Chapter 103 Pneumatic Retinopexy
Introduction
For more than a half century, the operation most favored for primary retinal detachment had been scleral buckling (SB). Between 75% and 88% of cases attain permanent reattachment with one operation with this procedure.1–3 However, it frequently results in tissue trauma, complications,4 relatively high expense, and use of a hospital or surgicenter operating room. In recent decades, pars plana vitrectomy has been used with increasing frequency for primary retinal detachments, particularly in pseudophakic eyes. This technique also presents its list of potential complications, and also requires an operating room with similar high expense.
Pneumatic retinopexy (PR) was developed in an attempt to minimize these problems. This outpatient procedure for retinal reattachment consists of an intravitreal gas injection with transconjunctival cryopexy or laser photocoagulation, followed by appropriate head positioning.5 No incisions are required. PR is substantially less expensive than scleral buckling or vitrectomy and has become widely accepted as the treatment of choice for selected retinal detachments, with the vast majority of vitreoretinal surgeons employing this procedure.6,7
A multicenter, randomized, controlled clinical trial has demonstrated that the anatomic success rate of PR is comparable to SB, with less morbidity and with significantly better visual results than with SB.8 Cataract surgery was required more often following SB than following PR.9 PR should be considered in patients who do not have detached inferior breaks, extensive retinal breaks, or significant proliferative vitreoretinopathy.
History
Ohm10 performed the first intravitreal air injection for retinal detachment in 1911. In 1938, Rosengren11 reported the use of intravitreal air with drainage of subretinal fluid in a series of 256 retinal detachments. In 1973, Norton12 reported intravitreal sulfur hexafluoride (SF6) injection used with SB or vitrectomy for various surgical problems, such as giant breaks, large posterior breaks, and fishmouthing. Blodi and Folk13 treated detachments due to a macular hole using intravitreal gas, and retinal detachments treated with “repeated insufflations of expansive gas” were described by Dominguez et al.14,15 Hilton and Grizzard5 introduced the term “pneumatic retinopexy” at the 1985 meeting of the American Academy of Ophthalmology, a procedure using only transconjunctival cryotherapy and gas injection without conjunctival incision.
Unlike SB or vitrectomy, PR affords no permanent relief of vitreoretinal traction. The fact that detachments can be cured without permanently relieving traction, demonstrated decades before by Ohm10 and by Rosengren,11 was reconfirmed in 1979 by Lincoff et al.,16 who introduced the use of a balloon to achieve temporary scleral buckling. After cryopexy to the tear, they inserted a deflated balloon directly over the tear through a conjunctival and Tenon’s capsule incision, inflated the balloon, and left it in place for a week. The efficacy of this procedure was demonstrated but it never caught on, and the orbital balloon is not commercially available worldwide. However, the principle of repairing a retinal detachment without permanently relieving vitreoretinal traction, well proven by Lincoff’s technique, combined with the demonstrated safety of intravitreal gas, formed the basis for PR.
In 1989, Tornambe and Hilton8 co-directed a multicenter, randomized, controlled clinical trial comparing PR with SB. Tornambe17 reviewed over 200 studies on PR, including statistical reports on over 1300 cases. The single-operation success rate in these combined series was 80%, increasing to 98% after reoperations18 – results which have been subsequently replicated.19 Chan et al.20 reviewed all published reports on PR cases from 1986 to 2007, totalling 4138 eyes. Single-operation success was 74.4%, and success with one or more operations was 96.1%.
Basic principles
Intraocular gases
Sulfur hexafluoride (SF6) and perfluoropropane (C3F8) are the gases most frequently used with PR. Success also has been reported with sterile room air.21 In 1993, the United States Food and Drug Administration approved certain SF6 and C3F8 products for use in PR.
The value of the intraocular bubble is based on three features: buoyancy, surface tension, and isolation of retinal tears from intraocular currents.22,23 Buoyancy applies upward pressure on the detached retina. The surface tension of the bubble closes the retinal break and prevents the bubble from passing into the subretinal space. With the break closed, the retinal pigment epithelial pump removes the subretinal fluid.
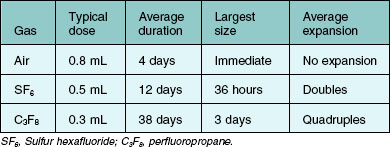
Because of their low solubility in water, SF6 and C3F8 tend to diffuse from the eye very slowly. However, the nitrogen and oxygen that are in solution in the surrounding tissues of the eye are much more soluble and pass relatively quickly into the gas bubble, following the law of partial pressures. The net result is the initial expansion of a bubble of pure SF6 or C3F8 within the vitreous, followed by gradual resorption. Characteristics of gas expansion and resorption for SF6, C3F8, and air are listed in Table 103.1.
SF6 and C3F8 are chemically inert, colorless, odorless, and nontoxic.24 SF6 has been studied extensively in experimental animals and has been found to be nontoxic as judged by electrophysiologic testing and electron microscopy.25 One study of rabbits concluded that “eyes only partially filled with C3F8 showed no permanent damage, with a total recovery of the cortical matrix, gel, and liquid vitreous.”26 Early concerns27 regarding PVR from intravitreal gas injection have not been substantiated. A 0.22 µm Millipore filter is sufficient to render gas sterile.25
Lincoff et al.28 noted that gas bubble contact with the crystalline lens can produce cataract after several days, but this is avoidable by appropriate positioning. Mougharbel et al.29 have demonstrated with Scheimpflug photography that PR does not cause cataract.
Retina–gas interface
Larger areas of tamponade require disproportionate increases in bubble volume. A 0.3 mL gas bubble in humans covers more than 45° of arc of the retina, but it takes approximately a 1.2 mL bubble to cover 80–90°.5 Because surface tension causes the gas to take on a relatively spherical contour, particularly with smaller bubbles, the extent of retina–gas contact is significantly less than that published from studies of model eyes.30 To cover the same arc of the retina, a highly myopic eye requires a larger volume of gas than an emmetropic eye. These factors are considered in deciding how much gas to inject.
Case selection
The multicenter clinical trial excluded cases with the following characteristics:8
1. Breaks larger than 1 clock-hour or multiple breaks extending over more than 1 clock-hour of the retina.
2. Breaks in the inferior 4 clock-hours of the retina.
3. Presence of PVR grade C or D (Retina Society Terminology Committee, 1983).31
4. Physical disability or mental incompetence precluding maintenance of the required positioning.
5. Severe or uncontrolled glaucoma.
A review of 1000 consecutive detachments5 revealed that 41% met these limited criteria. Subsequent experience has demonstrated that selected cases that do not strictly meet these criteria can also be successfully treated with PR.32,33 In our hands, about half of all primary retinal detachments are treated with PR.
Extent of breaks
Breaks spanning more than 1 clock-hour can be treated with PR. Single or multiple tears spanning up to 3 clock-hours pose no particular problem. Detached tears 6 clock-hours apart are difficult to fix with PR, although alternating positioning has been used successfully. Even detachments with giant retinal tears have been cured with PR.34 However, scleral buckling or vitrectomy is usually preferred for detachments with widely separated or giant tears.
Inferior breaks
Most cases involving breaks in the inferior 4 clock-hours of the eye have been difficult to treat with PR, despite various attempts.33 Even for flexible patients, it can be difficult to tilt the head below the horizontal plane for extended periods. Friberg and Eller35 first reported success with inferior breaks in eight of eight patients using dependent positioning. In 2003, Chang et al.36 reported single operation success in 9 of 11 patients with inferior breaks using PR with inverted positioning for only eight hours. Hilton et al.37 treated retinal detachment with inferior breaks by augmenting pneumatic retinopexy with in-office partial vitrectomy, allowing injection of a large enough gas bubble to close the breaks with side positioning. In 2011, Hwang et al.38 reported 10 out of 13 single-operation success with a 10–30 cm downward head tilt.
Lattice degeneration
In several studies, the presence of lattice degeneration did not adversely affect single operation success with PR,17,39 but in other studies, extensive lattice degeneration tended to decrease the chance of success with PR. It does not appear that mild to moderate lattice should be considered a contraindication.
Aphakia and pseudophakia
Retinal detachments in aphakic/pseudophakic eyes have a poorer prognosis than phakic eyes, no matter what surgery is performed.9 In some series, aphakic eyes did poorly with PR,40 especially if the posterior capsule was open,41 but in other reports this was not the case.8,9 Aphakic and pseudophakic eyes are prone to multiple, tiny, far-peripheral breaks and require an especially careful preoperative examination. In pseudophakic eyes, the view of the peripheral retina can be quite limited if much peripheral capsular opacity is present. PR should probably not be performed in these cases unless retinal detachment is rather limited. In our opinion, if the peripheral retina can be adequately examined, aphakia or pseudophakia is not a contraindication to PR.
Posterior vitreous detachment
Phakic eyes with complete posterior vitreous detachment tend to have a higher success rate with PR than those with partial vitreous detachment.42
Cases where pneumatic retinopexy presents a particular advantage
Compared with SB, PR is especially advantageous in the management of the following six situations:
1. Macular breaks and other posterior retinal breaks. Posterior retinal breaks are difficult to treat with SB, so PR is the procedure of choice in many of these cases, especially in phakic eyes. It also has been reported as an effective option in the treatment of optic pits with macular detachment.43,44
2. Redetachment or persistent detachment after scleral buckling. When subretinal fluid accumulates or persists because of a superior break after SB, PR may be much easier to perform than buckle revision.45 This is especially effective if the break is located on or anterior to the buckle.46
3. Isolated tears under the superior rectus. Placing a segmental buckle under a vertically acting muscle runs the risk of iatrogenic diplopia; this is eliminated with PR.
4. Filtering blebs. If a functioning filtering bleb is present, or if a filtering procedure may be necessary in the future, PR should be considered.
5. Impending macular detachment. Because PR can be performed promptly in the office without the delays required to prepare a patient for the operating room, and because the gas bubble can be used proactively to move the fluid away from the macula, a macula at imminent risk of detachment may be best served by PR.
6. Bullous detachment. When retinal detachment is highly bullous, retinal tears can be difficult to localize and treat with SB, a problem which is avoided by two-session PR.
Surgical technique
One-session versus two-session procedure
See Box 103.1 for indications for the one-session and two-session procedures.
Box 103.1
Indications for one-session versus two-session procedure
Cryopexy versus laser
If a laser indirect ophthalmoscope (LIO) is not available, tears in the far periphery may not be treatable with laser delivered by slit lamp, necessitating cryopexy. The LIO is ideal for PR because it allows treatment of the far periphery and facilitates maneuvering of the gas bubble out of the way.35 Although most tears can be treated with laser through a slit-lamp delivery system by tilting the patient’s head as necessary to move the bubble away from the tear, this is difficult with tears from 11 to 1 o’clock. It is often possible to apply laser treatment through a large gas bubble, but it can be difficult with the smaller bubbles utilized with PR because of optical factors. Application of laser through gas causes concentration of heat due to the insulating effect of gas, and excessively hot burns may result.
Chorioretinal adhesion may be quicker and firmer using laser rather than cryopexy.47,48 In addition, some authors believe that cryopexy may be associated with a higher incidence of proliferative vitreoretinopathy and other complications,49,50 although others could find no such association.51 Laser also has the advantage of less morbidity compared with cryopexy, especially if multiple or large breaks are present.
Transscleral diode laser photocoagulation offers some potential advantages over both cryopexy and transpupillary laser for retinopexy treatment.52 Like transpupillary laser photocoagulation, one can see exactly where the treatment has been and will be applied. As with cryopexy, it allows treatment in detached retina. Treatment can even be applied through a pre-existing buckle.
Amount and type of gas to inject
We believe that it is optimal for the gas bubble to cover the break(s) for 5 days and then disappear as soon as possible. However, successful reattachment has been reported after as little as 6–8 hours of tamponade, as with inverted positioning for inferior tears.36 The longevity of an air bubble is probably sufficient for most cases, but occasionally the chorioretinal adhesion may not be sufficiently mature when the air has resorbed. The use of air also forfeits the advantage of postinjection bubble expansion within the eye, necessitating injection of a larger volume, but it offers the advantages of decreased expense and universal availability.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree