CHAPTER 177 Neoplasms of the Posterior Fossa
Approach to Diagnosis of Neoplasms of the Posterior Fossa
Posterior fossa skull base neoplasms may be grouped into common CPA lesions, including lesions of the internal auditory canal; petrous apex lesions; rare CPA lesions; and intra-axial lesions. The signs, symptoms, and diagnostic procedures for tumors within the designated categories (Box 177-1) are described next.
Common Cerebellopontine Angle Neoplasms
In published series of CPA neoplasms, acoustic neuromas are the most common tumors, accounting for more than 90%. The remaining primary tumors were meningiomas (3%), primary cholesteatomas (2.5%), and facial nerve schwannomas (1%), with less common tumors composing the remaining lesions.1 When secondary tumors also are considered, paragangliomas constitute up to 10% of CPA neoplasms.2
Acoustic Neuroma
Type 2 neurofibromatosis, NF2, is the central form of the disease, characterized by bilateral acoustic neuromas in up to 96% of patients. Schwannomas of the other cranial nerves, meningiomas, and ependymomas also are observed much more commonly in patients with NF2.3 The precise frequency of NF2 is unknown, but it is far less frequent than NF1. The gene responsible for the condition encodes a membrane protein (merlin or schwannomin) and is located on chromosome 22. Acoustic neuromas in NF2 are characterized by an onset early in life, often before the age of 21 years, as opposed to unilateral lesions, most of which are diagnosed between the ages of 40 and 60 years. Thus, acoustic neuromas appearing before the age of 30 years mandate particularly close evaluation of the contralateral ear. Although the acoustic neuromas in NF1 and NF2 resemble the lesions in nonhereditary acoustic neuromas, they are technically more challenging to remove because of a tendency to adhere to nearby structures. The clinical presentation of acoustic neuroma in neurofibromatosis is identical to that of unilateral acoustic neuroma.
Malignant schwannomas may rarely occur. They often are associated with neurofibromatosis, but they also may occur with solitary schwannomas. Another very uncommon variant is a pigmented schwannoma.4
Natural History
The growth rate of acoustic neuromas is extremely variable. These tumors generally are slow-growing, with average reported growth rates of 0.2 cm per year; however, growth rates in excess of 2 cm per year have been documented. Acoustic neuromas that are not treated are potentially lethal. Gradual enlargement can lead to indentation of the brainstem, increased intracranial pressure, and death during a course of 5 to 15 years.5
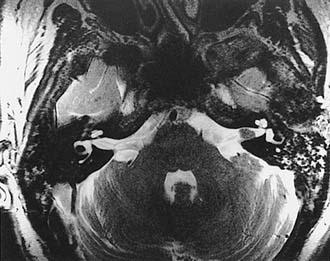
Growth of acoustic neuromas generally occurs in three phases: internal auditory canal, cisternal, and brainstem compression (Figs. 177-1, 177-2, and 177-3). Growth within the internal auditory canal results in acoustic and facial nerve compression and attenuation. Displacement of the seventh cranial nerve, the acoustic (eighth) nerve, and the anterior inferior cerebellar artery occurs just medial to the porus acusticus (cisternal portion). Medial tumor growth augments the blood supply of the tumor with bridging vessels from the brainstem surface. Fourth ventricle shift often occurs when the CPA component reaches 2 to 3 cm and total ventricle obstruction with resulting hydrocephalus occurs with continued tumor growth. Trigeminal compression occurs at approximately the 3-cm stage, which permits the superior portion of the tumor to abut the fifth cranial nerve. Massive tumor growth with hydrocephalus, brainstem compression, and cerebellar tonsil herniation may occur even with modern imaging. The otolaryngologist should remain vigilant for signs and symptoms suggestive of retrocochlear pathology.
Signs and Symptoms
Sensorineural hearing loss (SNHL), tinnitus, dysequilibrium, and facial hypesthesia are, in descending order, the most common symptoms of acoustic neuroma. Although progressive unilateral SNHL is the most common symptom, loss of speech discrimination is characteristic of retrocochlear dysfunction of the cochlear nerve presumably from pressure on the auditory nerve. The symptoms usually are slowly progressive, with a median duration of 2 years in one series.6,7 However, up to 20% of affected patients experience sudden SNHL, although complete recovery of hearing has been reported.8 Overall, only 1% of patients with sudden SNHL are found to have acoustic neuroma, whereas up to 5% of patients with acoustic neuroma have normal hearing.9 Thus, any patient with asymmetric or sudden hearing loss (even after total recovery) should be considered for further evaluation for a possible retrocochlear lesion.
Diagnostic Studies
Audiometry
Routine air, bone, and speech discrimination studies may suggest the possibility of an acoustic neuroma. Asymmetric SNHL or impairment of speech discrimination disproportionate to the pure tone loss requires specific investigation for a retrocochlear lesion. In the past, an extensive audiometric test battery was necessary for timely tumor diagnosis.6,10 In the magnetic resonance imaging (MRI) era, specialized audiometric tests are not sensitive or specific enough to detect small acoustic neuromas consistently.11
Auditory Brainstem Response Testing
Until recently, auditory brainstem (evoked) response (ABR) testing was considered the most sensitive modality for the detection of even small tumors, with detection rates of 95% to 100%.12 The brainstem response to an 83-dB broadband click is recorded while the contralateral ear is masked by 78-dB white noise.13 The latency for detection of wave V for the two ears is compared, and an adjusted interaural latency for wave V greater than 0.2 msec is considered abnormal. An approximately 10% false-positive rate has been reported for patients with SNHL who do not have a CPA tumor. Since the advent of gadolinium-enhanced MRI, however, the ABR false-negative rate (missing tumor diagnosis) has been 18% to 30% for intracanalicular tumors.14 The possibility of false-negative results with intracanalicular acoustic neuromas has lessened the role of ABR testing for routine screening.15 Stacked ABR testing is a modified ABR technique that offers improved sensitivity for detection of intracanalicular acoustic neuromas. It consists of testing the audiometric spectrum in a frequency-specific fashion followed by temporal alignment of the results; amplitude changes thus detected are much more sensitive for diagnosing small acoustic tumors than with routine ABR.16,17 Nonetheless, the technique has not yet been verified in widespread clinical practice.
Vestibular Tests
Vestibular testing is not a useful screening test for acoustic neuroma. Electronystagmography (ENG) and infrared video caloric testing are helpful in defining whether the tumor arises from the superior or the inferior vestibular nerve.18 Such information is valuable when hearing preservation surgery for acoustic neuromas is under consideration, because theoretically better results may be obtained with resection of tumors of the superior vestibular nerve.
Imaging
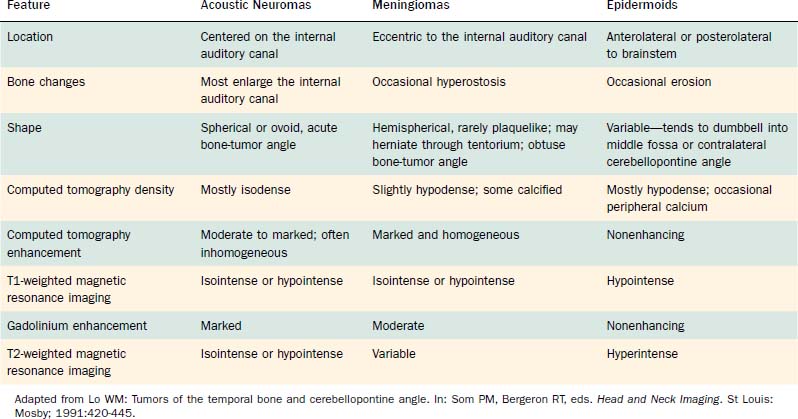
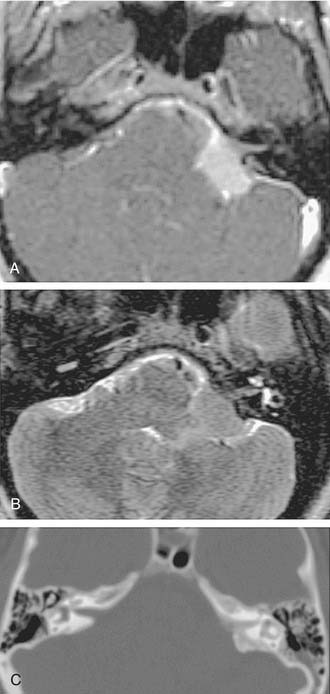
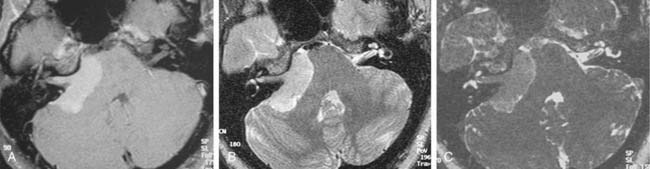
Computed tomography (CT) and MRI with the paramagnetic intravenous contrast agent gadopentetate (gadolinium-DTPA) are the principal imaging modalities for CPA lesions. Lo19 proposed a scheme for the CT or MRI differential diagnosis of CPA lesions by anatomic location (extra-axial, extradural, or intra-axial) and by incidence (rare or common) (Table 177-1). In adults and teenagers acoustic neuromas, meningiomas, and epidermoids are the three most common lesions. Acoustic neuromas are very rare in children. Instead, brainstem gliomas (which can enlarge the internal auditory canal) are the most common CPA lesion in younger children.20 Distinguishing among the three most common CPA lesions is based on specific imaging characteristics on CT and MRI (Table 177-2).
Table 177-1 Differential Diagnosis of Cerebellopontine Lesions by Imaging Location* and Incidence
| Location | Incidence | Type |
|---|---|---|
| Extra-axial | Most common | Acoustic neuroma |
| Common | Meningioma | |
| Common | Epidermoid (and other cysts: arachnoid, cysticercal, dermoid) | |
| Rare | Nonacoustic neuromas (cranial nerves V, VII, IX, X, XI, XII) | |
| Rare | Vascular lesions (loops, aneurysms, malformations) | |
| Extradural | Common | Paraganglioma (glomus jugulare, glomus vagale) |
| Rare | Bone lesions (benign or malignant; primary or metastatic) | |
| Intra-axial | Rare | Astrocytoma, ependymoma, papilloma, hemangioblastoma, metastasis |
* Computed tomography or magnetic resonance imaging.
Adapted from Lo WM. Tumors of the temporal bone and cerebellopontine angle. In: Som PM, Bergeron RT, eds. Head and Neck Imaging. St Louis: Mosby; 1991:420-445.
Although CT revolutionized the diagnosis of CPA tumors, gadolinium contrast MRI is sensitive and specific for detection of acoustic neuroma and is the diagnostic imaging technique of choice for the evaluation of acoustic neuroma and other CPA lesions (see Fig. 177-2).21,22 Acoustic neuroma images on MRI are isointense or mildly hypointense to brain on T1-weighted images and mildly hyperintense to brain on T2-weighted images.19 MRI for investigation of possible acoustic neuroma should be performed with intravenous contrast. Gadolinium increases the diagnostic sensitivity of MRI on T1-weighted images, and detection of lesions as small as 3 mm has been reported.21 In addition to improving sensitivity in diagnosing acoustic neuromas, MRI is noninvasive, and the patient incurs no radiation exposure.
Before MRI, CT was the principal study for CPA lesions. Now the role of CT is to provide adjunctive information regarding the osseous structures surrounding a CPA lesion. It also can be the primary imaging modality in patients who cannot undergo MRI because of medical reasons (e.g., cardiac pacemaker or cochlear implant) or unmanageable phobic reactions. The CT finding characteristic of an acoustic tumor is an ovoid lesion centered on the internal auditory canal with moderate enhancing qualities. The tumor often is not homogeneous, exhibiting areas of lesser and greater enhancement. Approximately 85% of acoustic neuromas show acute angles at the bone-tumor interface, in contrast with meningiomas, in which the interface is obtuse in 75% of tumors.19
Intravenous contrast CT can fail to detect acoustic neuromas smaller than 1.5 cm in greatest dimension. Oxygen cisternography has been used to diagnose small, mainly intracanalicular lesions. In this technique, 4 mL of oxygen was introduced into the subarachnoid space through the opening afforded by a lumbar puncture, to highlight the structures of the internal auditory canal. This technique was very sensitive for detection of small lesions; however, it was limited by a false-positive rate of 5% and the need for a lumbar puncture.23
The improved availability of MRI has led to the development of MRI-based internal auditory canal screening protocols, which in many centers are comparable in cost to ABR-based internal auditory canal screening.11 Special T2-weighted fast-spin echo sequences may achieve the accuracy of T1-weighted sequences with gadolinium without the need for contrast (see Fig. 177-1).24 Although the software for these special sequences is not generally available, initial findings with this technique are very encouraging.
Meningiomas
Meningiomas represent up to 18% of all intracranial tumors and approximately 3% of CPA tumors.25 The cells lining the arachnoid villi are the cells of origin. These cells are distributed throughout the intracranial space predominantly in relation to veins and dural sinuses. Meningiomas are benign but locally aggressive tumors, which occur at different anatomic sites in the following order of frequency: parasagittal region, falx, convexity, olfactory groove, tuberculum sellae, sphenoid ridge, petrous face (CPA), tentorium, lateral ventricle, clivus, and others.26
The gross appearance typically is that of a globular mass firmly adherent to the dura mater, with characteristic speckles scattered throughout the tumor that correspond to the microscopic psammoma bodies. The tumor displaces but does not invade adjacent neural tissue and has a thin investing capsule. Meningiomas can invade bone without destruction by extension along haversion canals. Adjacent bone is hyperostotic in 25% of cases.26
Many histopathologic classifications have been proposed, but a single, widely used system distinguishes among syncytial, transitional, fibrous, angioblastic, and sarcomatous types.27 The specific histopathologic subtypes and growth characteristics are reviewed elsewhere.28 In the posterior fossa, meningiomas usually arise on the posterior surface of the petrous bone, away from or at the edge of the internal auditory canal, or along the sigmoid sinus. Because they usually arise outside of the canal, these tumors may become large before producing signs and symptoms of cranial nerve VIII compression. Most meningiomas eventually involve cranial nerve VIII.
Signs and Symptoms
Audiovestibular symptoms usually are the first indication of a posterior fossa meningioma. Among patients presenting to neurosurgeons, a higher proportion first experienced symptoms referable to the trigeminal nerve.29 The signs and symptoms of meningiomas are similar to those of acoustic neuromas. Small tumors produce hearing loss, tinnitus, and imbalance. Larger tumors also produce signs and symptoms of involvement of other cranial nerves and hydrocephalus.
Diagnostic Studies
Imaging
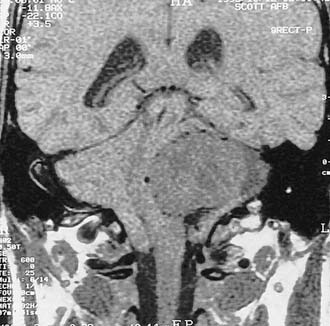
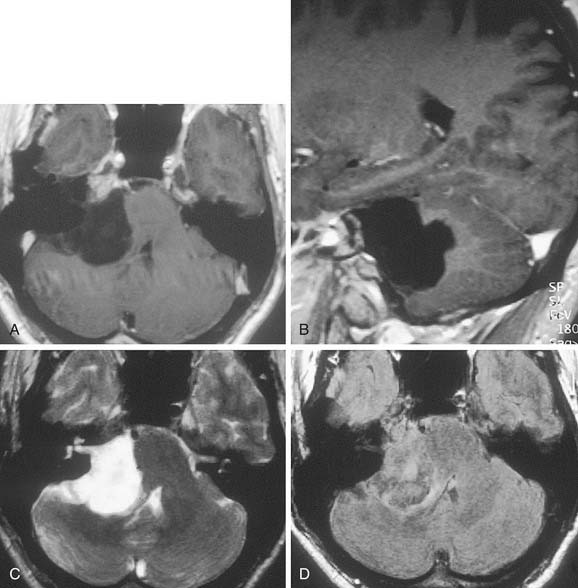
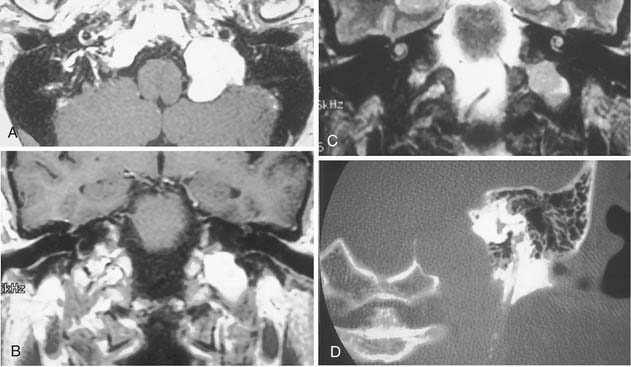
Table 177-2 summarizes features of meningiomas that assist in differentiating them from acoustic neuromas. Unlike acoustic neuromas, meningiomas usually are eccentric to the porus. Whereas acoustic neuromas seldom herniate into the middle fossa, approximately 60% of meningiomas extend to the middle fossa.19 Meningiomas usually are hemispheric because of their broad-based attachment to the posterior petrous wall, accounting for the obtuse bone-tumor angles in 75% of meningiomas. Unlike the site of origin of acoustic neuromas, that of posterior fossa meningiomas is varied (Figs. 177-4 and 177-5).
On MRI, meningiomas are of extremely variable intensity on T2-weighted images and isointense or slightly hypointense to brain on T1-weighted images. The different signal intensities among meningiomas correspond to different histopathologic subtypes.27 Surface flow voids on MRI correspond to marginal pial blood vessels, and arborizing flow voids represent active feeders to the tumor. Calcification and cystic foci cause heterogeneity on MRI images of meningiomas.
Primary Cholesteatomas
Diagnostic Studies
Auditory Testing
Auditory testing does not show any distinguishing features. As with other retrocochlear lesions, speech discrimination is poorer than would be expected from the degree of pure-tone loss. ABR also is frequently abnormal in primary cholesteatomas.30
Imaging
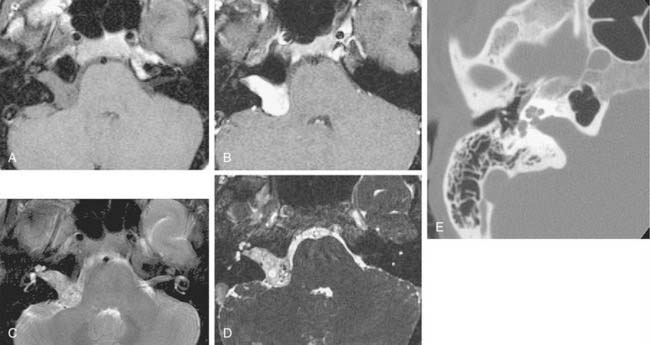
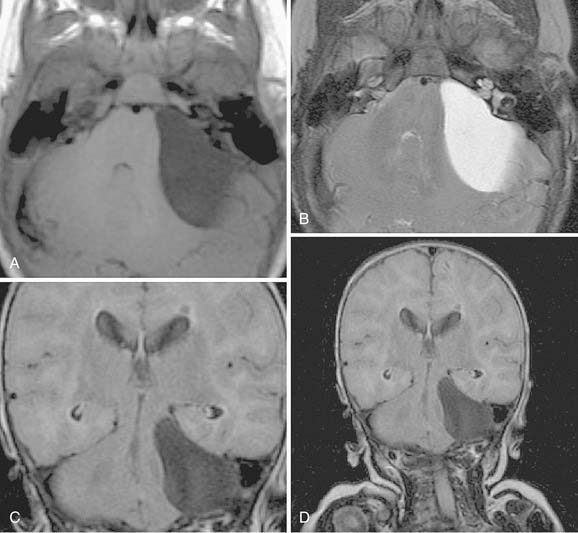
On CT, primary cholesteatomas are less dense than brain (having a density approximating that of cerebrospinal fluid [CSF]) and exhibit no enhancement with intravenous contrast. These lesions have irregular margins and are eccentric to the porus acusticus. Enhancing components suggest an associated malignancy.31 With MRI, primary cholesteatomas are inhomogeneous and hypointense to brain on T1-weighted images, and homogeneous and isointense or hyperintense to brain on T2-weighted images (Fig. 177-6). Schwannomas, meningiomas, and chondromas are similar to primary cholesteatomas by intensity criteria; however, epidermoids are differentiated because they are nonenhancing.
Arachnoid cysts of the CPA are difficult to distinguish from epidermoids on CT and MRI. Both lesions are of CSF density and nonenhancing; compared with primary cholesteatomas, however, arachnoid cysts have smoother surfaces, and special MRI sequences such as diffusion-weighted images (DWIs) may differentiate the intensity of the epidermoid from the CSF intensity of the arachnoid cyst (Fig. 177-7).
Facial Nerve Neuroma
Signs and Symptoms
Symptoms associated with these tumors depend on the portion of the nerve affected by the neoplasm. Peripheral involvement can manifest as a parotid mass, middle ear involvement can produce conductive hearing losses, and internal auditory canal or CPA involvement may result in SNHL. Unlike hemangiomas of the facial nerve, schwannomas do not produce facial weakness until they become very large. Sometimes a facial nerve tic is evident, which helps distinguish facial neuromas from acoustic neuromas but not from primary cholesteatomas. Of note, intratemporal lesions, with the possibility of neural entrapment, are more likely than CPA lesions to result in facial paralysis.32
Diagnostic Studies
Auditory Testing
The mechanisms for conductive hearing impairment and SNHL have already been described earlier in this chapter. Impedance testing may reflect motor fiber impairment on ipsilateral reflex testing or cranial nerve VIII involvement on contralateral reflex testing. ABR testing of tumors arising in the internal auditory canal shows abnormalities similar to those seen with acoustic neuroma.13
Electroneurography
Electroneurography, also known as electroneuronography (ENoG), measures the muscle response to a maximal bipolar stimulation of the facial nerve near the stylomastoid foramen. ENoG potentials may be reduced in facial nerve neuroma even when no facial weakness or tic is present, whereas ENoG findings remain normal in acoustic neuroma until the tumor becomes very large. Some reports suggest that the value of preoperative ENoG in patients with acoustic neuroma lies in predicting whether the lesion actually represents a facial nerve tumor.33,34
Imaging
Because facial nerve neuromas are histologically identical to acoustic neuromas, they have the same enhancement characteristics. It usually is impossible to identify these lesions using CT. Anterosuperior erosion of the internal auditory canal or erosion of the labyrinthine facial nerve canal if present may be the only diagnostic clue.19 More distal tumors enlarge the geniculate ganglion and fallopian canal.
As with CT, MRI of facial neuroma produces imaging characteristics identical to those of acoustic neuromas (Fig. 177-8). The preoperative diagnosis of intracranial facial neuroma is difficult. Early facial nerve symptoms are an obvious warning that a CPA lesion may rarely be a facial nerve neuroma. Patients diagnosed with CPA neuromas should be warned preoperatively of a 1% risk of facial nerve neuroma. This possibility is emphasized if findings on preoperative ENoG are abnormal on the tumor side. In some tumors, the lesion may extend from the CPA through the temporal bone course of the nerve to the extratemporal nerve in the parotid gland.
Other Cranial Nerve Neuromas
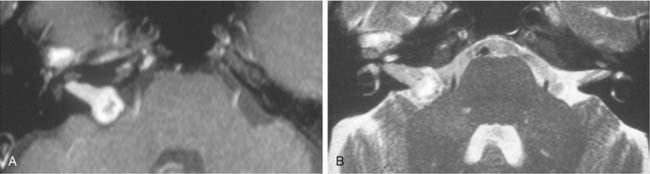
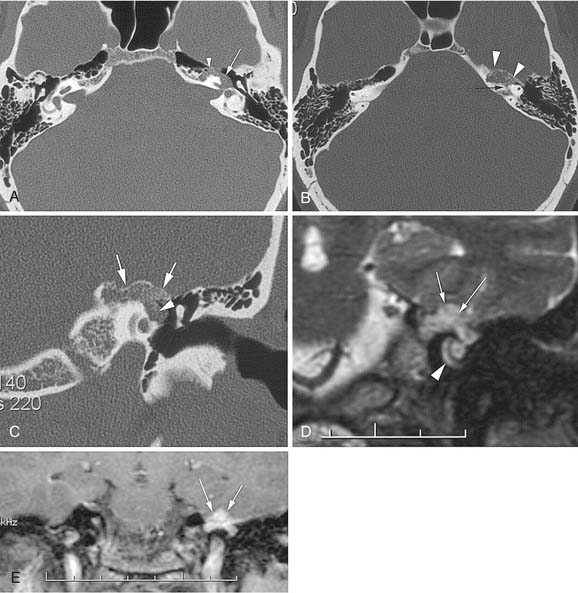
Trigeminal neuromas arise intradurally, from the nerve root in the CPA and Meckel’s cave, and extradurally, from the gasserian ganglion in the middle cranial fossa.35 Typically these lesions enlarge Meckel’s cave and produce hypesthesia of the face (Fig. 177-9).

Figure 177-9. Cystic trigeminal schwannoma, demonstrated by magnetic resonance imaging. A, T1-weighted image. B, Gadolinium T1-weighted image. C, T2-weighted image. The bulk of the tumor lies in the posterior fossa, with a small middle cranial fossa component enlarging the left Meckel’s cave (arrow). Note the similarity of the tumor to an arachnoid cyst (see Figure 177-7) on the noncontrast images (A and C). The tumor is nearly of CSF intensity because of the predominance of the intratumoral cystic components.
(From Jackler RK, Brackmann DE, eds. Neurotology. St Louis: Mosby; 1994.)
Neuromas of cranial nerves IX, X, and XI produce smooth enlargement of the jugular foramen and classically cause hypesthesia and weakness of the palate, vocal cord, and shoulder, respectively (Fig. 177-10). If they arise in the posterior fossa, these tumors may grow to a large size before producing predominantly acoustic or cerebellar signs. Accurate preoperative differentiation of these tumors from acoustic neuromas is important because residual hearing is more likely to be preserved with lower cranial nerve neuromas. Hypoglossal neuromas produce motor hemiatrophy of the tongue and enlargement of the hypoglossal canal on radiography.
Glomus Tumors
Glomus tumors (paragangliomas) are discussed in detail in Chapter 176. Because glomus jugulare tumors and glomus vagale tumors may extend into the posterior fossa, they constitute an important consideration in the differential diagnosis of skull base neoplasms.
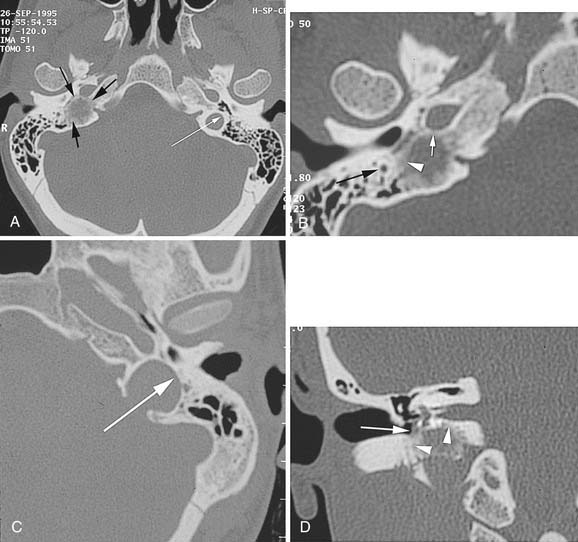
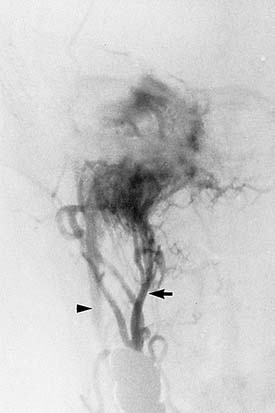
The characteristic appearance on CT with bone windows is irregular destruction of the jugular foramen, as compared with jugular foramen schwannomas, which produce smooth enlargement (Fig. 177-11). The vascular pattern of paragangliomas on angiography is characteristic (Fig. 177-12), and biopsy of these lesions is not indicated.36 Diagnostic angiography should be performed concomitantly with preoperative embolization when surgical resection is planned.
MRI produces a unique salt-and-pepper mixture of intensities on T1- and T2-weighted images. Arborizing flow voids reveal the prominent tumor vessels of this lesion. Lo19 described two limitations of MRI in evaluating paragangliomas: (1) bone changes and the relation of tumor to bone landmarks are not visualized; and (2) distinguishing tumor from bone marrow is difficult, especially on gadolinium-enhanced T1-weighted images. Thus, MRI may provide complementary information about infralabyrinthine and intracranial tumor extensions, but bone algorithm CT is the cornerstone of imaging evaluation in paragangliomas.
The combination of CT, MRI, and angiography can provide extensive information about involvement of the internal carotid artery. Magnetic resonance angiography (MRA) is an adjunct to intra-arterial catheterization and formal angiography (see Fig. 177-12). Angiography is a necessary step in preoperative embolization of glomus tumors; however, it is unlikely that MRA can replace direct intravascular angiography in glomus tumor management. For resection of skull base neoplasms involving the posterior fossa, if surgical resection requires manipulation of the artery, preoperative assessment of the adequacy of collateral flow by way of the circle of Willis is necessary. Temporary balloon occlusion of the carotid artery in combination with radioisotope imaging or xenon-enhanced CT can accurately quantify collateral blood flow.37
Arachnoid Cysts
Arachnoid cysts are thin-walled sacs that contain yellow, entrapped CSF. The current theory is that these lesions represent congenital developmental anomalies.38 Symptoms are produced by the mass effect of the cyst on surrounding structures and may be similar to the symptoms of acoustic neuromas. These patients may experience mild to profound hearing loss with a retrocochlear pattern.39
These lesions have CT and MRI characteristics similar to those of epidermoids. Enlargement of the internal auditory canal often is noted but is not characteristic of these lesions. The typical appearance is that of a smooth-surfaced lesion, which on CT approximates CSF and is nonenhancing and on MRI exhibits isointensity or hypointensity to brain on T1-weighted images and hyperintensity to brain on T2-weighted images (see Fig. 177-7). In most patients, no direct intervention is necessary. When treatment is necessary to control symptoms, management of these lesions is not total resection. Instead, surgical drainage by way of retrolabyrinthine exposure is the usual recommended therapy; however, diuretic therapy provides symptomatic relief in a few patients.
Hemangiomas
Capillary hemangiomas typically arise in the area of the geniculate ganglion in association with a perigeniculate capillary plexus.40 The lesion is characterized by a progressive facial weakness despite being much smaller than facial nerve neuromas. The expanding lesion with the production of pulsatile tinnitus may expose the upper basal turn of the cochlea. CT shows a smooth enlargement of the geniculate ganglion and enlargement of the labyrinthine portion of the fallopian canal by a soft tissue mass. Although capillary hemangiomas are enhancing, they produce facial weakness at such an early stage that only subtle findings may be apparent on CT, and a very small enhancement in the area of the labyrinthine segment may be the only finding. Other CT findings include honeycomb bone, irregular and indistinct bone margins, and intratumoral bone spicules (Fig. 177-13). These findings contrast with those in facial nerve neuromas, which are larger, more obvious lesions with sharp bone margins.