diffusion weighted methodology.8 The drawback of MRI is that it can be difficult for children to tolerate and thereby be subject to the risks of sedation or general anesthesia required to perform the procedure.
Sialendoscopy
Sialendoscopy is a novel technique that has gained acceptance as a tool for diagnosing and treating a variety of salivary gland lesions in adults.12 Since its development in the early 1990s, improvements in instruments and technique have allowed its application in the pediatric population. Semirigid sialendoscopes with external diameters between 1.1 mm and 1.6 mm are now available and can accommodate the pediatric salivary duct orifices. The endoscope includes two channels, one for irrigation and the other for instrumentation. Following routine dilation of the salivary duct, sialendoscopy is performed with continuous irrigation to maintain patency during exploration. This tool is best designed, and has proven applicable, to diagnose and treat obstructive pathology of pediatric unilateral salivary diseases. Diagnosis of salivary duct stenosis can be achieved with this technique, while wire baskets, lithotripsy, and YAG lasers can be simultaneously deployed to address small obstructing sialoliths.13
Inflammatory Diseases of the Salivary Glands
Inflammatory conditions of the salivary glands are rare in children. Acute processes occur in the majority of cases that involve either viral or bacterial disease. The peak incidence is during puberty with most cases occurring between 12 and 18 years of age (>40%).14 Submandibular disease is most commonly associated with proximal sialolithiasis and should be investigated appropriately. Granulomatous disease is a cause for a significant bulk of cases (primarily in toddlers) due to inflamed periglandular lymph nodes. Similar to adults, immunocompromised children are particularly prone to salivary gland infections and ductal stasis and obstruction and should be monitored closely. The most common clinical manifestation of salivary gland involvement is xerostomia and bilateral painless enlargement of the parotid glands.15 Autoimmune disorders in children are rare but often manifest as inflammation of the major salivary glands.
Viral
Once the most common cause of viral sialadenitis, the mumps virus has virtually been eliminated as a cause of parotitis in the United States since the institution of the mumps vaccine in 1982. However, a few cases emerge annually in local and migrating patients who are without prior vaccination. The reader is directed to review the current guidelines and contraindications for mumps vaccination, which is beyond the scope of this chapter. However, other viruses can mimic mumps sialadenitis and should be included in the differential of vaccinated children with diffuse bilateral salivary gland disease. Beyond mumps, the most common viral sources of childhood sialadenitis are Epstein-Barr virus (EBV, incidence = 14%), parainfluenza viruses (4%, types 1, 2 and 3), adenovirus, (3%), HHV-6, human immunodeficiency virus, coxsackievirus, and influenza.16 Most cases of viral sialadenitis are self-limited with conservative management of warm compresses, sialogogues, analgesics, anti-inflammatories, and hydration as the mainstay of therapy. The disease course of a few viral processes deserves special mention.
Epstein-Barr Virus
EBV is the cause of heterophil-positive infectious mononucleosis and has been implicated as a major cause of acute inflammation of the salivary glands in pubescent children.16 Disease symptoms range from an asymptomatic infection to fulminant lymphoproliferative disease. EBV is emitted in the saliva and has been colloquially termed the “kissing” disease due to its oral transmission. In fact, 10% to 20% of healthy seropositive persons can shed EBV into their oropharynx.
The long incubation period of EBV ranges from 30 to 50 days. A triad of fever, sore throat, and posterior cervical adenopathy occurs in more than 80% of patients. Lethargy can persist for months in infected patients. The cervical adenopathy can involve the periparotid or perifacial (submandibular) lymph nodes with subsequent involvement of adjacent glands. Again, there is no specific therapy for EBV infection, but improvements in serologic detection have helped practitioners identify disease early and accurately. Children often need weeks of isolation and rest before returning to normal daily activity. Corticosteroids can be used for life-threatening complications but are unlikely necessary in isolated salivary gland cases. Recently, nearly 50% of salivary gland tumors and oral malignancies of nonodontogenic origin have been associated with EBV. Thus in children with rare oropharyngeal or salivary malignancies, isolation of EBV genome by polymerase chain reaction may help families come to terms with the source of such rare tumors.17
Human Immunodeficiency Virus
Salivary gland disease is a common manifestation of HIV in infected children. More than 50% of cases of pediatric HIV present with head and neck masses with the majority of patients having major salivary gland enlargement.18 The enlargement can be lymphoproliferative, cystic, or both, with the parotid glands being the most frequently affected salivary entity. Parotid swelling of various degrees has been found in more than 10% of HIV-positive children.19 The submandibular and sublingual glands can be involved as well. The incidence of ranula has been shown to be increased in HIV pediatric patients.20 The high rate of salivary gland involvement is probably related to the presence of HIV within the saliva. Quantitative changes to the saliva can contribute to glandular hypertrophy, reduced salivary flow, stasis of secretions, and xerostomia.21 Slow continuous enlargement is the hallmark disease in pediatric AIDS patients. The pathophysiology remains elusive. However, in children, bilateral lymphoepithelial cystic enlargement or intraglandular lymph node enlargement of the parotid glands is common. Cystic disease can often be seen in combination with lymphocytic interstitial pneumonitis.22 Latent manifestation of EBV and cytomegalovirus in these patients may also contribute to salivary gland pathology.
Bacterial
Primary bacterial salivary gland infections are rare in the pediatric population. Dental infections, periglandular lymphadenitis, and floor of mouth and submandibular triangle processes need to be considered in the differential of acute bacterial sialadenitis because these may be the source of inflamed salivary tissues. When suppurative sialadenitis does present, it most commonly occurs acutely and within the parotid gland. The normally serous saliva of these glands contains limited bacteriostatic activity (lysozymes) compared with the mucinous secretions of the submandibular and sublingual glands and likely contributes to the predominance of bacterial disease occurring in the parotid. Submandibular gland infections are associated with proximal sialolithiasis more than 40% of the time.14 This is rare for the parotid gland.23 The primary cause of submandibular infections appears to be salivary stasis of dependent and viscous mucous secretions of the submandibular duct. Predisposing factors include congential Wharton’s duct stricture, ductal trauma, dehydration, autoimmune disorders, and congenital sialectasis. Similarly, recurrent sialadenitis has been associated with traumatic injury to the ductal orifices by repetitive biting of the buccal mucosa, dental appliances, and tooth eruption.24 Retrograde oral bacteria transmission via the salivary ducts is thought be a culprit in disease.
Signs and symptoms of acute bacterial sialadenitis include rapid onset of focal pain, edema, and erythema of the soft tissues overlying the affected gland. Disease is predominantly unilateral. Spiking fevers are often present with symptoms worsening during deglutition. Purulent discharge found draining from the involved gland’s duct is pathognomonic for disease. Most children remain nontoxic during acute episodes but, as the acute phase passes, residual induration and enlargement of the gland may persist for several months. Facial nerve paresis is extremely rare. The most common pathogens associated with bacterial sialadenitis are coagulase-positive Staphylococcus aureus, Streptococcus viridans, and Haemophilus influenzae. Less frequently identified bacteria include Streptococcus pneumoniae, Escherichia coli, Bacteroides melaninogenicus, and Streptococcus micros (Table 201-2).25,26 Cultures of aspirated material or swabs of the purulent ductal secretions will help guide antibiotic coverage.
Table 201-2 Acute Suppurative Sialadenitis
| Most common | Staphylococcus aureus (coagulase-positive) |
| Streptococcus viridans | |
| Haemophilus influenzae | |
| Peptostreptococcus spp. | |
| Less common | Streptococcus pneumoniae |
| Escherichia coli | |
| Bacteroides melaninogenicus |
Recurrent Sialadenitis
Repeated infections of the salivary glands have been shown to cause ductal metaplasia, parenchymal restructuring, and an increase in mucin production and quantity of viscous secretions that can lead to recurrent salivary stasis. Patients with recurrent disease present with low-grade edema and tenderness of the gland with slightly purulent secretions. Unfortunately, once the glandular architecture is altered, opportunistic oral flora are more likely to cause recurrent disease. These patients are frequently chronically infected with various organisms, and immunologic dysfunction such as IgA deficiency appears to play a role in its development.27,28
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree