Fungi are the causative agents of a number of pathologic conditions involving the nasal cavities.
8,9 Commonly, they are related to allergic rhinitis and thus may play some role in the development of chronic sinusitis and secondary complications from such. They also play a role in a number of other pathologic conditions of the sinuses, in which they can be either
noninvasive or
invasive.
Allergic Fungal Sinusitis
Allergic fungal sinusitis is the most common fungal disease of the sinuses. Clinically, patients with this disease are immunocompetent and may have concomitant asthma, allergic rhinitis, and nasal polyps.
10,11 and
12 The disease is
suspected after a protracted history of sinusitis that has been refractory to treatments for bacterial infection. It may be secondary either to hyaline molds such as
Aspergillus or
Fusarium species or to various dematiaceous molds.
10,11,12 and
13Surgically collected specimens will grossly have tan-brown fragments of mucosa with green-brown mucoid material and have evoked for some the culinary impressions of cottage cheese and peanut butter.
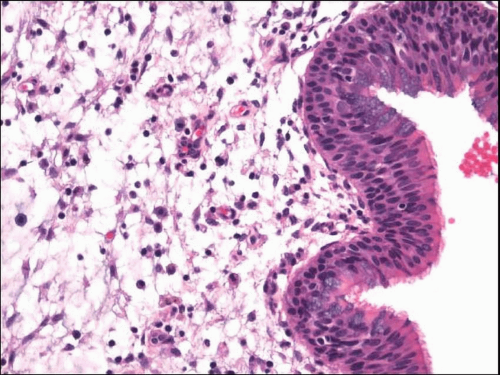
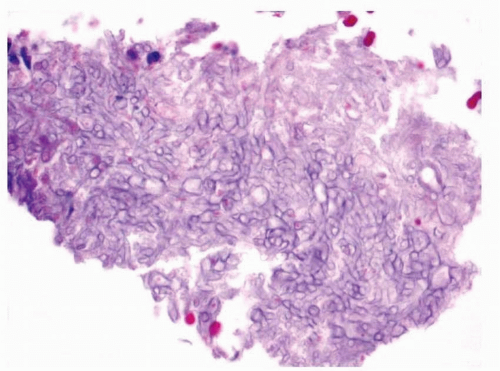
8 Microscopically, samples show abundant mucus with entrapped eosinophils and Charcot-Leyden crystals
(allergic mucus) (
Fig. 13.3, e-
Fig. 13.2).
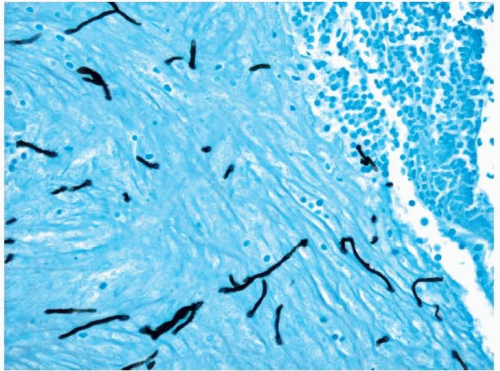
10,12 Although the fungal forms may be difficult to visualize with routine hematoxylin and eosin-stained material, silver staining will show numerous hyphal fragments (
Fig. 13.4, e-
Fig. 13.3). The hyphal fragments often branch at 45° angles and may have occasional conidia.
12 Fragments of edematous and inflamed respiratory mucosa will also be present, showing changes consistent with chronic sinusitis, and should not contain invasive organisms when viewed with special stains.
Sinus Mycetoma (Fungus Ball)
Sinus mycetomas most often occur in the maxillary sinuses of immunocompetent individuals.
8,14 Patients typically present with headache, facial pain, nasal obstruction, or a perceived musty odor. They sometimes have concomitant nasal polyps and chronic sinusitis, and they may even develop new-onset seizures. The disease is most often caused by
Aspergillus fumigatus.Removed material will appear mucopurulent or cheesy.
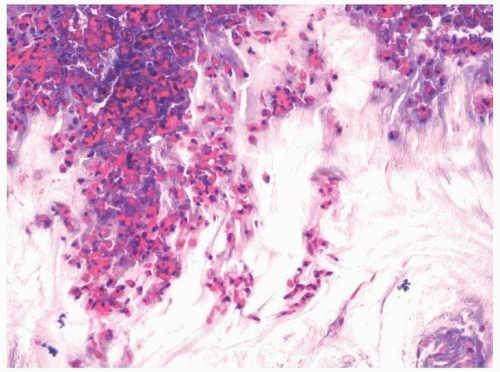
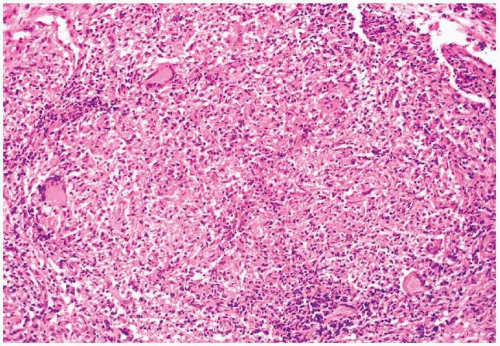
8 On microscopic examination, a dense collection of fungal elements will be seen, devoid of allergic mucus (
Fig. 13.5). Silver staining is rarely needed due to the abundance of the organisms (e-
Fig. 13.4). The fragments of the sinus tissue will show changes in chronic sinusitis, and fungi should not involve (invade) fragments of respiratory mucosa.
Invasive Fungal Sinusitis
Invasive fungal sinusitis is a disease that is generally restricted to immunocompromised individuals, although rare instances are noted in which the disease has affected apparently immunocompetent persons.
8,14,15,16 and
17 Acute or fulminant invasive fungal sinusitis may present as a painless, black, nasal septal, or palatal eschar. It is most often caused by fungi of the order Mucorales such as
Rhizopus and
Mucor, but may also be caused by
Aspergillus and
Fusarium species, as well as dematiaceous fungi.
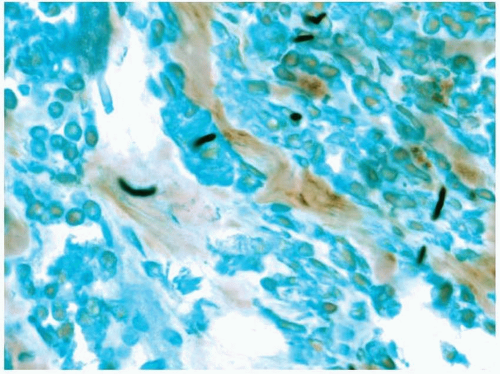
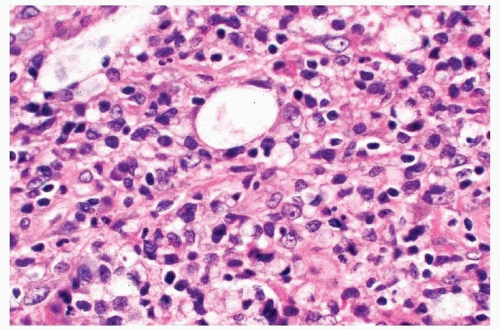
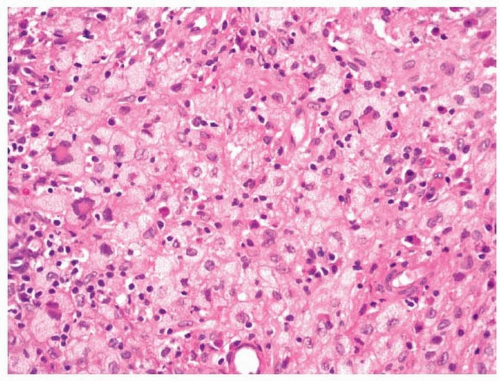
Microscopically, biopsy specimens from patients with acute fungal sinusitis will show invasion of tissue by the hyphal elements, often with involvement of vascular structures (
Fig. 13.6, e-
Fig. 13.5).
8,9,15 A secondary vasculitis and thrombosis with resultant hemorrhage and infarction are common. Surrounding inflammation is often minimal, a reflection of the immunocompromised state of the patient. Some have suggested the use of in situ hybridization for the typing of the etiologic agents.
18Chronic invasive fungal sinusitis follows a more protracted course than acute invasive fungal sinusitis.
8,9 Patients are immunocompromised, usually secondary to diabetes mellitus or previous treatment with corticosteroids. The disease may be associated with decreased vision and ocular immobility (orbital apex syndrome). Some believe that the lesion begins as a sinus mycetoma that eventually becomes invasive due to the immunocompromised state of the patient. The disease is most often secondary to infection by
A. fumigatus. As with acute invasive sinusitis, histologic sections show tissue and vascular invasion by hyphal elements, often with a limited inflammatory response.
Finally, paranasal granuloma or granulomatous invasive fungal sinusitis is a rare chronic fungal disease of immunocompetent patients that is associated with proptosis.
8 The disease is secondary to infection by
Aspergillus flavus. Most reports of this lesion come from Sudan.
Microscopically, tissue invasion is present, which induces nonnecrotizing granulomatous inflammation with intermixed plasma cells. At the centers of these granulomata, a small amount of fibrinoid necrosis with eosinophils may be present. An associated vasculitis may also be seen.