Chapter 43 Retinopathy of prematurity
Retinopathy of prematurity (ROP) was first reported by Terry.1 This condition, then known as retrolental fibroplasia, is a disorder of the immature retinal vasculature. It was extremely rare before the 1940s.2 The retinopathy developed after a normal fundus examination at birth.3 Clinical and experimental evidence supported a toxic effect of oxygen on the immature retinal vasculature4,5–7 which led to the restriction of oxygen use in preterm neonates.8 This resulted in a dramatic fall in the incidence of ROP, but even though oxygen plays a central role, many other factors contribute to the pathogenesis,8–11 which has been comprehensively reviewed.9,11–16
Retinal vascular development
Retinal vasculature develops to meet retinal metabolic demand except at the fovea which has a different vascular pattern.17 Early in development the retina receives all its nutrients from the choroid which is vascularized from about 6 weeks’ gestational age;18 vascularization of the retina begins at 14–15 weeks of gestation. Vascular endothelial cells, microglia, pericytes, and astrocytes migrate centrifugally from the optic disk, proliferate, and become aligned into vascular cords that develop lumina and differentiate into capillaries. The capillaries remodel and form a mature retinal vascular network with capillary-free areas.19 The retinal tissue responds to excessive or insufficient oxygen by trimming or inducing growth in microvasculature to match the retina’s metabolic requirements.20
Oxygen-dependent vascular endothelial growth factor (VEGF) plays a part in all stages of vascular development; other factors are also involved.10,16,21–24 One is oxygen-independent insulin-like growth factor (IGF-1) that controls VEGF activation of the Akt endothelial cell survival pathway; low levels result in reduced survival and growth of vascular endothelial cells.21–23
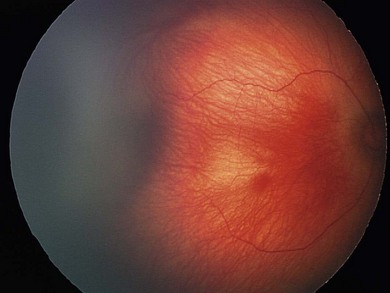
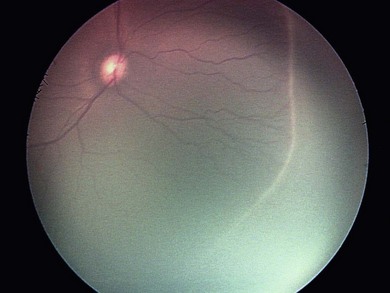
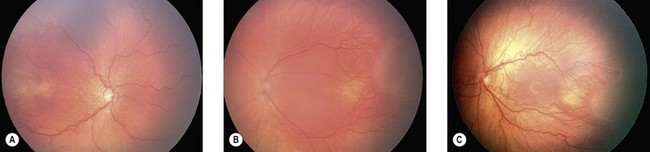
The nasal retina is vascularized by 32 weeks gestational age, the temporal retina just after term.25 The retinal vessels in the preterm infant are slender, relatively straight, and taper as they terminate toward the gray, avascular periphery (Fig. 43.1).The foveal region is differentiated ophthalmoscopically at around 40 weeks.26
Pathogenesis
Oxygen restriction does not abolish ROP; believing it would “seems naive in light of our current understanding.”24,27–29 At present, ROP is treatable, but not preventable.
According to the first – “classic” – theory,7,8 ROP consists of two phases of equal importance:
1. A hyperoxic phase leading to retinal arteriolar constriction.
2. Vaso-obliteration with death of retinal capillary endothelial cells on removal from a hyperoxic environment.
The second – “gap junction” – theory is based on the activity of mesenchymal spindle cell retinal capillaries precursors.30a Mesenchymal spindle cells migrate centrifugally from the optic disk and form capillaries just behind the advancing vanguard. In the relative hyperoxic extrauterine condition, tight bonds or gap junctions appear between adjacent spindle cells, interfering with normal cell migration and vessel formation. The angiogenic factors secreted by gap-junctioned spindle cells trigger the neovascular response.
Current concepts of ROP pathogenesis encompass both classic and gap junction theories.10,31b VEGF is secreted in response to physiologic hypoxia of the maturing avascular retina. Hyperoxia causes cessation of vessel growth and apoptosis of parts of the retinal vasculature and capillary regression with consequent retinal ischemia stimulating VEGF. This results in neovascularization known as ROP.10,31b–33
1. A vessel sustaining role, reduced by hyperoxia, leading to downregulation of VEGF-A with cessation of vessel growth and capillary regression.
2. Subsequent upregulation of VEGF-A induced by hypoxia, with vasoproliferation known as ROP.
There are two VEGF-A receptors in the mouse retina: VEGFR-1 supports retinal vessel survival (phase 1), and VEGFR-2 supports endothelial permeability and vasoproliferation.34
IGF-1, a somatic growth factor, controls VEGF activation: when IGF-1 is low, vessels do not grow.35 The levels of placenta-derived IGF-1 rise in the second and third trimesters and fall after preterm birth. Oxygen-independent IGF-1 and oxygen-dependent VEGF are synergistic, and IGF-1 permits VEGF to function maximally at low levels. A slow recovery to “normal” levels of serum IGF-1 may predict phase 2 proliferative ROP;10,15,21,23 causality has not been established35 but evolves36 with a number of exciting therapeutic options.37
Risk or associated factors
The major ROP risk factor is the degree of immaturity as measured by either birth weight or gestational age,38,39 the first being the more powerful predictor.27,30b,40,41
Clinical studies confirmed the relationship between oxygen and ROP,42–44 and the duration of transcutaneous PO2 over 80 mmHg and its incidence and severity.45–47 Oxygen levels are particularly critical for ROP development, within the first few weeks after birth.44,48 Safe levels of oxygen usage for clinical practice have yet to be detailed.48,49
ROP may develop in preterm infants who never received oxygen and in premature those with cyanotic heart disease.9 A relationship between neonatal hypoxia and ROP is suggested.9,49,50,51 That hyperoxia and hypoxia may be associated with ROP is not entirely contradictory.52a Relative hyperoxia may, via VEGF, lead to initial retinal capillary damage; subsequent ischemia stimulates VEGF overproduction and vasoproliferation explaining the association of recurrent apnea and cerebral ischemic events with ROP.53
The STOP-ROP trial tested the efficacy and risks of supplemental oxygen therapy for prethreshold disease but failed to show a beneficial effect.54 A meta-analysis noted that “late” (≥ 32 weeks PMA) high oxygen was associated with reduced incidence of severe ROP.44 Oxygen still plays a central role in ROP.55 Babies with target saturation levels of 94–98% (but not measured) had a much higher incidence of ROP requiring treatment compared to those with target oxygen levels of 70–90%, with no increase in neurologic morbidity in the latter.49,56 Looking at higher levels, a randomized controlled trial, comparing oxygen saturation ranges of 91–94% against 95–98%, reported no difference in infant growth, neurodevelopment, or rates of ROP.52b The risk of ROP should not be considered in isolation, but in the context of the wellbeing of the baby. Lower oxygen levels reduced the incidence of ROP blindness but at the expense of reduced survival, and higher morbidity.8,50
Acidosis is a risk factor for ROP,53 whereas hypercarbia is not.57,58 High PCO2, PaO2, and low pH in the first 3 days after birth were associated with severe ROP48 and hyperglycemia is a risk factor,59,60 perhaps related to insulin resistence. Antenatal steroids may be protective for ROP,61,62 but not when given after birth.63,64
There is no difference in ROP incidence in Hispanic compared to White non-Hispanic infants,65 while Indo-Pakistani infants are more likely to develop severe ROP than Caucasians.66,67 Afro-Caribbean infants may be less,40,68 or more,67 likely to develop ROP.
There is high concordance for ROP in monozygotic twins.69 ROP-associated mutations are all in the Wnt signaling pathway,70 which also encompasses the Norrie’s gene71–73 and familial exudative vitreoretinopathy; the importance of this is not yet understood.74
Multiple birth, per se, does not increase the risk but concordant twins behave similarly.75 In discordant twins, the smaller baby has the greater risk.76,77 Male gender is a risk factor78 as is assistive reproductive technology, the latter probably independent of multiple births.79
The antioxidant vitamin E31a is found at low levels in preterm neonates;81 initially, supplementation did not appear to reduce the frequency of ROP but reduced its severity.82,83 Due to side-effects of vitamin E its use prophylactically is not now recommended,80,84 although Raju et al.,85 in a meta-analysis of vitamin E prophylaxis, reported a reduction in the incidence of stage 3+ ROP, and suggested re-evaluation. Anemia is frequent in preterm infants; they may be transfused adult hemaglobin which binds oxygen less avidly than fetal hemoglobin so that more oxygen is delivered to the tissues giving hyperoxia, theoretically increasing the risk of ROP. There is a suggested association between ROP and blood transfusion,86–89 but it has not been confirmed.90,91 Recombinant erythropoietin, a cytokine which regulates fetal erythropoiesis and is important in angiogenesis, may now be used to treat neonatal anemia. Its role in ROP has yet to be clarified,92,93 including its timing.94,95
Surfactant has reduced mortality, the severity of respiratory distress syndrome, and chronic lung disease in very immature neonates96 but its effect is less clear with regard to ROP.97–99
Early exposure to light was suggested to be a risk factor.1,100,101 Studies by Hepner et al.10 and Locke and Reese102 did not provide supportive evidence; later studies did not support this10,102 at a time when supplemental oxygen could have swamped any effect of light. Light could, by damaging retinal tissues, generate free radicals and cause ROP. A reduction in neonatal unit illumination reduced the incidence and severity of ROP103 but this was not confirmed by the LIGHT-ROP study104 and another study.105
Antenatal exposure to infection and inflammation individually are not associated with ROP but, together, they carry an increased risk for severe ROP;106 postnatal systemic fungal infection is a risk factor for both ROP development and severity.107
Many factors are active simultaneously and differently according to the standard of neonatal care. Thus, the characteristics of babies developing ROP in some countries are different from those in others: larger and more mature infants are at risk in less developed countries,108 where the unrestricted use of oxygen and sepsis also need consideration.109
Classification
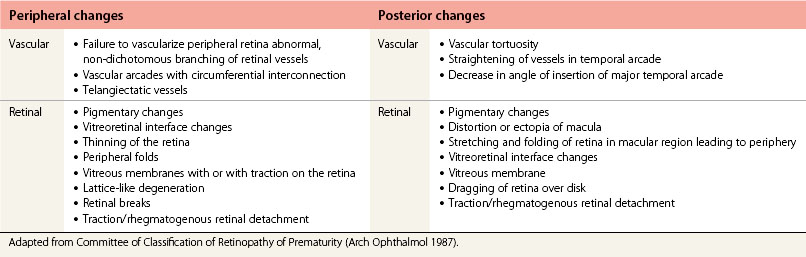
The international classification of acute ROP110 was expanded to include retinal detachment and ROP sequelae111 replacing the Reese et al. classification.112 All stages of ROP are now covered by a single classification (Table 43.1) allowing direct comparison between centers and countries. A revision was published,113 expanding two concepts, pre-plus disease and aggressive posterior ROP (AP-ROP).
Table 43.1 International classification of retinopathy of prematurity – revisited (2005)
| Severity | Stage of ROP | |
| Aggressive posterior ROP (ROP) | ||
| Anterior-posterior location | • Zone I: retina within a circle centered on the disk with a radius of twice the disk–foveal distance • Zone II: a doughnut-shaped area that extends from the edge of zone I to a circle which has a radius of the distance from the disk to the nasal ora serrata • Zone III: crescent-shaped retinal area peripheral to zone II | |
| Extent of disease | ||
| Posterior pole vessel abnormality | Pre-plus disease | |
| Plus disease |
The classification involves describing ROP by four parameters:
1. Severity by stage describing the retinopathy at the junction between the vascularized and avascular retina.
2. Location by zone describing the anterior–posterior position of the retinopathy.
3. Extent by clock hour of circumferential retinal involvement at the junction between the vascularized and avascular retina.
4. Plus disease, describing abnormally dilated or tortuous vessels of the posterior pole.
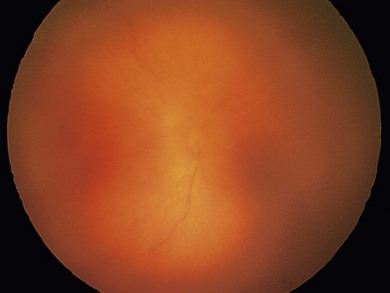
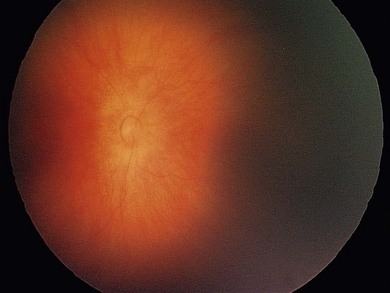
The normal fundus of the extremely premature baby can be difficult to visualize in detail (Figs 43.1 to 43.3). The retinal blood vessels are thin and straight. Later, they increase in caliber and tortuosity, but abnormally so with active ROP.
Severity of disease
Stage 1
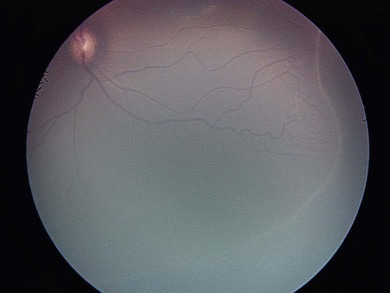
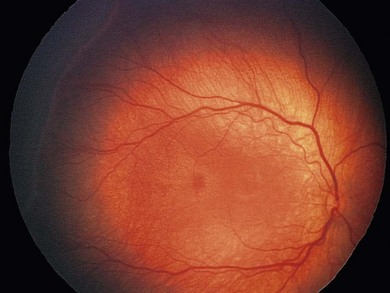
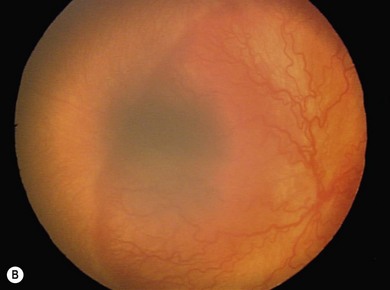
A flat gray-white demarcation line separates the vascularized from non-vascularized retina. Often faint, it can be difficult to identify. Retinal vessels run up to the line but do not cross it (Figs 43.4 and 43.5).
Stage 2
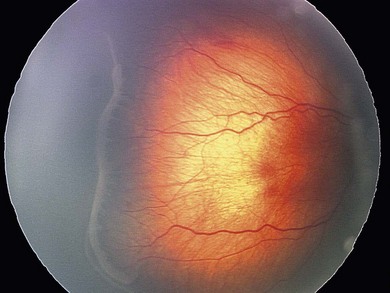
The demarcation line has increased in volume and extends out of the plane of the retina (Figs 43.5–43.7). The color of the ridge may be white or pink and small neovascular tufts may be seen posterior to the ridge. Differentiating stage 1 and early stage 2 is difficult but usually not prognostically important.
Stage 3
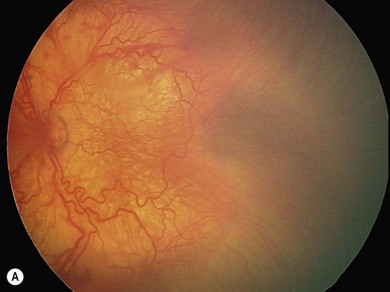
Stage 3 has the features of stage 2, but is characterized by extraretinal neovascularization (Figs 43.7 and 43.8). The new vessels may be continuous with, or disconnected from, the posterior border of the ridge, or extend into the vitreous. The extraretinal neovascularization may extend from the region of the ridge into the vitreous or, typically in more posterior disease, lies back across the surface of the vascularized retina.
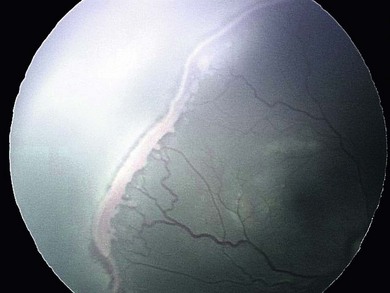
Fig. 43.8 Stage 3 ROP that has developed in the eye with stage 2 disease shown in Figure 43.6. Note the peripheral tortuosity and dilation as the vessels become close to the ROP lesion (compare with Figure 43.6). The shadow, just behind the lesion, indicates that the lesion is off the retinal surface.
Stage 5
There is a funnel-shaped total retinal detachment (Fig. 43.9). This stage is further defined according to the anterior and posterior characteristics of the funnel.
Aggressive posterior ROP (AP-ROP)
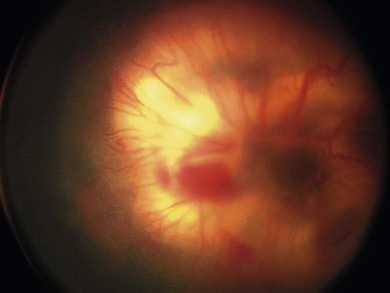
This severe form of ROP113 is characterized by a posterior location (zone I or posterior zone II) and a prominence of plus disease out of proportion with the rather innocuous appearance of the vascular abnormality at the vascular/avascular junction. The severity often makes it difficult to distinguish arterioles and venules (Fig. 43.10). Over time, the progression from stage 1 to stage 2 and stage 3 is not apparent; the flat network of new vessels can easily be missed.
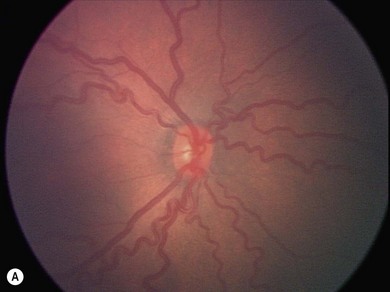
Plus disease
The first signs of plus disease (Figs. 43.11) are tortuosity of the retinal arterioles and congestion of the retinal veins close to the optic disk. Later, the vessels of the iris become engorged and the pupil fails to dilate to mydriatics. The vitreous becomes hazy. Signs of plus disease may appear at any ROP stage and occasionally as a precursor. The diagnosis is clinical comparing with a reference photograph; at least two quadrants must be involved and the peripheral changes of ROP must be present. Plus disease does not refer to vascular changes in the retinal periphery. The diagnosis of plus disease is critical; it is an indicator of severe ROP114 that may require treatment,115 and increased likelihood of an unfavorable outcome.66
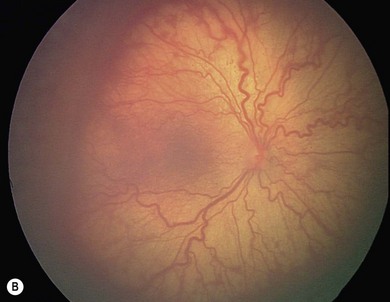
Pre-plus disease
The posterior vascular abnormalities that occur in ROP with plus disease are the most severe. The term “pre-plus”113 indicates posterior vascular abnormalities with insufficient arteriolar tortuosity and venular dilation to be termed plus disease although still abnormal (Fig. 43.12). It alerts to the possibility of peripheral retinopathy.
Location
Zone I is a circle that has a radius twice the disk–foveal distance – about 30°.
Zone II extends from the edge of zone I to the ora serrata on the nasal side and encircles the anatomic equator.
Zone III includes all retina temporally, superiorly, and inferiorly anterior to zone II. With no anatomic landmarks identifying zone III, only when the directly nasal retina is fully vascularized can it be stated that zone III has been entered.
Regression and resolution
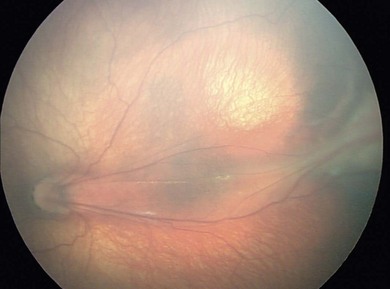
Resolution of acute retinopathy has been less well studied, because, once the need for surgical intervention has passed, there is less need for surveillance and recording.111 The rate of regression reflects PMA;116 the first signs are failure to progress and the lessening of any signs of plus disease. The ridge thins and breaks up. Later, vessels grow through the ridge into the peripheral avascular retina.117 Involution is characterized by vascular remodeling with gradual movement of vessels toward the periphery: in some eyes, vascularization to the ora serrata does not occur. The acute ROP lesion becomes white and vessels may be seen extending beyond the lesion into the periphery. Vasoproliferative lesions become fibrotic.109,111 All exclusively stage 1 and 2 ROP undergo complete resolution. Stage 3 may or may not resolve, depending on severity. A sign of incomplete resolution is retinal dragging (Figs 43.13 and 43.14). The mean time of onset of signs of involution in children with birth weights of less than 1251 g was 38.6 weeks; 74% of all ROP had begun to involute by 40 weeks of PMA 90% by 44 weeks.116
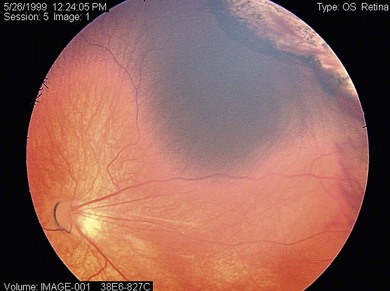
Fig. 43.13 Mild dragging of the retinal vessels. Note response to cryotherapy on upper border of picture.
Advanced cicatricial changes involving the anterior vitreous and retina (Table 43.2) may push the lens/iris diaphragm forward and cause shallowing of the anterior chamber, glaucoma, and corneal decompensation. In such cases, lensectomy is indicated to relieve narrow angle glaucoma.
Incidence and prevalence
Acute phase ROP
Early publications15,66,118,119,120 were mostly from single centers, but prospective geographic studies40,121 were undertaken, one of which was carried out after treatment was introduced.122
The multicenter study of cryotherapy for retinopathy of prematurity (CRYO-ROP study) was conducted in 23 nurseries in the USA.70 Over 4000 children with birth weights of less than 1251 g had serial eye examinations. ROP was observed in one or both eyes of 65.8% of these infants, with ROP observed in 90% of children with birth weights of less than 750 g, in 78% of those with birth weights of 751–1000 g, and in 47% of those with birth weights of 1001–1250 g. The CRYO-ROP study reported on the residua of ROP (the cicatricial phase in the eyes of 2759 children at age 1 year). None of these eyes had undergone cryotherapy. There was advanced scarring involving the posterior pole in approximately 4%, with 2% likely to have severe visual loss due to posterior pole scarring or retinal detachment.123
ROP may have declined in incidence,49,123–125 but not universally.126,127 The overall incidence of ROP in an intensive care nursery128 in infants with birth weights of less than 1251 g was 34% compared to 65.8% for CRYO-ROP study patients. For infants with birth weights of less than 1000 g, there was an incidence rate for ROP of 46%, compared to 81.6% for CRYO-ROP patients in the same birth weight group. However, there are several biases in single-center studies,129 including survival rates,130 ethnicity, and standard of care.
The ETROP study enrolled babies with birth weights of less than 1251 g in 26 neonatal intensive care unit (NICU) systems in the USA and imposed a rigorously controlled examination technique and frequency. The incidence of ROP in the ETROP study was 68% among infants of 1251 g birth weight, which is a very similar incidence to that for the CRYO-ROP study (65.8% among infants of similar birth weights). However, in the ETROP study, the incidence of more severe prethreshold ROP was 36.9% among infants with ROP while the incidence was 27.1% for patients in the CRYO-ROP study. The mean birth weight and gestational age for babies developing prethreshold ROP in the ETROP study were less than those in the CRYO-ROP study (740 vs. 831 g and 25.6 vs. 26.5 weeks),14 indicating that prethreshold ROP occurred among smaller and younger infants in the ETROP study cohort than in the CRYO-ROP study cohort.
Despite these caveats, in countries with well developed NICU systems, both the incidence and severity of ROP rise with the degree of prematurity, and more than 50% of babies under 1000 g at birth develop some stage of ROP; babies under 1251 g birth weight have an incidence of stage 3 ROP of around 18%. Approximately 6–8% of babies under 1251 g birth weight will develop ROP sufficiently severe to require treatment with peripheral retinal ablation.131
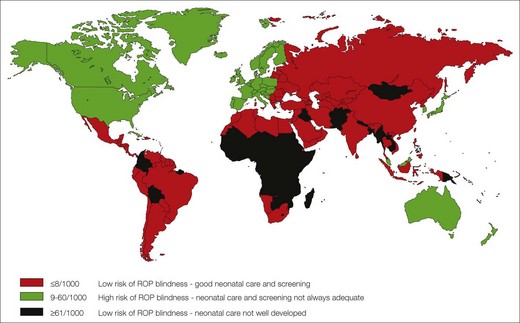
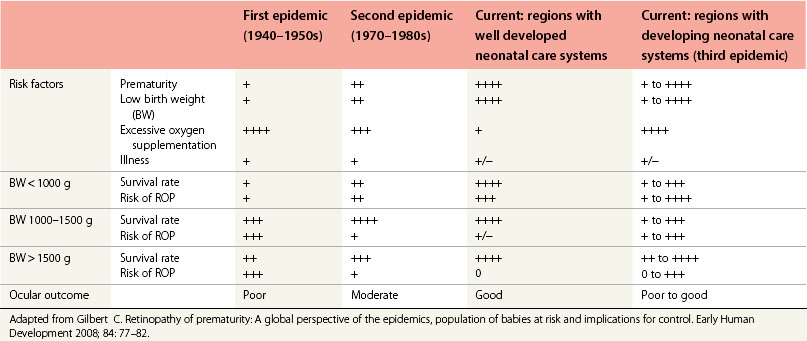
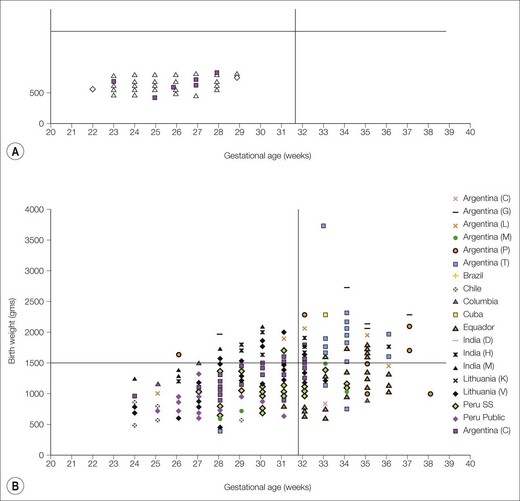
In industrialized countries, severe ROP is largely confined to infants with birth weights of less than 1000 g and gestational age of 31 weeks or less, and blinding disease is rare in larger babies.132 However, the prevalence in developing and industrializing countries appears to be increasing and blinding ROP is not confined to very low birth weight infants. When the infant mortality rates fall between 10 and 50 per 1000 births in an industrializing country, the risk for blindness due to ROP increases dramatically. The risk is very low in those countries with infant mortality rates of more than 50 per 1000 births133 (Fig. 43.15 and Table 43.3). In settings where only the premature babies with higher birth weights survive (e.g. the ROP epidemic of the 1940–1950s), extent of prematurity and low birth weight have minimal contributions because oxygen supplementation as a surrogate for the level of neonatal care is the prominent risk factor. Now that more babies with the lowest birth weights survive due to improvements in neonatal and perinatal care (as in the nurseries of the 1990s and 2000s), the extent of the infant’s prematurity becomes more important.
ROP disability: the three epidemics
There have been three ROP epidemics:108,133–137
1. The first in the 1940s was due to unrestricted, unmonitored oxygen and was brought to an end by oxygen restriction. The survival of neonates of < 1000 g birth weight was 5–8%, and most of the babies blinded during this period were of heavier birth weight. ROP was largely eliminated by advances in neonatal care.138
2. In the late 1960s, in countries with well developed neonatal intensive care services, the increased survival of the very immature neonate who did not previously survive (now around 50–60% < 1000 g birth weight) resulted in a second epidemic.
3. The term “third epidemic” describes the prevalence of ROP-induced visual disability worldwide and provides insights and opportunities for preventive interventions.108
Countries can be subdivided according to health–socioeconomic criteria into high-, middle-, or low-income according to the health provision (Fig. 43.16). There may be diverse health–socioeconomic communities within a single country. In high-income countries, where wealth and technology permit generally high-quality health care and neonatal intensive care, ROP-induced disability accounts for 3–8% of childhood vision impairment.139–142 In middle-income communities, technology permits increased survival of preterm babies, but limited health resources may limit the standard of care.133 Consequently, babies with higher birth weights and gestational ages are at risk of severe ROP; ROP-induced blindness contributes up to 39% of childhood vision impairment due to increased survival and associated limited resources with poor quality neonatal care108,143 (see Table 43.3). In low-income countries, very few preterm babies survive to develop ROP.108,109
The evidence that ROP-induced disability has been reduced by treatment comes from two sources. First, from studies such as the CRYO-ROP study, which demonstrated that the beneficial effects of treatment are maintained at least 15 years after treatment.144 Second, epidemiologic studies such as the report from the UK which showed that, between 1976 and 1985, ROP contributed 5% of childhood vision impairment; this proportion rose to 8% between 1986 and 1990140 and subsequently fell to 3% by 2000.145 Given the increase in survival of the most immature babies,143 in the absence of treatment, a considerable increase would have been anticipated.
Natural history
1 Age at onset and rate of progression
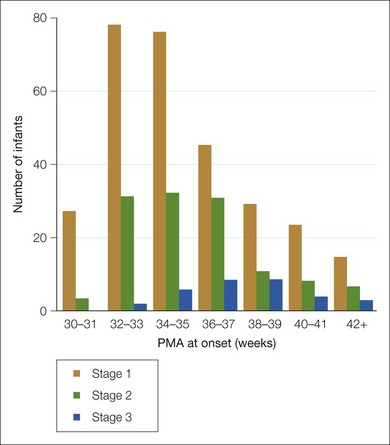
ROP affects only immature retinal vessels and cannot develop after retinal vascularization is complete. One would expect the most premature, often ill, neonate with a very immature retinal vascular system to develop ROP sooner postnatally than a larger and more mature baby. However, this is not so (Fig. 43.17); ROP develops over a relatively narrow PMA range.40,119,146,147 The time of ROP onset is linked more to the stage of development of the infant, by PMA, than neonatal events such as oxygen therapy or illnesses,40,119 acknowledging that the latter contribute to ROP occurrence and severity. The time during which ROP can develop is governed by PMA, but is terminated by complete retinal vascularization (also related to PMA). The narrowness of this “window” is exhibited by the occasional example of a baby of > 33 weeks gestational age and > 2000 g body weight (by which time the time remaining to develop ROP is very limited) who has received prolonged and excessive 100% oxygen and has reached stage 5 by 37 weeks PMA.
As with onset, the rate of progression is also governed predominantly by the stage of development (PMA) rather than by postnatal age, or neonatal events. Data119,130,132,148 show remarkable consistency in the behavior of ROP. The median PMA at which the various ROP stages and features develop is as follows:
In the CRYO-ROP study, babies were randomized for treatment (i.e. within 72 hours of diagnosis of threshold ROP) at a mean age of 37.7 weeks PMA (range 32–50 weeks).149 The mean age at treatment for prethreshold and threshold eyes in ETROP was 35.2 weeks and 37.0 weeks, respectively. The extremes of the age range at which eyes require treatment are notable, the earliest being 31 weeks PMA,119,130,149 Yet, almost all babies (99%) that develop severe ROP will have done so by 46.3 weeks PMA.148 The literature cited so far has all come from countries with high standards of neonatal care.
In countries with more variability in the standard of care, sight-threatening disease affects more mature and larger babies.108,109,150 In regions developing neonatal care systems, ROP progression also behaves according to PMA.
2 Zone of involvement
The propensity for severity of ROP is governed by the state of retinal vascularization, so that zone is the most important predictor of outcome.42,131 Incomplete vascularization in zone I carries a 54% risk of reaching threshold, but this risk falls to 8% when vessels have reached zone II. For ROP developing in zone III the risk of an adverse outcome is almost nil.151
3 Site of onset
ROP commences in the temporal retina in the more mature neonates when the nasal retina is already vascularized. However, in the immature neonate ROP commences preferentially in the nasal retina and later extends to other regions.39,152 The zones of vascular development were depicted by the international classification as being circular, but when this was revised in 2005113 their elliptical shape was recognized; retinopathy in the nasal retina is frequently closer to the optic disk than ROP in the temporal retina.153 The vertical retinal regions are less likely to be involved at onset and are only involved when ROP involves most of the circumference. The finding of ROP in these regions early in the course of the disease is a useful indicator of the possibility of future severity. The more premature the neonate, the more posterior by zone the location of the retinopathy and the greater the potential for progression. Thus, zone I disease is very likely to progress to stage 3, but ROP confined entirely to zone III rarely does.119
4 Plus disease
Plus disease is a critical sign of ROP activity and is now the main driver for treatment.132 Plus disease may be superimposed on any ROP stage and is a sign that ROP is, or is about to become, severe.40,114 Advanced plus disease is obvious, but if mild, is the least robust diagnostic aspect of ROP. To overcome this, attempts are being made to quantify some components of plus disease by digital imaging.
ROP detection programs
Severe ROP can be successfully treated.131 The ophthalmologist has a duty to examine babies to detect development of ROP requiring treatment. Understanding its natural history provides the basis for a logical and simple ROP examination protocol that is cost-effective in countries with well developed NICU systems.154–159
Which infants should undergo ROP examinations?
In high-income countries, severe ROP (stage 3 or more) is largely confined to infants of birth weight < 1501 g and < 32 weeks gestational age.159 Thus, in the UK, these are the current guidelines for those babies who require ROP examinations. In the USA, ROP examinations are recommended for all babies < 1501 g or with a gestational age of < 30 weeks and for babies between 1500 and 2000 g birth weight who had an unstable clinical course and are considered at risk of ROP.158
For high-income countries, the UK criteria include most, if not all, babies who develop severe ROP (stage 3 or more).160–165 Can we safely reduce the number of babies screened for ROP? Many of the larger and more mature babies who currently undergo examination are at very low risk of developing severe ROP. Of 16 000 babies, there was only one baby over 1200 g with severe ROP.166 Two studies from the UK have shown that the criteria of < 1251 g birth weight and/or < 30 weeks gestational age included all babies with stage 3;160,161 however, three US studies have reported stage 3 in larger babies.162,163,167
For low- and middle-income countries, larger babies also develop severe ROP, and the UK and US criteria may not include all babies at risk.133 It is essential in these communities for ROP examination guidelines to be based on local data.
Timing of examinations
Examinations can be timed on the principle that the onset and progression of ROP are predominantly determined by PMA rather than by neonatal events. While most guidelines recommend commencing ROP examinations between 4 and 7 weeks, Reynolds et al.148 developed an algorithm for examining the most immature baby later postnatally than the more mature baby. Timing of examinations is critical; there is only a narrow, imperfectly determined window for treatment. Onset of any stage of ROP is rare before 30 weeks PMA, and ROP commencing once vascularization has entered zone III and/or after 36–38 weeks PMA is most unlikely to reach severe disease.
After the initial ROP examination, weekly or every other week examinations are indicated, but as ROP progresses, more frequent examinations will be required. A weekly visit to the NICU allows examination of those babies likely to be discharged home soon and decreases the need for babies to be recalled, decreasing the likelihood of missed examinations and their potentially disastrous consequences.168 For the baby transferred to another hospital while still at risk, arrangements must be made for the continuation of ROP examinations.
Methods of examination
Eye examinations are stressful for the preterm baby,169–173 but this can be reduced by supportive neonatal care. Infants should be handled gently. It is helpful if a trained nurse is in attendance to help and to monitor the infant’s wellbeing. The ophthalmic examination should be performed using the indirect ophthalmoscope, with a 28- or 20-diopter lens, through dilated pupils, achieved by instilling dilating drops well prior to examination. A child’s lid speculum and scleral indenter (for ocular rotation rather than indentation), after anesthetic eye drops, may be used to permit complete evaluation of the peripheral retina. Although it can be argued that very peripheral disease so observed is of no clinical importance, identifying peripheral nasal retinopathy can be a clue to later progression. Only by observing the state of vascularization of the nasal retina can the ophthalmologist determine whether zone III has been entered.
Clinical findings
The ophthalmoscopic appearance of the neonate is different from that of the older infant.172 Before examining the retina it is important to use the indirect lens as a simple magnifier to examine the anterior segment, for instance, to see the amount of regression of the tunica vasculosa lentis as an indicator of maturity (regressing between 28 and 34 weeks gestational age).173
The retinal periphery is white-gray in its non-vascularized regions; the extent of this depends on the degree of immaturity (see Fig. 43.1). The optic disk frequently has a grayish and a double-ring appearance but the disk is a normal size. The macular area is relatively ill defined, there being no macular or foveolar reflexes until 36 and 42 weeks PMA, respectively.26 The retinal arterioles in the early neonatal period are not tortuous, tending to become so later, in part related to ROP severity.39