Pathology of Iris Atrophy
Deepak P. Edward
Michelle Y. Cho
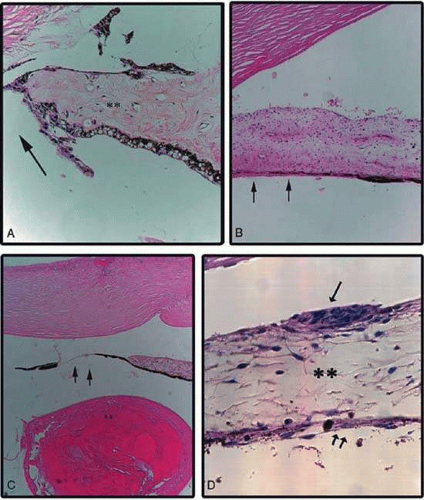
Pathologic changes in the iris and consequent clinical appearance may vary depending on the layer of the iris involved. Iris stromal atrophy causes stromal thinning and results in loss of iris crypts and the surface texture. Also, accompanying degeneration or loss of stromal melanocytes might alter the color of the iris. Degenerative changes may include loss of normal iris stroma being replaced with collagenous tissue that might appear whitish in color on slitlamp examination (Fig. 65-1A).
Iris atrophy may be limited to the iris pigment epithelial layer (Fig. 65-1B). This may be seen in conditions such as pigmentary glaucoma,1 diabetic changes2 in the iris, as well as chafing of the posterior iris surface after sulcus implantation of an intraocular lens3,4 (Fig. 65-1C). Such localized atrophy of the iris pigment epithelium would clinically show normal or slightly altered anterior iris and the presence of transillumination defects in the iris.
Iris atrophy may also involve both stroma and the iris pigment epithelium where there might be focal or generalized loss of both layers giving a moth-eaten appearance to the iris or polycoria such as is seen in iridocorneal endothelial syndrome5 (Fig. 65-1D).
CAUSES AND CLINICAL FEATURES
Table 1 lists the specific forms and causes of iris atrophy. Specific forms of iris atrophy and their clinical presentations are described in this chapter.
Table 1 Causes of Iris Atrophy and Degeneration | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Age-Related Changes
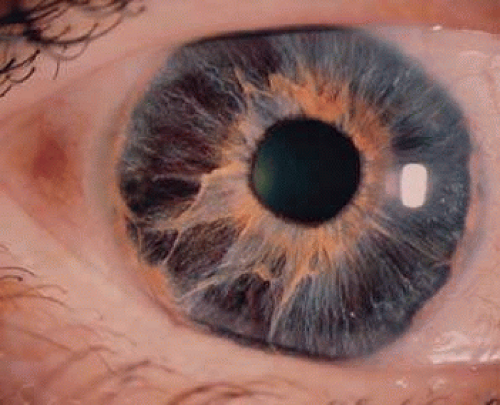
Atrophy of both the iris stroma and the pigment epithelial layer occurs as a natural aging process. Elderly patients may note a change in the iris to a more blue color, but these changes are usually not symptomatic. Age-related changes typically present as bilateral, symmetric iris stromal atrophy. There is a normal pupillary contour with increased prominence of the pigment epithelium and the pupillary sphincter, which may appear like a tan ring6 (Fig. 65-2). The absence of associated ocular findings may differentiate this acquired condition from other pathologic causes of iris atrophy.
Clinical Features
On slitlamp biomicroscopy, a generalized thinning of the iris stroma, flattening of the architecture, and a disappearance of the crypts, especially in the pupillary zone, are noted. The brown sphincter muscle becomes visible, and the entire pigment epithelial layer is easily seen. The pigment epithelial layer itself may show diffuse depigmentation and occasionally, focal areas of proliferation. The pupillary ruff often develops a moth-eaten appearance; granules of pigment may be seen scattered over the anterior iris surface, the anterior lens capsule, the endothelial surface of the cornea, and the trabecular meshwork. The red fundus reflex may be seen through the patchy areas of iris atrophy.
Functional changes are not clinically significant, although the so-called senile miosis and relative immobility of the pupil are common findings. The causes of these findings are probably related to hyalinization of the stroma and preferential atrophy of the dilator muscle, which arises from the iris pigment epithelium. These senile changes are believed to occur more frequently and earlier in eyes with exfoliation syndrome with or without glaucoma.
TREATMENT
Complications do not result from these changes, and no therapy is warranted except for reassuring the patient that the change in iris color does not represent any disease process.
PRIMARY IRIS ATROPHIES
Iridoschisis
Clinical Features
Iridoschisis is a rare condition that involves the iris stroma. The term iridoschisis is used when there is a split causing a cleft to occur in the iris stroma. The iris pigment epithelium and dilator muscle posteriorly are unremarkable. Histopathology including transmission electron microscopy shows marked atrophy of the iris stroma with separation of its anterior and posterior layers. The iris vasculature appears unremarkable.7,8
Iridoschisis occurs mainly in the elderly,9,10,11 but isolated cases have been reported in young patients after concussive injury.12 It has been reported to occur as a familial condition13 and has been associated with microphthalmos,14,15 interstitial keratitis,15,16 use of miotics,9 keratoconus,17 lens subluxation,18 and periocular eczema.19
Slitlamp examination reveals iris stromal fibers and blood vessels that remain attached to one portion of the iris with the loose ends floating freely in the anterior chamber, producing the so-called shredded-wheat appearance. The location of iridoschisis is often inferior (Fig. 65-3). The free-floating iris fibers may bow forward, leading to iridocorneal touch, corneal endothelial decompensation, and bullous keratopathy.8,20,21
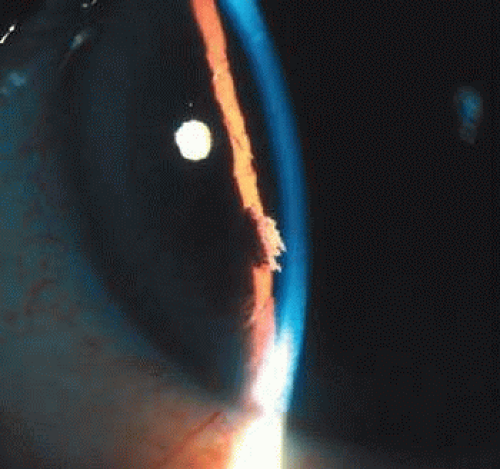 Figure 65-3. Iridoschisis located inferiorly. Note the split stromal fibers floating in the anterior chamber. (Copyright University of Illinois at Chicago. Reproduced with permission.) |
Iridoschisis may be associated with glaucoma in approximately 50% of the patients.9,22 The role of glaucoma in the pathogenesis of iridoschisis is, however, controversial. It has been hypothesized that iridoschisis may result in a secondary form of glaucoma in which the ruptured stromal fibers form anterior synechiae that block trabecular outflow.23 Others believe that the schisis in the iris is a result of iris stromal ischemia following acute elevation of intraocular pressure. Ultrasound biomicroscopy demonstrates narrowing of the angle, diffuse or discrete iris stromal changes with intact pigment epithelium in familial iridoschisis, and it is suggested that such patients may be predisposed to angle-closure glaucoma.24
Management and Treatment
Except for being alert to the possible relationship to angle-closure glaucoma, no specific therapy is warranted for iridoschisis. Cataracts are often seen concurrently in patients with iridoschisis since both conditions are associated with aging. Complications may arise from the free-floating iris fibers that obstruct the phacoemulsification tip of the irrigation/aspiration needle during cataract extraction. Various surgical techniques have been devised to avoid intraoperative injury to the iris.25,26,27,28,29
Iridocorneal Endothelial Syndrome
Etiology
Iridocorneal endothelial syndrome (ICE) denotes a spectrum of ocular disorders that includes progressive essential iris atrophy, Chandler syndrome, and iris-nevus syndrome of Cogan-Reese.5,30,31,32,33,34,35 It is included in this chapter because these ocular disorders were previously referred to as the essential iris atrophies.
The defect appears to be an abnormality of the corneal endothelium that results in endothelial cell proliferation causing a monolayer of endothelial cells with an underlying basement membrane to extend across the anterior chamber angle and onto the anterior surface of the iris.5,36,37 The contracture of the membrane results in iris atrophy and defects, as well as the peripheral anterior synechiae that causes secondary glaucoma.
An inflammatory and/or viral process has been implicated in the pathogenesis of the syndrome. Alvarado et al.38 used the polymerase chain reaction to detect herpes simplex virus DNA in corneal specimens with ICE syndrome, and the herpes simplex virus (HSV) was also isolated from the aqueous humor of a patient with ICE syndrome.39 Endothelial cell metaplasia is demonstrated in corneal specimens with ICE syndrome as shown by expression of various cytokeratins that are expressed in the corneal endothelium.40,41
Clinical Features
Iridocorneal endothelial syndrome, an acquired unilateral condition, is predominantly seen in middle-aged females.32 However, ICE syndrome resulting in secondary glaucoma has been described in a child,42 an adolescent with Down syndrome,43 and in occult cases status post laser in situ keratomileusis.44,45
The appearance of the iris is the main factor that divides this spectrum of diseases into its clinical variations.46 The most familiar form is progressive iris atrophy or essential iris atrophy in which there is marked corectopia, extensive and progressive atrophy of the iris producing a pseudopolycoria (Fig. 65-4A).5,31,36 In Chandler syndrome, the iris shows only mild stromal atrophy and/or corectopia with predominant corneal changes.34 In the iris-nevus syndrome of Cogan-Reese, nodular or diffuse pigmented lesions are present on the iris surface in association with varying degrees of corectopia and atrophy of the iris (Fig. 65-4B).33,35
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree