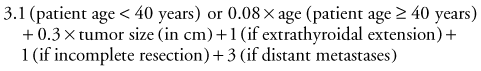CHAPTER 124 Management of Thyroid Neoplasms
Thyroid neoplasms represent almost 95% of all endocrine tumors, although they are relatively uncommon, accounting for approximately 2.5% of all malignancies. In 2008, the estimated annual incidence of thyroid cancer in the United States was 37,340 cases, and approximately 1590 patients (4.3%) were expected to die from thyroid cancer.1 The incidence of thyroid cancer has been steadily increasing over the past 2 decades, and thyroid cancer has the fastest increasing incidence of all major cancers in the United States (approximately 4% increase annually).2 Mortality rates have remained stable, and evidence suggests that improved detection has primarily contributed to the increased incidence.
Although thyroid cancer is rare, the incidence of thyroid nodules is significantly greater, affecting approximately 4% to 7% of the U.S. population.3 Although most of these nodules are benign, the challenge is to identify the approximately 5% of patients with a malignant lesion. A subset of thyroid cancers is particularly aggressive with a potential for devastating morbidity. No reliable indicators are currently available to determine which patients will develop aggressive or recurrent disease, although risk categories based on clinical and pathologic criteria yield important prognostic information.
Most thyroid carcinomas are well-differentiated tumors of follicular cell origin.4,5 These lesions are histologically defined as papillary carcinoma, follicular carcinoma, and Hürthle cell carcinoma. A survey of 53,856 patients described the overall incidence of thyroid cancer in the United States.5 In this report, approximately 79% of cases were papillary carcinoma, 13% were follicular carcinoma, and approximately 3% were Hürthle cell carcinoma. A small proportion (6%) of patients with these lesions have a family history of thyroid cancer. Medullary thyroid carcinoma (MTC), which arises from parafollicular C cells, accounts for about 4% of thyroid carcinomas. Approximately 30% of patients with these lesions have a strong genetic contribution. Anaplastic carcinomas, lymphoma, and metastatic disease constitute a small portion of thyroid malignancies.
The most common presentation of a thyroid cancer is the development of a thyroid mass or nodule. Assessment of the lesion requires a careful history, physical examination, fine-needle aspiration cytology (FNAC), and perhaps imaging studies. With correct diagnosis and management, most patients with well-differentiated thyroid carcinomas have an excellent prognosis. The 10-year disease-specific mortality rate is less than 7% for papillary thyroid cancer and less than 15% for follicular thyroid cancer.5–8 Controversy regarding the treatment of thyroid carcinomas and the extent of thyroidectomy to be performed arises because of the indolent course of most thyroid cancers. Interventions for thyroid cancer have been difficult to evaluate because of the long follow-up and the large number of patients needed to determine differences in survival. The morbidity that may accompany any aggressive intervention needs to be balanced with the generally good prognosis of patients with thyroid cancer.
Surgical Anatomy and Embryology
The thyroid medial anlage derives from the ventral diverticulum of the endoderm from the first and second pharyngeal pouches at the foramen cecum.9,10 The diverticulum descends from the base of the tongue to its adult pretracheal position through a midline anterior path with the primitive heart and great vessels during weeks 4 to 7 of gestation. The proximal portion of this structure retracts and degenerates into a solid, fibrous stalk; persistence of this tract can lead to the development of a thyroglossal duct cyst with variable amounts of associated thyroid tissue. The lateral thyroid primordia arise from the fourth and fifth pharyngeal pouches and descend to join the central component. Parafollicular C cells arise from the neural crest of the fourth pharyngeal pouch as ultimobranchial bodies and infiltrate the upper portion of the thyroid lobes.11 Because of the predictable fusion of the ultimobranchial bodies to the medial thyroid anlage, C cells are restricted to a zone deep within the middle to upper third of the lateral lobes.12
The thyroid gland is composed of two lateral lobes connected by a central isthmus, weighing 15 to 25 g in adults. A thyroid lobe usually measures about 4 cm in height, 1.5 cm in width, and 2 cm in depth. The superior pole lies posterior to the sternothyroid muscle and lateral to the inferior constrictor muscle and the posterior thyroid lamina. The inferior pole can extend to the level of the sixth tracheal ring. Approximately 40% of patients have a pyramidal lobe that arises from either lobe or the midline isthmus and extends superiorly (Fig. 124-1).
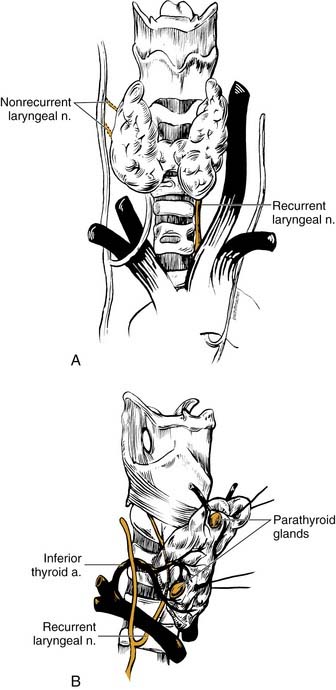
Blood supply to and from the thyroid gland involves two pairs of arteries, three pairs of veins, and a dense system of connecting vessels within the thyroid capsule. The inferior thyroid artery arises as a branch of the thyrocervical trunk (Fig. 124-2). This vessel extends along the anterior scalene muscle, crossing beneath the long axis of the common carotid artery to enter the inferior portion of the thyroid lobe. Although variable in its relationship, the inferior thyroid artery lies anterior to the recurrent laryngeal nerve (RLN) in approximately 70% of patients.13 The inferior thyroid artery is also the primary blood supply for the parathyroid glands.
The superior thyroid artery is a branch of the external carotid artery and courses along the inferior constrictor muscle with the superior thyroid vein to supply the superior pole of the thyroid. This vessel lies posterolateral to the external branch of the superior laryngeal nerve (SLN) as the nerve courses through the fascia overlying the cricothyroid muscle. Care should be taken to ligate this vessel without damaging the SLN. Occasionally, arteria thyroidea ima may arise from the innominate artery, carotid artery, or aortic arch, and supply the thyroid gland near the midline.13 Many veins within the thyroid capsule drain into the superior, middle, and inferior thyroid veins, leading to the internal jugular or innominate veins. The middle thyroid vein travels without an arterial complement, and division of this vessel permits adequate rotation of the thyroid lobe to identify the RLN and parathyroid glands.
The RLN provides motor supply to the larynx and some sensory function to the upper trachea and subglottic area. Careful management of thyroid carcinomas requires a thorough knowledge of the course of the RLN (see Fig. 124-2). During development, the RLN is dragged caudally by the lowest persisting aortic arches. On the right side, the nerve recurs around the fourth arch (subclavian artery), and on the left side, the nerve recurs around the sixth arch (ligamentum arteriosum).
The right RLN leaves the vagus nerve at the base of the neck, loops around the right subclavian artery, and returns deep to the innominate artery back into the thyroid bed approximately 2 cm lateral to the trachea. The nerve enters the larynx between the arch of the cricoid cartilage and the inferior cornu of the thyroid cartilage. The left RLN leaves the vagus at the level of the aortic arch, and loops around the arch lateral to the obliterated ductus arteriosus. The nerve returns to the neck posterior to the carotid sheath and travels near the tracheoesophageal groove along a more medial course than the right RLN. The nerve crosses deep to the inferior thyroid artery approximately 70% of the time and often branches above the level of the inferior thyroid artery before entry into the larynx.14 The RLN travels underneath the inferior fibers of the inferior constrictor (i.e., the cricopharyngeus muscle) and behind the cricothyroid articulation to enter the larynx. A “nonrecurrent” laryngeal nerve may rarely occur on the right side and enters from a more lateral course (see Figs. 124-2 and 124-4C).15 Typically, an aberrant retroesophageal subclavian artery (arteria lusoria) or other congenital malformation of the vascular rings is present.
The SLN arises beneath the nodose ganglion of the upper vagus and descends medial to the carotid sheath, dividing into an internal and external branch about 2 cm above the superior pole of the thyroid.16 The internal branch travels medially and enters through the posterior thyrohyoid membrane to supply sensation to the supraglottis. The external branch extends medially along the inferior constrictor muscle to enter the cricothyroid muscle. Along its course, the nerve travels with the superior thyroid artery and vein. The nerve typically diverges from the superior thyroid vascular pedicle about 1 cm from the thyroid superior pole.
Proper management of the parathyroid glands during thyroid surgery is crucial to avoid hypoparathyroidism. The superior parathyroid glands are derived from the fourth pharyngeal pouch, whereas the inferior counterparts originate from the third pharyngeal pouch. The parathyroid glands are caramel-colored glands weighing 30 to 70 mg. The subtle distinction of tan and yellow coloration permits differentiation from adjacent fatty tissue, although with trauma, the glands can become mahogany in color. Four parathyroid glands exist in 80% of patients, and at least 10% of patients have more than four glands.17 The glands are situated on the undersurface of the thyroid gland in predictable locations. The superior glands are located at the level of the cricoid cartilage, usually medial to the intersection of the RLN and the inferior thyroid artery.17 The inferior glands are more variable in location than their superior counterparts. These glands may be on the lateral or posterior surface of the lower pole. In many patients, the position of the parathyroid glands on one side is similar to the other side and should be a useful guide.
Molecular Basis for Thyroid Neoplasms
Alterations noted in the development of thyroid carcinomas include changes in total cellular DNA content. The loss of chromosomes, or aneuploidy, has been noted in 10% of all papillary carcinomas, but is present in 25% to 50% of all patients who die as a result of these lesions.18 Similarly, the development of follicular adenomas is associated with a loss of the short arm of chromosome 11 (11p), and transition to a follicular carcinoma seems to involve deletions of 3p, 7q, and 22q.19,20 Loss of heterozygosity involving multiple chromosomal regions is much more prevalent in follicular adenomas and carcinomas than in papillary carcinomas.21
Several oncogenes, altered genes that contribute to tumor development, have been identified in early thyroid tumor progression. Mutations in the thyroid-stimulating hormone (TSH) receptor and G-protein mutations are found in hyperfunctioning thyroid adenomas.22 These changes can lead to the constitutive activation of cell-signaling pathways, such as the adenylate cyclase–protein kinase A system. Point mutations of the G-protein Ras found in thyroid adenomas and multinodular goiters are believed to be an early mutation in tumor progression.23 Somatic Ras mutations are associated with follicular adenomas, and to a lesser extent with follicular carcinomas. The resultant activation of the phosphatidylinositol 3′-kinase (PI3K) signal transduction pathway and AKT, a PI3K-related serine/threonine kinase, also seems to be specific to follicular thyroid carcinoma.24
Other genetic changes have also been associated with certain types of thyroid carcinoma. Mutations within the mitogen-activated protein kinase (MAPK) pathway are involved in malignant transformation to papillary thyroid cancer. Additionally, rearrangements or activation of RET or BRAF proto-oncogenes, which can also activate MAPK, are often found in papillary thyroid cancer.25 Gene rearrangements involving tropomycin-receptor-kinase A, also known as neurotropic tyrosine receptor kinase type 1, a receptor for nerve growth factor, are associated with papillary carcinomas. Mutations in MET/hepatic growth factor have been linked to papillary and poorly differentiated thyroid carcinomas. Other growth factors, such as fibroblast growth factors, epidermal growth factor, and vascular endothelial growth factor, and their cognate receptors may have increased expression in thyroid tumors and contribute to tumor progression.
Different types of galectin, a carbohydrate-binding protein, seem to be differentially expressed in papillary and anaplastic carcinomas, and may be useful in distinguishing benign from malignant thyroid lesions.26,27 In Cowden’s disease (familial goiter and skin hamartomas), inactivating mutations of the phosphatase and tensin homolog (PTEN) gene have been identified.28 PTEN may inhibit phosphorylation and kinase activity of AKT1, leading to the development of follicular adenomas and carcinomas.24 The PAX8/PPARγγ1 (peroxisome proliferator-activated receptor) rearrangement seems to be unique to follicular thyroid carcinoma.29 PAX8 is expressed at high levels during thyroid development, and the PAX/PPARγγ1 gene product seems to function as a dominant negative, blocking the activation of wild-type PPARγγ1. Mutations in the tumor-suppressor gene p53, a transcriptional regulator, seem to be involved in insular thyroid carcinomas and the progression from papillary to anaplastic carcinoma.30,31
The role of mutations of the RET oncogene in the development of papillary carcinoma and MTC has been extensively studied.32 Located on chromosome 10, RET codes for a transmembrane tyrosine kinase receptor that binds glial cell line–derived neurotrophic factor. During embryogenesis, RET protein is normally expressed in the nervous and excretory systems. Abnormalities in RET expression result in developmental defects, including the disruption of the enteric nervous system (Hirschsprung’s disease). Presumably, RET gene mutations result in the activation of the Ras/JNK/ERK1/2 signaling pathways, resulting in further genomic instability and prevention of entry into the apoptotic pathway.33
MTC and pheochromocytoma arise from neural crest cells containing RET point mutations. These point mutations have been well documented in patients with familial MTC, multiple endocrine neoplasia (MEN) type IIA, and MEN IIB.34,35 The aggressiveness of the MTC that develops is linked to the specific RET mutation identified.36 Somatic mutations of ret are also found in approximately 25% of sporadic MTCs. Many of these are identical to the codon 918 mutation found as a germline mutation in MEN IIB, although other codons are more infrequently involved.37
Rearrangements of the RET gene by fusion with other genes also create transforming oncogenes. Although more than 10 rearrangements have been described, three oncogene proteins—RET/PTC1, RET/PTC2, and RET/PTC3—account for most of the rearrangements found in papillary thyroid cancers, and are more frequently associated with childhood thyroid carcinomas.38 Not all patients with papillary carcinomas express a RET/PTC gene however.39 There are marked geographic differences, and the gene rearrangement is strongly associated with radiation exposure. After the Chernobyl nuclear disaster, 66% of the papillary thyroid cancers removed from affected patients had RET/PTC1 or RET/PTC3 rearrangements.40 The RET/PTC3 rearrangement is most commonly associated with a “solid” follicular variant of papillary thyroid carcinoma, whereas RET/PTC1 is associated more often with the classic or diffuse sclerosing variants of papillary thyroid cancer.41,42
Risk Factors and Etiology
Although the specific molecular events related to the development of thyroid carcinomas remain incompletely defined, several patient and environmental factors have been closely examined. Women are three times more likely than men to develop differentiated thyroid cancer, and two times more likely to have anaplastic thyroid cancer. The median age at diagnosis is 47 years, with a peak in women at 45 to 49 years and in men at 65 to 69 years.2 Epidemiologic studies have not shown a clear association between dietary iodine and thyroid carcinomas.43 Also, there does not seem to be a simple relationship between benign goiter and well-differentiated thyroid carcinomas. Although papillary thyroid carcinomas are not associated with goiter, follicular and anaplastic thyroid carcinomas occur more commonly in areas of endemic goiter. Additionally, two particularly important risk factors, exposure to radiation and a family history of thyroid cancer, have been studied extensively.
Exposure to ionizing radiation increases patient risk for the development of thyroid carcinoma.44,45 Ionizing radiation exposure is the only established environmental risk factor for thyroid cancer.46 Low-dose ionizing radiation treatments (<2000 cGy) were used in the treatment of “enlarged thymus” to prevent “sudden crib death,” enlarged tonsils and adenoids, acne vulgaris, hemangioma, ringworm, scrofula, and other conditions. The risk increases linearly from 6.5 to 2000 cGy, and typically has a latency period lasting 10 to 30 years. Although higher doses of ionizing radiation typically lead to the destruction of thyroid tissue, patients with Hodgkin’s disease who receive 4000 cGy also have a higher incidence of thyroid cancer. Palpable thyroid nodularity may be present in 17% to 30% of patients exposed to ionizing radiation.47 A patient with a history of radiation exposure who presents with a thyroid nodule has a 50% chance of having a malignancy.48 Of these patients with thyroid cancer, 60% have cancer within the nodule, and the remaining 40% have cancer located in another area of the thyroid. The thyroid carcinoma tends to be papillary and frequently multifocal. There is also a higher risk of cervical metastases.
Similarly, patients exposed to radiation from nuclear weapons and accidents have a higher incidence of thyroid cancer. Children near the Chernobyl nuclear power facility had a 60-fold increase in thyroid carcinoma after the nuclear accident in 1986.49 Most of these children were infants at the time of the accident, and many of these cases developed without the typical latency period. The thyroid gland seems to be particularly vulnerable to ionizing radiation in children and yet relatively insensitive in adults. In the life span study of atomic bomb survivors in Hiroshima and Nagasaki, the risk of thyroid cancer was associated with patient age at the time of the bombings.50 The risk was greatest for individuals younger than 10 years, and no increased incidence of thyroid cancer was seen in individuals older than 20 years at the time of exposure.
Finally, familial and genetic contributions need to be fully evaluated. A patient with a family history of thyroid carcinoma may require specific diagnostic testing. Approximately 6% of patients with papillary thyroid cancer have familial disease. Papillary thyroid cancer occurs with increased frequency in certain families with breast, ovarian, renal, or central nervous system malignancies.51 Gardner’s syndrome (familial colonic polyposis) and Cowden’s disease are associated with well-differentiated thyroid carcinomas. Patients with a family history of MTC, MEN IIA, or MEN IIB warrant evaluation for the RET point mutation.
Tumor Staging and Classification
TNM Classification
The American Joint Commission on Cancer (AJCC) and the Union Internationale Contre le Cancer (UICC) adopted a tumor-node-metastasis (TNM) classification system (Table 124-1). In this system, patient age at presentation influences the clinical staging of a thyroid carcinoma. Of patients with stage I disease, 82% had a 20-year survival of nearly 100%, whereas the 5% of patients with stage IV disease experienced a 5-year survival of only 25%.52
Table 124-1 TNM Staging for Thyroid Cancer
| Primary Tumor (T) | |
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| T1 | Tumor = 2 cm in greatest dimension, limited to thyroid |
| T2 | Tumor >2 cm and = 4 cm in greatest dimension, limited to thyroid |
| T3 | Tumor >4 cm in greatest dimension, limited to the thyroid or |
| Any tumor with minimal extrathyroid extension (e.g., extension to sternothyroid muscle or perithyroid soft tissues) | |
| T4a | Tumor of any size extending beyond the thyroid capsule to invade subcutaneous soft tissues, larynx, trachea, esophagus, or recurrent laryngeal nerve |
| T4b | Tumor invades prevertebral fascia or encases carotid artery or mediastinal vessels |
| All Anaplastic Carcinomas Are Considered T4 Tumors | |
| T4a | Intrathyroidal anaplastic carcinoma—surgically resectable |
| T4b | Extrathyroidal anaplastic carcinoma—surgically unresectable |
| Regional Lymph Nodes (N) | |
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Regional lymph node metastasis |
| N1a | Metastasis to level VI (pretracheal, paratracheal, and prelaryngeal/Delphian lymph nodes) |
| N1b | Metastasis to unilateral, bilateral, or contralateral cervical or superior mediastinal lymph nodes |
| Distant Metastasis (M) | |
| MX | Distant metastasis cannot be assessed |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| Stage Grouping | ||
|---|---|---|
| Age <45 Years | Age ≥45 Years | |
| Papillary/Follicular | ||
| Stage I | Any T any N M0 | T1 N0 M0 |
| Stage II | Any T any N M1 | T2 N0 M0 |
| Stage III | T3 N0 M0 | |
| T1-T3 N1a M0 | ||
| Stage IVA | T4a N0 M0 | |
| T4a N1a M0 | ||
| T1-4a N1b M0 | ||
| Stage IVB | T4b any N M0 | |
| Stage IVC | Any T any N M1 | |
| Medullary | ||
| Stage I | T1 N0 M0 | |
| Stage II | T2 N0 M0 | |
| Stage III | T3 N0 M0 | |
| T1-3 N1a M0 | ||
| Stage IVA | T4a N0 M0 | |
| T4a N1a M0 | ||
| T1-4a N1b M0 | ||
| Stage IVB | T4b any N M0 | |
| Stage IVC | Any T any N M1 | |
| Anaplastic | ||
| Stage IVA | T4a any N M0 | |
| Stage IVB | T4b any N M0 | |
| Stage IVC | Any T any N M1 | |
From American Joint Committee on Cancer: AJCC Cancer Staging Manual. 6th ed. New York: Springer; 2002.
AMES
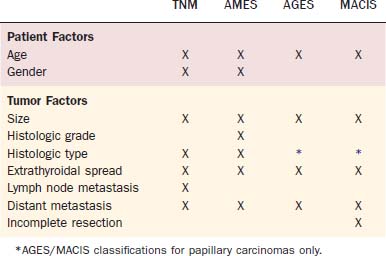
In the AMES system, patient age, the presence of metastases, extent of tumor invasion, and tumor size were used to stratify patients into low-risk and high-risk groups (Table 124-2). Low-risk patients were young (men, <41 years old; women, <51 years old) without distant metastases and all older patients without extrathyroidal papillary carcinoma, without major invasion of the tumor capsule by follicular carcinoma or with a primary tumor less than 5 cm in diameter. In a review of 310 patients from 1961-1980, low-risk patients (89%) had a mortality of 1.8% compared with a mortality rate of 46% in high-risk patients (11%). Recurrence in low-risk patients was 5%, and in high-risk patients was 55%.53 In DAMES, nuclear DNA content was added to the AMES system to improve risk-stratification for papillary thyroid carcinoma.54
AGES and MACIS
In the original AGES system, age at diagnosis, histologic tumor grade, extent of disease at presentation, and tumor size were used to calculate a prognostic score.55 Because of the infrequent practice of tumor grading, a more recent modification of the system eliminated histologic tumor grade and incorporated metastasis and extent of resection. The MACIS system accounts for metastasis, age at diagnosis, completeness of surgical resection, extrathyroidal invasion, and tumor size.56 The MACIS score is calculated as follows:
Other risk-classification systems with similar diagnostic criteria have been described.57–59 Although numerous multivariable prognostic scoring systems have been developed, none is universally accepted. Additionally, none of these classifications has shown clear superiority, and application of these systems to a single population has shown incompatible findings compared with the original studies.59,60 These systems do not apply to patients with poorly differentiated and more aggressive thyroid carcinomas.
Nevertheless, some general conclusions can be drawn from these studies regarding the prognosis of patients with well-differentiated thyroid carcinomas. Low risk for tumor recurrence and disease-specific mortality is noted in patients who are younger at diagnosis, have smaller primary tumors that lack extrathyroidal extension or regional/distant metastases, and have complete gross resection of disease at the initial surgery. Delay in treatment negatively affects prognosis. The most significant overall indicator of a poor prognosis is distant metastases, however, especially to bone.6 Although a single risk-classification strategy is unavailable, these criteria should guide physicians to use therapeutic strategies that are directed toward the particular disease and risk for an individual patient, rather than applying a general treatment strategy for all patients with a particular form of thyroid carcinoma. More recent management guidelines from the American Thyroid Association (ATA) have recommended use of the AJCC/UICC staging system for all patients with differentiated thyroid cancer.
Evaluation of a Thyroid Nodule
The incidence of thyroid nodular disease is quite high, spontaneously occurring at a rate of 0.08% per year starting in early life and extending into the eighth decade.47 Although thyroid nodules represent a wide spectrum of disease, most are colloid nodules, adenomas, cysts, and focal thyroiditis, with only a few (5%) being carcinoma. With a lifetime incidence of 4% to 7%, the annual incidence of thyroid nodules in the United States is about 0.1%, which is approximately 300,000 new nodules each year.61,62 Most of these nodules are benign and do not require removal. With approximately 37,000 new thyroid cancers each year, about 1 in 20 new thyroid nodules contains carcinoma, however, and approximately 1 in 200 nodules is lethal. The challenge in treating patients with thyroid nodules is to identify the patients with malignant lesions and to balance the potential morbidity of treatment with the aggressiveness of their disease.
Clinical Assessment: History and Physical Examination
Numerous findings should raise suspicion of malignancy in a patient presenting with a thyroid nodule. Younger and older patients are more likely to have a malignant thyroid nodule. Patients younger than 20 years have an approximately 20% to 50% incidence of malignancy when presenting with a solitary thyroid nodule.63 Nodular disease is more common in older patients, usually men older than 40 years and women older than 50 years. Although children may present with more advanced disease and even cervical metastases, malignancy in older patients has a considerably worse prognosis. Men often have more aggressive malignancies than women, but the overall incidence of thyroid nodules and malignancy is higher in women.
The physical examination of a patient with a thyroid nodule begins with careful palpation of the thyroid to assess the lesion. One should determine whether the lesion is solitary or the dominant nodule in a multinodular gland, although the risk of carcinoma in either setting is the same.3,48 Having the patient swallow assists in the examination because nonthyroid pathology does not typically elevate with the thyroid during swallowing. Palpable nodules are typically 1 cm or larger. Smaller nodules can be found incidentally on radiographic studies for other reasons and may be monitored. Lesions greater than 1 cm in size warrant a complete workup. The firmness of the nodule may be associated with an increased risk of carcinoma by twofold to threefold.64 Nodules greater than 2 cm in diameter and solid lesions have an increased incidence of harboring carcinoma. The evaluation of larger lesions also requires more caution because the rate of false-negative results during FNAC also increases.65
Further assessment of the patient may reveal the extent of involvement of a thyroid lesion. Palpable cervical nodes adjacent to the thyroid nodule increase the suspicion for malignancy, and may be the only presenting sign of a thyroid carcinoma. Adenopathy may be present, however, in a patient affected by Hashimoto’s thyroiditis, Graves’ disease, or infection.66,67 Large lesions can potentially shift the larynx and trachea within the neck. The mobility of the nodule relative to the laryngotracheal complex and adjacent neck structures should be evaluated. Malignant lesions are more likely to be fixed to the trachea, esophagus, or strap muscles.
Despite the importance of the initial clinical assessment, the history and physical examination are unreliable in predicting carcinoma. Many of the clinical signs of malignancy are manifest late in the course of disease. Additionally, many of these same findings may be caused by events associated with benign disease (e.g., hemorrhage into a benign nodule). The clinical assessment should provide a justification and a context for the interpretation of diagnostic studies, such as FNAC. Of particular note would be any patient and thyroid nodule features that might be concerning for aggressive carcinoma behavior (Table 124-3).
Table 124-3 Risk Factors for Aggressive Behavior of Well-Differentiated Thyroid Carcinomas
| History |
| Age |
| Younger—<20 yr |
| Older |
| Men—>40 yr |
| Women—>50 yr |
| Gender |
| Male > female |
| History of radiation exposure/therapy |
| Family history of thyroid carcinoma |
| Physical Examination |
| Hard, fixed lesion |
| Rapid growth of mass |
| Pain |
| Lymphadenopathy |
| Vocal cord paralysis |
| Aerodigestive tract compromise |
| Dysphagia |
| Stridor |
| Histopathologic Factors* |
| Size (>4 cm) |
| Extrathyroidal spread |
| Vascular invasion |
| Lymph node metastasis |
| Distant metastasis |
| Histologic type |
| Tall-cell variant of papillary carcinoma |
| Follicular carcinoma |
| Hürthle cell carcinoma |
Diagnostic Studies
Laboratory Studies
Most patients who present with a thyroid nodule are euthyroid. The finding of hypothyroidism or hyperthyroidism tends to shift the workup away from thyroid carcinoma to a functional disorder of the thyroid gland, such as Hashimoto’s thyroiditis or a toxic nodule.68 Although many thyroid hormone tests are available, few are needed in the initial patient evaluation. TSH measurement serves as an excellent screening test. Full thyroid function tests can be performed if the TSH level is abnormal.
Measurement of thyroglobulin is generally not performed initially because thyroglobulin is secreted by normal and malignant thyroid tissue, and it is not recommended in the 2006 ATA guidelines.69 Levels of thyroglobulin cannot differentiate between benign and malignant processes, unless levels are extremely high, as in metastatic thyroid cancer. Antithyroglobulin antibodies can also interfere with the assay. Thyroglobulin levels may be useful in studying patients who have undergone total thyroidectomy for well-differentiated thyroid cancer.
Fine-Needle Aspiration Cytology
FNAC has become the procedure of choice in the evaluation of thyroid nodules.69 The findings are highly sensitive and specific, although the accuracy of FNAC is related to the skill of the aspirator and the experience of the cytopathologist.70 The procedure is minimally invasive and may be performed quickly with little patient discomfort. In contrast to large-bore needle biopsies such as the Tru-cut or Vim-Silverman needle, there are fewer complications. With the advent of this technique, the number of patients requiring surgery has decreased by 35% to 75%, and the cost in managing patients with thyroid nodules has been substantially reduced.71–73 Also, the yield of malignancies has almost tripled in patients who have had thyroid surgery after FNAC.73,74 The accuracy of FNAC diagnosis of papillary carcinoma is 99% with a false-positive rate of less than 1%.75
FNAC should be one of the initial steps in the surgical evaluation of a thyroid nodule. Approximately 15% of all aspirates are inadequate or nondiagnostic, largely because of the sampling from cystic, hemorrhagic, hypervascular, or hypocellular colloid nodules. Repeat aspiration of such a nodule is crucial because a nondiagnostic finding should never be interpreted as a negative finding for carcinoma. Surgical diagnoses after repeated nondiagnostic aspirations revealed malignant nodules in 4% of women and 29% of men.76 Nodules that are difficult to localize and nodules that have yielded nondiagnostic aspirates on previous attempts may benefit from ultrasound-guided aspiration. FNAC is increasingly being performed with ultrasound guidance to improve diagnostic accuracy and yield. Cystic nodules with multiple nondiagnostic FNAC studies require close observation or surgical excision. Also, surgery should be more strongly considered for a solid nodule that is cytologically nondiagnostic.69
Successful FNAC categorizes a nodule into the following groups: benign, malignant, or suspicious. In 60% to 90% of nodules, FNAC reveals a benign or “negative” diagnosis. The likelihood of malignancy (false-negative rate) is 1% to 6%.71,77 The diagnosis of malignancy, particularly papillary (including follicular variant), medullary, and anaplastic carcinomas and lymphomas, can be determined in about 5% of nodules. The likelihood of a false-positive finding is less than 5%.71,77 Frequently, false-positive results occur because of difficulties in interpreting cytology in patients with Hashimoto’s thyroiditis, Graves’ disease, or toxic nodules. A benign cytology is a macrofollicular lesion or a colloid adenomatous nodule. The remaining “suspicious” samples are composed of lesions that contain abnormal follicular epithelium with varying degrees of atypia. This finding needs to be evaluated in the context of patient history and physical findings that may be suggestive of malignancy. A complete report of the FNAC detailing specimen adequacy and pathologic findings is crucial, and efforts have been made to standardize this information.78,79
Follicular neoplasms cannot be classified by FNAC alone. The presence of hypercellular, microfollicular arrays with minimal colloid increases the concern for carcinoma. The differentiation between follicular adenoma and follicular carcinoma depends on the histologic finding of capsular or vascular invasion, which requires evaluation of the entire thyroid nodule. Occasionally, patients with a diagnosis of follicular neoplasm on FNAC have an iodine-123 (123I) thyroid scan. If the suspicious nodule is “cold,” surgery is indicated. If the nodule is hyperfunctioning compared with the surrounding thyroid, surgery can be avoided. Overall, 20% of nodules diagnosed as follicular neoplasms by FNAC contain thyroid carcinomas.80
Similarly, Hürthle cell (oxyphilic) neoplasms can be difficult to evaluate. The presence of Hürthle cells in an aspirate may indicate an underlying Hürthle cell adenoma or carcinoma, but these cells can also be present in thyroid disorders, such as multinodular goiter and Hashimoto’s thyroiditis. Carcinomas can be found in 20% of nodules identified as follicular and oxyphilic neoplasms.81 Because of the risk of underlying carcinoma in these cases, surgery is recommended.
Imaging
A systematic ultrasound examination can be extremely valuable in the assessment of a patient with thyroid cancer, including color and power Doppler examination of the thyroid, specific nodules, and lymph nodes.82,83 Examination of the nodal basins should be bilateral and include the jugular, submandibular, supraclavicular, paratracheal, and suprasternal regions. These studies may detect cervical nodes that may contain early clinically occult metastatic disease that would not otherwise have been included in a surgical dissection.69,84 Characteristics of lymph nodes suspicious for metastatic deposits include loss of the fatty hilum, increased vascularity, rounded node configuration, hypoechogenicity of a solid nodule, and microcalcifications.83,85,86 Ultrasonography is also useful in the evaluation of cervical lymph nodes in patients with a history of thyroid cancer who present with adenopathy or increasing thyroglobulin levels. These studies are not useful, however, in the evaluation of substernal extent of disease or the involvement of adjacent structures.
In a patient with multiple thyroid nodules, FNAC should be performed in conjunction with a diagnostic ultrasound study. Aspiration of the largest or “dominant” nodule alone may miss a thyroid malignancy. In the presence of two or more thyroid nodules larger than 1 to 1.5 cm, nodules with a suspicious ultrasound appearance should be aspirated preferentially. If none of the nodules has suspicious ultrasound characteristics, and multiple sonographically similar coalescent nodules are present, aspiration of the largest nodule only is reasonable.69
Currently, there is no role for ultrasonography in screening asymptomatic patients for thyroid nodules. Preoperative ultrasound evaluation of the lateral cervical lymph nodes is recommended for all patients with papillary and Hürthle cell thyroid cancer before initial thyroidectomy because operative management may be altered in 20% of patients.87 In addition, intraoperative ultrasound examination may be useful in the localization of nonpalpable lesions in the thyroid bed or nodal metastases.
Computed tomography (CT) and MRI scans are usually unnecessary in the evaluation of thyroid tumors except for fixed or substernal lesions. Although these studies are not as effective as ultrasonography in the evaluation of thyroid nodules, they are more reliable in evaluating the relationship of the thyroid lesion to adjacent neck structures, such as the trachea and esophagus. These studies are useful in determining substernal extension, identifying cervical and mediastinal adenopathy, and evaluating possible tracheal invasion.88 Anatomic imaging should be obtained when visceral compartment invasion is suspected and for localization in patients with nodal disease. Also, CT or MRI can supplement ultrasound imaging, which cannot visualize the regions behind the sternum, trachea, and esophagus. Caution must be exercised in the use of iodine-containing contrast material in patients with multinodular goiter if a hyperthyroid state is suspected, and in patients with well-differentiated thyroid cancer. In the latter group, iodinated contrast media preclude the use of postoperative radioactive iodine therapy for 2 to 3 months. Finally, MRI is more accurate than a CT scan in distinguishing recurrent or persistent thyroid tumor from postoperative fibrosis.
Thyroid Isotope Scanning
Radionuclide scanning with 123I or technetium 99m (99mTc) sestamibi assesses the functional activity of a thyroid nodule and the thyroid gland. Nodules that retain less radioactivity than the surrounding thyroid tissue are termed “cold,” nonfunctioning, or hypofunctional. These cold nodules are thought to have lost functions of fully differentiated thyroid tissue and to be at increased risk of containing carcinoma. In a meta-analysis of patients with scanned nodules that were surgically removed, 95% of all nodules were cold.66,67 The incidence of malignancy in cold nodules was 10% to 15% compared with only 4% in hot nodules.
With the evolution of FNAC, radionuclide scanning is not routinely performed in the evaluation of a thyroid nodule. More frequently, “cold” nodules are detected in patients during evaluation for hyperthyroid disorders. Patients who present initially with a thyroid nodule, however, and are found to be hyperthyroid on preliminary thyroid function testing should have radionuclide scanning to differentiate between a toxic nodule and Graves’ disease and a nonfunctioning nodule. Also, after indeterminate FNAC, 123I thyroid scan should be considered. Surgical treatment should be contemplated if a concordant autonomously functioning nodule is not seen.69
Rational Approach to Management of a Thyroid Nodule
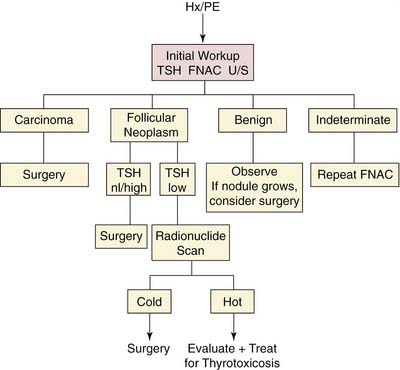
Numerous diagnostic algorithms have been proposed for the evaluation of a thyroid nodule (Fig. 124-3).70,89 Evaluation generally begins with a thorough history and physical examination to identify significant risk factors. Surgery may be deemed appropriate based solely on high-risk factors, such as age, sex, history of radiation exposure, rapid nodule growth, upper aerodigestive tract symptoms, and fixation.
Baseline TSH screening determines the diagnostic course. Patients with hyperthyroidism (suppressed serum TSH level) should receive radionuclide scanning to determine the presence of a toxic “hot” nodule, or Marine-Lenhart syndrome, or Graves’ disease with a concomitant “cold” nodule.90 A patient with hypothyroidism (elevated serum TSH level) should be appropriately treated by an endocrinologist, and then FNAC should be performed. Most patients are euthyroid (normal serum TSH level), and FNAC should be performed. Ultrasound examination can provide valuable diagnostic information, especially in the selection of a nodule for biopsy in a patient with multiple nodules, and may facilitate FNAC. In a patient with a thyroid malignancy, evaluation of the nodal basins may detect early clinically occult disease and alter surgical management. Patients with cytologic findings that are diagnostic or strongly suggestive of malignancy should be referred to a surgeon for removal of the lesion.
Review of Thyroid Neoplasms
Thyroid Adenoma
Pathology
The revised histologic classification of thyroid tumors divides epithelial tumors into the categories of follicular adenoma and other rare tumors (Table 124-4). Follicular adenomas are the most common benign thyroid lesions. Atypical follicular adenomas may show atypical microscopic features, including excess cellularity, increased mitotic figures, and necrotic foci. Although most of these lesions are benign, they may metastasize even in the absence of microinvasion.91
Table 124-4 World Health Organization Revised Histologic Classification of Thyroid Tumors
I. Epithelial tumors |
From Hedinger C, ed. Histological Typing of Thyroid Tumours. 2nd ed. Berlin: Springer-Verlag; 1988.
Management and Prognosis
Thyroid nodules that are determined to be benign require follow-up because of a low false-negative rate (approximately 5%) with FNAC.92,93 Nodule growth alone is not an indication of malignancy, but growth is an indication for repeat biopsy. The 2006 ATA guidelines recommend serial clinical examination for easily palpable benign nodules at 6- to 18-month intervals.69 All other benign nodules should be followed with serial ultrasound examinations 6 to 18 months after initial FNAC. Patients with nodules that remain stable in size may have subsequent examinations at longer time intervals. Patients with evidence of nodule growth should have repeat FNAC, preferably with ultrasound guidance.
Autonomously hyperfunctioning thyroid adenomas are usually anatomically and functionally stable. Although most patients do not develop thyrotoxicosis, 20% of patients with lesions greater than 3 cm may develop thyrotoxicosis. Surgery and radioiodine therapy can be used to manage these lesions, although many physicians prefer surgery for patients younger than 40 years. These patients may require preoperative medications to control thyrotoxic symptoms. The lesions are typically removed with a unilateral thyroid lobectomy. The remaining thyroid tissue typically returns to normal function after several months. Ethanol injection has become increasingly common, especially in Europe, to manage these lesions.94
Thyroid Cyst
Clinical Presentation
Although a thyroid cyst is not a specific diagnosis, this entity is frequently encountered in clinical practice. Approximately 15% to 25% of all thyroid nodules are cystic or have a cystic component.47 The presence of a cyst does not signify a benign lesion because papillary carcinomas and parathyroid tumors may manifest with cystic masses. Papillary carcinoma may be present in 14% to 32% of all cystic nodules, although most of these lesions are benign adenomas or colloid nodules.95,96
Pathology
A thyroid cyst can result from congenital, developmental, or neoplastic causes.95 Many cysts result from intranodular ischemia causing tissue necrosis and liquefaction. True epithelial-lined cysts are rare. Occasionally, parathyroid or thyroglossal duct cysts can be mistaken for thyroid nodules. A parathyroid cyst contains high parathyroid hormone levels within the clear fluid, and a thyroglossal duct cyst contains columnar epithelium. These lesions may also be differentiated by ultrasound imaging.
Management and Prognosis
When encountered during FNAC, a thyroid cyst should be drained completely. This procedure may prove curative in most simple cysts, although one or two additional drainage procedures may be required. If a cyst persists after three drainage attempts or reaccumulates quickly, the suspicion for carcinoma should increase. Brown fluid withdrawn from a cyst may represent old hemorrhage into an adenoma, but red fluid is more suspicious for carcinoma.64 Clear, colorless fluid may be withdrawn from a parathyroid cyst and can be assessed for parathyroid hormone.97 In suspicious cases, the surgeon and patient should consider ultrasound-guided FNAC to sample a solid component of the lesion or a unilateral thyroid lobectomy to obtain a definitive diagnosis. Because of the potential for thyroid carcinoma in cystic lesions, surgical excision for diagnosis is preferable to the injection of sclerosing agents.
Papillary Carcinoma
Clinical Presentation
Papillary carcinoma is the most common form of thyroid malignancy, accounting for 60% to 70% of all thyroid cancer.58,98 This lesion typically occurs in patients 30 to 40 years old, and is more common in women, with a female-to-male ratio of 2 : 1. This ratio has decreased steadily over the past 40 years as the incidence in men has increased.99 Papillary carcinomas are the predominant thyroid malignancy in children (75%). Although children more commonly present with advanced disease, including cervical and distant metastases, their prognosis remains quite favorable.
Most cases of papillary carcinoma occur spontaneously. Patients with a history of low-dose radiation exposure tend to develop papillary carcinomas (85% to 90%).100 These lesions also are more common in patients with Cowden’s syndrome (familial goiter and skin hamartomas), Gardner’s syndrome (familial colonic polyposis), and familial polyposis. Only 6% of papillary carcinomas are associated with familial disease.
Papillary carcinoma can be classified into three categories based on size and extent of the primary lesion.101,102 Minimal carcinoma or occult carcinoma/microcarcinoma tumors are 1.5 cm or smaller in size, and show no evidence of invasiveness through the thyroid capsule or to cervical lymph nodes. These lesions are typically nonpalpable and are usually incidental findings during operative or autopsy examination. Intrathyroid tumors are greater than 1.5 cm in diameter, but are confined to the thyroid gland with no evidence of extrathyroid invasion. Extrathyroid tumors extend through the thyroid capsule to involve the surrounding viscera. This latter form of papillary carcinoma is associated with substantial morbidity and decreased survival.7,101
Most patients present with a slow-growing, painless mass in the neck and are often euthyroid. Often, the primary lesion is confined to the thyroid gland, although 30% of patients may have clinically evident cervical nodal disease.103,104 Histologic studies have shown the strong lymphotropic nature of papillary carcinoma, leading to multifocal disease within the thyroid and regional lymphatics. Microscopic disease has been identified in the cervical nodes of 50% to 80% of patients and in the contralateral lobe in 80% of patients with papillary carcinoma at the time of surgery.105 The significance of this microscopic disease is unclear, however, because clinical recurrences in the neck and in the contralateral lobe occur in less than 10% of patients.106 More likely, the prevalence of microscopic disease suggests that most papillary carcinomas have an indolent course that only occasionally becomes clinically evident. Definite predictors of the clinical course for papillary carcinoma are not well defined, however.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree