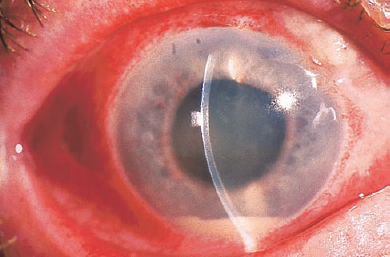
22 Endophthalmitis is a severe ocular condition involving infection of the internal structures of the eye by replicating microorganisms. Most often, these organisms are bacteria, but fungi, protozoa, and viruses can produce this disease. Most endophthalmitis follows surgery or trauma, at which time these organisms gain access to the inside of the eye. This is known as exogenous endophthalmitis. Less commonly, infectious organisms enter the eye through a hematogenous route, producing endogenous endophthalmitis, a disease more typically found in debilitated or immunosuppressed patients, intravenous drug abusers, and patients with indwelling catheters. Postoperative endophthalmitis can be acute (within 6 weeks of surgery) or delayed onset (more than 6 weeks). The onset of endophthalmitis has diagnostic and prognostic implications and directly impacts the initial management of the patient. The incidence of postoperative endophthalmitis has been studied in several large series. In the United States, the incidence was reported as 0.072% in a series of 23,625 cases.1 The estimates are somewhat higher for secondary intraocular lens (IOL) implants and penetrating keratoplasty (0.3%). It is distinctly unusual after vitrectomy surgery (0.02%). After penetrating trauma, the incidence varies between 3 and 30%, depending on the setting (nonrural is lower compared with rural) and whether an associated intraocular foreign body is present. The most common symptom is decreased vision, noted in 94% of patients in the endophthalmitis vitrectomy study (EVS), a randomized controlled clinical trial assessing the efficacy of intravitreal antibiotics, intravenous antibiotics, and vitrectomy in postsurgical endophthalmitis.2,3 Pain is not universal but was reported in 75% of patients. Unlike the typical postoperative course, the pain may become progressively worse rather than progressively better with time. The major signs of endophthalmitis are hypopyon and vitreitis (Fig. 22–1). Signs of periocular inflammation, such as eyelid and conjunctival erythema and corneal edema, may also be present and are typically more fulminant than that typically seen postoperatively. In the EVS, hypopyon was reported in 85.7% of patients. Membrane formation can occur around the IOL implant. A variable vitreitis is present, sometimes completely obscuring a view of retinal detail. A red reflex was present in only 32% of patients. Bleb-associated endophthalmitis can occur anytime after filtering surgery and is more common in blebs located inferiorly. Pus in the bleb may be the presenting sign. In chronic endophthalmitis caused by Propionibacterium acnes, a white plaque may be seen on the posterior capsule. Chronic granulomatous uveitis is often present. The distribution of signs and symptoms is summarized in Table 22–1. Bacteria are the most common cause of endophthalmitis, with gram-positive organisms accounting for 60 to 80% of acute infections. Generally, the organisms involved in acute postoperative endophthalmitis derive from the normal lid flora.Staphylococcus, particularly coagulase-negative varieties (70%), Staphylococcus aureus (10%), and Streptococcus species (11.5%) are the most common of these. Gram-negative bacteria accounted for 6% of cases in the EVS and are considerably more virulent than the coagulase-negative Staphylococcus. Delayed-onset endophthalmitis is typically due to P. acnes, a gram-positive bacillus, but can be caused by Candida parapsilosis, Staphylococcus epidermidis, and Corynebacterium species. Bleb-associated endophthalmitis is often a streptococcal infection; and these organisms can be particularly virulent. Traumatic endophthalmitis, in which organic foreign material is involved, has a greater likelihood of involving Bacillus cereus, which can have a dramatically virulent course. Fungal infection is reported in 10 to 15% of cases. Endogenous endophthalmitis is more commonly associated with fungal infection, particularly Candida and Aspergillus, but it can be bacterial, with Neisseria meningitidis, S. aureus, and Haemophilus influenzae being important causes. A thorough investigation for a systemic source should be undertaken in any suspected case of endogenous endophthalmitis and any infection addressed. FIGURE 22–1. Hypopyon is one of the common signs of endophthalmitis. See Table 22–1 and the text for full details.
Retinal Disease and the Anterior Segment Surgeon
Endophthalmitis
What Is Endophthalmitis?
What Is the Incidence of Endophthalmitis?
What Are the Symptoms of Endophthalmitis?
What Are the Signs of Endophthalmitis?
What Microorganisms Are Responsible for Endophthalmitis?
| Signs and Symptoms | Percent |
|---|---|
| Visual acuity <5/200 | 90 |
| Visual acuity LP | 26 |
| Pain | 75 |
| APD | 12 |
| Corneal infiltrate | 5 |
| Hypopyon | 86 |
| Inability to visual retinal vessels | 80 |
| Red reflex present | 23 |
LP, light perception; APD, afferent pupillary defect.
How Do You Categorize Endophthalmitis?
Endophthalmitis can be grouped into several categories with prognostic and therapeutic importance. These categories help determine the responsible organism and guide the initial choice of medical or surgical therapy. The most common form is acute postoperative endophthalmitis, which occurs within 6 weeks of intraocular surgery. Delayed-onset postoperative endophthalmitis occurs more than 6 weeks to several months after surgery and often presents with chronic indolent inflammation. Bleb-associated endophthalmitis can follow filtering surgery (or occur in eyes with inadvertent blebs) anytime. Infection isolated to the bleb (i.e., without endophthalmitis) can often be managed with intensive topical or periocular antibiotics. Traumatic endophthalmitis can follow penetrating or perforating eye injuries. In a rural setting, endophthalmitis has been reported in as many as 30% of cases, with a substantial portion of these being Bacillus species. In a nonrural setting, the incidence is considerably lower. Visual recovery is often limited by associated traumatic damage to ocular structures as well as the virulence of the microorganisms involved. Endogenous endophthalmitis refers to the hematogenous spread of microorganisms from a distant site. Patients are typically debilitated or immunosuppressed. Infections are generally fungal in nature but can be bacterial from a variety of sources.4
What Has the Endophthalmitis Vitrectomy Study Taught Us about Optimizing Treatment for Endophthalmitis?
The EVS was a randomized, multicenter clinical trial sponsored by the National Eye Institute to help determine the optimal management of acute postoperative endophthalmitis.2,3 The efficacy of vitrectomy versus vitreous tap, both with intravitreal antibiotic injection, was assessed. The effect of concomitant intravenous antibiotics was also studied. Patients with endophthalmitis within 6 weeks of cataract surgery or secondary IOL implantation were included. Vision between 20/50 and light perception was allowed, and some clarity of the cornea and anterior chamber sufficient to visualize part of the iris was required. Vitreous or anterior chamber opacity obscuring visualization of second-order arterioles was required. Study patients were randomized to one of four groups, all of whom received intravitreal antibiotics: (1) initial vitrectomy with intravenous antibiotics, (2) initial vitrectomy without intravenous antibiotics, (3) initial tap with intravenous antibiotics, and (4) initial tap without intravenous antibiotics.
For patients randomized to receive intravenous antibiotic therapy, ceftazidime 2 g every 8 hours was administered intravenously (or ciprofloxacin 750 mg twice daily administered orally for penicillin-allergic patients) and amikacin 7.5 mg per kilogram of body weight load then 6 mg per kilogram every 12 hours was administered.
Patients were reevaluated 36 to 60 hours after the initial procedure; 10.5% of patients required an additional procedure, of which 70.5% were reinjection of intravitreal antibiotics. Further intervention was recommended if the eye was clinically worsening and the following criteria were met: (1) vision 5/200 to light perception; (2) worsening media opacity; 3) at least equivocal growth from the initial cultures; and(4) 1-mm increase in hypopyon, corneal ring infiltrate, or worsening pain.
The primary study endpoints were visual acuity and clarity of the ocular media. No statistically significant difference in final visual acuity or media clarity was observed in patients treated with systemic antibiotics versus those without systemic antibiotics. No difference was observed in patients treated with initial tap and injection versus initial pars plana vitrectomy if the presenting vision was hand motions or better. An important difference in outcome occurred in patients who presented with vision of light perception only. Initial vitrectomy yielded a three times greater likelihood of achieving better than 20/40 final vision (33% versus 11%). Patients treated with vitrectomy in this group were twice as likely to achieve better than 20/100 (56% versus 30%) and less likely to have worse than 5/200 (20% vs. 47%) compared with the group of patients subject to initial vitreous tap and intravitreal antibiotic administration. A clinical pathway for the treatment of endophthalmitis from a variety of causes is shown in Figure 22–2.
What Is the Role of Cultures in Endophthalmitis?
Cultures yielded positive results in 69.3% of cases,12.8% were equivocal, and 17.9% yielded no growth. Cultures from the anterior chamber were positive in only 4% of cases. Importantly, gram-positive organisms were isolated in 94.2% of eyes. Coagulase-negative organisms were seen in 68%. All gram-positive organisms were sensitive to vancomycin. Six percent of eyes had gram-negative infections. Of these, 89% were sensitive to both amikacin and ceftazidime. Eleven percent were resistant to both of these antibiotics. Gram-negative organisms were never cultured from eyes in which a second-order retinal vessel was visible on initial examination.
What Drugs Are Indicated for the Treatment of Endophthalmitis?
Initial antibiotic therapy should cover a broad spectrum of microorganisms, have efficacy at concentrations achieved by intravitreal injection, and have low toxicity. Bactericidal agents are preferred over bacteriostatic drugs because the eye is an immune privileged site. Coverage to include methicillin-resistant Staphylococci and Bacillus species in trauma is necessary. Some antibiotics exhibit considerable retinal toxicity when introduced into the vitreous cavity. Aminoglycosides, particularly gentamicin, can cause vascular occlusion and retinal necrosis. The most commonly administered drugs are listed below:
Cephalosporins are synthetic penicillins. Cefazolin, a first-generation drug, has a relatively broad gram-positive coverage but weak activity against gram-negative, methicillin-resistant organisms and enterococci. Ceftazidime 2.25 mg in a 0.1 mL volume is typically administered.
Vancomycin is a bactericidal agent effective against most gram-positive bacteria, including Bacillus. It is generally regarded as first-line empiric therapy and is given as a 1 mg injection in a 0.1 mL volume.
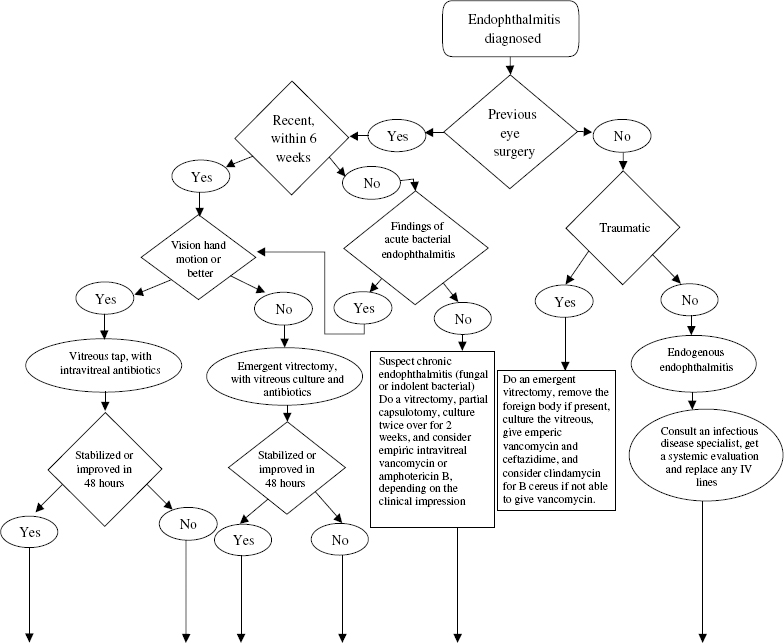
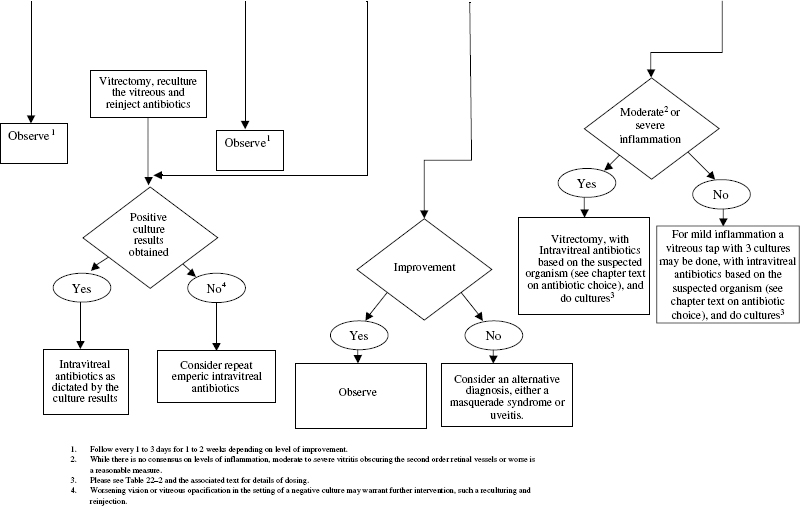
FIGURE 22–2. Clinical pathway for endophthalmitis. IV, intravenous.
Quinolones inhibit DNA synthesis and have broad-spectrum activity against numerous microorganisms. Anti-Streptococcal and anti-anaerobe activity is somewhat weak. Intravitreal administration can result in retinal toxicity, however. Ciprofloxacin administered systemically does achieve significant levels in the vitreous cavity.5
Antifungal drugs include amphotericin B and the azoles. Amphotericin B dosed at 5 to 10 μg in 0.1 mL has been used intravitreally with reasonable success in cases of fungal endophthalmitis. Moreover, in some instances of Aspergillus endophthalmitis, amphotericin B can be combined with 5-fluorocytosine.
A common initial approach to the treatment of bacterial endophthalmitis is the administration of intravitreal vancomycin 1 mg in 0.1 mL and ceftazidime 2.25 mg in 0.1 mL. Subconjunctival vancomycin and ceftazidime might be given as well. A postoperative drop regimen consisting of topical vancomycin, ceftazidime, and a steroid such as prednisolone acetate is commonly used.
Intravitreal antibiotics and antifungal agents commonly used in the treatment of endophthalmitis and their doses are summarized in Table 22–2.
What Are the Routes of Administration?
Antibiotics can be given topically, subconjunctivally, intraocularly (either intracameral or intravitreal), and systemically. Intraocular administration, particularly intravitreal injection, achieves therapeutic dosing for a period of approximately 24 to 48 hours. Systemic administration generally does not achieve therapeutic levels in the vitreous cavity, with the exception of ciprofloxacin.
| Antibiotic | Dose (in 0.1 mL) |
|---|---|
| Vancomycin | 1.0 mg |
| Amikacin | 0.4 mg |
| Ceftriaxone | 2.0 mg |
| Ceftazidime | 2.25 mg |
| Clindamycin | 0.5 to 1.0 mg |
| Amphotericin B | 5 or 10 μg |
How Do You Follow Up on Endophthalmitis?
Successful treatment often results in a dramatic improvement in pain by the first postoperative day. Worsening inflammation in the face of persistent pain may indicate active infection and the need for further intervention. Repeat tap and injection or vitrectomy surgery would be warranted for eyes that are clinically worsening. If cultures suggest resistance to the antimicrobial agent used, repeat injection with an appropriate antibiotic is necessary. Difficulty assessing the retina is a frequent problem because of media opacity; so serial ultrasounds are sometimes needed to identify retinal detachment, which was observed in 8% of eyes in the EVS. Additionally, membrane formation and traction detachment resulting from severe intraocular inflammation can result. Persistent corneal edema might require eventual penetrating keratoplasty. Elevated intraocular pressure, however, often can be managed medically. More difficult to control is postoperative hypotony. Vigorous control of intraocular inflammation can be helpful. A through evaluation for a wound leak is necessary.
When Would You Consider Re-Treating?
Retreatment of endophthalmitis is indicated for cases where stabilization or improvement is not noted after the first 36 to 48 hours. Signs of worsening include increased hypopyon, decreased vision, and an increase in vitreous opacification (which can be documented on ultrasound in situations where the vitreous is not ophthalmoscopically visible). In eyes previously treated with tap and injection, consideration of pars plana vitrectomy and reinjection should be given. In eyes previously vitrectomized, repeat vitrectomy or reinjection of antibiotics should be performed. If possible, retreatment should be guided by culture results from the initial vitreous or aqueous specimen.
How Can Endophthalmitis in Silicone-Filled Eyes be Treated?
Endophthalmitis in the silicone-filled eye can be difficult to detect because the eye may seem quieter than expected; however, successful treatment of endophthalmitis in this setting has been reported. The technique involves removing the silicone oil, washing the vitreous cavity with antibiotic for 15 minutes, performing an 80% fluid–air exchange, so that approximately 20% of the antibiotic solution remains in the eye, then replacing the oil.6 Oil itself may have some intrinsic antimicrobial activity against some bacteria and fungi.7
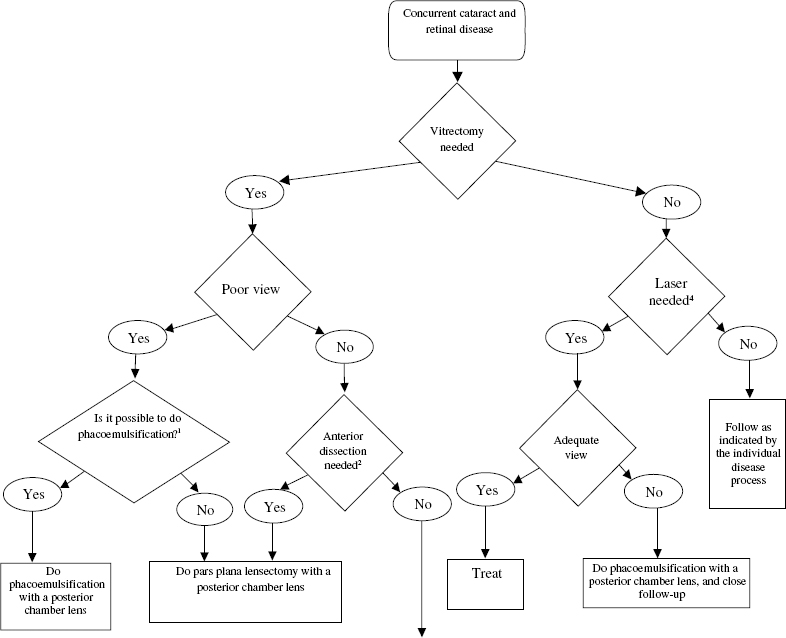
When to Remove a Cataract in the Face of Retinal Disease
What Are the Considerations for Removing a Cataract in a Patient with Retinal Disease?
The management of the lens in patients with underlying retina disease depends on the visual potential of the eye and how readily the goals of retina surgery or laser can be achieved. In some cases, it may be perfectly reasonable to leave a relatively dense cataract untouched in a patient with an underlying disciform scar and little chance of significant visual improvement, particularly if the fellow eye is in good condition. On the other hand, extracting a clear lens may be indicated if an anterior dissection of membranes is necessary to reattach the retina. Whether cataract extraction worsens preexisting retinal disorders is known for only a few conditions and is discussed here. Studies are under way to help address this issue for other diseases. Some investigators believe that the lens may serve as a barrier to diffusible substances such as vascular endothelial growth factor and may protect against rubeosis in proliferative retinopathies.8 There is always, of course, concern for the development of cystoid macular edema (CME) following cataract surgery. Macular edema may be particularly exuberant and aggressive in some patients with underlying retinal vascular disease. A second consideration is the likelihood of worsening cataract after a vitreoretinal procedure such as pars plana vitrectomy and membrane peeling for epiretinal membrane. In selected cases, simultaneous cataract extraction and IOL implantation concurrent with the posterior segment surgery are advisable.
There are a variety of techniques for the removal of the lens, and limited data are available to suggest which of these might be the best approach. Intracapsular cataract extraction (ICCE) was associated with a high incidence of CME and progression of diabetic retinopathy in the series reported by Aiello and colleagues.9 ICCE is decreasing substantially in popularity, however, and may produce problems with visual rehabilitation. Moderate levels of progression of retinopathy (20%) and macular edema (8%) have been reported in a small uncontrolled series after extracapsular cataract extraction (ECCE) with nuclear expression.10 Data from phacoemulsification cases have been reviewed in number of series. Mittra and colleagues reported similar postoperative findings after phacoemulsification in a retrospective series of 150 patients, compared with other modalities of cataract extraction.11 Seventy-eight percent had improvement in visual acuity of two or more lines. One quarter had progression of retinopathy. Risk factors for progression included preexisting nonproliferative or proliferative retinopathy and longer surgical times. Zaczek and colleagues reported similar data.12 Additionally, manipulation of anterior structures in patients with severe diabetic retinopathy can lead to the formation of a postoperative fibrin reaction that can be quite aggressive, sometimes leading to synechiae formation and hypotony.
Finally, pars plana lensectomy should be considered in specialized circumstances, for example, when anterior dissection of membranes is needed. In some situations, it is possible to leave the anterior capsule intact and place an IOL in the sulcus, either at the conclusion of surgery or as a second procedure. Figure 22–3 summarizes these considerations and how one might approach concurrent cataract and retinal disease.
The decision as to whether to implant a lens and the type of lens use is an important issue with little in the way of data to support one type over another in the literature. We recommend using a large-diameter optic (at least 6.5 mm) acrylic foldable lens for cases where small incision phacoemulsification is performed or an all polymethylmethacrylate (PMMA) lens with similar dimensions. Silicone lenses and plate-haptic lenses are not recommended. Silicone lenses are incompatible with silicone oil, which may be adsorbed onto the surface or absorbed into the lens resulting in irregular refractive changes.
How Does Retinal Surgery Impact on Cataract Formation?
Cataract formation is common after vitrectomy surgery; the incidence of progressive nuclear sclerosis at 2 years is 70 to 80%. The exact cause is unknown, but numerous hypotheses exist, including changes in the lens due to the infusion, local alterations in oxygen concentration, and ultraviolet or heat exposure from the operating microscope. Some “feathering” of the posterior lens capsule is common with intravitreal gas tamponade and is sometimes reversible. Eyes requiring silicone oil tamponade, for example, for complicated retinal detachments or the repair of macular holes where appropriate postoperative positioning cannot be achieved, inevitably develop cataracts. The incidence approaches 100% between 6 and 18 months postoperatively and can develop even after silicone oil removal.13–18
In cases of retinal disease with good visual prognosis, such as some epiretinal membranes and macular holes, it is reasonable to consider concurrent cataract extraction, IOL implantation, and vitrectomy surgery. We often extract the lens in eyes with relatively mild cataract, particularly if protracted gas or oil tamponade is anticipated, expecting the lenticular opacity to worsen rapidly in the postoperative period and become visually significant. The approach to the cataract varies between surgeons and sometimes involves collaboration between the anterior segment specialist and the vitreoretinal surgeon. In many cases, phacoemulsification and IOL implantation are performed first, followed by vitrectomy and membrane peeling. The cataract surgery may be performed through a scleral tunnel approach or a clear-cornea approach. We have found that suturing the wound, even if it appears self-sealing, greatly facilitates further manipulation of the posterior segment and maintains a formed anterior chamber. One disadvantage is that the anterior chamber may become somewhat turbid and the cornea mildly edematous following the lens extraction, occasionally compromising the view for the posterior-segment work. We have not found this to be a particularly significant problem, however. It has the advantage of allowing in-the-bag placement of an IOL. An alternative is to perform the cataract surgery as a separate procedure before vitrectomy, but this of course has the disadvantage of exposing the patient to two surgeries and doubling the anesthetic risk. Finally, pars plana lensectomy, sparing the anterior capsule allows for sulcus placement of the IOL.