Chapter 71 Polypoidal Choroidal Vasculopathy
Introduction
PCV is more common in non-white populations (including blacks, Hispanics, and Asians).1 The incidence of PCV in Chinese and Japanese patients in exudative AMD has been reported to be much higher than in Caucasians.2–7 The true prevalence and consequences may be underestimated if indocyanine green angiography (ICGA) is not performed. The widespread availability of ICGA has improved the diagnosis of PCV.
Polypoidal choroidal vasculopathy was first described as polypoidal, subretinal, vascular lesions associated with serous and hemorrhagic detachments of the retinal pigment epithelium (RPE) in a series of patients (10/11 were women) by Yannuzzi et al. at the Annual Meeting of the American Academy of Ophthalmology in 1982.8 The entitity was initially called idiopathic polypoidal choroidal vasculopathy (IPCV). Kleiner et al. in 19849 described a peculiar hemorrhagic disorder of the macula, characterized by recurrent subretinal and subretinal pigment epithelium bleeding in middle-aged black women, which they termed posterior uveal bleeding syndrome (PUBS).10 Later, a study from the same group of authors showed an expanded clinical spectrum for PCV, affecting various ages, both genders, and several racial populations.11 For the remainder of the chapter, I will use the PCV nomenclature; the IPCV and PUBS terms are mentioned for historical benefit.
The past decade has witnessed dramatic improvements in the understanding of exudative maculopathy and recognition of the importance of PCV, especially in the Asia–Pacific regions. A PCV Roundtable meeting panel of experts suggest that PCV is defined angiographically as the presence of single or multiple focal nodular areas of hyperfluorescence arising from the choroidal circulation within the first 6 minutes after injection of indocyanine green, with or without an associated choroidal interconnecting vascular network. The presence of orange–red subretinal nodules with corresponding indocyanine green hyperfluorescence is pathognomonic of PCV.12 The implication of the definition is obvious but bears repetition. The definition of PCV relies on ICGA. Retina specialists who do not generally utilize ICGA tend not to diagnose PCV. Later in the chapter, there is a clinical definition of PCV as well.
Pathogenesis
PCV is regarded as a primary abnormality of the choroidal circulation, characterized by an inner choroidal vascular network of vessels ending in an aneurysmal bulge or outward projection, often visible as a reddish-orange, spheroid, polyp-like structure. PCV primarily involves the inner choroidal vasculature that is well differentiated from the middle and larger choroid vessels by histology.13
Histopathological features have been reported by Kuroiwa from surgically excised specimens from five patients with PCV, who had been diagnosed by ICGA. The results indicated that arteriosclerosis appears to be an important pathological feature in the choroidal vessels of PCV subjects.14 In another histopathologic report, by Nakashizuka et al., who examined specimens surgically extracted from five eyes of five PCV patients, the pathologic findings revealed little granulation tissue formation in any of the specimens; on the other hand, all the specimens exhibited a massive exudative change and leaking, all the vessels exhibited hyalinization, and choriocapillaris had disappeared, even in the cases in which RPE had been preserved.15 This group also demonstrated by immunohistochemistry that PCV is not the same as choroidal neovascularization (CNV). CD-34 is a marker of vascular endothelial expression and CD-34 staining revealed discontinuity in the vascular endothelium, smooth muscle actin (SMA) was negative in hyalinized vessels, and there was disruption and injury of smooth muscle cells causing dilation of vessels. VEGF antibody was negative in vascular endothelial cells. These histopathologic findings indicated that hyalinization of choroidal vessels, like arteriosclerosis, was characteristic of PCV.15
The genetic studies with CNV and PCV have been controversial in published papers.16,17 Recently, we investigated the relationship between CNV of AMD and PCV by performing a series of meta-analyses. We found that many genes have common associations with PCV and CNV. For example, the pooled odds ratio (OR) between SNP rs10490924 (TT : GG, within the ARMS2 gene, previously identified as LOC387715)18 and CNV is 4.23 (95% CI 3.535.06), while the pooled OR between this SNP and PCV is 5.13 (95%CI 3.40–7.75) (data unpublished). SNP rs9332739 within complement factor 3 (C3) gene also have a common association with CNV (GG : CC, pooled OR 2.12, 95%CI 1.81–2.47) and PCV (pooled OR 3.52, 95%CI 1.43–8.69). Moreover, a similar trend was found in complement factor H (CFH, rs1061170, CC : TT) and SERPING1 (C1 inhibitor, rs2511989 GG : AA). The elastin gene identified by Kondo et al. in 2008 was shown to disrupt the elastic area of the Bruch’s membrane. He found that a common elastin gene (ELN) variant was significantly associated with susceptibility to PCV.19
Furthermore, a series of meta-analyses was performed to investigate the pooled OR between the risk factors for CNV and PCV. We found that CNV and PCV have many common risk factors, such as smoking and diabetes. For example, the pooled OR between smoking and CNV is 1.78 (95%CI 1.52–2.09),20 while the pooled OR between smoking and PCV is 1.51 (95% CI 1.06–2.16, data unpublished). The pooled OR between diabetes and CNV is 1.66 (95% CI 1.05–2.63),20 while the pooled OR between diabetes and PCV is 1.94 (95% CI 1.29–2.92, data unpublished). However, the pooled OR between hypertension and CNV is 1.02 (95% CI 0.77–1.35),20 while the pooled OR between hypertension and PCV is 1.60 (95% CI 1.17–2.18, data unpublished). Therefore, hypertension is only associated with PCV. This is consistent with pathology findings: the PCV lesion has thickened and hyalinized vessel walls, which is similar to that seen in hypertension.21
Clinical features
Demographics
The prevalence of PCV in presumed neovascular AMD was reported as 7.8% in US,1 4.0% in Belgian,22 9.8% in Italian,23 8.2% in Greeek.24 23.0–54.7% in Japanese,25 22.3% in Chinese,26 and 24.6%27 in Korean populations. The prevalence varies with age.28 In summary, PCV is more prevalent in blacks, Japanese and other Asians than in whites, while the incidence of AMD is very high in whites, and low in blacks. The incidence of both diseases is high in Asians.
Although early reports suggested that PCV was a condition predominantly of middle-aged women,11 and that typically PCV presents one to two decades earlier than classical AMD, it is most commonly diagnosed in patients between the ages of 50 and 65.29 However, the age of the subjects was reported with a mean of 66.1 ± 9.6 years in a Chinese population.28 Caucasian patients usually present with PCV at an older age.30 It has subsequently been established that PCV occurs in both genders (and more commonly in Asian men than women).27 Although women are involved more often than men in some reports, more men still have manifestations of the disorder among Chinese patients.26,28,30,31 PCV is usually a bilateral disease. The majority of the patients with evidence of PCV in one eye eventually develop similar lesions in the fellow eye.
The natural course of PCV is variable: it may be relatively stable or there may be repeated bleeding and leakage with vision loss and chorioretinal atrophy, with or without fibrotic scarring. Reddish-orange nodules alone, or nodules and small subretinal hemorrhage and absence of hard exudates, may still allow a benign clinical course and stable vision.32,33
The association of PCV with other conditions is not certain. PCV with severe thrombocytopenia and massive hemorrhage has been reported,34 and PCV has also been associated with sickle cell disease and irradiation.35 Some experts believe it is advisable to screen PCV patients for hypertension and platelet count, but hypertension is common in older patients and is already monitored during medical visits, and the platelet data are not firmly established. The role of hereditary and environmental factors in its etiology is inconclusive and needs further study.
Clinical findings
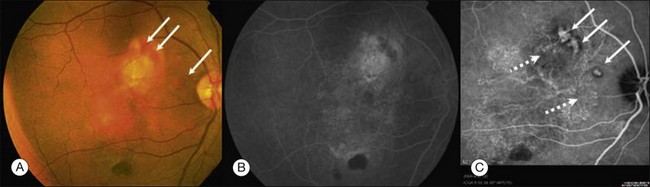
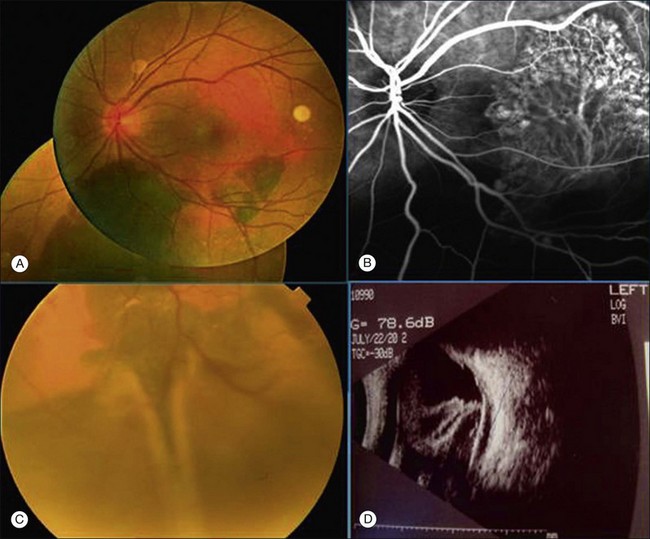
Clinically, PCV is characterized by protruding orange-red elevated lesions, often with nodular elevations of the RPE, that can be seen during routine fundus examination using ophthalmoscopy and contact lens slit-lamp biomicroscopy. The nodular elevations of the RPE are easily visible by optical coherence tomography (OCT). PCV is also characterized by polypoidal lesions only observed when using ICGA. The lesions appear as polyp-like or grape-like clusters (Fig. 71.1).11,15 The nodular lesions are associated with serous exudation and hemorrhage that may lead to detachment of the RPE and sometimes the neurosensory retina.32 Associated features are recurrent subretinal hemorrhage and vitreous hemorrhage (Fig. 71.2).
Polypoidal lesions are located mainly in the macular area, although this may be ascertainment bias since ICGA tends to look at that area. In one report, 69.5% polypoidal lesions were located in the macular area, 15% PCV lesions were located under the temporal retinal vascular arcade, and 4.5% PCV lesions were located peripapillary (within one disc diameter of the disc edge). PCV lesions were also located in the midperipheral area.28
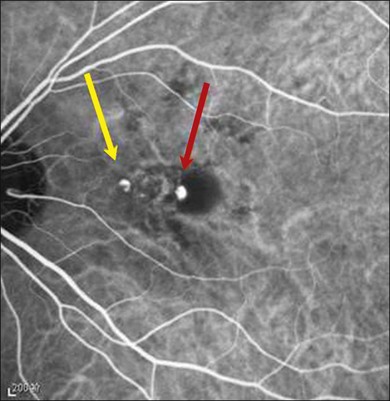
PCV lesions may be active or inactive (Fig. 71.3). PCV is considered as active if there is clinical, OCT or fluorescein angiography (FA) evidence of any one of the following: vision loss of 5 or more letters (ETDRS chart); subretinal fluid with or without intraretinal fluid; pigment epithelial detachment; subretinal hemorrhage; or fluorescein leakage (Box 71.1). There are currently no universally recognized criteria for defining disease activity in PCV; treatment should be initiated for active and symptomatic PCV and can be considered for active, asymptomatic PCV.12
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree