CHAPTER 50 Medical Management of Nasosinus Infectious and Inflammatory Disease
The incidence of rhinosinusitis as defined by health surveys in the United States has been estimated at 14% of the adult population.1 A review of the Canadian National Health Survey from 1996 and 1997 found a 5% overall incidence in that country.2 A history of rhinosinusitis was more common in women than in men and in adults than in children. Lower socioeconomic status, cigarette smoking, allergic rhinitis, and asthma were all associated with a higher incidence of sinusitis history. Sinusitis ranks in the “top 10” diagnoses associated with loss of productivity for U.S. businesses (just behind spine and back conditions).3 Approximately 200,000 patients per year are estimated to undergo sinus surgery in the United States for rhinosinusitis symptoms that are unresponsive to medical therapy.4
In the patient presenting with infrequent episodes of acute rhinosinusitis complicating viral upper respiratory infection, the disease is usually easily managed with short courses of antibiotics administered by the primary care physician. Unfortunately, chronic or recurrent rhinosinusitis implies a multifactorial etiology and is not solved simply by an empiric selection of antibiotic. Predisposing factors for recurrent or chronic rhinosinusitis include viral illness, allergic rhinitis, anatomic obstruction, immunodeficiency, ciliary dyskinesia, cystic fibrosis, and environmental irritants. Genetic factors obviously play a role, although they are poorly understood at present for allergic diathesis and other key predisposing conditions. The role of bacteria and fungi, obvious in cases of culture-proven acute bacterial or invasive fungal rhinosinusitis, is much less clear in chronic disease. This form of disease was previously thought to be mostly infection aggravated by underlying host factors, but the pendulum of thought is now swinging toward the view that a large percentage of cases of chronic or recurrent sinus disease represent underlying inflammation at times aggravated by infection.5
Diagnosis
Definitions
The symptoms of rhinosinusitis overlap with predisposing conditions such as viral illness and allergic rhinitis. Current consensus-defined, symptom-based definitions of chronic rhinosinusitis (CRS) usually include congestion and nasal discharge (anterior or posterior) possibly with facial pain or pressure and loss of smell, continuing beyond the expected 7- to 10-day course of a viral illness. Supporting evidence includes rhinoscopically determined presence of purulent rhinorrhea, polyps, or mucosal edema and imaging findings of sinus or ostiomeatal mucosal inflammation.6
Because of the ambiguities in defining recurrent or chronic rhinosinusitis, the U.S. Food and Drug Administration (FDA), in November 2006, issued guidelines for industry studies.7 The FDA has defined chronic rhinosinusitis as symptoms lasting more than 8 weeks and recurrent rhinosinusitis as more than three episodes of rhinosinusitis per year. In contrast to historic views, the FDA made the statement that “chronic sinusitis is not usually caused by bacterial infection” (Table 50-1).7
Table 50-1 List of Abbreviations
| FDA | Food and Drug Administration |
| CT | Computed tomography |
| IL | Interleukin |
| GM-CSF | Granulocyte-macrophage colony-stimulating factor |
| CRS | Chronic rhinosinusitis |
| ARC | Acute rhinosinusitis |
| VAS | Visual analog scale |
| ECP | Eosinophilic cationic protein |
| QOL | Quality of life |
| RSV | Respiratory syncytial virus |
| IFN | Interferon |
| TGF | Transforming growth factor |
| CYSLTS | Cysteinyl leukotrienes |
| PG | Prostaglandin |
| ASA | Acetylsalicylic acid |
| SE | Staphylococcal enterotoxin |
| LPR | Laryngopharyngeal reflux |
| NO | Nitric oxide |
| MRSA | Methicillin-resistant Staphylococcus aureus |
| AERD | Aspirin exacerbated respiratory disease |
Imaging
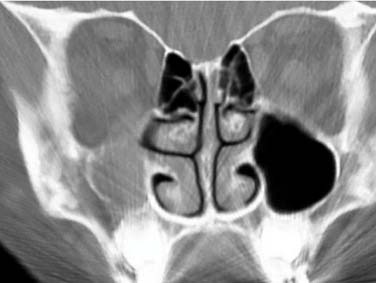
Plain radiographs have fallen out of favor in many clinical centers because of their low sensitivity compared with computed tomography.8 However, mucosal inflammation too minimal to be apparent on plain films probably correlates well with absence of clinically relevant rhinosinusitis. Specifically, plain films, if findings are normal, can help to rule out rhinosinusitis as a cause of congestion in a patient with allergic rhinitis or as a cause of headache in a patient with a family history of migraine (Fig. 50-1).
Studies have documented the incidence of incidental sinus mucosal abnormalities on computed tomography (CT) to be 20% in normal adults9 and to be as high as 60% in children younger than 8 years in temperate latitudes in winter.10 Presumably, these findings relate to the fact that viral upper respiratory infection causes rhinosinusitis with inflammation of nose and sinuses that can persist radiographically up to several weeks.11
Results of studies correlating CT scores with symptom scores and outcomes are mixed. Stewart and colleagues,12 in a prospective study of 57 adults, found that high preoperative CT scores correlated with more symptom improvement after sinus surgery. In a retrospective study of children, Kim and associates13 reported that high preoperative CT stage, along with nasal polyposis and cigarette smoke exposure, was associated with worse outcomes after surgery. Other studies have found an association between bony hypertrophy14 and frontal and sphenoid disease (factors that presumably indicate a more severe level of inflammation) and symptom scores. On the other hand, other prospective studies in adults have not shown correlation between preoperative CT scans by any (Lund-Mackay, Kennedy, or Harvard) staging system and symptom scores or outcomes.15,16
Differences in study conclusions regarding the extent of CT evidence of sinus inflammation and patient symptom scores and outcomes are probably due in part to patient selection biases. Presumably, patients with more severe disease in general have higher symptom scores and more measurable response to therapy, but the final word will have to await larger and better-designed studies. Some of the lack of correlation between symptoms and imaging is due to differences in underlying patient perception. Davis and colleagues, who used the SNOT-16 (Sinonasal Outcome Test 16), SF-36 (36-item Medical Outcomes Short Form Survey), BSI, (Brief Symptom Inventory) and PHQ (Patient Health Questionnaire)17 to assess adult patients with CRS selected for surgery, found a high rate of somatization, anxiety, and depression in the study population. Somatization in particular was associated with higher SNOT-16 scores (independent of imaging findings), although all groups of patients showed improvement after surgery.17 Facial pain and headache, in particular, among sinusitis symptoms correlate poorly with imaging findings.18,19
Rhinoscopy
Nasal endoscopy is generally considered a necessary part of any comprehensive assessment of rhinosinusitis. Rigid or fiberoptic endoscopes provide excellent visualization of the nasal cavities, but in their absence, an otoscope placed in the nostril can give an excellent magnified view up to the middle meatus and beyond. Polyps, rhinorrhea, turbinate hypertrophy, and other types of congestion can be evaluated and roughly quantified. Also, many authorities have shown fairly good correlation between culture results for endoscopically guided middle meatus specimens and maxillary sinus aspirates.20
Smell and Nasal Airflow
Smell can be measured objectively, and subjective responses on validated outcome measures correlate fairly well with objective test results.21 Subjective reports of nasal obstruction correlate fairly well with objective measures of nasal airflow such as rhinomanometry but less well with acoustic rhinometry, a measure of nasal airway volume.22
Outcome Measures and Quality of Life
The FDA recommends that the following measures be considered for new trials evaluating efficacy of medical therapies for rhinosinusitis: symptom profile with visual analog scale (VAS; mild, moderate, severe), nasal endoscopy with a scoring system, imaging with scoring, documentation of other medications, cigarette smoking, comorbid conditions such as asthma, and additional tests such as smell and mucociliary function.6
Several quality of life instruments have been utilized in studies of acute and CRS, although not all have been validated. The most commonly utilized general health status instrument has been the Medical Outcomes Study Short Form 36 (SF-36). Several disease specific instruments have been developed for rhinosinusitis; a sample of studies utilizing these instruments is shown in Table 50-2.23–30
Predisposing Factors
Viral Illness
Upper respiratory viruses are probably the most common predisposing factors for acute rhinosinusitis, especially in children. The link between viruses and CRS is less clear.31 A multiplicity of viruses has been implicated in the common cold, including influenza, parainfluenza, adenovirus, rhinovirus, and respiratory syncytial virus. Because of the number and antigenic variability of potential causative agents, viral vaccine therapy is not likely to become an effective weapon in the battle against the common cold.
Most spread of viral illness occurs via hand-to-hand contact, and studies have demonstrated reduced viral illness with protocols of hand washing and alcohol gel.32 Parents of children with recurrent sinusitis are encouraged to find child-care settings with the fewest possible children in order to reduce their children’s viral exposure. Sometimes coordinating vacations or child-care with relatives to remove the patient from the daycare setting for several weeks allows enough mucosal recovery to break a cycle of recurrent infection.
Vitamin C has long been proposed as both prevention and therapy for the common cold, but studies have failed to demonstrate significant benefit. Similarly, zinc gluconate may have a mild effect in reducing cold symptoms, but its mechanism of action is poorly understood.33
Antiviral agents have received much attention for the prevention and possible treatment of the common cold. A 2007 Cochrane review concluded that although interferons had some efficacy, their side effects were often equal to the cold symptoms.34 Other antiviral agents, including dipyridamole, palmitate, and pleconaril, show potential efficacy with fewer side effects. Currently there are no antiviral agents with FDA approval for prevention or treatment of the common cold.
Of particular interest in CRS is the possible association between early viral infection and a subsequent shift from an immune response dominated by type 1 helper T cell (TH1) cytokines (nonallergic) to TH2 cytokines (allergic). Respiratory syncytial virus (RSV) and rhinovirus infection at an early age have been associated in some studies with subsequent reactive airways disease and asthma.35,36 An individual’s susceptibility to a shift to a TH2 immune response and future refractory respiratory disease is probably related to several factors, including genetic predisposition, timing of the initial and subsequent viral infections, and exposure to environmental antigens.37
Environmental Irritants
More than half of the people in the United States are estimated to live in areas where exposure to ozone, sulfur dioxide, nitrogen dioxide, carbon monoxide, lead, and particulates exceeds the National Ambient Air Quality Standards. The reunification of Germany has provided an interesting laboratory for examining the possible effects of air pollution on incidence of rhinosinusitis. As levels of particle emissions and sulfur dioxide dropped with the modernization of the former East German industry, the incidence of rhinosinusitis reported on standardized questionnaires for pediatric patients has also fallen significantly.38
Air pollution may impair respiratory mucociliary protection and potentially augment sensitivities to common aeroallergens.39 Exposure to outdoor and indoor pollutants is therefore one possible factor explaining the dramatic increase in rates of immunoglobulin (Ig) E–mediated disease, including asthma and allergic rhinitis, reported from most industrialized areas over the past 20 years.
A careful history of environmental exposures should be a part of the evaluation of every patient with recurrent or chronic rhinosinusitis. Indoor sources include cigarette smoke,40 smoke from wood-burning stoves, formaldehyde, copier fluid, latex, chlorine, paint, and perfumes.
Allergy
Allergy may be a predisposing factor, a comorbid state, or even part of the definition of rhinosinusitis. Up to 30% of the adult population of the United States is allergic according to skin test results, and the percentage appears to be greater overall in populations of patients with rhinosinusitis. Studies have shown a higher prevalence of allergic rhinitis in patients with acute rhinosinusitis41 and more CT evidence of mucosal inflammation with viral illness.42 The association between allergic rhinitis, defined as atopy, and CRS is less clear. For example, Gelnick and colleagues43 studied 115 adults with CRS and found that allergy was not significantly associated with asthma or chronic sinusitis. Interestingly, CT scores and global health outcome measures were worse in the nonatopic group. However, in children, some studies have shown a strong association between a diagnosis of rhinosinusitis and allergic rhinitis.41
Inflammation
The last few years have witnessed a dramatic increase in the number of published studies linking chronic mucosal inflammation in general to chronic or recurrent rhinosinusitis. In aggregate, studies show a spectrum in inflammation from a more TH1 cytokine profile in nonatopic CRS to a more TH2 profile with atopic CRS associated with nasal polyps.6,44 These studies have led to a fundamental shift in treatment philosophy away from exclusively antimicrobial strategies towards a more balanced approach that includes anti-inflammatory treatments also.
Histologic studies of adult patients with long-standing CRS demonstrate sinus mucosal changes similar to those seen with remodeling in asthma, with greater basement membrane thickness, increases in goblet cells and submucosal mucous glands, and eosinophilic infiltrates.45 Epithelial damage from eosinophil-mediated cytotoxicity may promote a vicious circle of increased antigen interaction and/or infection, which in turn leads to even greater inflammation. Studies have shown that pediatric patients with CRS also demonstrate increases in basement membrane thickening and eosinophilic infiltrate in comparison with controls, but not as much as adult patients with CRS.46 The assumption is that the adult patients exhibit more remodeling as a function of more damage and repair over a longer period.
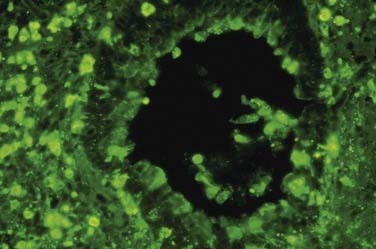
Peripheral eosinophilia has been associated with sinus CT stage, history of asthma, and presence of polyps in patients with CRS.47,48 Tissue eosinophilia has also been related to greater severity and worse prognosis in patients with CRS.49,50 Cytokine expression is up-regulated in patients with CRS in comparison with controls, showing a TH1 profile in nonatopic CRS with increases in interferon-γ and tumor growth factor-β and a TH2 profile in patients with nasal polyps showing increases in interleukins IL-5 and IL-13.51 Nasal polyps exhibit a special inflammatory profile, with decreased tumor growth factor β1, increases in IL-5, eotaxin, and eosinophilic cationic protein, and an imbalance of metalloproteinases (Fig. 50-2).52,53
Eicosanoids and Aspirin Sensitivity Triad
Abnormal regulation of arachidonic acid metabolism pathways with elevated cysteinyl leukotrienes has been associated with allergic rhinitis, asthma, CRS, nasal polyposis in general and aspirin triad (steroid-dependent asthma, nasal polyposis, and aspirin intolerance). Cysteinyl leukotrienes promote airway inflammation and fibrosis. The extent of up-regulation of these leukotrienes in such disorders appears to be linked to the level of eosinophilic inflammation as indicated by IL-5 and eosinophilic cationic protein measurements.54 Patients with nasal polyps in particular may also have a down-regulation of the cyclooxygenase pathway with decreased production of prostaglandin E2, an anti-inflammatory mediator.55 These abnormalities do not seem to be related to allergy (Fig. 50-3).
There appears to be a spectrum of severity with aspirin sensitivity, and up to 20% of patients who have steroid-dependent asthma without overt nasal polyps can have exacerbation of asthma with aspirin exposure.56 Also, some patients without overt asthma have respiratory symptoms with ASA exposure. Perhaps as much as 20% of patients with CRS and tissue eosinophilia experience upper and lower airway disease flare-ups with ASA exposure. The average age of onset of aspirin-exacerbated respiratory disease (AERD) is young adulthood, and a majority of patients are women. The definitive diagnosis is made with an oral challenge of aspirin in a controlled setting (anaphylaxis management), during which the physician looks for symptoms such as flushing, sneezing, wheezing, conjunctivitis, rhinorrhea, and periorbital edema. Oral aspirin challenge is being replaced in some centers by nasal challenge with lysine aspirin, which has nearly the same sensitivity and specificity but is much safer.
Superantigens
In 2001, Schubert57 wrote that “superantigen amplification of preexisting immunopathology is the proposed mechanism for disease induction and maintenance” in a large proportion of patients with CRS. In other words, microbial infection in a patient with an underlying hyperinflammatory state leads to amplified inflammation with chronicity and recurrence. Staphylococcal bacterial antigens such as enterotoxins A and B (SEA, SEB) and toxic shock toxins could potentially bind at the T-cell V beta site, thus bypassing normal antigen processing and dramatically up-regulating IgE-mediated inflammation.58,59 A significant percentage of people have staphylococcal colonization at any given time, affording a source for inflammation amplification.60 Similarly, antigenic fungi such as Alternaria species could potentially act as superantigens.61 Ferguson and associates,62 in a study of patients with CRS and eosinophilic mucin, found that more than half of the patients had histologic evidence of bacteria within the mucin.62 These researchers hypothesized that perhaps a significant proportion of patients with culture-nonspecific CRS may have bacterial superantigen inflammation amplification.
Laryngopharyngeal Reflux
Laryngopharyngeal reflux could potentially inflame nasal mucosa and predispose to CRS. On the other hand, gastroesophageal reflux in general can be associated with an allergic diathesis, and laryngopharyngeal reflux may be more of a comorbid state than a cause of CRS. Helicobacter pylori has been detected within sinuses of patients with CRS, but a causal relationship has not been demonstrated.63
Nitric Oxide
Nitric oxide, which is produced by nasal and sinus mucosa, has a role in inflammation and presumably also in local immune defense. In fact, healthy sinuses contain a higher concentration of nitric oxide than the nasal cavities and may serve in part as reservoirs. Nitric oxide metabolites have been found to be elevated in sinus lavage fluid from patients with CRS, and nasal nitric oxide levels have been found to decrease with successful treatment of CRS.64–66
Bacteria
Acute Rhinosinusitis
It has been estimated that up to 2% of all cases of routine viral rhinosinusitis progress to acute bacterial rhinosinusitis (ABRS). In culture studies, the most common organisms are Streptococcus pneumoniae, nontypeable Haemophilus influenzae, and Moraxella catarrhalis (more common in children). A 2008 study reported that the use of polyvalent pneumococcal vaccines in children with secondary herd immunity effects in adults may be causing an overall decrease in the number of S. pneumoniae infections.67 The subsequent loss of bacterial interference from Streptococcus species may in turn be causing a rise in disease and colonization secondary to Staphylococcus aureus, with implications also in terms of superantigen inflammation. Studies also point to an increase in the recovery of methicillin-resistant S. aureus (MRSA) in acute and chronic rhinosinusitis.68 A significant percentage of S. pneumoniae isolates show intermediate penicillin resistance, and a growing percentage of Haemophilus isolates are positive for beta-lactamase.69
Chronic Rhinosinusitis
The role of bacteria in CRS is uncertain. A 2003 study comparing middle meatus and maxillary sinus culture specimens from patients with CRS and from healthy controls showed an 80% correlation between findings in middle meatus and maxillary specimens as well as recovery of similar bacteria in similar frequencies in the two populations.70 In the CRS group, the most common isolates were S. aureus (36%), coagulase-negative Staphylococcus (20%), and S. pneumoniae (17%). In a 2007 study, the researchers found no microbiologic culture difference between patients with CRS and controls but the patients with CRS did have more white blood cells in their secretions.71
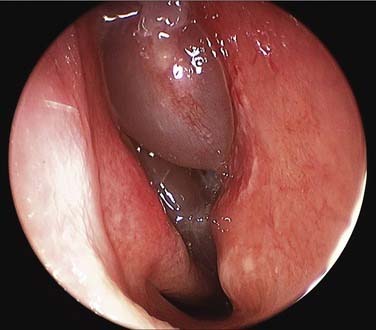
Studies from cohorts of patients undergoing surgery for CRS sometimes show a greater prevalence of gram-negative rods.72 Not surprisingly, writers are reporting steady increases in antibiotic resistance among cultured organisms, including MRSA.73,74 And one study has shown an association between intracellular presence of S. aureus, as determined by confocal microscopy of surgical specimens, and persistent clinical disease (Fig. 50-4).75
Biofilms
Biofilm formation may explain in part some of the mysteries of CRS, including indolent course and poor response to antibiotic therapy. Several studies in humans and in animal models of sinusitis have demonstrated evidence of biofilm formation by confocal microscopy and other techniques.76 Biofilm formation is probably more likely with gram-negative rods such as Pseudomonas species. In one study, researchers performed in vitro assays of nasal secretions from patients with CRS and found that 28% had bacteria with a “severe biofilm-forming capacity.”77