CHAPTER 189 Pediatric Facial Fractures
After the first month of life, trauma is the number one cause of pediatric deaths and a major contributor to long-term morbidity.1,2 Nearly 15,000 children die and 100,000 others are disabled worldwide each year from trauma resulting from motor vehicle crashes, sports injuries, falls, altercations, and abuse.2 More than $15 billion is spent annually in the United States for the treatment of childhood injuries.1,3 Despite this epidemic of injuries, facial fractures in children remain uncommon.
Epidemiology
Pediatric maxillofacial injuries account for approximately 5% of all facial fractures, although the incidence is reported as being between 1.5% and 15%.4–18 Children younger than 5 years of age account for only 0.9% to 1.2%, whereas those 6 to 14 years of age account for 4% to 6% of all cases of maxillofacial trauma.19,20 Bamjee and colleagues19 found that 7% of pediatric traumatic injuries occur in children younger than 6 years, whereas 70% befall those between 13 and 18 years of age, indicating the increased vulnerability of the maturing face as well as the greater risk-taking behavior of a growing child.
In children, males are more prone to maxillofacial fractures than females, with an overall ratio of 1.5 : 1 to 2.3 : 1.18,21–24 In children younger than 8 years, no difference between genders has been noted.25 With approaching adolescence and greater involvement in sports and violence, the male-to-female ratio approaches 4.5 : 1.25 In South Africa, male children are three times more likely to be involved in violence resulting in facial fractures; however, other types of injuries resulting in facial fractures are gender-neutral.19 Nasal fractures are the most common fractures of the facial bones in children. Because most nasal fractures are treated in an office setting, precise statistics on their frequency are difficult to determine. Limited studies have reported the incidence to be between 45% and 60% of all facial fractures.26,27
Mandible fractures are the most common facial fractures in children requiring hospitalization. Depending on whether nasal fractures are included, mandible fractures account for between 5% and 50% of reported pediatric facial fractures.1,16,21 The condyle is the most common site, accounting for 40% to 70% of mandibular fractures.11,18,22,28 Mandibular body (0 to 20% of fractures, depending on the series), angle (3% to 17%), ramus (3% to 10%), and symphyseal fractures (2% to 30%) occur less frequently in young children.9,12–14,22,25–27,29–37 As adolescence approaches, the incidence of fractures of the arch and body of the mandible reflects a more adult pattern.38 Dentoalveolar fractures, like nasal fractures, often are treated in an office setting; hence, it is difficult to gauge their true frequency, which probably is higher than the 14% incidence reported in one large study.29
Complex facial fractures are rare in children. The literature shows a wide range of anatomic distribution. Reported incidence rates for orbital fractures range from 0.2% to 30%; for zygomatic complex fractures, 2.7% to 13.5%; and for Le Fort type fractures, 0 to 16%.9,17,26,27,39,40 The sporadic distribution of midface injuries, especially among preadolescent children, highlights their uniqueness and emphasizes how limited a particular surgeon’s experience can be with these complex injuries.
Associated injuries are a common feature of childhood maxillofacial trauma. In the pediatric age group, intracranial, intra-abdominal, and orthopedic injuries are associated with more severe types of facial fractures in 30% to 60% of the cases.8,9,38,41 Temporal bone and soft tissue injuries also are commonly associated with facial fractures.9 In young children, up to 86% of orbital roof fractures are associated with frontal skull fractures and concomitant intracranial injuries.7,42 In 30% of orbital fractures, there are other associated facial injuries.43 These statistics emphasize the importance of a complete initial assessment in children with facial trauma and highlight the dilemma of the timing of repair when medical instability conflicts with the limited period during which optimal reconstruction is best accomplished.
Etiology
Motor vehicle collisions constitute the main cause of serious pediatric facial trauma.4–15,17,18 Fortunately, less than 1% of traffic accidents result in pediatric facial bone fractures.44 The risk of injury is 1.6 times higher for children who are inappropriately restrained in the vehicle, and children seated in the front seat have nearly twice the risk of fracture as those sitting in the rear seat.44 Air bags have been shown to decrease the mortality rate in the adult population; however, children younger than the age of 10 years have a 34% increased risk of dying in frontal crashes with air bag deployment.45 The current recommendations for protection during motor vehicle travel by the American Academy of Pediatrics and the National Highway Traffic Safety Administration are use of child restraints for children weighing less than 40 lb, belt-positioning booster seats for children from 40 to 80 lb and up to 57 inches in height, and lap-shoulder belts for children weighing more than 80 lb and more than 57 inches tall, and placement of children younger than 13 years in the rear seat.46–48
Although a majority of serious pediatric facial fractures are incurred in motor vehicle collisions, childhood play is the most common cause of facial trauma (15% to 30%) when all forms of fractures are considered, including orbital, nasal, and dentoalveolar fractures.49 Bicycle accidents remain an important cause of injury to children, and although wearing helmets has greatly reduced midfacial and intracranial injuries, the current designs of helmets do not protect against mandibular and dentoalveolar injury.50–53 Other causes include falls (20% to 30%), aggravated assault (8% to 30%), and sports injuries (2% to 30%).* An uncommon cause of facial fracture is trauma from dog bites.56,57 Isolated orbital fractures most commonly are seen in sports, with violence recognized as a significant risk among those 12 years and older.43,58 Child abuse deserves special mention. Facial fractures, in particular, of the mandible, can be the result of child abuse.8,14,33,59 It is important to consider the possibility of child abuse, especially in cases of facial injury among children younger than 5 years, because this is the age group at highest risk for battering.
Understanding the difference in the patterns of fractures between children and adults requires familiarity with the development of the facial skeleton. At birth, the craniofacial ratio is 8 : 1, with the face located in a recessed position relative to a large skull. The cranium and forehead effectively shield the smaller lower and middle thirds of the face from injury. By the age of 2 years, the cranium has achieved 80% of its mature size. Facial growth also is rapid during this period, but it is only after the second year that facial growth outpaces cranial growth. Brain and ocular growth are near completion by age 7; however, facial growth continues into the second decade of life.60,61 In adults, the craniofacial ratio is 2 : 1.61,62 The consequence of the higher craniofacial ratio is that the cranium, especially the prominent forehead overhanging the face, absorbs the major portion of the impacting force. This accounts for the higher proportion of pediatric skull fractures relative to facial fractures and the rarity of serious midface fractures in young children, in whom the force necessary to cause major maxillary disruption often results in brain injury and death.14,35
Another resilient feature of young children is the makeup of the face with thick soft tissue, lack of sinus pneumatization, and elasticity of the bones. The greater proportion of cancellous to cortical bone explains the higher incidence of “greenstick” fractures in children.63 The presence of unerupted and mixed dentition in the maxilla and mandible increases the resistance to fracture, although fractures can occur through developing tooth crypts. As the child enters adolescence and adulthood, the concomitant pneumatization of the paranasal sinuses, maturation of the dentition, hardening of bone, and thinning of the facial soft tissues, synchronized with higher-energy injuries associated with increased risk-taking behavior, all lead to a higher risk of fracture.29,60,64–66
Emergency Management
Orotracheal intubation is necessary in patients with concomitant cranial trauma, severe bleeding associated with midfacial fractures, or oropharyngeal obstruction and posterior retrusion of the mandible. Orotracheal intubation ideally is accomplished after the cervical spine is secured. Optimally, when oropharyngeal or laryngeal injuries are present, intubation is performed in the operating room with rigid instrumentation. “Crash” tracheotomy and particularly cricothyrotomy in the emergency department are ideally avoided in favor of orotracheal intubation. Once the airway is secured, elective controlled tracheotomy can be performed in the operating room when deemed necessary by the extent of injury. In one series of 73 patients with mandibular fractures, only one patient required airway control for obstruction; a tracheotomy was not necessary.14 However, a controlled tracheotomy is a safe option in patients with panfacial fractures and associated major systemic trauma.38
Facial Growth and Trauma
Craniofacial bone growth is a complex process that is incompletely understood, and even less is known about the effects of trauma on this process.67 See Chapter 184 for a detailed review of facial growth.
It is clear that facial growth is a complex process. What is unclear is the mechanism of control and coordination responsible for overall growth.68 Practical concepts for the repair of facial fractures in children are based on the known effects of trauma in the growing face, as well as on an understanding of the operational mechanics of facial development (see Chapter 185, Craniofacial Surgery for Congenital and Acquired Deformities). The mandible appears to be generally resistant to abnormalities of growth as a result of trauma, unless its function is altered as a result of injury in or near the TMJ. Early return of mandibular mobility is desirable to facilitate the restorative bony changes that result from normal function.
Rigid Fixation
Controversy still exists over the use of rigid fixation in the developing face. See Chapter 184 for detailed discussion of rigid fixation in children.
Nevertheless, with the recognition that each reconstructive technique has its strengths and its weaknesses, the use of rigid fixation is highly desirable in complex fractures in which the original features are difficult to restore. The alternative of no correction is unacceptable, and the use of interfragmentary wires requires tedious surgery and can lead to reconstruction of uncertain stability. As Rahn69 has written, “Open reduction of a fracture may sometimes appear to be an aggressive approach. It can be justified when late reconstruction and secondary surgery is avoided. Thus an approach that appears initially to be more aggressive may be more conservative.”
Permanent versus Resorbable Plating Systems
As noted, controversy is ongoing regarding the use of permanent plates and screws in the developing face. Although these techniques are excellent for fixation, leaving a metallic foreign body in a growing child has several drawbacks: Transcranial migration of metallic plates is a well-recognized problem.70–72 Growth disturbances in both animal and human studies have been shown with use of permanent rigid fixation.41,73–75 Metal hypersensitivity, potential for bone atrophy, interference with CT and MRI, allergy, and palpability all are potential problems with use of metallic plates. Accordingly, many surgeons routinely remove plates once the fracture line is well healed, especially in children.76–80 However, others suggest that watchful waiting is appropriate and have found the need to remove plates for the aforementioned reasons in approxiamtely 8% of the pediatric population.81 Of interest, in a rabbit study, Connelly and Smith82 found that the removal of rigid plate fixation had a more deleterious effect on growth than if the rigid fixation was left in place. An alternative solution to this quandary is the use of absorbable plates and screws, the safety and efficacy of which have been shown by extensive animal research and subsequent use in craniofacial surgery and facial trauma reconstruction.72,76,83–95 A study using resorbable copolymer in infant minipigs looked for local reaction as well as the complication of passive intraosseous translocation of plates. The investigators found the tissue reaction to be minimal and demonstrated that although the plates were embedded in the growing bone, this did not hinder degradation of the plates or screws.95 Bioabsorbable plates are reasonable in cost and clinically safe. With experience in their application, resorbable systems are comparable in efficiency to metal plating systems.
The most commonly used craniofacial bioabsorbable materials are high-molecular-weight polyalphahydroxy acids: polylactic acid (PLA), polyglycolic acid (PGA), and polydioxanone (i.e., the suture material PDS) and their copolymers. Lactic acid has two enantiomeric forms: L-lactic acid (PLLA) and D-lactic acid (PDLLA). PLA and PGA are metabolized into carbon dioxide and water and are eliminated by respiration; the degradation products of PDS are excreted primarily in the urine.93 The absorption occurs initially by hydrolysis and is completed by phagocytosis.96 The physical properties of the polymers are outlined in Table 189-1.90 Pure polymers have been shown to have disadvantages in strength, time to degradation, or local tissue reaction.86,93,96 PGA has the best initial strength properties, equal to those of stainless steel.97 PGA’s mechanical properties are lost in 6 weeks, and resorption is complete within 1 year.49,57 However, the use of pure PGA has resulted in adverse reactions in up to 60% of cases, including swelling, draining fistulas, and osteolysis.98,99 In vivo sheep studies have shown that PLLA takes 5 years to resorb.94 Reports on pure PLLA have demonstrated late tissue responses up to 4 years after implantation.100
Table 189-1 Physical Properties of the Common Monomeric Polymers Currently Used in Fabricating Bioresorbable Plates and Screws
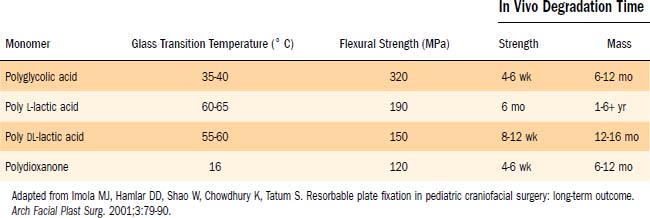
Current resorbable plating systems take advantage of copolymers of PLA and PGA, which allow for variable duration of adequate mechanical strength and biodegradation time. LactoSorb was the first absorbable system approved by the U.S. Food and Drug Administration (FDA) in 1996, and at present there are five commercially available resorbable plating systems with different properties based on their individual polymer chemistry, as outlined in Table 189-2.90 Because each plating manufacturer uses slightly different mixtures of copolymers, plate strength, absorption, and ease of handling will differ between the systems. Ideally, the plate used will retain full strength for 4 to 6 weeks, the time it takes for facial fractures to ossify.101 The absorbable plates currently on the market maintain 60% to 90% of their strength through the 3-month mark.96
Table 189-2 Commercially Available Craniomaxillofacial Resorbable Plating Systems and Their Properties
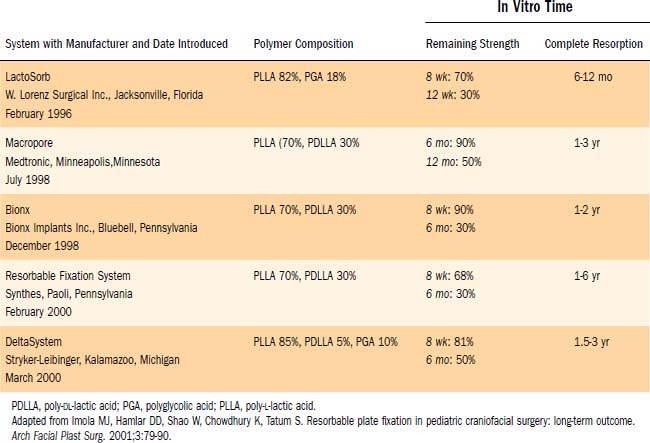
Complications with resorbable systems are comparable with results with metal plates, with the exception of the potential for long-term translocation with metal implants.20,90,93,102–104 The most common adverse reactions are transient peri-implant edema and a high rate of temporary visibility or palpability of hardware, both of which resolve with time. Foreign body reaction in the surrounding tissue typically is minimal.96 To date, the long-term studies on absorbable plating systems have exclusively used LactoSorb, which has a faster resorption time than some of the more recently released systems. Studies have shown that the available resorbable plating systems (1.5- and 2.0-mm screw diameters) provide flexural and tensile strength comparable with that of the microplate titanium systems (1.0- to 1.3-mm-diameter screws).105 Definitive long-term studies in facial trauma have yet to conclude whether resorbable plates cause problems with growth restriction.
At this time, absorbable plates are not recommended for all types of pediatric facial fractures. As indicated by preliminary data on sagittal split osteotomy plated with absorbable systems, excellent results were obtained without the use of maxillomandibular fixation (MMF) in a 15-month follow-up study.93 However, the use of absorbable plates in the mandible and “load-bearing” bone is still investigational in children, and long-term results are limited.90,92 At this time, the indications for the use of absorbable systems in pediatric trauma are in non–load-bearing regions in the upper and middle thirds of the craniofacial skeleton.
Maxillomandibular Fixation
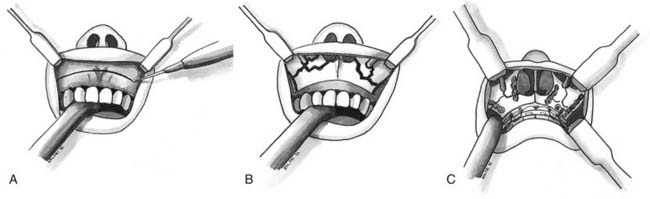
In decisions regarding techniques to achieve MMF, primary considerations include the age and development of the teeth. In children who lack teeth or in whom the poor retentive shape of the deciduous teeth makes the use of arch bars and interdental wiring impractical, alternative methods of MMF need to be considered. Fortunately, 2 to 3 weeks of mandibular immobilization in children younger than 12 years is adequate.38,106
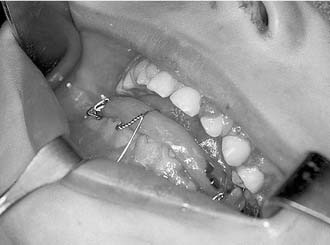
One approach is the use of an overlay acrylic mandibular splint held in place by circum-mandibular and transnasal wires. The splint’s occlusal surface is placed in normocentric relation to the maxilla, and immobilization is accomplished by suspending a wire from the piriform aperture and tightening it around the mandibular midline wire that is holding the splint to the mandible (Fig. 189-1).
An alternative technique that adds more interocclusal stability is the placement of resorbable screws in the symphysis at the inferior border as well as that of the zygoma. The monofilament suture is tied between the two screws, thus avoiding the need for a circum-mandibular suture. The use of this MMF method in teenagers and adults is not advised, owing to the higher forces generated by the masticatory muscles.107
In children between the ages of 2 and 5 years, the deciduous incisors have firm roots, and if the deciduous molars have formed, they can be used for cap splints or arch bars. In general after age 10, the development of permanent teeth provides for safe anchors.29 However, children develop at different rates, and the strength of the teeth should be carefully examined before the placement of any type of MMF instrumentation.
Surgical Approaches
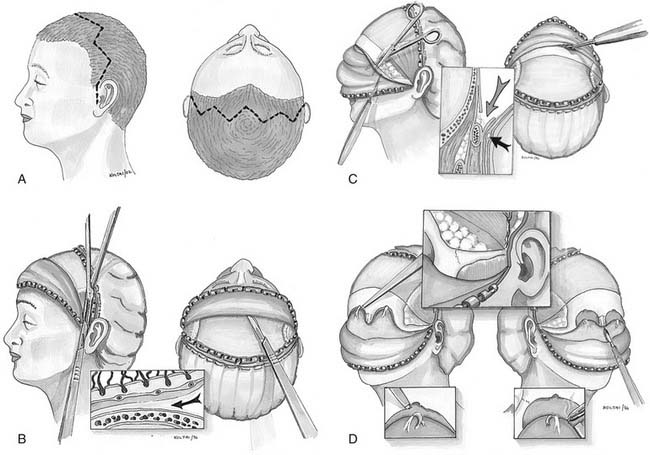
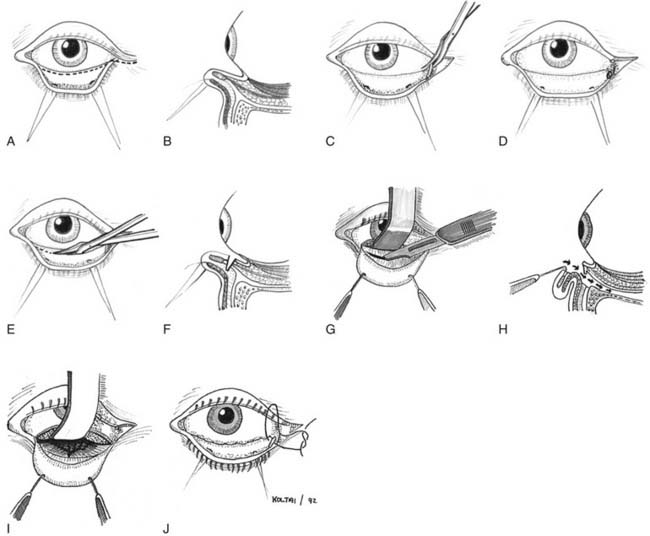
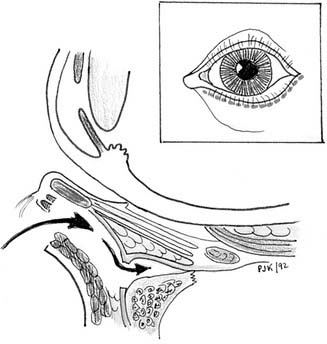
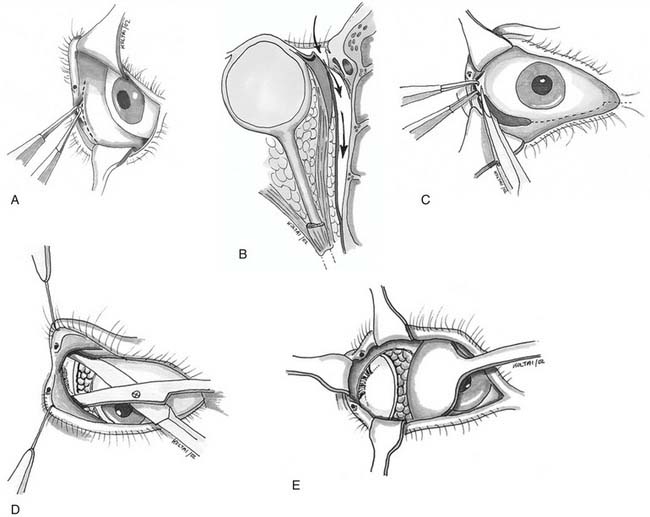
Miniplate and microplate screw fixation systems have made open reduction with internal fixation (ORIF) the preferred method of treatment for midface fractures. The entire facial skeleton can be accessed and reconstructed using a combination of six incisions, which allows for rigid interfragmentary fixation of the entire midface108 (Figs. 189-2 to 189-6). The bicoronal incision provides exposure of structures of the upper third of the face, including the zygomatic arches; lateral, medial, and superior orbital rims; and the nasoethmoidal region. The bicoronal approach, which is subperiosteal over the cranium and subfascial over the temporalis muscle, permits access to the superior and medial orbital rims. Orbital roof and nasoethmoid exposure can be obtained by mobilizing the supraorbital neurovascular bundle. Detachment of the temporalis fascia from the lateral orbital rim and zygomatic arch reveals the bones of the entire upper face from the zygomatic root on one side completely around to the other. The exposure provides a means to realign and rigidly fix the frontozygomatic suture, the entire zygomatic arch, and the nasal bones. It also allows for harvesting of cranial bone grafts for orbital reconstruction and cantilever nasal reconstruction in complex nasoethmoid fractures. The inferior orbital rims and floor can be exposed by either a subciliary or transconjunctival incision. The medial orbit and apex can be exposed by a transcaruncular approach. The gingivolabial sulcus approach provides access to the entire maxilla, laterally to the malar bone and zygoma, anteriorly to the infraorbital nerve, and medially up to the lacrimal fossa. The exposure provides for the reestablishment of both the lateral zygomaticomaxillary buttress and the medial nasomaxillary buttress. The entire nasoethmoidal region can be exposed by a midfacial degloving incision.
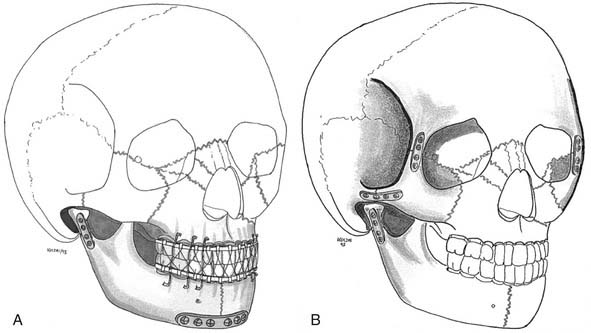
The sequencing of procedures for repair of severe midfacial fractures, especially when the mandible is fractured, is important. Reestablishing occlusion by MMF and repair of the mandible establishes a solid base for upper face reconstruction. Different strategies with respect to the sequencing of maxillary fractures have evolved (Fig. 189-7). Traditionally, the external frame of the face is realigned, and then the work proceeds toward the central core. This approach emphasizes control of the facial width and projection, which is the function of the zygomatic arch.
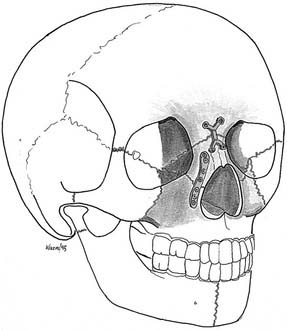
Our own approach to pediatric injuries is based on the recognition that the face is composed of component units connected by their associated buttresses and that the most prominent and most challenging aesthetic unit is the nasoethmoidal area. First, occlusion is established, and if necessary, the mandible is repaired. The central core is then reconstructed, followed by positioning of the orbits and the outer facial frame to the central core (Fig. 189-8).