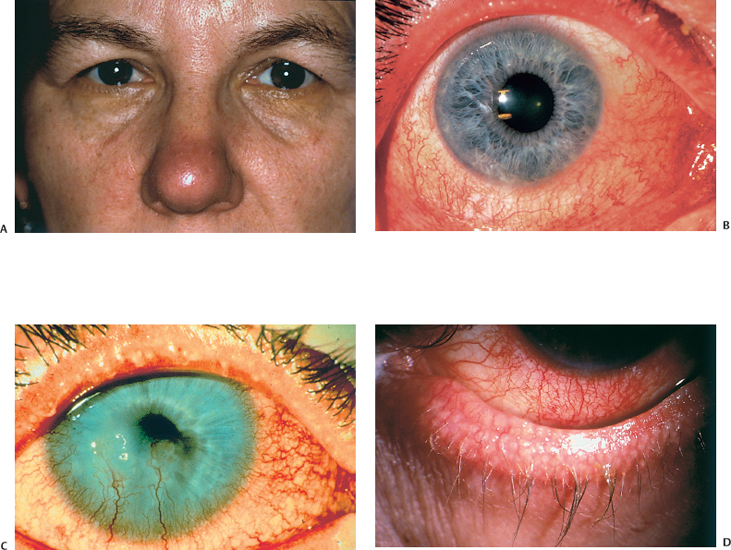
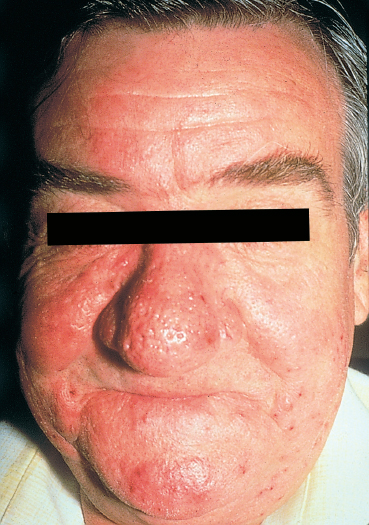
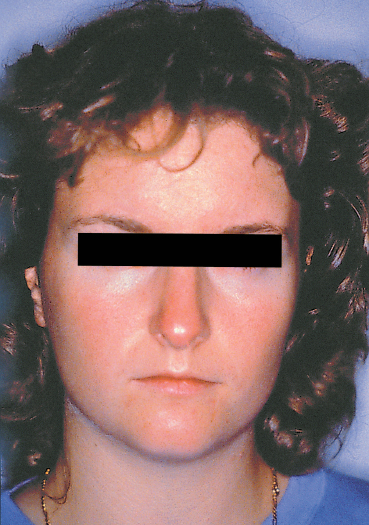
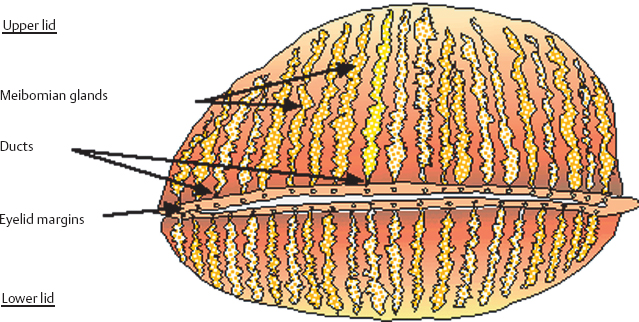
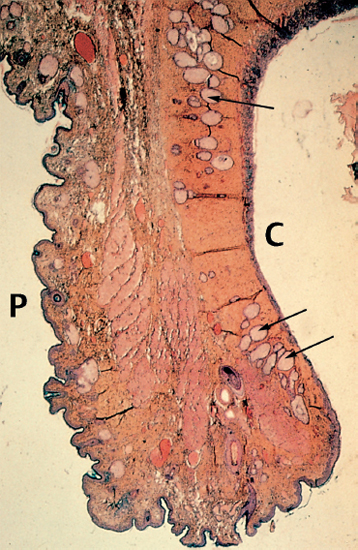
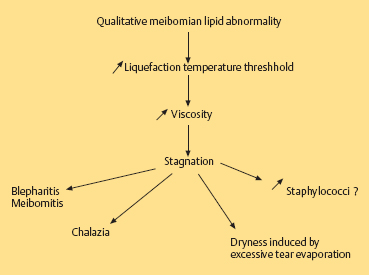
Chapter 5 Ocular Rosacea Rosacea generally affects middle-aged adults, between 40 and 60 years, but can also occur during childhood.1–3 It affects all races, but is more frequent—or at least more visible—in fair-skinned northern Europeans.4,5 It is estimated that more than 10% of the general population is affected. Females appear to be more susceptible than males,6 but rosacea, and especially ocular rosacea, seem to be more severe in males.7 There are many explanations for the lack of epidemiological studies on ocular rosacea. In particular, ophthalmologists often fail to identify the cutaneous signs of rosacea, because rosacea is defined by highly pleomorphic criteria. The rhinophyma is highly evocative, but it is rare and occurs only in certain advanced forms (Fig. 5.1). A consensus seems to have emerged that a combination of facial telangectasia, flushes, and papules or pustules is a reliable diagnostic triad (Fig. 5.2). Another explanation for the lack of epidemiological studies is that ocular rosacea is more readily identifiable when it occurs simultaneously with, or following, the cutaneous signs, but in a quarter of cases it precedes the cutaneous involvement, sometimes by several years, and may remain isolated.8 This explains why the reported frequency of ocular involvement in rosacea varies so widely from one publication to another (3-58 %).7,9 Finally, while meibomian dysfunction is almost always present, it is not synonymous with ocular rosacea, and the underlying primary meibomitis is only one possible cause of the dysfunction. Several authors have attempted to classify meibomian dysfunction, using different criteria.10 It has been estimated that 20-40% of patients consulting ophthalmologists have meibomian dysfunction, which is the main cause of blepharitis.11–13 In another study, blepharitis or conjunctivitis was diagnosed in three quarters of the 2-6% of patients who consulted general practitioners or emergency departments for eye problems.14,15 Also, more than half of all patients with a history of chalazion have cutaneous stigmata of rosacea.16 Taken together, these reports suggest that the prevalence of ocular rosacea is probably underestimated. Fig. 5.1 Advanced-stage rosacea with rhinophyma Fig. 5.2 Rosacea: facial erythema associated with flushing Fig. 5.3 Rosacea in a 12-year-old The cause of rosacea is unknown, but it is thought that the cutaneous manifestations may be of vascular origin.6,17 Other triggering factors—or possibly causes—of cutaneous rosacea8 include colonization of the skin by certain microorganisms, climatic factors (especially ultraviolet radiation), psychosomatic disorders, and gastrointestinal disorders, in which Helicobacter pylori17 and Demodexfollicularum may play a role. In contrast, the ocular complications appear to result, in large part at least, from abnormalities of the meibum. Thus, to understand the pathogenesis of ocular rosacea it is necessary to recall the anatomy of the meibomian glands and the physicochemical characteristics and physiology of meibomian secretion. Anatomy of the Meibomian Glands The meibomian glands bare the name of Heinrich Meibom, who was the first to describe them precisely in 1666.18 More numerous in the superior tarsus (30-40) than in the inferior tarsus (20-30), they are aligned in a single row and oriented perpendicularly to the lid margin (Fig. 5.4). Their orifices are located just in front of the mucocutaneous junction; the latter is visible at the junction of the anterior three quarters and posterior quarter of the lid margin, is slightly rounded, and is about 2.5 mm wide. With age, the orifices have a tendency to move posteriorly. As the meibomian glands lie immediately beneath the transparent conjunctival palpebral mucosa, their contours are visible to the naked eye when the eyelid is everted. They form whitish parallel lines that can be clearly identified by slitlamp examination.19,20 The meibomian glands are modified sebaceous glands with a partially keratinized duct and acini whose centrally located cells degenerate and form the lipidic holocrine secretion (Fig. 5.5). Normally, about 45% of the glands are functional and only about ten glands per tarsus are expressible (the number tends to decrease with age).21,22 Seventy-four percent of patients with blepharitis have a reduction in the number of meibomian glands, compared with only 20% of healthy controls.13 Fig. 5.4 Diagrammatic representation of the meibomian glands Fig. 5.5 Histological section of the lid: meibomian glands (arrows) (HES staining). P: cutaneous surface; C: palpebral conjunctiva Physiology Role Meibomian secretions compose the superficial lipid layer of the tear film and have multiple roles: —They prevent tear evaporation and nocturnal dessication, and slow tear evacuation23–26; —They participate in the spread of the tear film on blinking27–29; —They prevent contamination of the tear film by cutaneous sebaceous secretions (which would otherwise accelerate tear film rupture); —They contribute to smoothening of the corneal surface for refraction of the incident light rays. Laboratory Investigations —Meibometry is the measurement of the amount of lipids at the lid margin, which is reduced in patients with meibomian dysfunction.30,30a —Video-imaging tear film interferometry is used to assess the dynamics of the lipid film during blinking.31 —Meibography or videomeibography using in vivo transillumination biomicroscopy with infrared photography visualizes the glandular ducts and acini.32 Regulatory Mechanisms Meibomian secretion decreases with age.21 Some authors have reported that meibomian secretion may be under hormonal regulation—androgens stimulating it33 and estrogens inhibiting it.34 Only estrogen receptors have been found in the meibomian glands.34a The role of the autonomic nervous system is unclear. Biochemistry The most recent analytical techniques include thin layer chromatography and gas liquid chromatography coupled to mass spectrometry. Normal meibomian secretions differ from cutaneous sebaceous secretions.35,36 They are mainly composed of neutral lipids, waxes and especially esterified sterols, which are twice as abundant as in sebum. In contrast, they contain very small amounts of triglycerides, fatty acids and free sterols, and no squalene. Patients with keratoconjunctivitis due to meibomian dysfunction have an abnormal meibum composition, including: —An increase in the ratio of sterol esters to waxes,37 —An increase in mono-unsaturated fatty acids,38 —An increase in free fatty acids and the presence of abnormal fatty acid peroxides. The former are toxic and irritant for the epithelium; they sometimes give a foamy aspect to the tear meniscus and destabilize the tear film.39,40 The presence of epoxides suggests that certain lipid abnormalities encountered in meibomitis result from abnormal peroxidase function in the peroxisomes and endoplasmic reticulum.41 Increased chain length, an increase in the sterol ester fraction, a shift from cholesterol ester to cholesterol, and a reduction in branching or unsaturation of lipid components are all factors that raise the melting point of lipids, thereby increasing the viscosity of meibomian secretions, leading to their stagnation and meibomian gland obstruction (see Table 5.1). Cholesterol esters are necessary for meibomitis to develop, and also favor staphylococcal proliferation.42 However, marked interindividual variations in meibomian lipid components mean that such findings must be interpreted with care.43 Table 5.1 Meibum in ocular rosacea Models There are several animal models of meibomian gland dysfunction: Polychlorinated biphenyl This compound provokes meibomian hypersecretion in humans. Histolopathological studies of the meibomian glands of rhesus monkeys after ingestion of polychlorinated biphenyl revealed characteristics identical to those of human meibomitis.44 This suggests that environmental pollutants may explain some cases of meibomian dysfunction.36 Topical epinephrine Topical epinephrine administration to rabbits interferes with meibomian secretion by obstructing the glands ducts through hyperkeratinization and epithelial cell desquamation.20,45,46 Obstruction of meibomian orifices Lesions of the ocular surface identical to those of keratoconjunctivitis sicca are observed in rabbits.47 Isotretinoin and irradiation Both these agents provoke glandular atrophy and an increase in tear viscosity and osmolarity, partly because of ductal hyperkeratinization.48,49 Mechanisms All these models point to an obstructive mechanism involving duct hyperkeratinization, leading to obstruction of the glandular orifices, tear film instability, excessive tear evaporation, ocular surface lesions, and a dry eye syndrome.50 Meibomian dysfunction can lead to blepharitis, and more precisely to meibomitis and keratoconjunctivitis. This is what is currently designated by the term ocular rosacea, the diagnosis of which does not require the presence of cutaneous involvement.7 Also, it should be remembered that ocular rosacea is not synonymous with meibomian dysfunction, which also occurs in other conditions such as seborrheic dermatitis and inflammatory oculopalpebral disorders (especially ocular allergies).10 Fig. 5.6 Ocular rosacea: immunoperoxidase staining shows a marked inflammatory infiltrate (helper T cells) in the conjunctival epithelium and subepithelial stroma (original magnification x 20) Table 5.2 Therapeutic guidelines for ocular rosacea
| I. Well-tolerated symptoms (dryness, irritation, transient conjunctival redness, etc.): —Daily lid hygiene (life-long) —Repeated ocular lubrication with preservative-free saline |
| II. Severe incapacitating symptoms and/or fibrosing conjunctivitis and/or severe meibomitis associated with recurrent chalazia and/or corneal complications (epithelial punctate keratitis, ulcerations, scars, neovascularization) —Lid hygiene 1-3 times per day —Repeated ocular lubrication with preservative-free saline + —Oral antibiotic therapy, 2- to 6-week cycles, the number of cycles depending on severity and the response to treatment (patients must be warned about of potential adverse effects and drug interactions) Drug options: Tetracycline 250 mg q.i.d. Doxycycline 50 mg b.i.d. Minocycline 50 or 100 mg b.i.d. Children under 8 years: erythromycin 30-50 mg/kg per day —Adjunctive therapy: Punctal plugs Short-course topical corticosteroid therapy for acute ocular surface inflammatory flare-ups Topical antistaphyloccocal therapy in case of positive smears and cultures |
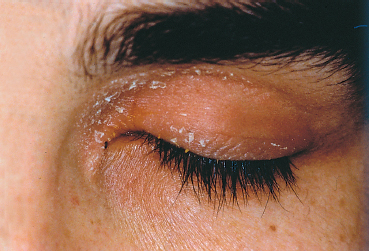
Fig. 5.7 Blepharitis in a patient with seborrheic dermatitis (note the characteristic pink skin covered with scales)
Ocular rosacea results from primary meibomitis, in which meibomian lipids are modified by the commensal flora. The flora is richer in this setting, containing, in order of decreasing frequency, Staphylococcus epidermidis, Propionibacterium acnes, Corynebacterium, and Staphylococcus aureus.51,52 The li-pases produced by these bacteria appear to be indirectly responsible for meibomitis, by favoring lipolysis of the cholesterol esters present in meibum and the release of toxic free fatty acids. Direct lipase toxicity and an immune reaction (delayed cell-mediated hypersensitivity) to an exotoxin produced by commensal bacteria may also play a role in the inflammation associated with ocular rosacea.53
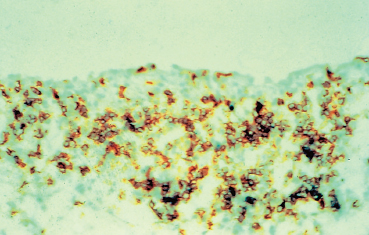
Few histological or immunopathological studies have been done on the conjunctiva in ocular rosacea.54–57 An im-munoenzymatic study with monoclonal antibodies directed against surface antigens and cell receptors suggested that the inflammation of the lids and ocular surface in rosacea was mainly due to the presence in the tear film of inflammatory mediators and antigenic stimuli stemming from the eyelid margins (Fig. 5.6).57
As Demodex folliculorum feeds on sebum and attracts a few lymphocytes when it degenerates, it has often been held responsible for meibomitis.58–60 Yet it is found in 50% of the normal population, and in 90% of the very elderly. Lash epilation for parasitological examination therefore appears unnecessary.61–63
Classification
Ocular rosacea is classified under the term blepharitis, which covers a wide variety of conditions. There is no consensus on the classification of blepharitis, and this explains the multitude of terms used in the different publications to designate what may in fact be the same condition, such as chronic blepharitis, chronic keratoconjunctivitis, meibomitis, seborrheic meibomitis, meibomian conjunctivitis, seborrheic conjunctivitis, and seborrheic blepharokerato conjunctivitis. Some classifications focus on the clinical aspect of the eyelids, others on the qualitative and quantitative nature of the secretions or the aspect of the orifices, and still others on the result of tests such as the Schirmer test, tear osmolarity, or meibography.10,13,64–69 All are useful, but they are not comparable because they are not based on the same criteria. All authors agree, however, on the key pathogenic role of meibomian gland dysfunction.
Generally, staphylococci are responsible for blepharitis that predominates on the anterior lid margin and is accompanied by madarosis, scales, and crusts at the base of the lashes. In contrast, chronic inflammatory meibomian dysfunction affects the posterior lid margin. McCulley’s classification (1982)10 schematically distinguishes the following types of meibomian dysfunction:
—Meibomian hyperseborrhea, with or without meibomitis;
—Primary meibomitis associated with ocular rosacea and seborrheic dermatitis (Fig. 5.7), that is responsible for potentially severe keratoconjunctivitis;
—Meibomitis secondary to other conditions such as atopy and psoriasis.
It is therefore important to assess the meibomian secretions (hypersecretion or hyposecretion), the presence of inflammation (meibomitis) and/or an associated dry eye syndrome, and the skin (especially the face).
The Eyelid-Conjunctival Complex
Qualitative disorders of meibomian secretion manifest themselves clinically at the level of the eyelids as nonspecific signs such as telangiectasia of the lid margins, which are hyperemic, irregular, and thickened (Fig. 5.8). Meibomian hyperseborrhea, which takes the form of large clear lipid droplets at the gland orifices, is very frequently complicated by meibomitis. The orifices of the meibomian glands are occluded by yellowish, solidified meibum plugs mixed with epithelial debris. These concretions take on the aspect of spicules or domes that can only be removed with the tip of a needle. The meibomian orifices are often displaced backward, and are less numerous and visible than in healthy subjects. Digital pressure to the lid margin expresses a more or less viscous and cloudy, sometimes granular or doughy material from the meibomian orifices (Fig. 5.9), while the meibum is normally fluid and transparent. This maneuver may be unproductive is some cases. There is often a history of chalazia (Fig. 5.10), recurrent erosions and contact lens intolerance with giant papillary conjunctivitis and deposits on the lens.68
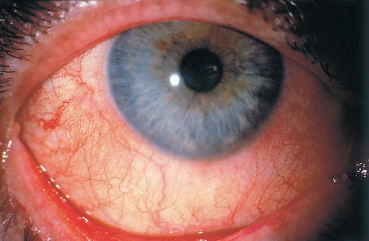
As the lipid film no longer fulfills its stabilizing effect on the tear film, excessive evaporation occurs, leading in turn to symptoms of dryness, which are by far the most frequent manifestation of ocular rosacea. Not all patients with rosacea have a quantitative tear deficiency, but Lemp et al. reported that 36.6% of 60 patients had an abnormal Schirmer test without anesthesia, versus only 4.1 % of 120 healthy subjects.70 The intensity of symptoms is not always proportional to the severity of morphological changes in the eyelid margins. Ocular manifestations are nonspecific, and may include a burning sensation, a foreign body sensation, irritation, and sometimes itching and visual fluctuations. Many patients themselves complain of a sensation of dryness. The conjunctiva may be white or, on the contrary, diffusely hyperemic, especially the bulbar part (Fig. 5.11). Most patients have a history of chronic conjunctivitis with exacerbations or remissions.71,72 Cases of nodular conjunctivitis corresponding histologically to granulo-matous inflammation and phlyctenular conjunctivitis have also been reported.73
Complications
Dryness is not the only consequence of chronic meibomitis. More or less severe palpebral and corneoconjunctival complications can also occur, and carry a risk of permanent functional visual loss.
The most serious conjunctival complication is fibrosis (Fig. 5.12), yet it is so often overlooked that it is sometimes omitted from the list of classic signs in publications on ocular rosacea, the principal cause of fibrosis being ocular cicatricial pemphigoid.74 Fibrosis presents clinically as subepithelial white lines, which are often branched. It can sometimes lead to fornix foreshortening and symblepharon formation. These complications are always accompanied by changes in the architecture of the lid margins, which are erythematous, thickened, and irregular. Secondary ectopic eyelashes are frequent, perpetuating conjunctival inflammation and causing keratitis.
The main corneal complication of ocular rosacea is keratitis, which can have various clinical aspects.8,72 Epithelial punctate keratitis is the most frequent, classically predominating in the lower part of the cornea. A form of more specific corneal involvement, perhaps pathognomonic for some,55 is interstitial keratitis composed of multiple inferior corneal opacities, vascularized by superficial neovessels coming radially from the limbus (Fig. 5.15). In children, this type of corneal lesion occurs in the setting of phlyctenular keratoconjunctivitis (Fig. 5.16).1,75 Inflammatory relapses are painful and can even lead to corneal perforation. The other corneal complications of rosacea are catarrhal infiltrates (Fig. 5.17), with or without ulceration, peripheral neovascularization of all forms (Figs. 5.18 and 5.19), with or without lipid exudation, and peripheral ulcerative keratitis resembling Mooren’s ulcer (Fig. 5.20). Scleritis or episcleritis (Fig. 5.13) is relatively frequent, and may be associated or isolated.73
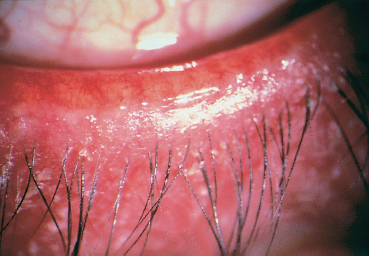
Fig. 5.8 Meibomitis
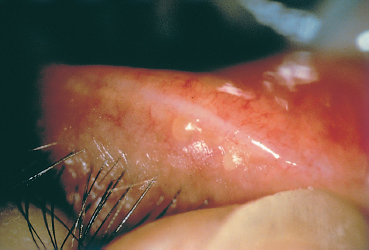
Fig. 5.9 Digital pressure to the lids expresses thick yellow meibomian secretions
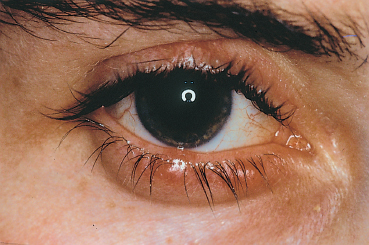
Fig. 5.10 Blepharitis and chalazia
Fig. 5.11 Ocular rosacea: bulbar conjunctival hyperemia
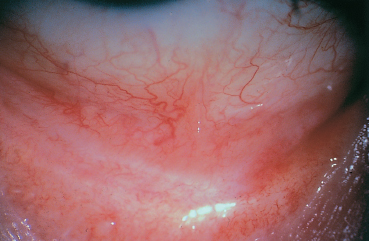
Fig. 5.12 Conjunctival fibrosis secondary to ocular rosacea
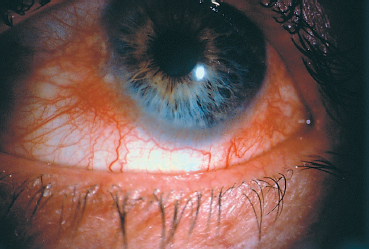
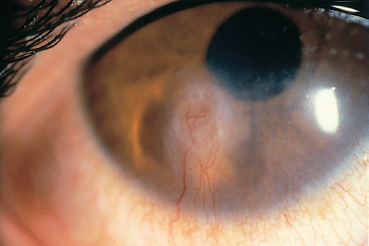
Fig. 5.13 Ocular rosacea: episcleritis
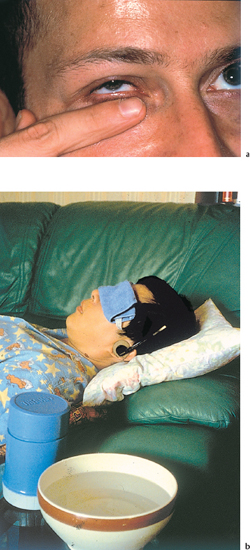
Fig. 5.14a and b Lid hygiene
Fig. 5.15 Ocular rosacea: corneal vascularization and scarring
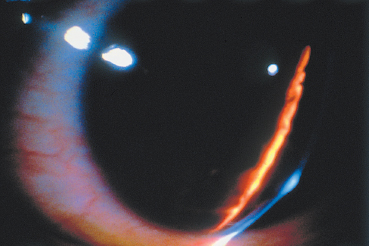
Fig. 5.16 Corneal phlyctenule
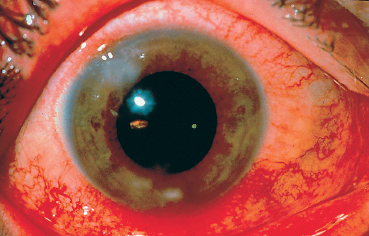
Fig. 5.17 Catarrhal corneal infiltrates
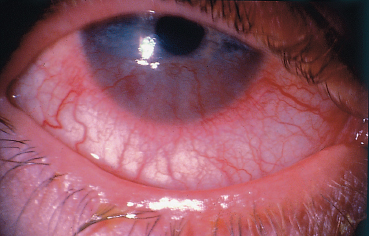
Fig. 5.18 Ocular rosacea: peripheral corneal neovascularization
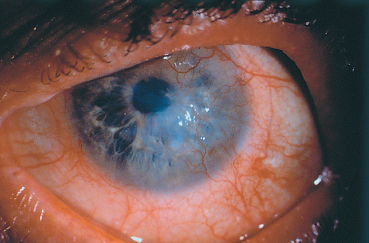
Fig. 5.19 Ocular rosacea: peripheral corneal neovascularization
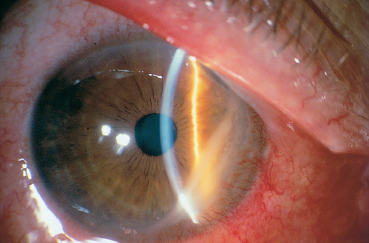
Fig. 5.20 Ocular rosacea: peripheral ulcerative keratitis
Differential Diagnosis
Too often ignored or underestimated as a cause of conjunctival inflammation, ocular rosacea can, on the contrary, be overdiagnosed or be considered, wrongly, as the only cause of the clinical manifestations.
For example, discrete erythema of the eyelid margins and obstruction of a few meibomian orifices are common in aging subjects, but this does not necessarily mean that the meibum is impaired.76
The principal differential diagnoses are dryness, allergy, and toxic conjunctivitis. These diagnoses are particularly difficult to rule out because they can be associated with ocular rosacea.
Tear Deficiency-Induced Dry Eye Syndrome
In ocular rosacea, inadequate meibomian secretion due to mei-bum hyperviscosity leads to tear film instability, as demonstrated by the shortened tear film break-up time. The consequence is dryness due to excessive tear evaporation, a condition that must be distinguished from the dry eye syndrome due to tear insufficiency.24,77 The Schirmer test is not very useful for distinguishing between the two mechanisms. Tear production can appear to be increased because of reflex tearing, even though the meibomian glands are less numerous.78 However, chronic meibomitis may be associated with an authentic tear deficiency in 25-40% of cases.75,79,80
Ocular Allergy
Conjunctival allergy and ocular rosacea are often confused, especially as they share almost the same symptoms and they can be associated. For instance, secondary meibomitis is common in atopic keratoconjunctivitis,10 and patients with ocular rosacea sometimes develop authentic allergic hypersensitivity reactions to eyedrops (especially their preservatives), which are excessively prescribed in this setting. The hypersensitivity manifestations will be amplified when the eyes are dry, as this increases the allergen concentration on the ocular surface. Slit-lamp examination of a patient with ocular rosacea must always include inspection of the palpebral conjunctiva, especially the upper tarsal one, to detect any marked papillary hyperplasia and hypertrophy reflecting an allergic hypersensitivity reaction. Some authors have reported that moderate papillary conjunctivitis is a common feature of ocular rosacea.81 IgE detection in tears can be helpful to ascertain the diagnosis of ocular allergy.82
Toxic Conjunctivitis
The preservatives contained in eyedrops are known not only for their allergizing properties but also for their conjunctival toxicity.83 The latter can cause chronic conjunctivitis and fibrosis. Benzalkonium, one of the most widely used eyedrop preservatives, is also a cationic detergent that destabilizes the tear film.84 Toxic conjunctivitis is often difficult to diagnose because of its frequent association with allergy and dry eye syndrome. Toxic conjunctivitis should be suspected in the presence of numerous follicles in the inferior palpebral conjunctiva and fornix, epithelial punctate keratitis predominating in-feriorly, and punctate dye-staining of the medial bulbar conjunctiva.
Treatment (see Table 5.2)
The patient must be informed from the outset that ocular rosacea is a chronic condition, that treatment will no doubt be lengthy (perhaps life-long), and that inflammatory reactivations may still occur, even when treatment is correctly conducted.
Lid Hygiene
Daily lid hygiene is the cornerstone in the treatment of ocular rosacea.85 Patients must understand the underlying principles if they are to carry out these relatively time-consuming care procedures correctly. As the meibomian secretions lack fluidity, the eyelids must be warmed by applying for several minutes a facecloth or packs soaked in warm water. The patient must be shown how to perform a firm and effective finger massage of the four eyelids (Fig. 5.14). Finally, the eyes are rinsed with preservative-free saline. The lid hygiene procedure may be repeated twice or three times a day depending on the severity of the condition. Lid hygene should be ideally performed in the morning, because of the nocturnal meibum accumulation. Some authors recommend cleansing the lid margins with a cotton tip dipped in baby shampoo. However, this is often irritant and there is a risk of corneal traumatic erosion. Interestingly, in manuscripts written in the first half of the 20th century, ophthalmologists also advised patients with blepharitis to massage their eyelids to express the staphylo-cocci contained in the glands.86–88
Antibiotic Treatment
Many publications in the dermatological and ophthalmological literature56,90–92 have confirmed the efficacy of oral tetracy-cline in rosacea, as first reported by Sneddon in 1966.89 The tetracycline derivatives oxytetracycline93 and doxycycline,94,95 and also topical tetracycline,96 have since proved their efficacy in ocular rosacea. Minocycline also seems safe and effective.96 a Recommended oral daily doses are 250 mg q.i.d. tetracycline, 50 mg b.i.d. doxycycline, and 50 or 100 mg b.i.d. minocycline, and the duration of treatment is empirical, usually at least 15 days to one month.
The mode of action of the cyclines is not clear. They appear to act by reducing lipase production by staphylococci present in the commensal flora, thereby reducing the release of toxic free fatty acids through meibomian lipid hydrolysis.97 They also have anticollagenase activity, suppress neovascularization in rabbits, and have an antichemotactic effect.98–100
Tetracycline and its derivatives must be reserved for severe forms of ocular rosacea, especially those with recurrent corneal involvement, symblephara, and chalazia modifying the lid architecture. The cyclines in no way obviate or reduce the need for regular lid hygiene. Relapses are frequent on antibiotic withdrawal (25% after 1 month and 66% after 6 months),92 necessitating reinduction or chronic therapy.
The contraindications of tetracycline must be respected,91 namely age less than 8 years, pregnancy, breast-feeding, hepatic and renal failure, drug allergy, and chronic bronchitis. Er-ythromycin (30-50 mg/kg per day) can be used instead of cyclines for children under 8 years of age. If treatment with one cycline fails, another should be tried, given the frequent absence of cross-resistance. Pigmentation of the skin, teeth, conjunctiva, and sclera is a risk with minocycline,101,102 as well as benign intracranial hypertension.
Metronidazole gel has shown relative efficacy in combination with lid hygiene.85 Topical corticosteroid eyedrops are only indicated to control an acute inflammatory exacerbation, and only for a brief period. Fusidic acid 1 % gel, through its immunosuppressive action, may also be beneficial in ocular rosacea.103 Note that isotretinoin, which is very effective against acne, can worsen blepharoconjunctivitis. Finally, permanent or temporary occlusion with punctal plugs can relieve the symptoms of dryness.
References
3. Drolet B, Paller AS. Childhood rosacea. Pediatr Dermatol 1992; 9: 22-6
4. Browning DJ, Rosenwasser G, Lugo M. Ocular rosacea in blacks. Am J Ophthalmol 1986; 101: 441-4
5. Rosen T, Stone MS. Acne rosacea in blacks. J Am Acad Dermatol 1987; 17: 70-3
6. Berg B, Liden S. An epidemiological study of rosacea. Acta Derm Venereol 1989; 69: 419-23
8. Browning DJ, Proia AD. Ocular rosacea. Surv Ophthalmol 1986; 31: 145-58
9. Starr PA. Oculocutaneous aspects of rosacea. Proc R Soc Med 1969; 62: 9-11
16. Lempert SL, Jenkins MS, Brown SI. Chalazia and rosacea. Arch Ophthalmol 1979;97 : 1652-3
18. Meibomius H. De Vasis Palpebrarum Novis Epistola Muller. Helmstädt 1666
21. Norn MS. Expressibility of meibomian secretion. Acta Ophthalmol 1987; 65 : 137-42
22. Weingeist TA. The glands of the ocular adnexa. Int Ophthalmol Clin 1973;13 : 243-61
23. Mathers WD. Ocular evaporation in meibomian gland dysfunction. Ophthalmology 1993; 10 : 347-51
25. Andrews JS. The meibomian secretion. Int Ophthalmol Clin 1973; 13 : 23-8
26. Browning DJ. Tear studies in ocular rosacea. Am J Ophthalmol 1985; 99 : 530-3
32. Tapie R. Etude biomicroscopique des glandes de Meibomius. Ann Ocul 1977; 210 : 637-48
39. Norn MS. Foam at outer palpebral canthus. Acta Ophthalmol 1963; 41 : 531-7
43. Tiffany JM. The lipid secretion of the meibomian glands. Adv Lipid Res 1989; 22 : 1-62
52. Groden LR, Murphy B, Rodnite J, Genvert GI. Lid flora in blepharitis. Cornea 1991; 10 : 50-3
60. Rufli T, Büchner SA. T-cell subsets in acne rosacea lesions and the possible role of Demodex folliculorum. Dermatologica 1984; 169 : 1-5
61. Marks R, Harcourt-Webster JN. Histopathology of rosacea. Arch Dermatol 1969; 100 : 682-91
64. Gifford SR. Meibomian glands in chronic blepharoconjunctivitis. AmJ Ophthalmol 1921; 4 : 489-94
65. Cowper HW. Meibomian gland seborrhea. AmJ Ophthalmol 1922; 5 : 25-30
71. Goldsmith AJB. The ocular manifestations of rosacea. Br J Dermatol 1953; 65 : 448-57
72. Jenkins MA, Brown SI, Lempert SL, Weinberg RJ. Ocular rosacea. Am J Ophthalmol 1979; 88 : 618-22
73. Akpek EK, Merchant A, Pinar V, Foster CS. Ocular rosacea. Ophthalmology 1997; 104 : 1863-7
74. Foster CS. Cicatricial pemphigoid. Trans Am Ophthalmol Soc 1986; 84 : 527-663
77. McCulleyJP, Sciallis GF. Meibomian keratoconjunctivitis. Am J Ophthalmol 1977; 84 : 788-93
83. Baudouin C. Encycl Méd Chiru (Elsevier, Paris), Ophtalmologie, 21-150-A-20 1998, 8p.
87. Wolff J. Hypersecretion of the meibomian glands. Arch Ophthalmol 1912; 41 : 383
89. Sneddon IB. A clinical trial of tetracycline in rosacea. Br J Dermatol 1966; 78 : 649-52
90. Marmion VJ. Tetracyclines in the treatment of ocular rosacea. Proc R Soc Med 1969; 62 : 11-2
91. Salamon SM. Tetracyclines in Ophthalmology. Surv Ophthalmol 1985; 29 : 265-75
96. Roper Hall MJ. Ocular aspects of rosacea. Trans Ophthalmol Soc UK 1966; 86 : 727-32.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree