 Metastatic Tumors to the Uvea, Retina, and Optic Disc
Metastatic Tumors to the Uvea, Retina, and Optic DiscMetastatic Tumors to the Intraocular Structures
General Considerations
Metastatic cancer probably represents the most common form of intraocular malignancy. There are many large series, reviews, and case reports on metastatic neoplasms to the intraocular structures, and only selected reports are cited here (1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56). Even though uveal metastasis is the most common intraocular malignancy, in a practice of ocular oncology it is not encountered so frequently as uveal melanoma, possibly because many affected patients have advanced systemic cancer and do not come to the attention of an ophthalmologist. Metastatic cancer reaches the intraocular structures through hematogenous routes and most commonly develops in the uveal tract, with >90% involving the posterior aspect of the choroid and <10% arising in the iris and/or ciliary body. Metastasis to the retina, optic disc, and vitreous are relatively uncommon.
Most intraocular metastases are carcinomas, with sarcomas and melanomas being less common. The majority of uveal metastases originate from breast cancer in women and lung cancer in men. Less often, the primary malignancy arises from carcinoma of the alimentary tract, kidney, thyroid gland, pancreas, prostate, and other organs. Cutaneous melanoma and bronchial carcinoid tumors occasionally metastasize to the uveal tract and have distinctive features. Of patients who present to the ophthalmologist with uveal metastasis, about 25% have no known history of systemic cancer. After subsequent systemic evaluation, about 10% have no detectable primary cancer, the primary being occult (3). Hence, the clinician should be familiar with the clinical manifestations of intraocular metastatic disease.
Clinical Features
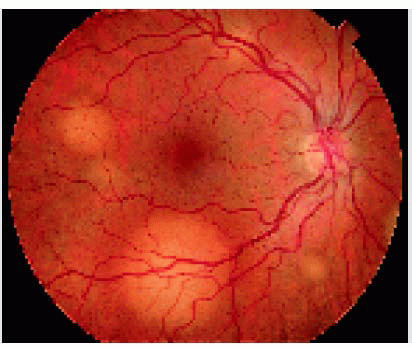
The clinical features of an intraocular metastasis vary with the location of the tumor (1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56). Iris metastasis appears as single or multiple yellow, white, or pink nodules in the iris stroma. It can appear as one or more circumscribed tumors, or it can be friable and seed cells into the aqueous, producing a clinical picture of intraocular inflammation. A ciliary body metastasis is often more difficult to detect clinically. It can appear as a solitary mass, or it can produce inflammatory signs, simulating iridocyclitis. Choroidal metastasis usually appears as one or more yellow lesions. It has a tendency to affect the posterior choroid, frequently in the macular area. In contrast to iris and ciliary body metastasis, choroidal metastasis tends not to produce inflammatory signs, but it usually causes a quiet secondary serous retinal detachment. Although choroidal metastasis usually has a yellow color, metastasis from melanoma often has a gray or brown color and metastasis from carcinoid tumor, thyroid cancer, and renal cell carcinoma often has an orange color. Retinal metastasis, which is extremely rare, can simulate an occlusive vasculitis and can seed into the vitreous. Vitreous metastasis is also rare and probable derives from the retina. It generally presents with tumor cells in the vitreous, resembling a primary inflammatory process of primary lymphoma. Optic disc metastasis can develop by contiguous spread from juxtapapillary choroidal metastasis, or it can involve only the optic nerve, where it produces a unilateral elevation of the optic disc (9). Secondary glaucoma frequently occurs, particularly with iris and ciliary body tumors.
Diagnostic Approaches
The diagnosis of intraocular metastasis is generally made by taking a history for prior cancers and by careful slit lamp biomicroscopy and ophthalmoscopy. Ancillary studies such as fluorescein angiography and ultrasonography can be of assistance in diagnosis. Fluorescein angiography of a choroidal metastasis generally shows beginning hyperfluorescence of the mass in the late venous phase, usually later than with choroidal hemangioma or melanoma. With ultrasonography it usually shows high internal reflectivity with A-scan and acoustic solidity with B-scan, a pattern similar to that seen with choroidal hemangioma. In rare instances, a choroidal metastasis can assume a mushroom configuration similar to that of a choroidal melanoma (26). In difficult cases that cannot be diagnosed with the aforementioned methods, fine needle aspiration with cytologic evaluation of aspirate can be used to establish the diagnosis (6).
Pathology
Most intraocular metastases are diagnosed clinically and no histopathologic material is available. However, uveal metastasis can assume many gross and microscopic patterns (26). Grossly, it is usually white or yellow and sessile, nodular, or diffuse. Histopathology of uveal metastasis varies considerably, depending on the type, primary site, and degree of differentiation (1,2). Some tumors are so poorly differentiated that the primary site is difficult to determine based on examination of the ocular tissue. In such instances, immunohistochemistry may be of some value in classifying the neoplasm and in determining the primary site.
Management
Management options for uveal metastasis vary with the clinical situation (1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11,54, 55, 56). Small, asymptomatic tumors or those that have responded to prior or present chemotherapy may require no immediate treatment and can be followed periodically. Larger, symptomatic tumors may require external beam irradiation or plaque radiotherapy.
The systemic prognosis varies with the type of tumor. Patients with choroidal metastasis from breast cancer often have a more favorable prognosis, whereas patients with metastasis from lung cancer or melanoma have a worse prognosis. Patients with metastasis from carcinoid tumor often have a much better prognosis, and metastatic foci from this tumor can remain relatively dormant for months or years (10,23).
Selected References
1. Ferry AP, Font RL. Carcinoma metastatic to the eye and orbit. I. Clinicopathologic study of 227 cases. Arch Ophthalmol 1975;92:276-286.
2. Shields JA, Shields CL. Metastatic tumors to the intraocular structures. In: Shields JA, Shields CL, eds. Intraocular Tumors. A Text and Atlas. Philadelphia: WB Saunders; 1992;207-238.
3. Shields CL, Shields JA, Gross N, et al. Survey of 520 uveal metastases. Ophthalmology 1997;104:1265-1276.
4. Shields JA, Shields CL, Kiratli H, et al. Metastatic tumors to the iris in 40 patients. Am J Ophthalmol 1995;119:422-430.
5. Mewes L, Young SE. Breast carcinoma metastatic to the choroid: analysis of 67 patients. Ophthalmology 1982;89:147-151.
6. Shields JA, Shields CL, Ehya H, et al. Fine needle aspiration biopsy of suspected intraocular tumors. The 1992 Urwick Lecture. Ophthalmology 1993;100:1677-1684.
7. Kindermann WR, Shields JA, Eiferman RA, et al. Metastatic renal cell carcinoma to the eye and adnexae. A report of 3 cases and review of the literature. Ophthalmology 1981;88:1347-1350.
8. DePotter P, Shields CL, Shields JA, et al. Uveal metastasis from prostate carcinoma. Cancer 1993;71:2791-2796.
9. Shields JA, Shields CL, Singh AD. Metastatic neoplasms in the optic disc: the 1999 Bjerrum Lecture: part 2. Arch Ophthalmol 2000;118:217-224.
10. Harbour JW, De Potter P, Shields CL, et al. Uveal metastasis from carcinoid tumor: clinical observations in nine cases. Ophthalmology 1994;101: 1084-1090.
11. Demirci H, Shields CL, Chao AN, et al. Uveal metastasis from breast cancer in 264 patients. Am J Ophthalmol 2003;136:264-271.
12. Brown GC, Shields JA. Tumors of the optic nerve head. Surv Ophthalmol 1985;29:239-264.
13. Shields JA, Shields CL, Ehya H. Lung cancer diagnosed by fine needle biopsy of the optic disk. Retina 2001;21:665-666.
14. Amichetti M, Caffo O, Minatel E, et al. Ocular metastases from breast carcinoma: a multicentric retrospective study. Oncol Rep 2000;7:761-765.
15. Shields JA. Metastatic tumors to the uvea. In: Shields JA, ed. Update on Malignant Ocular Tumors. International Ophthalmology Clinics. Boston: Little, Brown; 1993;33:155-161.
16. Stephens RF, Shields JA. Diagnosis and management of cancer metastatic to the uvea. A study of 70 cases. Ophthalmology 1979;86:1336-1349.
17. Shields JA, Shields CL, Shakin EP, et al. Metastasis of choroidal melanoma to the contralateral choroid, orbit, and eyelid. Br J Ophthalmol 1988;72: 456-460.
18. Lieb WE, Shields JA, Shields CL, et al. Mucinous adenocarcinoma metastatic to the iris, ciliary body and choroid. Br J Ophthalmol 1990;74: 373-376.
19. Catalano RA, Litoff D, Shields JA. Breast carcinoma metastatic to the choroid. JAMA 1990;264:1032.
20. DePotter P, Shields JA, Shields CL, et al. Unusual MRI findings in metastatic carcinoma to choroid and optic nerve. Int Ophthalmol 1992;16:39-44.
21. Hykin PG, Shields JA, Shields CL, et al. Carcinoid tumor metastatic to the choroid. Br J Ophthalmol 1996;80:8452-8453.
22. Gunduz K, Shields JA, Shields CL, et al. Ewing’s sarcoma metastatic to the iris. Am J Ophthalmol 1997;124:550-552.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


