Definitions and Diagnosis
Microbial keratitis can be defined as corneal inflammation associated with replicating microorganisms, and this definition reflects the importance of microbial investigations. In hospital-based case series of presumed microbial keratitis, however, where corneal scrapes are routinely taken, approximately 50% of cases are culture-proven.
5,6,7 A culture-negative response may occur for several reasons: Where there is a small amount of tissue available for culture, viable organisms present deeper in the cornea which is not amenable to recovery on a corneal scrape, and prior treatment with antibiotics may reduce the viability of invading organisms. Frequently, cases of presumed microbial keratitis are managed empirically, without scraping, particularly in mild disease.
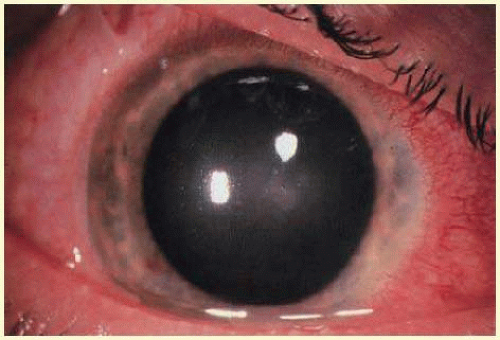
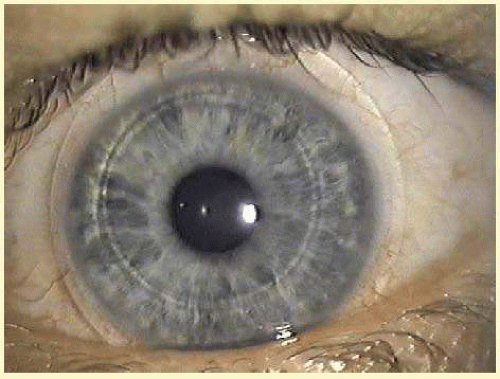
The appearance of focal corneal inflammation is, however, nonspecific and can also be associated with sterile keratitis in the absence of replicating organisms. Sterile keratitis is a heterogeneous group of inflammatory conditions including responses to microorganisms or their products, excipients of care solutions, or mechanical stimuli. Clinical and laboratory investigations may be used to differentiate between microbial and sterile disease, although sterile disease is generally a diagnosis of exclusion. Relevant clinical features in diagnosis would include the size, shape and position of the lesion, involvement of other ocular structures such as an anterior chamber response and lid swelling, disease history, management, and patient symptoms. The initial symptoms of microbial keratitis vary and are similar to the symptoms experienced with other contact lens-related complaints, including pain, foreign-body sensation, and redness.
8 Sterile keratitis is generally self-limiting upon removal of the contact lens, but microbial keratitis is almost invariably associated with progressive signs and symptoms.
8,9,10 Table 53.2 describes the relevant clinical criteria in presumed microbial and sterile keratitis, and
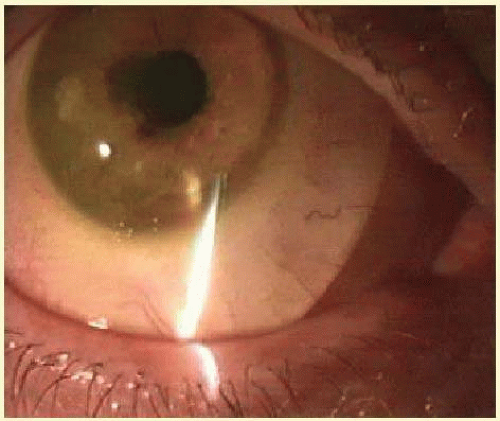
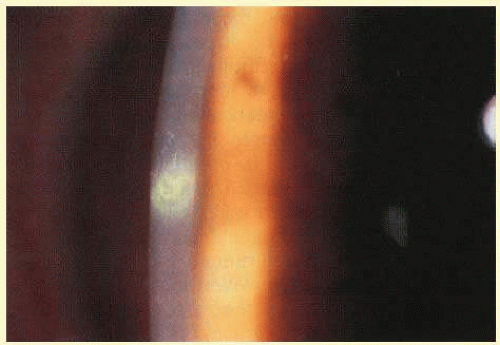
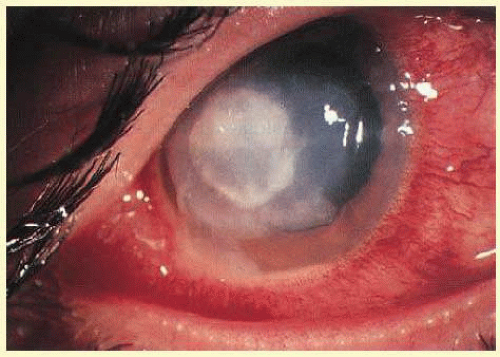
Figures 53.6,
53.7,
53.8 and
53.9 show examples of sterile and microbial disease.
The use of clinical criteria has been supported by epidemiologic data showing that these definitions can distinguish between distinct disease processes (
Table 53.2).
3,4,11,12,13,14,15 Various approaches have been described in such studies to exclude sterile keratitis, given the subjective nature of making a diagnosis. One recent approach has been to use a scoring grid to grade relevant clinical parameters for keratitis cases.
9 This has been applied to cases presenting to a hospital casualty department where cases have been classified as “severe” and “nonsevere keratitis” based on the numerical score.
16 The authors suggest that “severe” keratitis is analogous to a diagnosis of presumed microbial keratitis as reported in other studies. In a postmarket evaluation of overnight use of silicone hydrogel contact lenses, an endpoint committee comprising a panel of experts graded focal infiltrates using a predetermined classification scheme to identify those with a “high probability of microbial keratitis”.
17 Similarly, in the Australian surveillance studies, cases were considered by two independent reviewers and consensus was reached on the diagnosis of presumed microbial or sterile keratitis.
18 In epidemiological studies, the criteria for inclusion of an event as microbial keratitis need to be rigorously defined as this has a direct impact on estimates of incidence; however, in practical terms it is important to distinguish microbial keratitis from more common sterile inflammatory conditions to ensure appropriate and timely management. Delays in diagnosis of microbial keratitis of more than 12 hours are associated with poorer final visual outcome and morbidity.
19,20
Epidemiology
Epidemiology is the study of distribution and determinants of disease in a population, which requires study of both those with the disease and unaffected controls. Other than contact lens wear, risk factors include trauma; ocular surgery; ocular surface disease, particularly postherpetic disease, bullous keratopathy, neurotrophic disease, and exposure; and systemic conditions such as diabetes or immunocompromise.
14,21,22,23 Lens wear may account for 50% to 65% of new cases presenting to acute care facilities,
14,24 and in a tertiary referral center in urban Australia, contact lens wear and trauma comprised the two major preventable predisposing factors, each accounting for one-third of cases in a working-age population.
25Well-designed large-scale epidemiological studies have informed our understanding of the incidence of disease and predisposing factors relevant in the pathogenesis of contact lens-related microbial keratitis.
Table 53.3 summarizes the incidence estimates from a range of studies with hydrogel, silicone hydrogel, and daily-disposable contact lenses. While study design has varied between studies, in population-based studies where cases and community-based controls have been identified contemporaneously, daily use of hydrogel lenses is broadly associated with an incidence of disease of 2 to 4 per 10,000 wearers per year and extended wear 5 to 10 times higher at 20 per 10,000 wearers per year. Rigid lens use is associated with a risk of 1 to 2 per 10,000 wearers per year and this appears to be lower than with daily use of hydrogel contact lenses. Differences between extended-wear and daily-wear hydrogel in early studies led to the hypothesis that increased oxygen availability with silicone hydrogel contact lenses would reduce the incidence of disease in overnight lens use.
26 Based on the most recent evidence, incidence rates appear not to be appreciably different with contemporary contact lens materials and wear modalities. However, there are two observations of note; first, that the incidence rate for microbial keratitis with
overnight use of silicone hydrogel lenses is no different to that of hydrogel contact lenses at 20 per 10,000 or 1 per 500 wearers per year. Second, that daily-disposable contact lenses are not associated with a lower risk for all microbial keratitis than daily-wear frequent-replacement contact lenses.
Analysis of risk factors for disease and their impact on risk provides information on factors relevant to both pathogenesis and prevention. Independent risk factors for microbial keratitis do show some variation between studies, probably due to differences in study design and analysis, differences in wear practices and prescribing modalities, and the power to detect differences. The timing of epidemiological studies relative to the introduction of new modalities may also be an important consideration.
27 There is evidence that early experience with new modalities may be associated with a higher risk of disease. It is conceivable that the small proportion of wearers who initially adopt new modalities and technologies show different behaviors and risks to those who adopt later, when such modalities have become more mainstream and are used by a greater proportion of wearers.
27Risk factors for contact lens-related microbial keratitis disease may be considered as either modifiable due to wear habits and lens hygiene or nonmodifiable related to duration of wear and wearer demographics. Modifiable risk factors conceivably may be impacted by education of wearers and improved compliance with recommendations for safe wear. Those consistently reported include extended or overnight wear
14,28; a longer continuous duration of extended wear
11,28,29; occasional overnight lens use
3; and poor lens hygiene,
11,28 including omission of or infrequent lens disinfection,
12,14 omitted or infrequent case cleaning,
3,30 omission of handwashing prior to handling lenses
4 and smoking.
3,11,31 Nonmodifiable risk factors include younger age; gender, with males having a higher risk than do females; and socioeconomic class.
4,13,14,31 Systemic risk factors include self-reported poor general health,
8 diabetes,
28 and thyroid disease.
8 Seasonality may also play a role. Most recently, studies in contemporary lens use have identified an increased exposure (number of days of wear per week) in daily wear, hypermetropia, obtaining lenses via the Internet or mail order, and the early period of lens wear as additional risk factors.
3,4 One surveillance study estimated a higher unadjusted incidence rate for
daily use of silicone hydrogel contact lenses compared with hydrogel contact lens use (
Table 53.3).
3 However, multivariable analyses have not identified lens material as an independent risk factor.
3,4 In a UK case-control study, a higher relative risk of microbial keratitis was also reported with daily-disposable contact lens use compared with planned-replacement soft contact lens use. This excess risk persisted in multivariable analysis, and brand-related differences were identified.
4 Using these approaches to assess independent risk factors, risk factors identified typically account for 70% to 80% of the (variance) total risk. It is conceivable that other behavior traits, not evaluated in these studies, including risk-taking propensity,
32 early adopter characteristics,
27 virulence characteristics of causative organisms, and individual differences in susceptibility also contribute to this unexplained risk of disease.
Somewhat disappointingly, new lens modalities have not reduced the overall incidence of microbial keratitis. An alternative approach may be to examine disease severity and to identify independent risk factors that may predispose to a more severe disease profile. Identifying and limiting factors with a large impact on disease load may reduce the likelihood of vision loss or a more severe outcome. Within any definition of microbial keratitis, severity of eligible cases may be described using a clinical grading system of presenting features of the lesion, including size and position of the lesion, anterior chamber response, and nature of symptoms.
8,33 Disease outcome can be described as the frequency of loss in best corrected spectacle acuity (usually two or more lines), cost of treatment, and disease duration.
18 Where a clinical grading system has been applied prospectively, there is generally good agreement between disease outcome measures and disease severity.
18Vision loss of two or more lines of best corrected spectacle acuity is reported in 12% to 14% of cases of contact lens-related microbial keratitis (
Table 53.1),
15,20,29 representing 6 per 100,000 wearers per year. The rate of vision loss varies with lens type with 3 to 4 per 10,000 wearers per year using extended-wear lenses affected. Vision loss is highest in the first 6 months of contact lens use. Vision loss is strongly associated with keratitis caused by an environmental organism (such as Gram-negative bacteria,
Nocardia sp., fungi or
Acanthamoeba), rather than with other gram-positive bacteria or a culture-negative result (11.4X, 95% CI, 4.2 to 30.9), with remoteness to health care (5.1x, 95% CI, 1.6 to 16.6).
20 Disease costs based on direct and indirect costs of disease vary widely (median AU$1,200, range AU$500 to >AU$10,000) and an increased disease cost was associated with the corneal culture result as described above and with a delay in receiving appropriate treatment.
20 Disease duration (median 7 days, interquartile range 11 days) was more prolonged in individuals over 28 years of age.
Although the incidence of microbial keratitis in daily-disposable contact lenses use is of a similar magnitude to that of other daily use lenses, the low rate of moderate and severe disease in daily-disposable contact lenses (0.5 [CI, 0.5 to 0.6] per 10,000 wearers per year) compared with frequent-replacement daily-wear contact lenses (1.1 [CI, 1.1 to 1.2] per 10,000 wearers per year), and low risk of vision loss with this modality is of interest.
3,4 This is perhaps consistent with these lenses being disposed of after each wear rather than being exposed to risk factors associated with hygiene procedures, particularly those involving lens disinfection and case hygiene. A preliminary study has described a greater proportion of culture-negative lesions in daily-disposable wearers compared with other daily-wear contact lens users.
34 This low rate of severe disease particularly caused by environmental organisms when daily-disposable lenses are worn on a strict dailydisposable-wear basis would suggest an advantage of reduced morbidity with this wear modality.
Of importance in the understanding of the disease is consideration of those factors which might be anticipated to be relevant in the development of microbial keratitis but which have not been proven to do so. Lens age or lens-replacement frequency has been linked with
Fusarium keratitis in one study,
35 but has not been consistently reported in contact lens-related microbial keratitis. This is at odds with certain laboratory studies showing an increased microbial adhesion to deposits on worn lenses (see below). There is some evidence of a survivor effect in wearers with a higher risk in the early period of wear, but no change in the risk with longer exposures.
3 Other compliance behaviors such as rubbing and rinsing lenses which would be hypothesized to reduce the risk of infection have not been proven to do so.
Based on the early studies showing increased risk with overnight wear, it would have been expected for lenses with improved oxygen availability and improved corneal epithelial physiology to have been associated with a lower risk of keratitis.
36 Lens material type, however, does not appear to be associated with infection risk,
36 or the risk of vision loss, although there is some evidence that the disease duration may be slightly shorter in culture-proven disease
20 with silicone hydrogel contact lenses. In a study of hospital presenting corneal infiltrates, less severe infiltrates were seen with extended-wear silicone hydrogel compared with hydrogel use.
31 Conversely, laboratory studies have demonstrated higher levels of bacterial adhesion with silicone hydrogel compared with hydrogel materials,
37 which might be expected to result in a higher risk of microbial keratitis. There has been no epidemiological evidence to support this hypothesis, and there appears to be limited impact of both material and microbial adhesion to lens deposits, implying that other factors are more important.
An analysis of independent risk factors for moderate and severe microbial keratitis among daily-wear contact lens users has specifically indicated the importance of poor storage-case hygiene and infrequent case replacement.
38 Other specific risk factors included occasional overnight wear (sleeping in lenses for up to one night once per fortnight or once per month), high socioeconomic class, smoking, and multipurpose solution type. When assessing the contribution of specific risk factors to disease load, attention to storage case cleaning and replacement could reduce the disease load by more than 60%.
38 The significance of storage case hygiene practice in limiting severe disease would suggest the importance of microbial contamination of the storage case in microbial keratitis. Despite storage case contamination remaining common among asymptomatic wearers,
39 there is evidence that the causative organism can be recovered from the storage case in microbial keratitis
40,41,42,43 and indeed in sterile keratitis.
44 A recent study examining nonculturable organisms from the storage case has demonstrated a link between the number of bacterial species recovered and increased severity of keratitis.
45 This study also suggested that specific gram-negative species such as
Stenotrophomonas, Delftia, and
Acinetobacter were overrepresented in storage cases from wearers with infiltrates and infections and may have a role in the pathogenesis of disease. Taken together, this evidence would imply that reducing microbial contamination of storage cases through eliminating the need for a case in daily-disposable contact lens use or using antimicrobial surface technologies, easily cleaned case designs, frequent case replacement, or simplified case hygiene practice would be effective approaches to limit disease severity.
Causative Organisms
Causative organisms in microbial keratitis vary with climate zone and with predisposing factor. Gram-positive bacteria are more frequently recovered in temperate climate regions
5,25,46 and gram-negative bacteria and fungi in tropical or subtropical climates.
7,47,48,49 Fungi account for 5% to 40% of culture-proven infections.
In noncontact lens-related keratitis, gram-positive bacteria predominate, specifically
Staphylococcus aureus, coagulase-negative staphylococci,
Streptococcus pneumoniae, and
viridans streptococci.5,25,46 However, the gram-negative species,
P. aeruginosa, is the most commonly recovered causative organism in contact lens-related disease, with recent studies from the USA,
50 UK,
51 Holland,
52 India,
53 and Australia
54 reporting the frequency of
Pseudomonas sp. in contact lens-related disease as 20%, 29%, 22%, 52%, and 35%, respectively. In Japan,
Pseudomonas sp. make up only 1% to 3% of isolates.
55,56 Other causative organisms include gram-negative bacilli (such as
Serratia marcescens, Kleibsiella pnemoniae). Gram-positive bacteria are isolated in approximately 25% of cases, mainly
Staphylococcus aureus, coagulase-negative staphylococci, and
Streptococcus spp., followed by fungi and
Acanthamoeba less commonly.
54,57,58,59,60 The strong association between
P. aeruginosa and contact lens-related keratitis is discussed below.
Severe microbial keratitis with vision loss in contact lens wearers is more likely to be caused by an environmental pathogen
20,61 and to occur in tropical regions in association with high daytime temperatures.
54 There is a further strong association between
Acanthamoeba and contact lens-related disease, with up to 95% of
Acanthamoeba keratitis cases attributed to contact lens wear.
61 Non-contact lens-related
Acanthamoeba keratitis is associated with a delayed diagnosis and more severe disease phenotype evidenced by a greater need for surgery and poorer visual outcome with 44% of cases with reduced vision.
Acanthamoeba Keratitis
The incidence of
Acanthamoeba keratitis in contact lens wearers is harder to estimate than for bacterial keratitis; numbers are small and the range in estimates is wide, particularly due to regional variations. Broadly rates in Europe, the US, and Australia are in the order of 1 to 5 per million daily-wear users per year.
61,62,63,64,65 Incidence rates in the UK vary between 0 and 85 per million wearers per year depending on regional variations in hard water distribution, where limescale in the domestic plumbing appears to favor colonization by
Acanthamoeba.66 Widespread use of bathroom water storage tanks to supply bathroom water in the UK may also promote proliferation of the organism.
67The epidemiology of
Acanthamoeba keratitis in contact lens wearers is somewhat different to bacterial keratitis.
Acanthamoeba disease is predominantly a disease of daily contact lens wear, and recent studies have further elucidated the impact of care solution, lens hygiene, and environmental factors. Historically,
Acanthamoeba keratitis was associated with the use of home-made saline in lens care and contact with contaminated water through, for example, hot tub use, well water use, or water sports, particularly swimming.
68,69,70 A large case series of
Acanthamoeba keratitis in the UK suggested that planned-replacement soft contact lens wearers were overrepresented compared with the penetrance of these lenses in the community.
71 A subsequent case-control study confirmed the impact of poor hygiene, specifically failure to disinfect lenses, had a 56x higher relative risk (95% confidence intervals [CI], 10 to 302) compared with disinfection using peroxide following lens removal.
72 Chlorine release systems, even used for disinfection following every removal, were associated with a 14x (95% CI, 7 to 232) higher risk than disinfection using peroxide. The excess risk with planned-replacement lenses was largely due to inadequate hygiene, but there may also have been some impact of the early adopter phenomenon, given the recent introduction of the modality during the period of study.
In 2006 to 2007, there was increased reporting internationally of
Acanthamoeba keratitis in daily-wear soft contact lens users.
73,74,75 Independent risk factors were identified in two case-control studies and are shown along with the historical risk factors for comparison in
Table 53.4. Both studies demonstrated an excess risk associated with Complete MoisturePlus (Advanced Medical Optics, Santa Ana, California).
76,77 However, not all cases were associated with this product and the disease has persisted subsequent to an international recall of this product, which would suggest the contribution of other risk factors, including water disinfection protocol.
78,79