CHAPTER 100 Malignant Neoplasms of the Oropharynx
About 10% to 12% of all malignancies of the upper aerodigestive tract occur in the oropharynx. Approximately 130,000 new cases were detected worldwide in 2002.1 Histopathologically, most malignancies found in the oropharynx (>90%) are squamous cell carcinomas (SCCs) with an incidence of 1 to 3/100,000 in the United States and Europe per year. Although the male population has a higher prevalence of oropharyngeal cancer, future counts will depend on tobacco and alcohol consumption behavior, which is the major risk factor for oropharyngeal carcinoma, followed by occupational and environmental pollutants and endogenous risks.
Anatomy of the Oropharynx
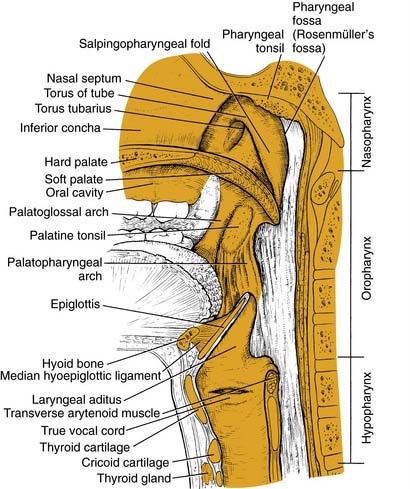
The structure of the oropharynx is complex and can be subdivided into the soft palate, tonsils, and base of tongue respecting different site-specific characteristics. The oropharyngeal borders are the soft palate superiorly and the hyoid bone and the vallecula inferiorly. The ventral border is the base of tongue, which ends at the circumvallate papilla. Laterally and dorsally the oropharynx contains the tonsillar region and the lateral and dorsal pharyngeal wall, respectively. In the following sections, the anatomic sites are described separately, accounting for their specific characteristics (Fig. 100-1).
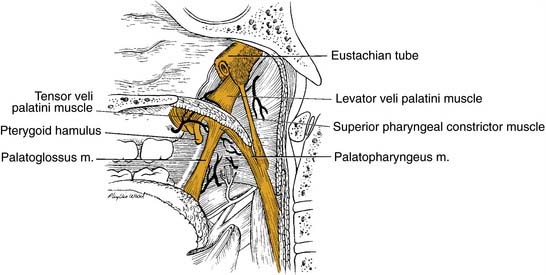
Tonsillar Fossae
Base of Tongue
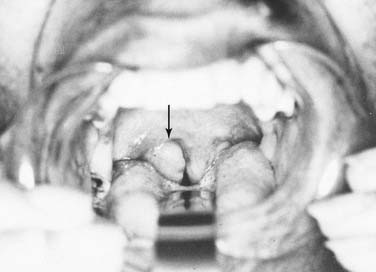
The base of tongue reaches from the circumvallate papilla to the vallecula and to the glossopalatine sulci laterally. The tongue base is rich in lymphatics and contains part of Waldeyer’s ring. The blood supply is covered by the lingual arteries. Innervation of the tongue musculature is provided by the hypoglossal nerve (Fig. 100-2).
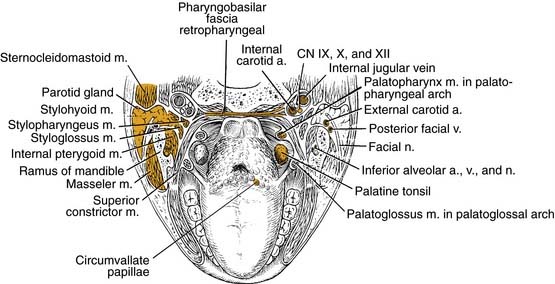
Oropharyngeal Wall
The posterior pharyngeal wall of the oropharynx reaches from the region of the soft palate to the epiglottis and borders the tonsillar fossae and the lateral aspect of the piriform sinuses laterally. The wall is composed of superior constrictor muscle and the buccopharyngeal and pharyngobasilar fascia covered by the pharyngeal mucosa (Fig. 100-3).
Lymphatics
The lymphatic pattern of the oropharynx is complex, and lymphatic spread from malignant lesions depends on size and location of the primary malignancy. Its understanding is of high importance for the outcome of surgical and/or radiotherapy treatment of oropharyngeal lesions. A number of previous studies have defined the routes of lymphatic spread in the head and neck.2–4 The clinical neck levels are defined by levels I to VI with subdivisions A and B for levels I, II, and V (Fig. 100-4).5
Oropharyngeal carcinoma is predisposed to drain to levels II, III, and IV with possible further spread to other regions in extensive disease.6 However, primary drainage is to the jugulodigastric nodes in the upper deep jugular chain (level II) and to the retropharyngeal and parapharyngeal nodes in the retropharyngeal and parapharyngeal space, respectively. Other metastatic spread may involve midcervical (level III) and lower cervical nodes (level IV). Skip metastases to other clinical levels are extremely rare. Shah described in a retrospective study of 1119 radical neck dissection specimens that tumors of the oropharynx characteristically metastasized to lymph node levels II to IV. There was no positive node involvement of level V when levels II to IV were negative.7
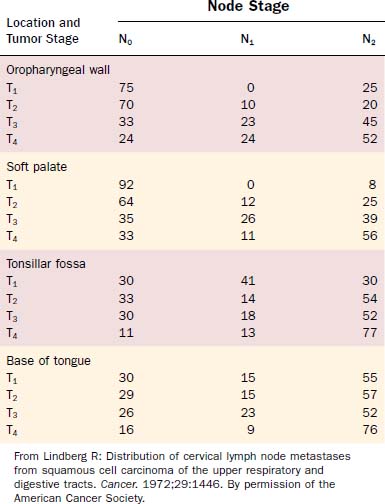
Attention should be spent on the location of the primary malignancy near the midline. Tumors of the base of tongue, the soft palate, and the posterior pharyngeal wall have a higher incidence of bilateral lymphadenopathy. These considerations should influence therapeutic planning to optimize therapy and minimize patient morbidity (Figs. 100-5 and 100-6, Table 100-1).
Pathology
SCC is the most common malignancy of the oropharynx, comprising more than 90% of all malignant tumors of that origin. Other malignant transformations are lymphomas, lymphoepithelial carcinoma, minor salivary gland tumors, malignant melanomas, and other rare malignancies. The World Health Organization Classification of Tumours outlines the presently known malignancies of the oropharynx (Table 100-2).8
Table 100-2 World Health Organization Classification of Malignant Tumors of the Oropharynx
Epithelial Precursor Lesions
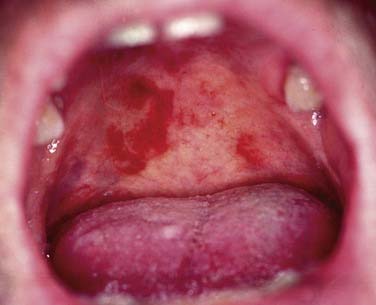
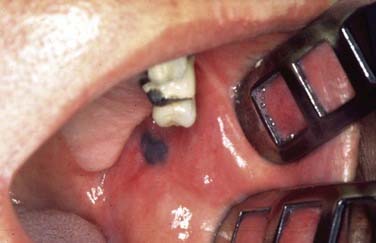
In general, precursor lesions of the oropharynx clinically present as white patches (leukoplakia) (Fig. 100-7) or red patches (erythroplakia). Some lesions appear as mixed variants with white and red components. Most white lesions do not show dysplastic cells and relate to hyperplasia. However, erythroplakia or mixed lesions frequently display dysplasia. Whereas leukoplakia rarely undergoes malignant transformation and may even regress after elimination of underlying etiologic factors, erythroplakia can often lead to malignancy.
Carcinoma in situ is described as malignant transformation without invasion, which is difficult to distinguish morphologically. WHO experts recommend that CIS be diagnosed when full-thickness architectural abnormality and severe cytologic atypia is found.9
Squamous Cell Carcinoma
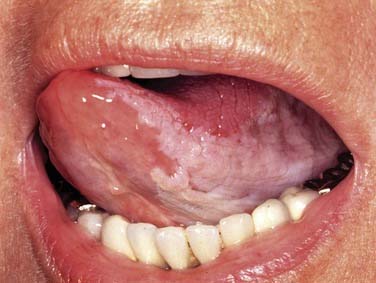
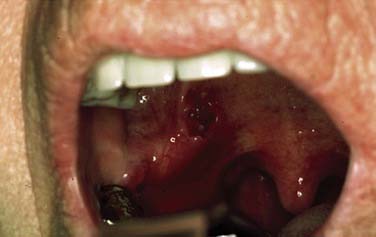
SCC is defined as an invasive epithelial neoplasm with varying degrees of squamous differentiation and a propensity to early and extensive lymph node metastases, occurring predominantly in alcohol- and tobacco-using adults in the fifth and sixth decades of life.10 The clinical appearance varies and presents as exophytic, flat, ulcerated, verrucoid, or papillary in growth (Fig. 100-8). Independent from the gross appearance, histopathologic patterns of invasion demonstrate multiple variations as well. SCC can vary from keratinizing to nonkeratinizing and well differentiated to poorly differentiated. Severe dysplasia and CIS are common elements found in association with invasive SCC. Invasive carcinoma eliminates the regular architecture and can include lympho-vascular space invasion, neurotropism, and infiltration of other tissue components such as muscle or cartilage, demonstrating aggressive behavior of the tumor.
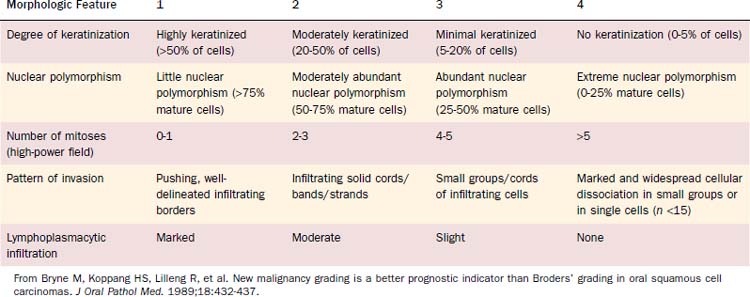
Malignancy grading can be a prognostic marker, although different grading systems have been proposed with diverse impact on prognosis. Broders suggested a four-tiered grading system for carcinoma of the lip in 1929.11 His grading system allows assessment of tumor differentiation with some predictability of prognosis because poorly differentiated tumors are more likely to recur.12,13 However, the grading system has also been shown to lack consistent prediction of prognosis using multivariate analyses.14,15 A subsequent multivariate analysis on 77 patients with oropharyngeal cancer unveiled only the invasive pattern of the tumor to be an independent predictor for patients’ survival.16 Anneroth and colleagues17 proposed that grading should include six morphologic features: degree of keratinization, nuclear polymorphism, number of mitoses, pattern of invasion, stage of invasion, and lymphoplasmacytic infiltration. Bryne and colleagues18 revised this grading method, applying Anneroth’s criteria to the most anaplastic fields in the areas of highest infiltration. This invasive cell grading was demonstrated to be a highly significant predictor for oral SCC patients’ outcome and survival.18–20 Table 100-3 summarizes the invasive cell grading system (ICG) by Bryne and colleagues.
The present WHO grading system has been recommended for histopathologic typing of oropharyngeal cancers by the American Joint Committee on Cancer (AJCC).21 This grading system is based on Broders’ system and includes three grades, which are described in Table 100-4.
Table 100-4 World Health Organization Grading System for Oral Squamous Cell Carcinoma
| Grade | Description |
|---|---|
| 1 | Well differentiated. Histologic and cytologic features closely resemble those of the squamous epithelial lining of the oral mucosa. There are varying proportions of basal and squamous cells with intercellular bridges; keratinization is a prominent feature; few mitotic figures are seen and atypical mitoses or multinucleated epithelial cells are extremely rare; nuclear and cellular polymorphism is minimal. |
| 2 | Moderately differentiated. This is a neoplasm with features intermediate between well differentiated and poorly differentiated. Compared with well-differentiated squamous cell carcinoma, these tumors have less keratinization and more nuclear and cellular pleomorphism; there are mitotic figures and some are abnormal in form; intercellular bridges are less conspicuous. |
| 3 | Poorly differentiated. Histologically and cytologically there is only a slight resemblance to the normal stratified squamous epithelium of the oral mucosa. Keratinization is rarely present, and intercellular bridges are extremely scarce; mitotic activity is frequent and atypical mitoses can readily be found; cellular and nuclear pleomorphism are obvious and multinucleated cells may be frequent. |
From Pindborg JJ, Reichert PA, Smith CJ, et al, eds. Histological Typing of Cancer and Precancer of the Oral Mucosa. 2nd ed. Berlin: Springer-Verlag; 1997:11. By permission of the publisher.
Lymphoepithelial Carcinoma
The lymphoepithelial carcinoma is defined as a poorly differentiated SCC or undifferentiated carcinoma, accompanied by a prominent reactive lymphoplasmacytic infiltration. The lymphoepithelial carcinoma is rare and diagnosed in 0.8% to 2% of all oral and oropharyngeal malignancies.22 More than 90% of all lymphoepithelial carcinoma in the oral and oropharyngeal region is found in the tonsils and the base of tongue. Some are found in the buccal mucosa and the palate.
The histopathologic pattern is described as invasive, comprising syncytial sheets and clusters of carcinoma cells with prominent nucleoli, ill-defined cell borders, and often intact surface. The tumor sites are accompanied by rich lymphoplasmacytic infiltrate. These tumors are radiosensitive and local control can be achieved in a high percentage of cases.23
Salivary Gland Tumors
Among malignant salivary gland tumors, 9% to 23% are found in the oral cavity and oropharynx.24–26 Nearly half of the salivary gland tumors in the oral cavity and oropharynx are malignant.24 However, most neoplasms of minor salivary glands arise in the oral cavity. The most common oropharyngeal sites are the (soft) palate, the tonsillar fossa, and base of tongue. Oropharyngeal salivary gland tumors are rare with only 1.1% to 3.3% of all minor salivary gland tumors.24,25,27 All malignant salivary gland tumors that can possibly arise in the oropharynx are listed in Table 100-2. Among those, adenoid cystic carcinoma and the mucoepidermoid carcinoma are most frequent.
The adenoid cystic carcinoma is relatively common in minor salivary glands. A large series describes 42.5% of this lesion to be found in minor glands and 20.5% of minor salivary gland adenoid cystic carcinoma in the oral/oropharyngeal space.28 They usually present as slow-growing masses, sometimes with ulceration of the palate. Symptoms like pain, signaling nerve invasion, suggest progressive disease. The histopathologic pattern is cylindromatous or cribriform, although some can show tubular areas or appear solid.29 Cribriform lesions have a more favorable prognosis than solid tumors. The spread of adenoidcystic lesions is commonly hematogenous, typically in the lung and bones, and lymph node involvement is rather rare. Therefore neck dissection is generally reserved for patients with palpable disease.30 In case of radiation therapy for these rather moderately radiosensitive tumors, neutron beam radiation is recommended.31 Prognosis is negatively influenced by minor salivary gland as origin, tumor size greater than 4 cm, osseous invasion, advanced stage, and local recurrence.32,33
Mucoepidermoid carcinoma is the most common salivary gland malignancy and accounts for 9.5% to 23% of all minor salivary gland tumors.25,34,35 Half of the often asymptomatic cases arise in the palate, whereas the base of tongue and other oropharyngeal locations are rare.36 They are commonly well differentiated and appear as a bluish swelling. Some show granulation or a papillary surface. Oropharyngeal tumors can cause symptoms of dysphagia. Histopathologically, a mixture of epidermal epithelium and mucous membrane-producing cells is found. Treatment usually includes wide surgical excision, and neck dissection appears to be beneficial in patients with suspected or clinically obvious metastases. Olsen and colleagues reported on a series of oropharyngeal mucoepidermoid carcinoma at Mayo Clinic. Based on a follow-up of at least 10 years, recurrence rate was significantly reduced after wide local excision, including bone if necessary compared with simple excision with closer margins. Of the patients in the series, 24% presented with positive lymph nodes.37 Patients treated with primary radiation or postoperative radiation did not profit in terms of local control or survival in this evaluation; however, numbers might be too small to draw final conclusions and radiotherapy might be beneficial in case of adverse effects in those patients.
Soft Tissue Tumors
Kaposi’s sarcoma is a locally aggressive growing neoplasm, which might present as cutaneous but also as mucosal lesion, showing multiple patches and nodules (Fig. 100-9). It can also affect lymph nodes and visceral organs. It rarely metastasizes and is a member of intermediate-type vascular tumors. It is associated with human herpes virus 8 (HHV 8) infections. Kaposi’s sarcoma occurs as an indolent variant in elderly men in the Mediterranean and Eastern Europe, as endemic disease in equatorial Africa, and in immunosuppressed patients after organ transplantation or in case of human immunodeficiency virus (HIV) infection.38
Patients might be treated with surgery, radiation, and chemotherapy, depending on the epidemiology of the disease. The incidence and course of the disease has improved with antiretroviral therapy in HIV-infected patients.38
Hematolymphoid Tumors
On the basis of the complex and widespread lymphatic tissue in the oropharynx, lymphoid malignancies often occur in this area. Non-Hodgkin’s lymphomas (NHLs) are found in the palatine tonsils, palate, base of tongue, and other oropharyngeal sites. Etiologic factors are yet unknown in most patients, although some lymphoma patients might suffer from an immunodeficiency. Clinical symptoms can be fullness of the throat, dysphagia, snoring, and pain. Systemic symptoms are rare.39 The lesions present as exophytic mass, submucosal swelling, and sometimes ulcerations (Figs. 100-10 and 100-11).
Most NHLs in the oropharynx are B-cell lymphomas, with diffuse large B-cell lymphoma being the predominant type. Histopathologically the stroma is densely infiltrated with lymphoma cells, which vary depending on the histologic type. Treatment is based on radiotherapy and/or chemotherapy depending on the histologic type and stage. Studies have demonstrated a benefit for adjuvant chemotherapy along with radiation compared with radiation treatment only.40,41 The 5-year survival rate for localized disease has been reported from 50% to more than 80%.41–45
Mucosal Malignant Melanoma
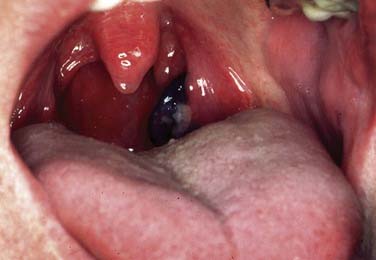
Although malignant melanoma (MM) most commonly manifests as skin lesions, it may also arise from melanocytes in the mucosa, but only 1.3% of all melanomas are mucosal melanomas (Figs. 100-12 and 100-13). Of those, 55.4% are mucosal MMs of the head and neck.46 They are characterized as malignant transformed melanocytes at the epithelial–connective tissue interface with migration into the epithelium and the connective tissue, respectively. Oropharyngeal mucosal melanoma is exceedingly rare, with more mucosal MM of the head and neck occurring in the oral cavity (50%).46,47
No known etiologic factors exist for mucosal MM of the head and neck. Clinically they can present as black, gray, or reddish, and they are rarely amelanotic. They typically consist of widespread, multiple, pigmented lesions with or without nodules. Ulcerations and bone infiltration are common and oral bleeding, dysphagia, and sensations of pain can be symptoms before diagnosis. Mucosal MM is usually diagnosed in an advanced stage with most tumors showing invasive character. However, 20% are in situ lesions.47 Aggressive surgical resection is still the primary treatment of choice, although many adjuvant modalities including different chemotherapy regimens and radiotherapy have been applied. Despite surgically achieved possible local control, 5-year survival of pharyngeal melanoma is only 13%.46,48,49 Developments for improved treatment options are limited because mucosal MM is a rare disease lacking clinical treatment trials. By early detection, melanoma patients have the best chance for cure and physician’s threshold for biopsy of a suspected lesion should be low.
Squamous Cell Carcinoma
Etiology
Predominant and synergistic acting risk factors for SCC of the oropharynx are tobacco and alcohol consumption.50 These factors account for about 75% of oral and oropharyngeal carcinoma in Western countries.50,51 For the highest consumption levels, relative risks from 70 to 100 have been reported compared with lowest consumption levels. A super-multiplicative effect in case of contact with tobacco and alcohol substances has been shown for oral and oropharyngeal cancers in case-control studies.52
Tobacco chewing is a common habit, particularly in India, parts of Southeast Asia, the Middle East, China, and Taiwan. Tobacco is often consumed in betel quids containing areca nut, which has been classified as human carcinogen by the IARC in 2003. Smokeless tobacco is a major risk factor for oropharyngeal cancer in these parts of the world.53 In India about 50% of oropharyngeal cancers in male and 90% in female are caused by chewing.54
Human papillomavirus (HPV) infection is another well-established cancer risk factor. Genotypes HPV 16 and 18, known causes for uterine cervix and skin cancer, are found in 50% of tonsillar and oropharyngeal SCC, and studies suggest that up to 40% of oropharyngeal cancer cases may be due to HPV infection.55 The incidence of HPV-associated oropharyngeal primaries is rising according to recent reports—up to 70% of all presenting cases in some series. The cohort of patients harboring this form of the disease is younger by approximately 10 years and has a distinctly favorable prognosis. Epidemiologic studies propose that having multiple sexual partners is significantly associated with HPV-related tumors.1
In addition to exogenic risk factors, multiple endogenic risks have been identified and associated with head and neck cancer.56 DNA repair, differences in mutagen sensitivity, and alteration of genes such as epidermal growth factor receptor (EGFR) have shown their impact on head and neck cancer and influenced modern therapy and prevention strategies.
Clinical Presentation, Patterns of Spread
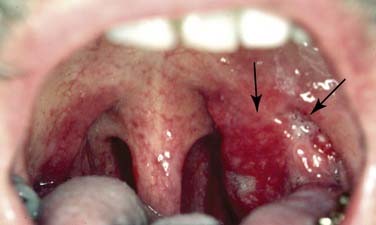
The most frequent location for oropharyngeal tumors is the tonsillar fossa, with the palatine tonsil and the anterior tonsillar pillar. They commonly present as foreign body, dysphagia, otalgia, or impeded jaw mobility caused by infiltration of the periosteum or bone of the mandible or the pterygoid muscles in extended cases. Physical examination can show exophytic or ulcerated lesions, dysplasia, and/or inflammation reaction. Extension into the base of tongue inferiorly and the soft palate superiorly is common, with 55% and 60% in a study by Perez and colleagues, respectively.57 Lymphatic drainage is directed primarily to level II nodes but can involve level III parapharyngeal and retropharyngeal nodes level I, IV, and V, depending on stage of presentation.58–60 Lindberg described nodal metastasis for cancer of the tonsil in 76% of cases compared with 45% in patients with cancer of the tonsillar pillar, with most common node involvement in level II for both locations. Contralateral nodal disease was found in 11% of patients with cancer of the tonsil and 5% with primary malignancy of the pillar.61 However, in these studies no mention is given of proximity to the midline, which is the most frequent reason a lateralized tumor epicenter in the oropharynx would present with “contralateral” neck metastasis. Differences might be based on higher-staged groups with tonsillar lesions, and likelihood of contralateral lymphatic spread is less than 5% for T1 and T2 cancers.
Cancer of the base of tongue is particularly difficult to detect and often becomes clinically evident in an advanced stage. This is due to relatively late clinical symptoms because the base of tongue is nearly without pain fibers. Moreover, the assessment of the base of tongue can be more difficult during physical examination due to prominent lingual tonsils or submucosal location, and deep basal areas might not completely present even using operative endoscopy. As in the palatine tonsil, occult carcinoma with clinically prominent metastatic disease, typically in neck level II, is often found in the base of tongue and is often the presenting feature of this disease. As with neck metastases primarily clerived from tonsils, some of these neck masses can be misdiagnosed as branchial cleft cysts or, on biopsy, as branchiogenic carcinomas.62 To rule out such findings, meticulous microendoscopic examination (see Fig. 100-12) and biopsy of suspected lesions are mandatory. Base of tongue cancer has a higher frequency of bilateral metastases (up to 20%), which is mainly due to proximity to the midline but enhanced by a rich lymphatic drainage of the base of tongue and tendency to late clinical presentation with upstaging. Ipsilateral node involvement can be detected in more than 70% of cases.63
Diagnostic Evaluation
Anamnesis
A thorough anamnesis is part of any comprehensive evaluation of head and neck cancer patients. Symptoms at first presentation of oropharyngeal cancer patients are commonly dysphagia and/or odynophagia, oral bleeding, otalgia, or changes in speech. In addition to the history of risk factors such as alcohol and tobacco consumption, xenobiotic exposure at working places and in the environment should be evaluated. Dietary and social habits are also worthwhile to evaluate. Knowing about possible risk factors for head and neck cancer in individual patients enables for future elimination and enlarges the chances for prevention of recurrent disease. Also, better results and improved treatment tolerance are obtained for patients who quit smoking.64 Other factors such as social aspects, family support, and general health status have great impacts on treatment and outcome and should therefore be consequently evaluated.