Radiation Retinopathy
Neal H. Atebara
John H. Drouilhet
Gary C. Brown
Katie Atebara
First described by Stallard1 in 1933 in the fundi of patients with retinal capillary hemangiomas and retinoblastomas after radon seed implantation, radiation retinopathy has become increasingly more common as additional radiotherapeutic techniques have come into use. Ionizing radiation appears to have a pathologic effect on the small blood vessels of the retina and optic nerve. Characteristically, months to years after exposure to radiation, there is a slowly progressive dysfunction of the vasculature of the retina and optic nerve, leading to alterations in structure and permeability, occlusion, and neovascularization. The clinical picture of capillary telangiectasia, microaneurysms, intraretinal hemorrhages, hard exudation, macular edema, capillary nonperfusion, and neovascularization can be identical to that of diabetic retinopathy, which is suggestive of a common pathophysiologic mechanism of damage to capillary endothelial cells. Visual loss usually is caused by macular edema, macular hard exudates, macular ischemia, or optic nerve damage.
Pathophysiology
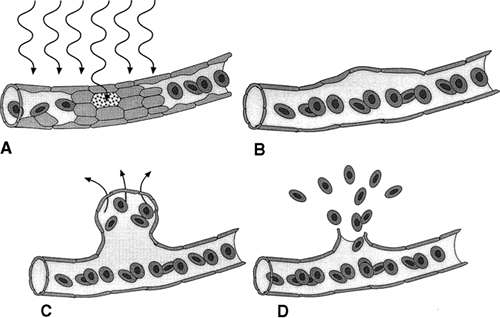
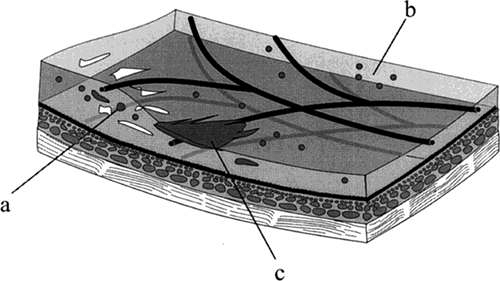
Although it is not entirely clear how ionizing radiation causes radiation retinopathy, the primary site of damage appears to be the endothelial cells of the retinal and optic nerve vasculature.2,3,4,5,6 Although the neural retina, like the central nervous system, is resistant to radiation damage,5,7 the blood vessels within them are not.8 Ionizing radiation interrupts cell processes during interphase, and as the cells attempt division in the ordinary course of repair and replacement, radiation also interrupts mitosis (Fig. 1A). The primary event is probably endothelial cell damage with secondary loss of pericytes. This histologic picture is similar to that seen in diabetic retinopathy.
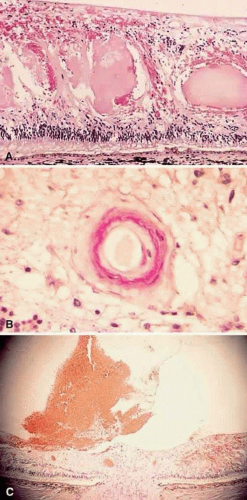
Irvine and Wood9 irradiated monkey eyes with up to 30 Gy and examined them histologically. The first changes, seen 12 to 24 months after irradiation, were focal loss of capillary endothelial cells and pericytes (see Fig. 1B). Archer et al.10 examined human eyes enucleated after radiation treatment and found endothelial cell loss. Fusiform capillary dilations and microaneurysms develop in areas of poorly supported capillaries (see Fig. 1C). Telangiectatic channels also develop, presumably caused by alterations in hemodynamics.
Lower degrees of radiation damage cause the vascular endothelial cells to remain viable, but lose the integrity of their intercellular tight junctions. The blood vessels become abnormally permeable to larger molecules, leading to leakage of fluid and proteins (see Fig. 1C). Archer et al.10 performed electron microscopic studies that revealed new intraretinal vessels containing fenestrated endothelium, suggesting an alternative mechanism of vascular leakage. Light microscopic examination of the retina reveals intraretinal blood from burst microaneurysms (see Fig. 1D) and eosinophilic exudates in the outer plexiform and inner nuclear layers (Fig. 2A).
In more severe cases of radiation damage, the endothelial cells die, and the vascular channels become occluded, leading to capillary dropout and ischemia. As areas of capillary loss become confluent, cotton-wool spots are seen clinically. These subsequently fade as large areas of retinal capillary nonperfusion develop.9 Later in the course, larger vessels become affected, possibly related to thickening of the vessel walls from a deposition of fibrillary or hyaline material10 (see Fig. 2B). Retinal ischemia, presumably mediated by vasoproliferative factors, leads to neovascularization of the retina and optic disc (see Fig. 2C), vitreous hemorrhage, and neovascular glaucoma.
Symptoms
Radiation retinopathy often is asymptomatic in its early stages. The disease process usually does not become manifest symptomatically or ophthalmoscopically for months to years after the irradiation. The first symptoms a patient typically notices is blurred or dimmed central vision. This usually results from macular edema or macular ischemia. Less commonly, abrupt and severe loss of vision can occur from central retinal artery occlusion,11 central retinal vein occlusion,12 ischemic optic neuropathy,13 or vitreous hemorrhage. In advanced cases of radiation damage, the eye can become blind and painful as a result of radiation-induced ocular ischemia or neovascular glaucoma.
Ocular Features
The first signs of radiation retinopathy typically occur months to years after the exposure to ionizing radiation. Brown et al.2 found that an average of 14.6 months passed from the time of radioactive plaque therapy until the onset of radiation retinopathy with a range of 4 to 32 months. The findings for external beam radiation were similar with a mean period of 18.7 months and a range from 7 to 36 months. Guyer et al.13 report an even more delayed onset of signs after proton beam radiation that ranged from 5 months to 15 years with a median of 40 months. Although Flick in 194814 described acute retinal changes in survivors of the atomic blasts at Hiroshima and Nagasaki, these acute lesions correlated with low white blood cell counts and were thought to result from the radiation-induced pancytopenia, not direct injury to the retina.
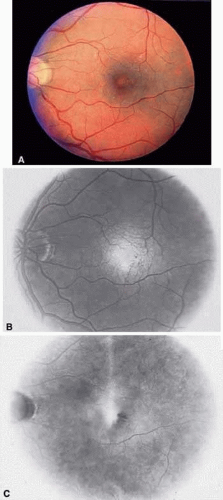
The first ophthalmoscopic features of radiation retinopathy reflect vascular endothelial damage. Guyer et al.13 found that the earliest and most common finding was macular edema that was present in 87% of patients within 3 years of radiation. Leakage of fluid and proteins into the neurosensory retinal tissue causes retinal edema and hard exudates. Structural decompensation of the vessel wall leads to intraretinal microaneurysms and intraretinal microvascular abnormalities. Hemorrhage of these microaneurysms result in dot and blot intraretinal hemorrhages.2,3,4,15,16,17 This funduscopic appearance is similar to that of diabetic retinopathy except that in most cases fewer microaneurysms are seen. Recognizing the similarity in disease progression patterns with background diabetic retinopathy, this early stage of the disease has been called “background radiation retinopathy.”18 The fundus photographs and fluorescein angiograms shown in Figures 3 and 4 demonstrate the findings seen in background radiation retinopathy. Figure 5 also reviews these findings.
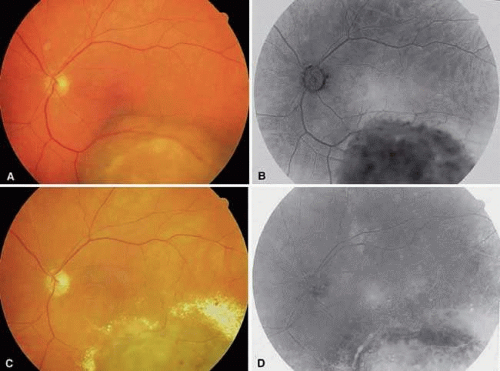
The later ophthalmoscopic features of radiation retinopathy are reflective of vascular occlusion at various layers of the retina and choroid. These changes may be considered “preproliferative radiation retinopathy.” Capillary endothelial damage from ionizing radiation results on patches of capillary nonperfusion. Ischemia in the nerve fiber layer (perfused by the peripapillary capillary retinal network) results in retinal cotton-wool spots. Alterations in retinal vascular blood flow result in retinal venous beading. Less commonly, occlusion of the larger retinal vessels occurs. Retinal arterial and vein occlusions can be seen, which is possibly related to the vessel wall sclerosis seen histologically. Uncommonly, occlusion of the choroidal circulation can be seen, detected on fluorescein angiography, or seen later as retinal pigment epithelial alterations. The fundus photographs and fluorescein angiograms shown in Figure 6 demonstrate the findings seen in preproliferative radiation retinopathy. Some prefer the term nonproliferative radiation retinopathy rather than distinguishing “background” from “preproliferative” radiation retinopathies.
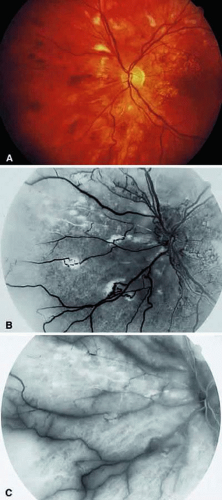 FIGURE 6. Preproliferative radiation retinopathy in a 23-year-old man who underwent radiation teletherapy with 30 Gy 18 months previously for a brain tumor. The patient underwent focal macular laser photocoagulation for macular edema 5 months earlier, but the final visual acuity was limited to 20/60 because of macular ischemia. A. Fundus photograph of the left eye reveals cotton-wool spots, intraretinal hemorrhages, and macular scars from prior laser treatment. B. Fluorescein angiography reveals broad areas of capillary nonperfusion in the nasal fundus. C. In the later phase of the angiogram, staining of the major vessels is apparent.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|