(1)
Department of Ophthalmology, St. Thomas’ Hospital, London, UK
8.1 Introduction
8.2 Pathogenesis
8.3.1 Introduction
8.3.2 Grading
8.3.3 Risk of PVR
8.4 Surgery
8.4.1 Mild PVR
8.4.2 Moderate PVR
8.4.3 Severe PVR
8.4.4 Radial Retinotomy
8.4.5 Silicone Oil Injection
8.4.6 Applying Laser
8.4.7 ROSO Plus
8.4.8 Very Severe PVR
8.4.9 Choice of Endotamponade
8.4.10 Removal of Subretinal Bands
8.4.11 Adjunctive Therapies
8.4.12 Success Rates
8.4.13 Postoperative Complications
8.5 Summary
Abstract
Proliferative vitreoretinopathy (PVR) is a cellular proliferation producing ‘epiretinal membranes’ in rhegmatogenous retinal detachment. Primary RRDs which have been present for weeks or months are likely to develop PVR. At presentation, the rate of PVR in all patients with RRD varies depending on the ease of access to health care; where prompt surgery is available, PVR rates of 5 % are expected; however, where there is delay in receiving surgery, PVR rates are much higher, for example, 53 % in South America (Tseng et al. 2004) and 17.5 % in East Africa (Yorston et al. 2002). Failed surgery increases the risk of postoperative PVR which has been reported in 5 % of RRDs with U tears, 18 % with paravascular tears and 25 % of giant retinal tears (Bonnet et al. 1996).
Electronic supplementary material
The online version of this chapter (doi:10.1007/978-3-642-31872-6_8) contains supplementary material, which is available to authorized users.
8.1 Introduction
Proliferative vitreoretinopathy (PVR) is a cellular proliferation producing ‘epiretinal membranes’ in rhegmatogenous retinal detachment. Primary RRDs which have been present for weeks or months are likely to develop PVR. At presentation, the rate of PVR in all patients with RRD varies depending on the ease of access to health care; where prompt surgery is available, PVR rates of 5 % are expected; however, where there is delay in receiving surgery, PVR rates are much higher, for example, 53 % in South America (Tseng et al. 2004) and 17.5 % in East Africa (Yorston et al. 2002). Failed surgery increases the risk of postoperative PVR which has been reported in 5 % of RRDs with U tears, 18 % with paravascular tears and 25 % of giant retinal tears (Bonnet et al. 1996).
The onset of the proliferative response is variable with some eyes producing a response after short duration and others with chronic detachment remaining free from proliferation. Other conditions which cause retinal detachment are also at high risk of PVR such as severe ocular trauma and some inflammatory conditions, for example, acute retinal necrosis.
8.2 Pathogenesis
The retinal pigment epithelial cell appears to be the main source of the proliferation (Glaser et al. 1987). These cells are dispersed into the vitreous cavity through a retinal break. They change into myofibroblasts by the action of growth hormones released as a result of the retinal detachment. Types 1 and 3 collagen are laid down, and the cells contract in a similar fashion to a normal wound healing response.
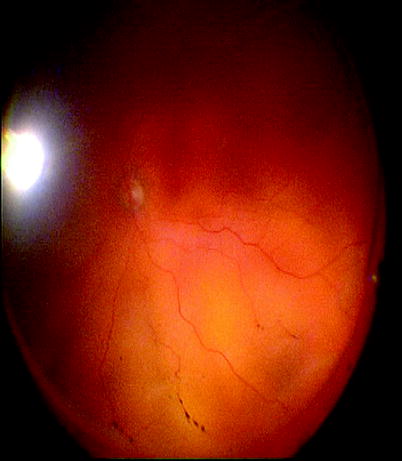

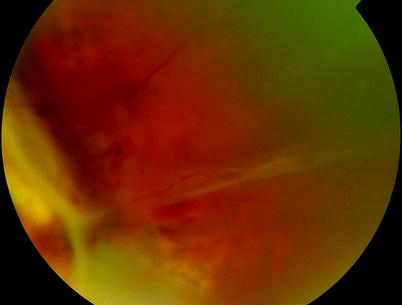
Fig. 8.1
Activation of RPE cells can be seen by clumping of the cells on retinal blood vessels
Note: Contraction of a wound on a planar surface usefully closes the wound by drawing the wound edges together. However, the same process is detrimental on the inside surface of a sphere such as the eye where the contraction tends to drag tissue (in this case, the neurosensory retina) into the centre of the sphere, exacerbating the retinal detachment process.
In addition, the retina becomes shortened and stiffened (between its anchor points, the optic disc and the ora serrata) by the PVR process so that the area of retina is not large enough to allow it to reattach to the inner surface of the posterior segment of the eye.
8.3 Clinical Features
8.3.1 Introduction
PVR is characterised by shortening, stiffening and folding of the retina progressing to a funnel retinal detachment which is immobile. Fibrosis can be seen on the inner and outer surfaces of the retina. PVR has the effect of reopening existing tears, creating new tears especially at the site of previous retinopexy but will also induce RD in eyes in which no open tears are found (Moisseiev and Glaser 1989). Patients who have preoperative PVR are at risk of developing furthermore severe postoperative PVR (Kon et al. 2000).
8.3.2 Grading
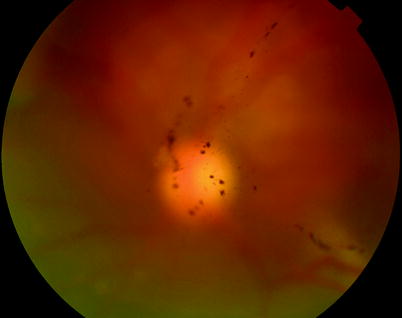
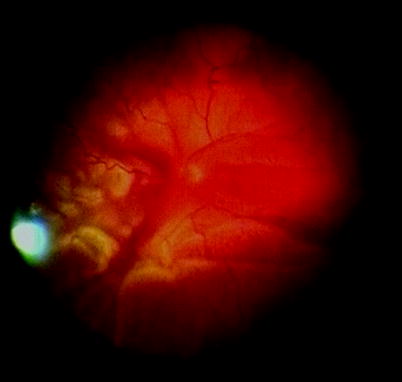
Fig. 8.2
Clusters of RPE cells can be seen in the vitreous of this patient with RRD indicating grade A PVR
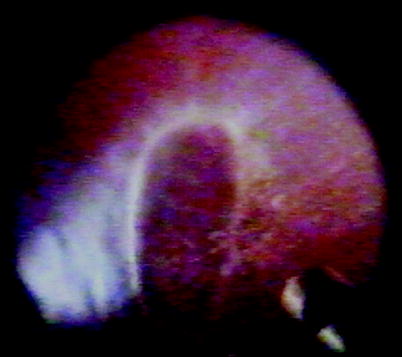
Fig. 8.3
Grade A PVR is diagnosed by stiffening of the vitreous clumps of pigmentation in the gel
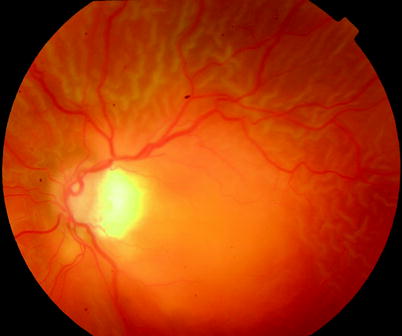
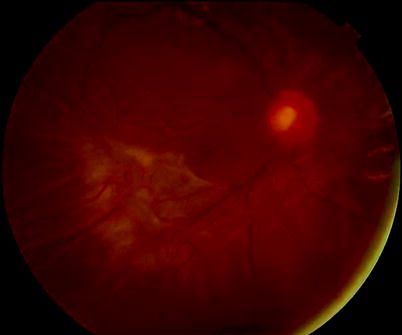
Fig. 8.4
B PVR, diffuse partial-thickness wrinkling is shown on the retina indicating grade B PVR in this patient
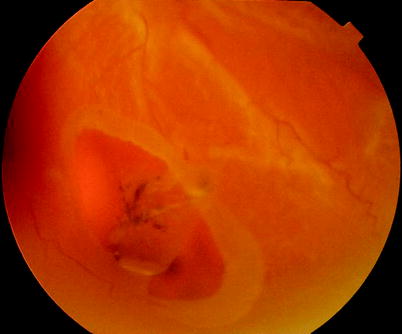
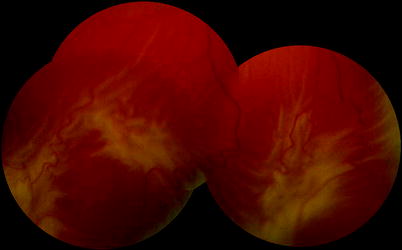
Fig. 8.5
A rolled edge to a retinal tear is an early sign of PVR
The response can be usefully graded (Machemer et al. 1991):
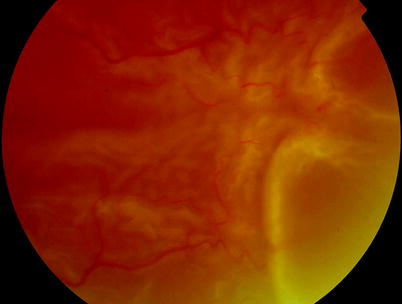
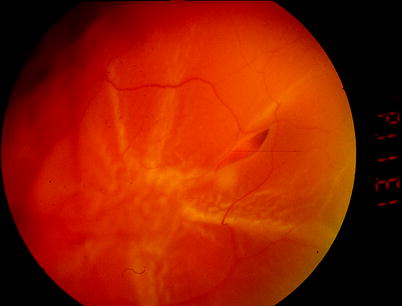

A.
Clumping of retinal pigment epithelial cells and stiffening of the vitreous
B.
Partial-thickness folding of the inner retina
C.
Full-thickness fixed folding of the retina, commencing as localised star folds and progressing to an open funnel formation and then a closed funnel in the final stages. C proliferative vitreoretinopathy is quantified by locating the area of folding either anterior (A) or posterior (P) to the equator and by indicating the number of clock hours of retina involved (1–12).

Fig. 8.6
This patient has CP2 PVR, but the membranes are very poorly formed and may be difficult to remove with forceps. The membranes can be stretched by insertion of heavy liquid to relieve the tension on the retina

Fig. 8.7
CP2: In this patient, a small focus of PVR is present over a previously failed non-drain retinal detachment repair

Fig. 8.8
This patient has PVR grade CP4 mostly on the nasal side which is early in its evolution and can present immature membranes which are friable and difficult to remove. Stretching out the retina with heavy liquids helps open out the retina

Fig. 8.9
CP4: In this patient, there is a focal PVR

Fig. 8.10
CP6: This patient shows full-thickness retinal folds over an extent of the retina of approximately 6 clock hours

Fig. 8.11
CA8: Anterior PVR is shown by shortening of the anterior retina with full-thickness retina folds

Fig. 8.12
CP12 PVR or a closed posterior funnel is present on this patient with RRD

Fig. 8.13
CP12: In this patient with severe PVR, the PVR is almost complete with a funnel retinal detachment

Fig. 8.14
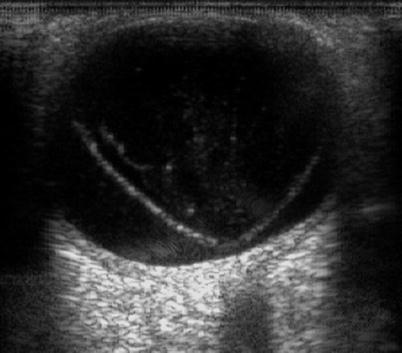
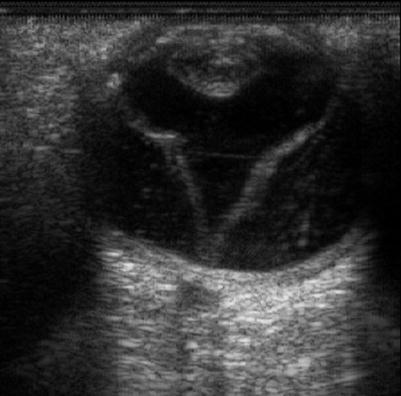
An open funnel RRD from PVR on ultrasound, CP12

Fig. 8.15
A closed ‘funnel’ RRD from PVR on ultrasound, CP12 and CA12
Table 8.1
Grading PVR
Proliferative vitreoretinopathy | ||
Grading | ||
A | Vitreous haze, pigment clumps, pigment clusters on inferior retina | |
B | Inner retinal wrinkling, retinal stiffness, rolled break edges, vitreous stiffness | |
CP1–12 | Full-thickness retinal folds or subretinal strands posterior to the equator (described as 1–12 clock hours of involvement) | |
CA1–12 | Full-thickness retinal folds or subretinal strands anterior to the equator (described as 1–12 clock hours of involvement), anterior displacement, condensed vitreous strands | |
Descriptive terms | ||
Type | Usual location | Features |
Focal | Posterior | Star folds in the retina |
Diffuse | Posterior | Confluent retinal folding |
Subretinal | Posterior | Fibrous strands, linear and purse string around the optic disc |
Fibrous sheets | ||
Circumferential | Anterior | Contraction of the retina inwards at the posterior edge of the vitreous base |
Anterior displacement | Anterior | Anterior traction on the retina at the vitreous base |
Ciliary body detachment and epiciliary membrane | ||
Iris retraction | ||
Occasionally, the proliferation is predominantly subretinal (Lewis et al. 1989) producing fibrous strands which elevate the retina like the guy ropes of a tent or even ‘purse string’ the retina around the optic disc.
8.3.3 Risk of PVR
Fig. 8.16
A subretinal band of fibrosis can ‘purse string’ the retina around the optic disc
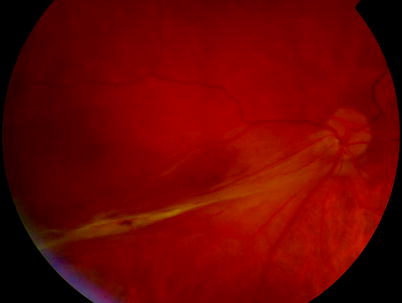
Fig. 8.18
See previous figure
PVR is particularly likely to occur if the patient has any of the following:
RRD for a prolonged period (Tseng et al. 2004).
Choroidal detachment (Cowley et al. 1989).
Previous surgery or cryotherapy which may aid the dispersion of the RPE cells (Glaser et al. 1993) and their stimulation by growth hormones.
Pre-existing PVR.
Aphakia.
Vitreous haemorrhage on PVR formation is uncertain, but there is an opinion in vitreous surgery that this may increase the risk of PVR (Duquesne et al. 1996).
Vascular abnormalities such as retinal telangiectasia or angiomas.
Uveitis.
Trauma.

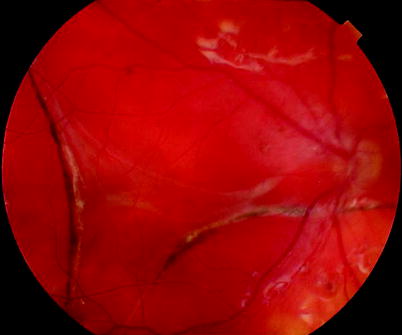
Fig. 8.19
Severe subretinal fibrosis postoperatively

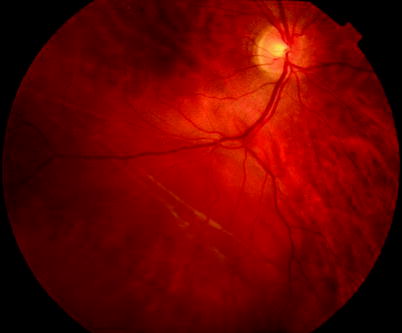
Fig. 8.20
Subretinal fibrosis in a chronic retinal detachment

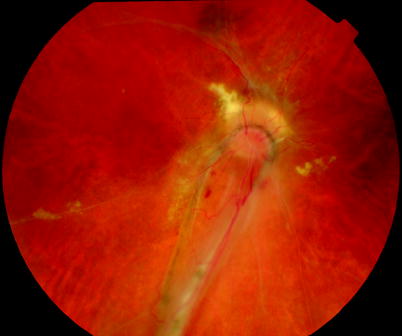
Fig. 8.21
This patient has a purse string subretinal band which is anchored in the periphery and is dragging retina downwards over the surface of the disc. Another instance of retina pulled over the disc surface is in ‘dragged disc’, for example, retinopathy of prematurity. The surgeon must be aware that retina overlies the disc and care must be taken when draining residual vitreous cavity fluid off the optic nerve head during PPV in these patients because the retina can be aspirated into the flute needle tip
8.4 Surgery
Surgery for PVR varies from performing conventional operations to retinectomy of the inferior retina.
8.4.1 Mild PVR
(Approximately C1–C2)
Table 8.2
Difficulty rating for surgery for mild PVR
Difficulty rating | Low |
Success rates | Moderate |
Complication rates | Low |
When to use in training | Middle |
Perform a non-drain procedure
or
PPV with additional surgical steps:
Insert heavy liquids.
Stain membranes with trypan blue.
Peel PVR membranes.
Use long-acting tamponade.
When dealing with PVR from C1 to C2, if the retinal breaks are far away from the PVR, for example, you have a superior horseshoe break with inferior CP2 PVR, often all that is required is to close the break, for example, a plombage can be placed on the tear. The normal physiological mechanisms will overcome the shortening to the retina from the PVR.
If the PVR is any more than this, however, you will have to perform a vitrectomy and attempt to peel any retinal membrane from the surface of the retina to allow the retina to open up and reattach itself to the back of the eye.
Note: Insert heavy liquids onto the posterior retina to stabilise the retina and act as countertraction as you pull on the membranes.
Use trypan blue dye to visualise the membranes and grasp with serrated forceps. Unfortunately, early membranes are often friable and difficult to remove. In this case, stretch and open out the contraction from the membranes using heavy liquid. The eye is primed to produce scar tissue, and the surgical intervention may induce further PVR formation; therefore, perfluoropropane gas or even silicone oil may be required.
Surgical Pearl of Wisdom
On the empirical use of silicone oil when no breaks are found:
Silicone oil that floats may be more effective at displacing aqueous from the upper fundus and heavy silicone oil that sinks may be better at tamponading inferior retinal breaks. But a slight under fill with either will lead to large area of retina unsupported. There is no such thing as a total tamponade. Successful re-attachment depends on identification of all retinal breaks, relief of traction and application of retinopexy. Use of silicone oil empirically (without identification of retinal breaks) invariably leads to recurrence of retinal detachment with oil in situ or upon its removal.
David Wong, Eye Institute, University of Hong Kong, Hong Kong, China
8.4.2 Moderate PVR
(Diffuse grade B, C3–C5)
Table 8.3
Difficulty rating for surgery for moderate PVR
Difficulty rating | Moderate |
Success rates | Moderate |
Complication rates | Medium |
When to use in training | Late |
PPV with additional surgical steps:
Insert heavy liquids.
Stain with trypan blue.
Peel PVR membranes.
Use silicone oil.

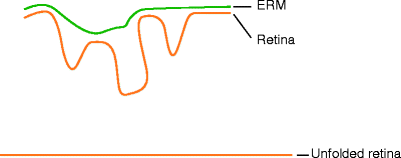
Fig. 8.22
Membrane (green) on the retina (orange) causes full-thickness folds; by removing the membrane, the retina can be unfolded, and, therefore, its length increased again, improving the chances of reattaching it to the curve to the eye
With levels of PVR C3–C5, peeling the membrane on its own may often be enough. Stabilise the retina with a heavy liquid, for example, perfluoro-n-octane (Han et al. 1994). This acts as a counterweight when pulling on the membranes; otherwise, the retina will be pulled with the membrane, and there is a risk of tearing. Use trypan blue stain to see the membrane more easily, and use either serrated forceps or forceps with a sharp tip to elevate the membrane. It is often easiest to engage the membrane in the centre of a star fold. Thereafter, deal with any breaks and fill the eye with silicone oil to allow reattachment of the retina. Leave the oil in until the proliferative process has had a chance to stop usually a few months. Superior holes can be treated with silicone oil. The management of inferior breaks is controversial. The placement of an inferior indention in a silicone eye may prevent adequate inferior silicone oil fill and may create a space behind the indent where shortening of the retina can easily form. I now do not use inferior buckles with moderate PVR and rely on the silicone oil fill on its own. Surprisingly, the inferior holes can flatten in this situation allowing silicone oil removal. This is despite the lack of inferior tamponade on the breaks from the oil (see sphere within a sphere in Appendix). If the inferior retina does not flatten, proceed to retinectomy.
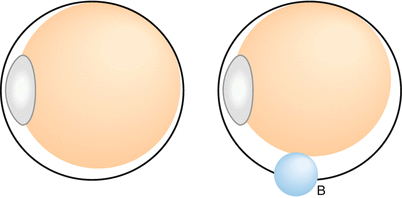

Fig. 8.23
Adding an inferior indent to try to close inferior breaks in a patient with moderate PVR often results in more shortened retina and elevated inferior breaks probably because the oil bubble cannot fill the inferior vitreous cavity at B. It is better to allow the oil to fill the eye after stretching or removing the PVR. Surprisingly, the breaks often close, most likely from reduced fluid currents inferiorly
Remove the silicone oil at 3–4 months, to avoid long-term oil-induced complications.
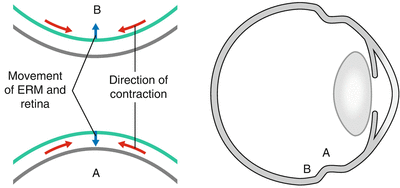
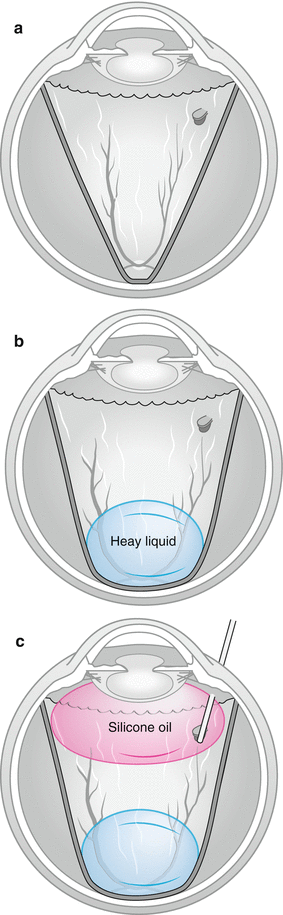

Fig. 8.24
Indentation under an area of contraction from PVR membrane (shown in green) here can be used to aid attachment of the retina at that site. However, the system only works if the PVR is on the apex of the indent point A. If the PVR is also present at point B, the indent will cause lifting of the retina

Fig. 8.25
In patients with funnel RRD from PVR (a) heavies are useful in patients with PVR to stabilise the posterior retina (b). Oil can be inserted on top of the heavies (c) whilst draining through the retinal break. As the oil bubble increases, the heavy liquid is compressed by the oil which pushes the peripheral retina flat. The SRF can drained from the meniscus between the oil and heavy liquids
8.4.3 Severe PVR
(C6–C8)
Table 8.4
Difficulty rating for surgery for severe PVR
Difficulty rating | High
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|