CHAPTER 108 Management of Early Glottic Cancer
Background
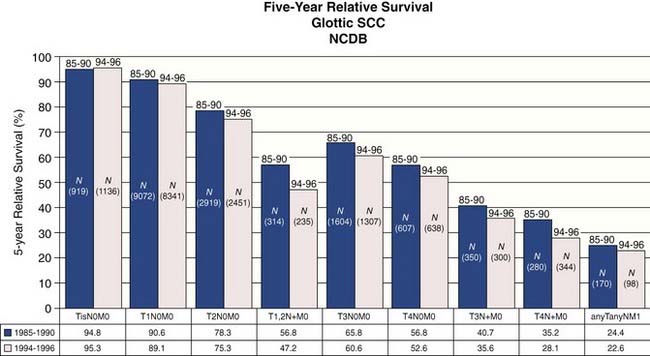
Statistics from the National Cancer Database (NCDB) provide insight into contemporary demographics and management of laryngeal cancer in the United States.2 The most common site for laryngeal squamous cell carcinoma is the glottic larynx (Fig. 108-1). The role for surgery as single-modality therapy for advanced laryngeal cancer is diminishing as combined chemoradiation has become more common in the initial management (Fig. 108-2). Five-year relative survival was best for early-stage (stages 0, I, and II) glottic cancer (Fig. 108-3).

Figure 108-1. Cancers isolated to the glottic larynx account for approximately one half of all laryngeal cancer cases in the United States.
(From Hoffman HT, Porter K, Karnell LH, et al. Laryngeal cancer in the United States: changes in demographics, patterns of care, and survival. Laryngoscope. 2006;116[9 Pt 2 Suppl 111]:1-13.)

Figure 108-2. General treatment type by year for laryngeal squamous cell carcinoma.
(From Hoffman HT, Porter K, Karnell LH, et al. Laryngeal cancer in the United States: changes in demographics, patterns of care, and survival. Laryngoscope. 2006;116[Suppl 111]:1-13.)
Analysis of this dataset demonstrated that squamous cell carcinoma is the dominant histologic type of laryngeal cancer in the United States, representing approximately 95% of cases (Fig. 108-4). Unless indicated otherwise, the remainder of the discussion in this chapter focuses on squamous cell carcinoma.3,4
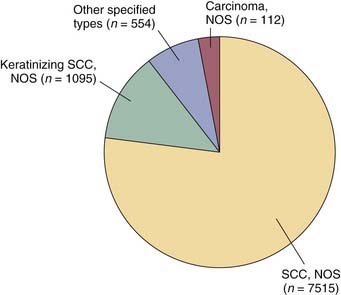
Figure 108-4. Histology of laryngeal cancer cases diagnosed in 2000 national cancer database.
(From National Cancer Database Benchmark Report: Histology of Laryngeal Cancer in the U.S. in 2000. © Commission on Cancer, American College of Surgeons. NCDB Benchmark Reports, v1.1. Chicago, 2002. [The content reproduced from the applications remains the full and exclusive copyrighted property of the American College of Surgeons. The American College of Surgeons is not responsible for any ancillary or derivative works based on the original text, tables, or figures.])
Etiology
Squamous cell carcinoma of the larynx is strongly associated with the use of tobacco and alcohol. An increase in the prevalence of cigarette use among females in the past has been linked to a proportionate increase in the number of female patients with laryngeal cancer. Other factors implicated in the development of laryngeal cancer include passive smoking, chronic laryngeal irritation from gastroesophageal reflux, and viral infection.5–9
Since 1979 gastric acid reflux has been identified as a risk factor for laryngeal and pharyngeal cancer.10,11 Alkaline reflux has more recently been identified as a laryngeal carcinogen by Galli and colleagues.12 These investigators identified a higher than expected number of patients with pharyngolaryngeal cancer associated with achlorhydria and presumed alkaline reflux after gastric resection. They concluded that periodic endoscopy of the upper aerodigestive tract should be performed on gastrectomized patients because of their higher risk for pharyngolaryngeal disorders in general and cancer in particular. These investigators also identified that, among patients with laryngeal squamous cell carcinoma with an intact gastric acid secreting mechanism, 81% showed abnormal acid reflux on 24-hour pH testing.
Human papillomavirus (most notably the oncogenic type 16) has been associated with head and neck cancer.13–15 The role for human papillomavirus (HPV) infection in laryngeal oncogenesis has been supported by previous study but does not have as strong an association as has been identified with cancer of the oropharynx and oral cavity.16,17 Initial study suggests that a difference exists in behavior between HPV and non–HPV-related squamous cell carcinoma of the upper aerodigestive tract, with a better prognosis afforded the HPV-positive cases.18
Differences in exposure to carcinogens or genetic susceptibility may explain the differences in the predominant types of laryngeal cancer throughout the world. Glottic cancers predominate over supraglottic cancers in the United States at a ratio of 2 : 1. In France the opposite occurs, where the number of supraglottic cancers exceeds that of glottic cancers at a ratio of 2 : 1.19 In Finland the incidence of supraglottic cancer decreased and the incidence of glottic cancers increased between the decades 1976 to 1985 and 1986 to 1995.20 Supraglottic cancers were more common than glottic cancers in the earlier decade (ratio of 1.4 : 1). Glottic cancers predominated in the later decade at a ratio of 2 : 1. The reason for this change in distribution of laryngeal cancers in Finland is not clear. A low exposure to alcohol and a high availability of medical services were factors Brenner and colleagues identified as important determinants to the characteristics of laryngeal cancer in Israel. These investigators linked these factors to the high proportion of laryngeal cancers treated at an early stage with an overall better outcome when compared with other countries.21
Staging
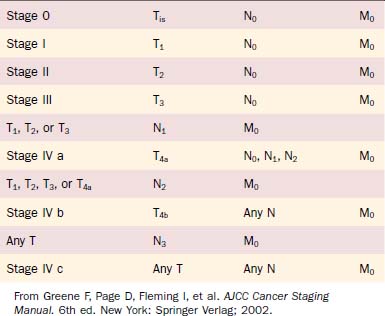
The most widely accepted system for classifying cancer in the United States, as is currently promoted by the American Joint Committee on Cancer (AJCC), had its beginning in 1959 with the organization of the American Joint Committee for Cancer Staging and End-Results Reporting (AJC).22 This staging system was first published in 1977 in the Manual for Staging of Cancer.23 The staging system has evolved to its current form as it is codified in the AJCC Staging Manual, Sixth Edition published in May of 2002 and implemented as of January 1, 2003.24 Major revisions made to the fifth edition in the head and neck section were directed primarily to the advanced-stage cases. Although the definitions for stages 0, I, and II laryngeal cancers remained unchanged, alterations to the T3 definitions in the sixth edition have impacted on the interpretation of T1 and T2 classification.
The anatomic definitions and boundaries between sites, as published in the AJCC Staging Manual, have remained constant across the multiple editions. The glottis has been defined as the true vocal cords including the anterior and posterior commissures. Unfortunately, the definition of the posterior commissure remains vague. It is difficult to discriminate among the glottis, the adjacent supraglottis (arytenoids), and the hypopharynx (postcricoid region) in the region of the posterior commissure.25 The superior border of the glottis is more clearly defined by a horizontal plane passing through the apex of the ventricle. The inferior limit of the glottis is defined by a line parallel to and 1 cm inferior to this plane.
Glottic cancers are staged primarily by clinical assessment of degree of vocal cord mobility (Table 108-1).24 Cancers confined to the glottis with normal mobility are staged T1. Subdivision of T1 vocal cord cancers is based on extension to involve the opposite cord. A tumor limited to one vocal cord is T1a. A horseshoe lesion that extends around the anterior commissure to involve both vocal cords without impairing mobility is T1b. Despite consistency across all previous editions of AJCC staging manuals indicating that T4 status is based on “tumor invasion through the thyroid cartilage …, ” it has been common clinical practice to consider any extent of thyroid cartilage invasion from a glottic cancer to be cause for T4 classification. Clinically apparent stage I glottic cancers (T1N0M0) were reassigned stage IV status (T4N0M0) when “microscopic invasion of adjacent cartilage” was identified.26 Revision to the AJCC Staging Manual, Sixth Edition identifies that minor cartilage erosion (e.g., inner cortex) should be classified as T3 rather than T4.
Table 108-1 Primary Tumor (T) Classification of Glottic Cancer
| Tx | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Tis | Carcinoma in situ |
| T1 | Tumor limited to the vocal cord(s) (may involve anterior or posterior commissure) with normal mobility |
| T1a | Tumor limited to one vocal cord |
| T1b | Tumor involves both vocal cords |
| T2 | Tumor extends to supraglottis or subglottis, or with impaired vocal cord mobility |
| T3 | Tumor limited to the larynx with vocal cord fixation or invades paraglottic space, or minor thyroid cartilage erosion (e.g., inner cortex) |
| T4a | Tumor invades through the thyroid cartilage or invades tissues beyond the larynx (e.g., trachea, soft tissues of neck including deep extrinsic muscle of the tongue, strap muscles, thyroid, or esophagus) |
| T4b | Tumor invades prevertebral space, encases carotid artery, or invades mediastinal structures |
From Greene F, Page D, Fleming I, et al. AJCC Cancer Staging Manual. 6th ed. New York: Springer Verlag; 2002.
Still further clarification is necessary to address inconsistencies that persist among publications describing the paraglottic space.12,27–29 Berman defined the borders of the paraglottic space as they have been commonly reported by others:
This definition fails to address the controversy as to whether all, part, or none of the thyroarytenoid muscle is part of the paraglottic space.30 An assessment by Weinstein and colleagues31 defines the medial border of the paraglottic space as the tissue lateral to the conus elasticus at the glottic level and lateral to the quadrangular membrane at the supraglottic level. By this convention, the thyroarytenoid muscle is considered part of the paraglottic space. Others define the glottic portion of the paraglottic space as the fat deep to the intrinsic laryngeal musculature. For the purposes of T classification according to the revised AJCC criteria, it is reasonable to consider that paraglottic space invasion (T3) occurs when the fat compartment deep to the intrinsic laryngeal musculature is involved with tumor. Impaired vocal cord mobility (T2) (without paraglottic space involvement) may be considered when the thyroarytenoid or lateral cricoarytenoid muscles are infiltrated but not fully transgressed.
Carcinoma in situ is classified as Tis. Accuracy in classifying premalignant changes affecting the laryngeal epithelium is compromised by the subjective nature of the assessment and by a lack of uniformity in terminology. Some pathologists discriminate between severe dysplasia and carcinoma in situ, whereas others group them together as type III intraepithelial neoplasia.32–34 The grading of laryngeal dysplasia as mild, moderate, and severe or as carcinoma in situ originated from a similar classification of preneoplastic diseases of the uterine cervix. Keratinizing dysplastic lesions of the upper airway are more common than in the cervix and are also more difficult to categorize. The differences that exist in the behavior of carcinoma in situ originating at these two distinctly different anatomic sites has also undermined the transfer of concepts governing uterine cervical carcinoma in situ to laryngeal carcinoma in situ.35 Despite the clearly stated pathologic principle that high-grade dysplasia and carcinoma in situ are equivalent terms in describing abnormal laryngeal squamous epithelium, clinical management is generally less aggressive for high-grade dysplasia than for carcinoma in situ.
The stage grouping of primary tumor, regional lymph nodes, distant metastatic (TNM) classifications for laryngeal cancer is the same as for other head and neck mucosal sites (Table 108-2). Although the stage groupings have been revised—with the sixth edition introducing the division of stage IV into IV a, IV b, and IV c—the staging of early glottic cancer has remained constant (stages 0, I, and II).
Definitions: Early Glottic Cancer
The appropriate definition of the term early as it applies to laryngeal cancer has been debated. Some investigators have loosely used the term to describe laryngeal cancer in the context of management options. By this convention, a laryngeal cancer is considered early if it can be treated by partial laryngeal (conservative) surgery without a neck dissection, by endoscopic excision, or by radiotherapy alone.36 Others apply the term early to stage 0, I, or II tumors and late to stage III or IV tumors. This definition of early may be inappropriate. Because the extent of disease—not the rate of growth—determines staging, the term localized is more accurate than the term early.37 Similarly, the term advanced may be more appropriate than the term late.38 The most accurate grouping uses the terms localized for any-TN0M0 disease, regionally spread or vicinally spread for any-TN+M0 disease, and widely disseminated for any-T-any-NM1 disease.39 Convention and tradition dictate that this chapter continue to use the terms early to refer to lower stage (0, I, and II) and late to refer to higher stage (III and IV).
Diagnosis
Endoscopic Imaging
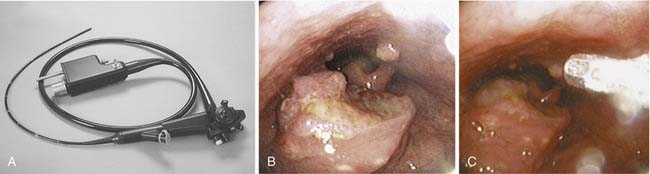
The development of flexible fiberoptic endoscopes designed for transnasal endoscopy has also permitted use of these instruments to evaluate the larynx and trachea.40–43 The improved optics of these instruments—coupled with the presence of a side port that permits the use of suction, placement of biopsy instruments, and instillation of topical anesthetics—has expanded the capacity to perform in-office laryngeal evaluations (Fig. 108-5A). Although direct laryngoscopy remains the most appropriate approach to evaluate and biopsy the majority of lesions suspected of being malignant, in selected cases biopsy can be effected in the clinic (see Fig. 108-5B and C).
When videostroboscopy was introduced as a common clinical tool, enthusiasm was high for its potential to help discriminate invasive cancer from epithelial atypia on the basis of the stroboscopic appearance of the vocal fold epithelial pliability.44,45 Subsequent experience of Colden and colleagues46 has identified that laryngeal stroboscopy is neither reliable in distinguishing vocal fold atypia from invasive cancer nor useful in determining the depth of invasion of vocal fold cancer. These investigators did suggest that the stroboscopic appearance of a relatively normal mucosal wave indicates that a lesion is superficial and does not extensively involve the underlying vocal ligament.
Advances in technology, coupling physical and biologic analysis to endoscopy, have identified several adjuncts to complement standard imaging of the larynx at the time of direct laryngoscopy. Lugol’s solution, as well as toluidine blue, have been used to stain laryngeal tissue in an effort to discriminate normal tissue from tumor.47,48 These approaches have not met with uniform success and therefore have not been widely adopted. Fluorescent tumor markers such as tetracyclines and hematoporphyrin derivatives are preferentially absorbed by tumor cells and can be used to discriminate tumor from adjacent normal tissue by the red fluorescence they emit when exposed to ultraviolet light.49 Additional cost and operating time, as well as problems with skin photosensitivity, have limited the usefulness of this technique. A more promising approach employing autofluorescence laryngoscopy has been reported. Zargi and colleagues50 have employed the lung imaging fluorescence endoscope (LIFE) system to evaluate laryngeal lesions. Their preliminary work identified differences in the intensity of autofluorescence between normal and tumor tissue when exposed to blue or violet light. They also identified shortcomings in the use of the LIFE system for this purpose, noting that further technical advances are necessary to make this endeavor a useful clinical tool.
Stone and colleagues51 reported that the fluorescence of biologic tissue actually may impede the use of Raman spectroscopic analysis to discriminate normal from malignant laryngeal lesions. These investigators have developed strategies that negate the impact of the natural biologic fluorescence to permit evaluation of the distinctive spectral signatures of tissues elicited when exposed to monochromatic laser excitation. This strategy may develop into a noninvasive tool in the early detection of cancerous and precancerous lesions.
Radiology
Chest X-Ray
A chest radiograph is necessary in the evaluation of patients with glottic cancers to assess for concurrent pulmonary disease and to screen for second primary lung cancers. The incidence of distant metastatic spread of an early glottic cancer to the lung is sufficiently rare that a preoperative chest radiograph is not generally intended to survey for that specific purpose. Annual chest x-rays should be obtained in the course of clinic follow-up examinations primarily because of the high incidence of second primary lung cancers in patients managed for laryngeal cancer.52
Magnetic Resonance Imaging and Computed Tomography
In our institution most early laryngeal cancers will be assessed clinically and with a fine cut computed tomography (CT) to determine invasion into or through the thyroid cartilage and to assess for spread into the paraglottic space. The appropriate role for magnetic resonance imaging (MRI) and CT in early glottic cancers is still being defined.53 These advanced imaging studies are of questionable value in the assessment of most superficial T1 lesions of the vocal cords. Selected T1 lesions with anterior commissure involvement may benefit from MRI or CT imaging if extension of the tumor superiorly into the preepiglottic space or anteriorly into the thyroid cartilage is detected. The more routine use of MRI or CT is advocated in the evaluation of T2 lesions not only to help determine tumor extent (and hence T classification) but also tumor volume. Some authors have suggested that CT or MRI of T2 lesions may help determine radiocurability by assessing tumor volume. Other investigators have not found the size of the tumor to be useful for predicting radiocurability.54
Wenig and colleagues55 contend that surgical treatment of laryngeal cancer with less than a total laryngectomy mandates preoperative MRI in almost all cases. These investigators identify that tumor involvement of the entire vocal cord or the anterior commissure, as well as the presence of vocal fold paresis, is considered an indication for the use of MRI. They relate that only the most superficial T1 lesions of the mobile portion of the vocal cord need not be imaged because MRI will not detect the extent of these lesions. This philosophy is distinctly opposite from that of Steiner and Ambrosch,56 who relate that advanced imaging of laryngeal cancer is fraught with artifact. Steiner and Ambrosch find little value in preoperative imaging of laryngeal cancers and relate that the most accurate assessment of tumor extent comes at the time of endoscopic laser microsurgical resection.
Debate continues as to whether CT or MRI offers the most useful information when imaging the larynx.57 Advances in the speed with which CTs can now be done offer an advantage to this technique. In 2002 Stadler and colleagues58 reported that use of rapid imaging spiral CTs of the larynx permits complete evaluation during the course of a single maneuver. This rapid imaging of the larynx may be accomplished as the patient performs either a modified Valsalva maneuver or phonation of the utterance “E.” This functional imaging (done during the maneuver) was compared with nonfunctional imaging (done with quiet breathing) and correlated with postsurgical pathology assessments and findings at microlaryngoscopy. Although the numbers in their study were small, Stadler and colleagues conclude that functional CT imaging appears to be more accurate in determining tumor extent than nonfunctional imaging.
Positron Emission Tomography
The role for positron emission tomography (PET) is continuing to expand. Although distant metastases from early laryngeal cancers are rare, the most common cause of death in patients with early laryngeal cancer is second primary tumor.59,60 The most common second primary tumors are lung followed by other head and neck primaries, which are more easily detected clinically and with endoscopy. It has become standard practice in our institution to order a PET scan in all but the earliest T1 laryngeal cancers.
F-18-fluoro-deoxy-glucose (FDG) PET scanning has been reported as useful in detecting subclinical recurrent or persistent cancer at a stage when it is highly curable.61 Terhaard and colleagues62 propose an algorithm for the follow-up of patients treated for laryngeal or hypopharyngeal cancers that emphasizes the use of PET imaging. Through a prospective study of 109 PET scans done on 75 patients with either laryngeal or hypopharyngeal cancer, these investigators concluded that an FDG PET scan should be the first diagnostic step when a recurrence is suspected after treatment with irradiation. As a result of this study, in which the majority of cases were glottic and classified as either T1 or T2, these investigators suggested that no biopsy is necessary if the scan is negative. In the face of a positive scan and a negative biopsy, a follow-up scan showing decreased FDG uptake would indicate that a recurrence is unlikely. Caution should be exercised in following these guidelines if other clinical indicators support the need for a biopsy. At this point in our experience, endoscopy with a biopsy to sample tissue suspicious in appearance for cancer remains the most critical means by which recurrence is evaluated.
Shortcomings of PET imaging have been noted. Most investigators suggest waiting at least 3 months after treatment to perform PET imaging because false-positive results are common if the imaging is done soon after radiotherapy is completed.63 False positives may also result from infection, radionecrosis, or accumulation of saliva in the vallecula.64,65 Some of these shortcomings of PET imaging are being addressed by use of pharmaceuticals other than FDG. The presence of viable tumor has been identified through PET imaging with 1-[1-11C]-tyrosine (TYR). TYR PET imaging analyzes protein synthesis activity and has been reported to be successful in detecting recurrent squamous cell carcinoma of the larynx.66 Further progress with PET imaging is anticipated with the introduction of other pharmaceuticals.
Treatment
Surgery
Physicians’ views vary on the indications for use of a specific procedure or radiotherapy technique. General guidelines for selection of an approach based on the extent of a lesion are listed in Table 108-3 and depicted in Fig. 108-6. The choice of therapeutic approach for a patient requires consideration of many other variables in addition to the anatomic extent of the cancer. Overall cure rates and laryngeal preservation figures are high for the surgical management of early glottic squamous cell carcinoma (Table 108-4).
Table 108-3 Selected Surgical Procedures for Glottic Cancer
| Procedure | Indications | Study |
|---|---|---|
| Microlaryngoscopy partial cordectomy with or without carbon dioxide laser (excisional biopsy) | T1 midcord leukoplakia, Tis/microinvasion | Strong and Jako (1972) |
| Kleinsasser (1990) | ||
| Shapshay, Hybels, and Bohigian (1990) | ||
| Cordectomy complete with or without carbon dioxide laser (endoscopy) | T1 midcord | Strong (1975) |
| Annyas et al (1990) | ||
| Shapsay, Hybels, and Bohigian (1990) | ||
| Eckel and Thumfart (1992) | ||
| Cordectomy (laryngofissure) | T1 midcord | DeSanto et al (1977) |
| Bailey (1985) | ||
| Frontolateral partial laryngectomy | T1 with extension to anterior commissure | Leroux-Robert (1975) |
| Hemilaryngectomy and extended hemilaryngectomy | T1/T2 with extension to arytenoid, without anterior commissure fixation (minimal) | Norris (1958) |
| Som (1975) | ||
| Mohr et al (1983) | ||
| Anterior partial laryngectomy with epiglottoplasty or with keel | T1/T2 with extension to anterior commissure (without arytenoid) | Tucker et al (1979) |
| Som and Silver (1968) | ||
| Subtotal laryngectomy with cricohyoidoepiglottopexy | T1b/T2 bilateral anterior involvement; may include removal of one arytenoid | Laccourreye et al (1990) |
| Charlin et al (1988) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree