Chapter 5 Management of the Patient with Asthma
Definition
Asthma is defined as a chronic inflammatory disorder of the airways causing recurrent episodes of cough, wheeze, shortness of breath, and/or chest tightness.1 These episodes are associated with increased airway sensitivity to a variety of stimuli leading to variable airflow obstruction that is often either preventable or reversible with proper treatment. Clinical manifestations of asthma can be controlled with appropriate management. With asthma controlled, no more than seldom flare-ups should occur, with severe exacerbations presenting rarely, if ever.
This definition of asthma has evolved over the past three decades. Prior to the 1990s asthma was considered primarily a bronchospastic disease.2 The role of inflammation was not appreciated, and treatment paradigms therefore focused on management of the bronchospasm without consideration of addressing the underlying progressive chronic inflammation. With a better appreciation of the pathophysiology of asthma, effective treatments have been developed that treat not only the acute symptoms of asthma but also the underlying inflammatory disease at its core.
Epidemiology
Asthma affects approximately 300 million individuals worldwide. The World Health Organization (WHO) has estimated 15 million disability-adjusted life years are lost each year due to asthma, representing a staggering 1% of the total global disease burden. Asthma mortality is at 250 000 lives lost yearly irrespective of disease severity.3 There is also significant international, national, and regional variability in asthma prevalence, a phenomenon which is not well understood. The economic impact of the disease is driven by level of control and extent of emergent care, highlighting the need for a systematic approach to management.
Pathophysiology
Airway inflammation is the hallmark of disease pathophysiology in asthma. As with other allergic conditions, activated mast cells, eosinophils, and T cells (Th2 and invariant natural killer T cells) are associated with disease expression. Inherent structural cells, such as airway epithelial and smooth muscle cells, also can play a role in the disease pathology.4
Airway narrowing is the final common pathway that leads to the symptoms of asthma. In a variable and complex interplay, smooth muscle contraction, airway edema, airway thickening, and mucous hypersecretion may each play a role in airway narrowing.5
Diagnosis
The clinical diagnosis of asthma includes symptoms of breathlessness, wheezing, cough, and/or chest tightness.1 These symptoms are usually episodic and recur with allergen or irritant exposure, seasonal variability, exercise, and/or concomitant respiratory infection. A family history of asthma and atopy is helpful, but not necessary, in making the diagnosis. Clinical and/or pulmonary function response to bronchodilation assists the diagnosis confirmation. Symptoms can occur at any time of day but are more frequently worse at night due to the circadian pathophysiology described above. A useful set of questions is important for all clinicians to consider in making the diagnosis.
Cough may present as the sole symptom of asthma and in most cases should not be treated differently than other symptoms. The differential diagnosis for cough, is of course, quite broad and therefore the clinician should carefully rule out other causes if the clinical diagnostic picture is unclear. In conditions where lung function and its variability are normal, diagnoses such as chronic sinusitis, allergic rhinitis, gastroesophageal reflux, chronic rhinitis with postnasal drainage, vocal cord dysfunction, eosinophilic bronchitis, and cough associated with angiotensin-converting enzyme (ACE) inhibitors should be considered.
▪ Pulmonary Function Testing
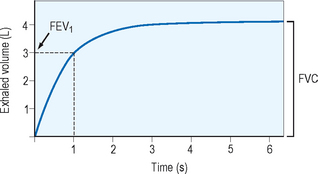
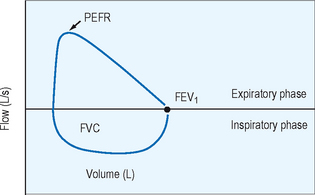
Spirometry provides the best assessment of airflow obstruction available to the clinician. Measurements of forced expiratory volume in one second (FEV1) in relation to the forced vital capacity (FVC), as well as forced expiratory flow and peak flow, are compared to population normative values to provide clinical comparison. Published recommendations for standardized testing are available. Spirometric testing quality is maximized by well and properly trained technicians and, for younger patients, interactive software. Because of anthropomorphic differences, population normative values chosen should consider ethnic variation. A normal spirometric tracing is represented in Figure 5.1. An example of a flow–volume loop is displayed in Figure 5.2.
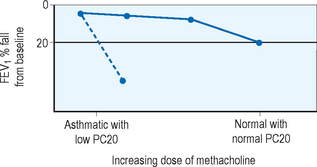
In patients who exhibit normal lung function without symptoms at the time of clinical evaluation, measurement of airway hyperresponsiveness is often helpful. Usually conducted within a specialist’s office or laboratory, the airway challenge is a sensitive tool which, if normal, helps to rule out the diagnosis of asthma. Various challenge agents are used such as methacholine, histamine, cold air, mannitol, or exercise to evaluate airway response. A 20% fall in FEV1 in relation to the quantity needed to provoke that fall gives the clinician a quantifiable value for airway sensitivity. An example of the change in FEV1 with methacholine challenge is displayed in Figure 5.3. Airway hyperresponsiveness has been described in illnesses other than asthma such as chronic obstructive pulmonary disease (COPD), cystic fibrosis, and bronchiectasis so this tool, while sensitive, is not specific and the clinician should consider other diagnostic possibilities when faced with a positive test result.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree