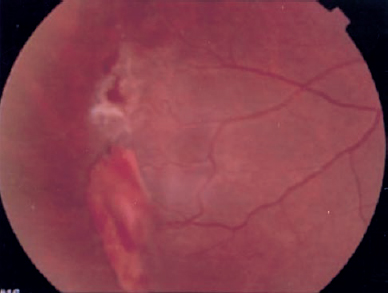
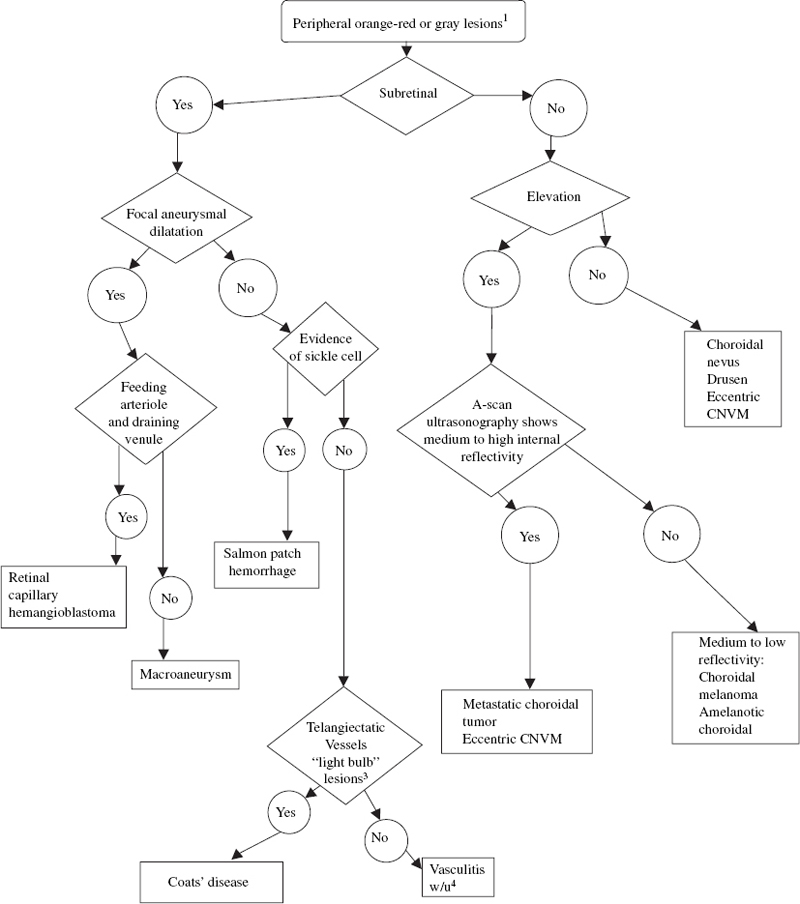
10 Gray and orange–red lesions in the fundus represent localized or, at times, diffuse pathologic changes often originating from abnormalities in the vasculature of the retina, retinal pigment epithelium (RPE), or choroid. When these lesions are subretinal or located deep in the retina and associated with intraretinal fluid, the inherent transparency of the retina and view to the RPE and choroid are affected, making the diagnosis of these conditions challenging. Evaluation requires identification of the origin of the lesion, a thorough knowledge of the posterior segment anatomy, and an orderly approach to examination. A treatment strategy may be designed. The color of the lesion seen on fundus examination depends on the composition of the tissue and its location. Vascular lesions and blood are orange–red because of hemoglobin. Acute macular neuroretinopathy (AMN) and photic retinopathy may have a deep red appearance and the mechanism of the color change is not clearly understood. Depending on the location within the retinal layers or choroid, the lesions will have subtle differences in appearance. Vascular lesions appear red or orange–red when they are located within the neurosensory retina or subretinal space as a result of the transparent nature of the retina. When blood is present underneath the RPE, the melanin of the RPE blocks some of the red of hemoglobin, causing the lesion to appear darker or even gray. Often there may be support cells associated with the abnormal vasculature that influence the color. An example is a choroidal neovascular membrane (CNVM), in which fibroglial cells may mask the red of the vasculature. The choroid is a large lobular vascular network that causes the fundus to appear orange–red. When there is an accumulation of fluid or blood underneath the RPE, the observer may see an orange–red nodular elevation. Examples of fluid beneath the RPE include central serous chorioretinopathy (CSCR) and posterior scleritis. Choroidal tumors, such as the nevus, melanoma, and osteoma, may appear orange–red at certain stages in their pathology. Depending on the nature and location of the lesion, patients may present without complaints or have a variety of symptoms, including painful loss of vision. An orderly approach is thus needed to limit the differential diagnosis. A comprehensive examination with a review of the patient’s medical, family, and social history is important. Additional history will identify patients at risk for signs of hypertension, neoplastic disease, and sickle cell retinopathy, for example. At times, the medical history may not be available. A review of systems may be useful for discovering important systemic symptoms. Pointed questions may uncover symptoms of diabetes mellitus, medication use, or connective tissue disorders, for example. Because of the different presenting conditions, no guideline will always yield a diagnosis. The following is a general approach. The diagnosis may depend on the input of experienced subspecialists. The examination begins with a methodical evaluation of the orbit and anterior segment structures, specifically looking for lid swelling, anterior chamber inflammation, iris nodules, or lens changes. Examination of the anterior and posterior vitreous for inflammatory cells, pigmented cells, or blood is performed. It is useful to localize the lesion based on region: the peripapillary region, the posterior pole, and the periphery. The peripapillary area denotes the area adjacent to and including the optic nerve. The posterior pole includes the region within the vascular arcades. The periphery encompasses the region outside the vascular arcades to the ora serrata. The key to diagnosis within the posterior segment is determining the location of a lesion within or under the retina. Because a number of orange–red and gray lesions arise from vascular abnormalities, distinction between choroidal and retinal aberrations helps pinpoint the diagnosis. For example, choroidal neovascularization typically causes subretinal hemorrhage, whereas retinal vascular abnormalities are associated with intraretinal and preretinal bleeding. Fundus abnormalities may derive from the choroidal or retinal vasculature. The two circulations are distinct. The retinal circulation supplies the inner two thirds of the retina, the choroidal circulation the outer third. Localization of a lesion and its depth within or below the retina can be facilitated by determining whether the lesion lies above or below the retinal vessels. Differentiating deep retinal, subretinal, and sub-RPE locations may be difficult, but signs such as blood, exudates, or fluid in the surrounding area can be helpful. Ophthalmoscopic examination techniques enable the examiner, through magnification and stereopsis, to localize the lesion. The use of imaging techniques, such as optical coherence tomography and scanning laser tomography, are also useful in determining the location with respect to the inner and outer retina and choroid. Depending on the size and nature of the lesion, more than one location may be involved. This refers not only to the location with respect to the retina and choroid but also to the topographic location of the lesion. For example, bleeding from macroaneurysms may occur at all layers in the posterior segment, including subretinal, intraretinal, and preretinal areas.1–3 Macroaneurysms may be located anywhere there are retinal vessels. This includes the peripapillary area, posterior pole, and periphery. Gray and orange–red lesions present in many ways. The diagnosis depends on recognizing patterns of signs and using them based on the history and symptoms of an individual patient. Although certain lesions classically occur in a particular location, an early stage of the pathology may present differently. Some lesions grow, whereas others remain dormant. A classic example is the differentiation between a choroidal nevus and a choroidal melanoma. A choroidal melanoma may reach a very large size if it is discovered late in its course. Choroidal nevi rarely demonstrate growth or malignant transformation.4 An early choroidal melanoma may appear to be a nevus; distinguishing between these two is critical. Follow-up examinations are important in making this distinction. Orange–red and gray lesions, often vascular, can be associated with hemorrhage or exudates. These signs represent a breakdown in the blood–retina and blood–RPE barrier. To establish the localization of the lesion, it is helpful to localize the associated leakage. Vascular abnormalities derived from the choroid tend to be associated with fluid, blood, or exudates in the sub-RPE or subretinal space. Retinal vascular abnormalities present with blood, fluid, or exudates within the retina. There are, however, exceptions to this. Macroaneurysms can bleed into the subretinal space, and some cases of choroidal neovascularization may bleed into the retina or vitreous, obscuring the subretinal pathology. Fluorescein angiography (FA) can help to determine the vascular properties of a lesion, such as the double circulation in choroidal melanoma, or the extent of the CNVM.28 We have learned from submacular surgery, however, that the neovascular membrane tends to be larger than seen on FA.8 FA is helpful to determine nonperfused retinal areas such as in Coats disease and sickle cell retinopathy. Angiographic features of RPE detachments aid in identifying patients with CSCR, large drusen, CNVM, and posterior scleritis. FA of the optic nerve may demonstrate disc leakage in Vogt– Koyanagi–Harada (VKH) syndrome, metastatic choroidal tumors, and posterior scleritis.27,61 Filters on the FA camera provide useful information: For example, macular changes in AMN may be identified with red-free photos, and autofluorescence properties of optic nerve head drusen may be demonstrated without injecting fluorescein.63,76 Ultrasonography provides a cross-section of the globe and orbit. It is used for differentiating tumors by determining the internal patterns of reflectivity and measuring the cross-sectional height. Several estimations of the height and extent of a lesion are important in determining growth since the last examination. In addition, ultrasonography may be helpful when media opacities obscure clear visualization of the posterior segment. The internal reflexivity of a choroidal mass can provide important diagnostic information. For example, a choroidal melanoma classically displays a high initial spike on A-scan with a medium to low internal reflectivity,22 whereas a choroidal hemangioma demonstrates a high initial spike with medium to high internal reflectivity.10 Guidelines for the acoustic characteristics are helpful, but these lesions should be evaluated by experienced specialists familiar with ocular oncology who can differentiate subtle differences. The reflectivity of other lesions may be useful in identification. Optic nerve head drusen display high reflectivity on B-scan, even at low gain.76 Lesions that contain calcium, such as choroidal osteoma, demonstrate a high initial spike with acoustic shadowing, or lack of acoustic waves, behind the lesion.67 Eyes with posterior scleritis may have a diffusely thickened choroid and edema within the Tenon capsule, creating the “T-sign.”60 Indocyanine green (ICG) angiography permits visualization of the choroidal and retinal vasculature, even in the presence of thin layers of blood. This technology may be useful in detecting CNVM, particularly occult CNVM. ICG is useful in evaluating conditions such as CSCR, idiopathic polypoidal choroidal vasculopathy, and inflammatory disorders of the choroid.14 Computed tomography (CT) scan may be helpful in evaluating scleral and choroidal thickening. In addition, disorders with calcification are readily visualized on CT scan images, such as choroidal osteoma and optic nerve head drusen. CT scanning also permits imaging of possible associated intracranial lesions, such as cerebellar masses in von Hippel–Lindau (VHL) syndrome. Certain blood tests can be useful in diagnosing the cause of inflammatory retinal vasculitis or choroiditis. A sickle prep may identify a patient with salmon patch hemorrhage. A simple finger stick for blood sugar may help to diagnose a patient with diabetes mellitus and intraretinal hemorrhage. Orange–red intraretinal hemorrhages may develop from localized or diffuse retinal vasculitis. Appropriate laboratory testing may reveal the etiology of inflammatory, neoplastic, or infectious pathologies. Subretinal lesions usually appear beneath the retinal vessels. Occasionally, associated bleeding into the inner retina may limit the ability to localize the lesion. Choroidal or uveal melanoma arises from the pigmented cells of the choroid. It is critical to differentiate a benign lesion, such as choroidal nevus, from choroidal melanoma. Examination and differentiation should be performed by an experienced specialist who is familiar with subtle differences in presentation. Choroidal melanoma typically appears as a dome-shaped, elevated, oval mass with variations in the degree of pigmentation. When the tumor reaches a large size, it typically assumes a mushroom shape as it passes through the Bruch’s membrane. Smaller tumors may not be so readily recognized. Although choroidal melanomas are commonly seen as dark pigmented masses, they can have varying degrees of pigmentation and may even be amelanotic. Some are dark red or pale, nodular lesions. Their size also varies.11 When melanomas are confined to the choroid, or relatively soon after gaining access to the subretinal space through the Bruch membrane, they may demonstrate overlying patches of orange pigment, some with indistinct borders, and evidence of subretinal fluid. Evaluation of the thickness or height of the mass and determining whether there has been progression since a previous examination are critical in distinguishing a melanoma from a benign nevus. In general, lesions 2 mm or smaller are considered nevi, whereas those larger than 2 mm in height are suspicious for choroidal melanoma.4–6 Ultrasonography is used to measure the tumor height and to reveal its internal acoustic reflectivity. Choroidal melanoma has an initial steep spike with medium to low internal reflectivity.11 In unclear cases, tumors are monitored for growth. Fluorescein angiographic characteristics have been described but are not pathognomonic.4,15 Classic presentations include multiple pinpoint leaks on the surface of the tumor and an intrinsic or double circulation of the tumor. The orange pigment patches tend to appear as hypofluorescent spots. The definitive treatment for choroidal melanoma is controversial. Treatment involves eradicating the tumor’s growth potential and, if possible, saving the vision and the eye. A large part of the treatment strategy is determined by the size and location of the tumor. Small tumors or suspected nevi in the periphery may be observed and measured with ultrasound to determine height and growth. Treatment options for melanoma include photocoagulation, radiotherapy, transpupillary thermotherapy, local resection, enucleation, and exenteration and are covered in greater detail in Chapter 15.11,18–20 Choroidal nevus is a benign growth within the choroid that may be confused with choroidal melanoma. In one series, it accounted for approximately one quarter of the lesions differentiated from choroidal melanoma.21 A nevus occurs from a benign proliferation of melanocytes within the choroid.9 As with choroidal melanoma, its presentation may be variable. The typical choroidal nevus, particularly when located in the peripheral retina, is discovered as part of a routine examination and is asymptomatic. It may be located anywhere in the fundus. The size is one to two disc diameters, but it may attain a larger size, simulating a medium to large malignant melanoma. There may be some elevation but usually less than 2 mm.22 The nevus ranges in color from gray to the amelanotic orange–red. The surface characteristics may include a homogeneous color or an irregular mixture of orange and yellow pigment and small, drusen-like surface changes.22,23 These clumps differ from melanomas that reveal larger patches of orange pigment. Variable amounts of RPE atrophy may surround the choroidal nevus, likely a reflection of the long-standing nature of this lesion. Subretinal fluid adjacent to the tumor is not a reliable distinguishing factor because other lesions may present with adjoining retinal detachment, including choroidal melanoma, choroidal osteoma, and others.15,21,67 As in choroidal melanoma, ultrasonography is used to measure the thickness and size of the nevus and detect growth in follow-up evaluations. FA plays a limited role but may demonstrate blockage of fluorescence with variable amounts of staining surrounding the lesion. If drusen are present on the surface of the lesion, these may also stain. At times, choroidal nevi are associated with choroidal neovascularization, and FA helps to detect this. Referral to a specialist for atypical or large lesions is advised. Nevi that do not develop into melanomas are followed up and need only treatment for visually threatening sequelae, such as adjacent choroidal neovascularization or exudative retinal detachment encroaching on the macula. These tumors should be followed up regularly because malignant transformation may occur.22 Metastases to the choroid may occur due to its rich lobular vascular network. Metastatic choroidal tumors account for the most common intraocular malignancies.26 The most common site of origin is the lung for men and the breast for women. At times, patients will be unaware of the presence of a primary tumor, and the ophthalmologist may be the first to identify evidence of neoplastic disease. Metastatic lesions may be multifocal or present as a single focus. Single metastatic tumors often are associated with an exudative retinal detachment. Metastatic lesions tend to have a creamy yellow to pale orange color and have indistinct borders. The size and location in the fundus are variable. Tumors may be bilateral. They do not grow to mushroom shape and may have an irregular elevation of the RPE. The surface of the tumor may present with coarse mottling and depigmentation of the RPE.27 Sometimes they are difficult to differentiate from a choroidal melanoma. As with all choroidal tumors, ultrasonography is important in the clinical evaluation. The ultrasound characteristics of metastatic lesions are variable. Many present with a high initial spike and high internal reflectivity. Although not diagnostic, ultrasonography provides information about the size and characteristics of these lesions. FIGURE 10–2. Late frame of the fluorescein angiogram demonstrates stippled hyperfluorescence in a metastatic choroidal tumor located along the inferior arcade. FA may be used, but there are no uniform fluorescein characteristics of these lesions. Many demonstrate hypofluorescence in the early transit frames with late hyperfluorescence (Fig. 10–2). Large choroidal vessels within the tumor are typically absent from the surface of the tumor.28 Both CT scan and magnetic resonance imaging (MRI) are useful in characterizing the extent of the primary tumor. Metastatic disease involving the central nervous system (CNS) can be detected with brain imaging. The prognosis for patients with metastatic choroidal tumors is poor. Median survival varies with the primary tumor, but most are on the order of months.27 Treatment of metastatic tumors needs to be considered because, despite the poor prognosis, some patients do achieve long-term survival. Treatment involves systemic treatment of the primary tumor along with local treatment to the ocular tumor. Small peripheral tumors may be followed for response to systemic treatment. Radiation therapy, transpupillary thermotherapy (TTT), and photocoagulation are also treatment options. Enucleation is reserved for eyes with blind, painful symptoms.26–28 Choroidal neovascularization may occur anywhere in the fundus. The most common location is in the macula where it interferes with vision. A choroidal neovascular membrane (CNVM) may form in the periphery of the fundus and result in an asymptomatic disciform scar (subretinal gliosis) with subretinal and/or intraretinal hemorrhage. However, sometimes these lesions bleed into the vitreous, resulting in reduced vision. Extramacular CNVM occurs rarely and usually is discovered on routine funduscopy. Because the location is away from the macula, vision is rarely affected. These lesions range in size from one to several disc diameters.33 They may arise from old chorioretinal scars or the pars plana region or in the periphery without any apparent abnormality.34 The amount of associated subretinal hemorrhage may vary, in part depending on how long the hemorrhage has been present. The surface of the lesion tends to have RPE mottling and may appear gray, with associated gliosis, or black, or dark orange with associated sub-RPE blood. Differentiating these lesions from choroidal melanoma is important.33–35 Fluorescein angiography is helpful in defining the extent of the lesion. In general, sub-RPE membranes will demonstrate early hyperfluorescence with pooling into the RPE detachment. Subneurosensory retinal membranes may demonstrate early hyperfluorescence with late leakage beyond the boundaries of the CNVM. When gliosis is present, the hyperfluorescence is associated with varying amounts of staining. Ultrasonographic findings vary. A-scan tends to show initially low reflectivity in early lesions and high reflectivity in older lesions. B-scan generally shows a broad, sessile thickening, with variable internal reflectivity.10 Treatment is reserved for lesions that compromise vision. Laser photocoagulation or transscleral cryotherapy in peripheral lesions have been effective in reducing leakage that encroaches the macula.9 Vitreous hemorrhage may be treated with pars plana vitrectomy if clearing does not occur spontaneously.33 Drusen are subretinal deposits that increase in frequency with increasing age. Histopathologic studies have shown that drusen are localized collections of eosinophilic material between the basement membrane of the RPE and the Bruch’s membrane.36 Drusen typically are pale gray to yellow in color on funduscopy. They are often present in the posterior pole but may be seen in the periphery. There may be a symmetric appearance in both eyes. Drusen are usually multifocal, and the size of each individual druse ranges from a 63 μm or smaller diameter (hard drusen) to 250 μm or larger (soft drusen). Drusen may coalesce into larger lesions, and, as drusen enlarge, they become localized detachments of the RPE. Peripheral drusen usually are discovered on routine examination and have no affect on vision.38 Peripheral drusen do not affect vision, and so diagnostic testing is not routinely performed. FA, when performed in macular drusen, can reveal hyperfluorescence from RPE atrophy and hypofluorescence from hyperpigmentation or viscous subretinal fluid. The drusen, when large, may demonstrate early well-defined hyperfluorescence consistent with an RPE detachment. Treatment is reserved for complications that arise as sequelae to drusen, such as CNVM. Treatment for peripheral drusen is not indicated. Macroaneurysms are acquired localized dilatations of the retinal arteriolar vasculature usually occurring within the first three orders of retinal arteriolar bifurcations, but rarely they occur outside the posterior pole. Typically, macroaneurysms arise in patients in the sixth to seventh decade of life who have had systemic hypertension. Women outnumber men three to one. Histopathology demonstrates linear breaks in the vascular wall and thrombus formation within a thin-walled outpouching of the arteriole.1–3,39 Peripheral macroaneurysms are discovered, most often as an incidental finding. Vision loss occurs as a result of leakage of blood and its constituents. When the macroaneurysm is located away from the macula, vision remains unaffected unless there is an associated breakthrough vitreous hemorrhage. Typical presentation is a variable amount of ring exudates, edema, and hemorrhage surrounding a gray or orange fusiform dilatation of a retinal arteriole. Often a distinct macroaneurysm may be difficult to find, leaving only the deposited lipoproteins (exudates) obvious. Variations in presentation will depend on the amount of leakage from the aneurysm. Bleeding may occur throughout all layers of the retina, subretinal space, and vitreous.39 FA is used to delineate the macroaneurysm. Blood may obscure the macroaneurysm, however, or it may be thrombosed and not appear on FA. Angiography delineates retinal microvascular abnormalities surrounding the lesion, such as capillary nonperfusion and cystoid edema of the retina. Photocoagulation is reserved for lesions threatening vision. Peripheral macroaneurysms may be observed. Pars plana vitrectomy for nonclearing vitrous hemorrhage affecting vision may be considered. Retinal capillary hemangiomas are congenital hamartomas that may be isolated or part of VHL syndrome associated with cerebellar hemangioblastoma. Patients with VHL are at risk for other organ involvement, such as tumors of the pancreas, adrenal glands, kidneys, liver, and epididymis. The familial disease has been mapped to chromosome 3.40–42 Retinal capillary hemangiomas are typically red or orange–red, well-circumscribed tumors that may occur in the retina or optic nerve. Optic nerve hemangiomas appear different from the peripheral type and are discussed later. Peripheral hemangiomas are accompanied by two dilated vessels: a feeding arteriole and draining venule. The size of the angioma is variable and may range from less than a disc diameter to several disc diameters. Larger lesions have larger feeding arterioles and draining veins. The angiomas may be single or multiple. There is often associated lipid or edema surrounding the angioma. Vitreoretinal traction may develop along the surface of the angioma or in the macula away from the angioma. The funduscopic findings are typical so that diagnosis usually can be made on presentation. FA may help to identify smaller lesions undetected on clinical examination. Ultrasonography may help in larger lesions where the diagnosis is uncertain. MRI and CT scanning are critical to rule out tumors of the CNS and organs. Treatment of these vascular tumors is important for lesions that are posterior in location and large enough to affect vision. Some physicians advocated treating all tumors because they tend to grow. Small lesions are easier to treat than larger ones.41 Photocoagulation for small lesions is accomplished by treating the vessels at the edge of the tumor and by direct tumor ablation. When treating the feeding vessels, it is important to treat the arteriole first to decrease the blood flow and the risk of bleeding. Larger lesions may require cryotherapy or radiation. Vitreoretinal complications, such as traction or rhegmatogenous retinal detachment, may need retinal detachment repair. Systemic involvement should be comanaged with appropriate specialists. Coats disease represents a spectrum of sporadic, unilateral, and congenital vascular telangiectatic and aneurysmal retinal vessels. Most cases present in males (3:1) before the age of 10 years. Patients with minimal changes in the macula often go unrecognized until adulthood, when the telangiectatic vessels are discovered on routine examination. Histopathologic studies have shown loss of retinal vessel endothelial integrity and lipid-laden macrophages in the subretinal space.43–45 Presentation in children may take the form of leukocoria or strabismus. Red–orange lesions in the adult form of Coats disease may be either focal aneurysmal dilatation of the retinal vessels or intraretinal bleeding from incompetent vessels. Telangiectasia of the retinal vessels is found more often in the periphery than in the posterior pole and can have associated edema, exudates, or bleeding. Exudation from peripheral lesions may appear in the macula. Retinal vessel sheathing tends to be absent. Fluorescein angiography delineates the extent of the retinal telangiectasia and corresponding retinal vessel nonperfusion. The classic “light bulb” lesion that represents a focal aneurysmal dilatation is pronounced on angiography. Fluorescein leakage from telangiectasia and aneurysms and hypofluorescence from the surrounding occluded microvasculature are features. In some childhood cases, CT scanning is used to distinguish Coats disease from retinoblastoma, which usually has calcification. Treatment is advocated for lesions that compromise or threaten vision. Visual loss usually occurs as a result of leakage of fluid or exudates into the macula. Photocoagulation or cryotherapy obliterates the incompetent vessels and aneurysms, permitting subsequent reabsorption of subretinal fluid. At times, extensive subretinal fluid may be drained to facilitate treatment. Multiple treatment sessions may be needed. Sickle cell retinopathy occurs from sickling of red blood cells within blood vessels as a result of abnormal hemoglobin molecules. The vessels become occluded and account for the clinical findings. Sickle cell retinopathy occurs almost exclusively in black patients. Most patients with retinopathy have the SC or S-thal variant.46 A characteristic of sickle cell retinopathy is the salmon-patch hemorrhage, named for its characteristic orange–red appearance. It may be one to a few disc diameters in size and is often well circumscribed and circular to oval. These hemorrhages are located in the preretinal or superficial retina and frequently are found in the midperipheral and peripheral fundus. Rarely, they may be located in the posterior fundus. The blood may track into the vitreous, creating a vitreous hemorrhage, or into the subretinal space. Unless associated with a vitreous hemorrhage, they are generally asymptomatic.46 They may be single or multiple. Other signs of sickle cell retinopathy include iridescent spots, remnants of blood products and macrophages within the retina, and black sunbursts that are reactions of the RPE to blood. Peripheral ghost vessels or sea-fan neovascularization may be present. Confirmation of sickle cell disease by blood testing, specifically, a sickle prep or hemoglobin electrophoresis, is used to identify the presence of abnormal hemoglobin. FA may detect areas of nonperfusion in the periphery. Salmon-patch hemorrhages will block fluorescence. Sea-fan neovascularization leaks fluorescein (Figs. 10–3 and 10-4) and borders retinal capillary areas of nonperfusion.
Orange–Red and Gray Lesions in the Fundus
What Are Gray or Orange Lesions in the Fundus?
Why Do Fundus Lesions Have a Gray or Orange–Red Color?
Where Does the Examiner Begin When Evaluating Gray or Orange–Red Lesions?
Why Is It Important to Determine the Anatomic Location of the Lesion?
Are Lesions Commonly Located in Only One Part of the Retina?
What Is the Significance of Associated Hemorrhage or Exudates?
In What Cases Is Fluorescein Angiography Helpful?
In What Cases Is Ultrasonography Helpful?
Are There Other Tests That May Be Helpful?
What Are Orange–Red and Gray Lesions of the Peripheral Retina?
Subretinal Origin (Fig. 10–1)
Choroidal Melanoma
CLINICAL PRESENTATION
TESTING
TREATMENT
Choroidal Nevus
CLINICAL PRESENTATION
TESTING
TREATMENT
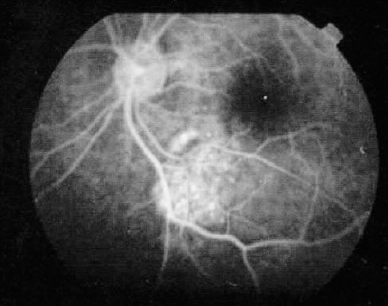
Metastatic Choroidal Tumors
CLINICAL PRESENTATION
TESTING
TREATMENT
Choroidal Neovascularization (Extramacular Location)
CLINICAL PRESENTATION
TESTING
TREATMENT
Drusen
CLINICAL PRESENTATION
TESTING
TREATMENT
Retinal Origin
Retinal Arterial Macroaneurysm
CLINICAL PRESENTATION
TESTING
TREATMENT
Retinal Capillary Hemangioma
CLINICAL PRESENTATION
TESTING
TREATMENT
COATS DISEASE
CLINICAL PRESENTATION
TESTING
TREATMENT
Salmon Patch Hemorrhage: Sickle Cell Retinopathy
CLINICAL PRESENTATION
TESTING
Ento Key
Fastest Otolaryngology & Ophthalmology Insight Engine