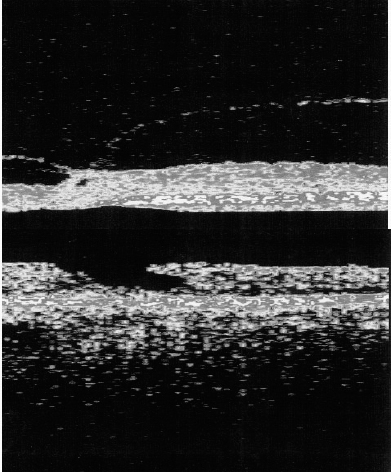
14 A macular hole is a central retinal hole that may be the end stage of an evolving full-thickness macular dehiscence, a contracting premacular membrane, macular trauma, or macular cyst formation either with or without associated macular edema. Idiopathic macular holes are more common than macular holes associated with trauma, inflammation, edema, or myopia. As a result, attention is focused here on idiopathic macular hole development and management. Because treatments for macular holes may be quite involved, they must be differentiated from macular-hole–like lesions, such as a macular cyst or macular pseudohole. Epiretinal membranes (ERMs) can mimic macular holes and may be idiopathic or the result of surgical intervention or inflammatory insult. Both ERMs and macular holes share some common pathogenic mechanisms, but the resulting structural changes and treatments are distinct and therefore are described separately. Patients note a unilateral decrease in central vision with or without metamorphopsia (Fig. 14–1). Some patients report an increase in floaters and a central scotoma. Often symptoms are noticed when a patient covers the fellow eye. A fully developed macular hole typically causes vision loss to the level of 20/60 or worse. Macular holes are full-thickness holes in the retina generally involving the central foveal tissue. They are usually easily diagnosed with a contact lens or with indirect slit-lamp biomicroscopy (typically using a 78- or 90-diopter lens). A cuff of retinal detachment surrounds a dark, round defect in the fovea. Macular holes must be distinguished from the masquerading condition of the pseudomacular hole, which is a hole in a premacular membrane. Both may benefit from surgery if there is associated vision loss. Features associated with idiopathic macular holes include a yellow central macular spot or ring in the case of a premacular hole. Once a full-thickness defect has developed, a pseudooperculum may be seen. This is not a true operculum but rather a cellular condensation of the posterior vitreous that has detached from the retinal surface. With time, intraretinal cysts may form at the edge of the hole, and fine striae or associated ERMs of variable size may be seen in association with the edge of the macular hole. In the end stage of a mature full-thickness macular hole, fine yellow deposits are seen on the retinal pigment epithelium surface (RPE) (Table 14–1). There are three basic concepts of macular hole formation: trauma, cyst development, and vitreous or retinal surface traction. In the traumatic theory of macular hole development, it is postulated that forces are transferred to the macula via the vitreous body along various attachment points, one of which is at the foveola. Abrupt rotation of the globe can affect tangential traction at the vitreomacular interface. The vitreous is most strongly attached to the retina at the vitreous base, along the major arcades, the optic nerve, and at 1500-μ diameter rim surrounding the fovea and the 500-μ diameter foveola. In addition, stretching of the globe during impact causes stretching of the retina. The foveal retina may be more susceptible to damage because it is relatively thinner. If tractional forces become greater than the cohesive forces, macular holes can result.1
Macular Membranes and Holes
Macular Holes
What Is a Macular Hole?
What Are the Symptoms of a Macular Hole?
What Are the Signs of a Macular Hole?
What Are the Historical Concepts of Pathogenesis of Macular Holes?
| ERM | VMT | |
|---|---|---|
| Age (yr) | 60–70 | 60–70 |
| Surgical anatomy | Complete PVD (80%) | Incomplete PVD |
| Pathology | RPE cells, glial cells, new collagen | Glial cells, new collagen |
| Traction vector | Tangential | Anteroposterior, tangential |
PVD, posterior vitreous detachment; RPE, retinal pigment epithelial.
The cystoid theory holds that aging causes cystoid changes within the fovea. The coalescence of these cystoid spaces is implicated in the formation of macular holes.2 The relatively weak associations between most macular holes and cystoid macular edema (CME) or epiretinal membranes argue in favor of newer theories involving the vitreous.
What Are the Current Ideas Regarding the Pathogenesis of Macular Holes?
Gass suggested that tangential tractional forces along the retinal surface from a shrinking cortical vitreous are a possible cause of macular holes.3 This theory is incorporated into his widely accepted staging of macular holes. Both the “tangential traction theory” and the staging of macular holes are discussed in a separate section in this chapter. Recent work with optical coherence tomography (OCT) has shed new light on the initial stages of macular hole formation. It appears that, in many cases, the inciting event is an incomplete posterior vitreous detachment (PVD) with residual vitreous attachment at the fovea. During this event, radial retinal striae and cystoid formations at the foveola are seen. Complete PVD results in one of the following events: (1) the cystoid spaces resolve; (2) unroofing of a cystoid space occurs, and a lamellar macular hole forms; or (3), if there has been a dehiscence of the floor of the cystoid space, a full-thickness macular hole develops (Fig. 14–2).4,5
Because of variations in local foveal anatomy, most eyes do not develop full-thickness macular holes or early premacular hole findings. Some associated factors in the formation of macular holes include the relative tensile strength and health of the native fovea (foveal susceptibility), the strength of the vitreofoveal attachments, and the nature of the PVD.5
What Is the Histology of Macular Holes?
Microscopically, macular holes are seen as round or oval retinal defects surrounded by rounded retinal edges and a cuff of detached neurosensory retina with subretinal fluid present. In one report, intraretinal cystoid edema and variable amounts of photoreceptor atrophy were present in most eyes (78%), especially near the edge of the hole.6 An ERM was present in 68% of these eyes.
The role of ERMs in the development of macular holes is not clear. Although ERMs may be associated with CME, most eyes with ERMs do not develop macular holes. The presence of ERMs on histologic specimens may represent a local inflammatory response to macular hole formation.6
Studies of removed pseudoopercula show that they are composed of contractile glial elements and RPE cells and do not contain displaced neurosensory photoreceptor cells. Fibrous astrocytes and Muller cells are the predominant components.7
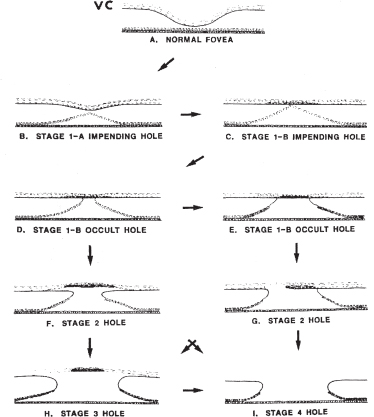
FIGURE 14–2. The Gass classification for macular hole staging.
What Is the Gass Classification for Macular Holes?
Gass and Joondeph are credited with recognizing the yellow spot and ring as the initial presentation of macular holes.3,8 They described a classification scheme that categorizes the development of a macular hole into several stages. This system of macular hole classification is widely used and correlates well with clinical observations (Fig. 14–2).
Stage 1 macular hole (foveolar detachment) is characterized by a central yellow spot (stage 1A, impending hole) or a yellow ring (stage 1B, occult hole) and loss of the foveal depression. These are not true holes because there is no full-thickness dehiscence of the retinal tissue. The cortical vitreous is attached to the fovea, and the yellow spot that develops is thought to be the result of xanthophyll pigment that has become more visible because of retinal detachment from the RPE. In a stage 1B occult hole (yellow ring), an occult dehiscence may occur under the contracted prefoveolar vitreous cortex and may be kept closed by the overlying vitreous. Visual acuities are in the range of 20/25 to 20/40. Most stage 1 holes do progress, although some show resolution with completion of the PVD.9
Stage 2 macular holes (early full-thickness macular hole) result from continued vitreous traction and enlargement of the yellow ring with the formation of a foveal hole or dehiscence associated with a cuff of subretinal fluid (Fig. 14–3). The posterior vitreous cortex is still attached to the retina, and radial striae may be seen around the edges of the hole at the level of the inner retina and the internal limiting membrane. The retinal defect inside the yellow ring may be oval, crescent, or horseshoe shaped. The full-thickness tear may begin at the center of the fovea and expand in a symmetric manner. Alternatively, a full-thickness tear may begin in an eccentric location in the fovea and extend in a can-opener manner to form a crescent-shaped hole. Visual acuity is in the range of 20/40 to 20/100. Most stage 2 holes gradually enlarge and progress to stage 3 holes within several months.
FIGURE 14–3. Early full-thickness macular hole (stage 2 by Gass classification).
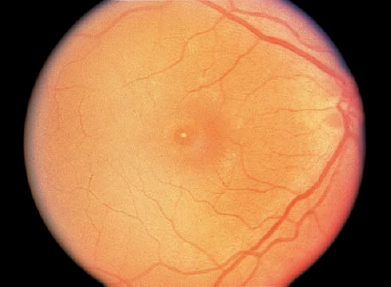
FIGURE 14–4. A stage 3 macular hole is shown with a white nodular deposit at the base of the hole.
Stage 3 macular holes (fully developed macular holes without complete PVD) are characterized by further enlargement of the retinal defect to 400 μ or more in diameter. A localized PVD overlying the hole may be detected if there is a pseudooperculum. As the hole enlarges, discrete white nodular deposits are seen at the base of the hole, representing RPE reaction (Fig. 14–4). Vision is in the range of 20/60 to 20/400.
Stage 4 macular holes (full-thickness macular hole with complete PVD) are round retinal defects with a rim of elevated retina. The presence of a Weiss ring confirms the completed PVD. The size of the hole and the level of vision loss will stabilize at this stage for most patients. If progressive, visual loss occurs, it is likely to be due to photoreceptor atrophy, cystoid changes at the border, or accumulation of subretinal fluid under the hole. A prefoveolar opacity may or may not be seen in the case of stage 3 or 4 macular holes. The presence of the pseudooperculum indicates a good prognosis because it indicates separation of the cortical vitreous from the fovea.
What Is a Lamellar Macular Hole?
A lamellar macular hole is a partial-thickness loss of the foveal retina. It resembles a round or petal-shaped red depression in the inner retinal surface. Lamellar macular holes may be due to the unroofing of a foveal cyst secondary to vitreous traction on the fovea. The floor of the cyst remains attached to the RPE; hence, it is a partial-thickness defect in the neural retina.4
What Are the Risk Factors for Developing Macular Holes?
The Eye Disease Case Control Study Group studied the risk factors for development of full-thickness macular holes and compared 198 subjects with macular holes with 1023 normal controls. Although the study group did not look at vitreomacular relationships or involutional macular thinning as factors, they found that older age and female sex were risk factors.10 Seventy-two percent of patients with macular holes were female, whereas fewer than 3% of macular holes were found in patients less than 55 years old. Data from recent studies suggest that an abnormally strong vitreomacular attachment combined with impending PVD substantially increases the risks for developing macular holes.5
WHAT IS THE RISK IN THE FELLOW EYE?
Risk of development of a macular hole in the fellow eye ranges from no risk to 28%, depending on the study. The normal fellow eye without a PVD in patients with a unilateral full-thickness macular hole has a 10 to 20% risk of developing a macular hole within 5 years. The presence of a complete PVD seems to confer protection and lowers the risk to less than 1%. In fellow eyes with an impending macular hole, the risk is much higher, in the range of 40 to 60%.9,11,12
What Should Be in the Differential Diagnosis for Impending Macular Holes?
The differential diagnosis should include lamellar holes and pseudomacular holes. In these cases, a raised retinal edge will not be seen. Alternate diagnoses that must be ruled out include central serous retinopathy, solar and inflammatory maculopathy, pigment epithelial detachment, a centrally located druse, CME with central macrocyst, subfoveal choroidal neovascularization, and vitreomacualar traction (VMT) syndrome.8,13 In VMT syndrome, the entire foveola may be elevated above the plane of the retina, visible on slit-lamp biomicroscopy or with the aid of OCT. In solar retinopathy and a centrally located druse, there will be preservation of the foveal depression. Although a yellow spot could be seen from other disease processes, the change from a yellow spot to a yellow ring is quite specific to patients developing a macular hole.
Some entities that mimic macular holes can be distinguished by fluorescein angiography (FA), including choroidal neovascularization, CME, and central serous retinopathy. In early central areolar choroidal dystrophy, mild atrophy of choriocapillaris under the fovea can give the appearance of a full-thickness macular hole, a round red lesion. Careful examination will reveal an intact neurosensory retina without the typical rim of subretinal fluid surrounding the red circular lesion.
How Can Epiretinal Membranes Mimic Macular Holes?
Epiretinal membrane contraction can cause foveal detachment, mimicking an impending macular hole. ERMs with a central round opening and pseudoholes have been mistaken for macular holes.8,13 The presence of a crinkled reflex from the inner retinal surface surrounding the hole and the absence of a perifoveal ring of retinal detachment, operculum, or yellow deposits at the level of the pigment epithelium centrally are the features of a hole in an ERM. If the hole is in a membrane rather than in the retina, visual acuity is often better than 20/40.
HOW DO YOU DIFFERENTIATE A LAMELLAR MACULAR HOLE FROM A FULL-THICKNESS HOLE?
Inner retinal lamellar (partial-thickness) holes typically have vitreofoveal separation and often are accompanied by an operculum, the presence of a foveal depression, and one sharply demarcated round, oval, or petal shaped depression in the inner retinal surface. In contrast, stage 1 lesions will have loss of foveal depression. Angiographically, lamellar holes are differentiated from stage 2 or 3 holes by minimal or a complete lack of hyperfluorescence, whereas fullthickness holes typically show moderately intense early hyperfluorescence in the area of the hole. Lamellar holes do not have a rim of detached retina, unlike full-thickness macular holes. The key to proper diagnosis and staging of macular holes is contact fundus biomicroscopy through an adequately dilated pupil. This will allow for better delineation of flat versus elevated structures and preretinal membranes as well as vitreous condensation. OCT may be a helpful test to correlate the physical findings.
What Are Some Conditions That Have Been Associated with Macular Holes?
Macular holes have been reported in association with other conditions. Some of these include proliferative diabetic retinopathy, pan-retinal photocoagulation for diabetic retinopathy, high myopia, choroidal neovascularization, Best disease, adult vitelliform macular degeneration, trauma, topical pilocarpine, scleral buckle operations, and pneumatic retinopexy for retinal reattachment.9,11
What Diagnostic Tests Would Be Appropriate?
Fluorescein angiography is nonspecific for stage 1 macular holes. FA may or may not demonstrate hyperfluorescence. When there is a defect in the neuroretina, as in stage 2 and beyond, early transmitted fluorescence may be seen in the area of the hole. Sometimes the early transmitted fluorescence may be blocked by the pseudooperculum, showing hypofluorescence. Although it is a nonspecific test for diagnosing macular holes, FA is useful to rule out other macular entities that have classic findings such as CME, choroidal neovascularization, and central serous chorioretinopathy. Other modalities, such as OCT, are more sensitive in defining the anatomic relationships between the cortical vitreous and retina and in showing intraretinal detail; however, OCT is not always available.
Perhaps the simplest test is the Watzke-Allen test.14 This is especially helpful in differentiating between impending or full-thickness macular holes and masquerade syndromes. A narrow beam of light from a slit lamp is shone passing through the foveola, and the patient is asked to describe what he or she sees. If the patient reports that he sees a break in the white line (positive Watzke sign), it can be assumed that a defect in the retina exists. Patients with impending macular holes and their mimickers will either report a straight line or a line with thinning in the middle.
Alternatively, a 50-μ aiming beam can be aimed at the lesion. Patients with macular holes will not see the aiming beam as it passes over the macular hole. The beam will seem to disappear once it passes over the lesion.
What Is the Management of Macular Holes?
Standard treatment of macular holes is pars plana vitrectomy with complete cortical vitreous detachment and injection of a nonexpanding mixture of long-acting intraocular gas with prone positioning for at least 1 week.15 Silicone oil can be used as the tamponade agent if the patient is unable to tolerate face-down positioning.16 Because about half of stage 1 lesions resolve with relatively good vision, most clinicians wait until the lesion has progressed to stage 2 before considering repair.17
The 1990s saw the development of adjuvant use in the treatment of macular holes. Usually, the adjuvant is added in the area of the macular hole at the end of vitrectomy in hope of either providing tamponade or acting as tissue adhesive by promoting wound healing. Anatomic closure rates range from 64 to 100%, with functional success from 33 to 100%. A recent meta-analysis showed a greater than 80% chance of success for macular hole surgery in general. Postoperative vision of greater than 20/40 has been reported in up to 49% of patients.17
Recently, internal limiting membrane removal in addition to the standard treatment has been shown to have slightly improved anatomic success.26,27 Good outcomes were obtained for repair of traumatic macular holes without use of adjuvants.18 It is interesting to note that patients’ vision can continue to improve beyond one year after successful anatomic repair.19
WHAT IS THE STABILITY OF THE REPAIR?
In a series of 390 operated eyes, there was a 4.8% rate of reopening of macular holes. In this series, the reopening occurred spontaneously. Ten of the 17 eyes had a history of cataract surgery, but only one cataract surgery case was thought to be directly responsible for macular hole reopening.20 Another study reported a reopening rate of 9.5% in a series of 116 patients.25 Reoperation resulted in good results for most patients.
WHAT CAN COMPLICATE MACULAR HOLE REPAIR?
The rates of anatomic and functional success after macular hole repair are very promising; however, posterior segment complications, such as endophthalmitis, rhegmatogenous retinal detachment, phototoxicity, enlargement of the hole, and RPE derangements have been reported. In addition, development of cataract or worsening of preexisting lens opacities is common. Secondary glaucoma, CME, visual field defects, and choroidal neovascular membranes also can complicate otherwise successful surgery.21
What Is the Natural History of Macular Holes?
Unoperated macular holes typically slowly enlarge, and the adjacent RPE atrophies. The vision stabilizes at a level of 20/200 to 20/400. The presence of an operculum, PVD, and ERM do not appear to be correlated with the visual morbidity.22
What Is the Prognosis for a Failed Macular Hole Repair?
For a macular hole that has reopened, the rates of success of a second operation are similar to that of the first operation. If the hole detaches a second time, 75% of patients can be expected to obtain anatomic improvement and 56% can be expected to attain both anatomic and functional improvement.23 If the initial two surgeries for repair have been unsuccessful, a third attempt at repair showed no benefit. For longstanding macular holes (longer than 6 months), anatomic closure was still effective in improving vision. Rates of anatomic closure are slightly less than repairs made for holes of less than 6 months’ duration. Alternatively, up to 98% achieve anatomic closure if repaired before 6 months. Long-standing cases are limited to about 70% closure rates by standard techniques.24
Epiretinal Membranes
What Are Epiretinal Membranes?
Different cells types can gain access to the inner surface of the retina and proliferate to form fibrocellular tissue, which forms thin membrane sheets that cover the retinal surface. Numerous and overlapping terms have been used to describe such proliferative membranes: ERM, premacular membrane, preretinal macular fibrosis, surface-wrinkling retinopathy, cellophane maculopathy, and macular pucker.28,29 ERM can be secondary to many ocular conditions, such as ocular inflammatory disease, retinal vascular disease, rhegmatogenous retinal detachment, and accidental or surgical trauma, particularly after retinal cryotherapy or photocoagulation. RPE cells are observed to be the primary cell type in this condition.30,31 ERM may also be idiopathic, unassociated with any ocular disorder. The primary cell type recognized in idiopathic ERM is the fibrous astrocyte.32
What Are the Symptoms of an Epiretinal Membrane?
Patients affected with ERM may be asymptomatic in most cases. Membranes that are asymptomatic do not cause distortion of underlying foveal photoreceptors or are eccentric to the fovea. If retinal distortion occurs, patients may recognize variable degrees of vision loss, metamorphopsia, and micropsia (Fig. 14–1). Some may complain of an acute loss of vision with accompanying distortion over a period of a several weeks, although the actual onset and duration of symptoms are difficult to ascertain.28,33
What Are the Signs of an Epiretinal Membrane?
The clinical features of an ERM vary, depending on the severity of the membrane, membrane thickness, membrane location, and degree of contraction. An irregular or glistening light reflex from the retinal surface may be the only sign in asymptomatic patients. This may be seen only with careful slit-lamp biomicroscopy. In some cases, the membrane may appear to cover the entire macula. In more advanced cases, in symptomatic patients with actual contraction or shrinkage of the membrane, distinct retinal findings can be appreciated, such as retinal striae radiating from the center of the ERM, retinal vessels straightened toward the membrane center, or tortuosity of retinal vessels and dilated retinal veins.28,34 Macular dragging and foveal ectopia can occur if the membrane is centered distal to the center of the fovea. Inner retinal punctate hemorrhages also can be seen and may be associated with retinal thickening resulting from retinal vessel leakage and retinal edema secondary to the tractional effects of the ERM. Such traction also may produce stasis on axoplasmic flow resulting in cotton-wool spots. Pseudoholes or lamellar holes also may be associated with ERMs and may simulate full-thickness macular holes.35 A PVD is present in the most eyes (75 to 90%) with idiopathic ERMs, although it is not a necessary prerequisite.34,36,37
What Techniques and Diagnostic Tests Are Helpful in the Diagnosis?
Traditionally, slit-lamp biomicroscopy with a contact lens evaluation has been considered the best method for evaluation of an ERM. It remains the most important diagnostic tool in such cases.
Fluorescein angiography is also a useful tool in the diagnosis of ERM. Anatomic features of retinal distortion may be better seen on FA, such as straightened retinal vessels, retinal vascular tortuosity, foveal ectopia, and macular dragging. Fluorescein leakage and macular edema secondary to ERM traction-induced vascular leakage also may be assessed. FA also may help to distinguish between a full-thickness hole and a pseudohole caused by an ERM or because transmitted fluorescence is not usually present in a pseudohole. Another important role of FA would be to detect the presence of other retinal pathology, such as a choroidal neovascular membrane.
Optical coherence tomography may be helpful in evaluating patients with ERM. OCT provides good visualization of the vitreoretinal juncture and actually may show the membrane on the retinal surface. It also may be helpful to distinguish a pseudohole from a full-thickness hole. OCT also may play a role in preoperative and postoperative evaluation of surgery for ERMs.38
How Are Epiretinal Membranes Classified?
There are many classification schemes for ERM. A commonly used method of classification is by severity of retinal distortion proposed by Gass.39 Another method of classifying ERM is according to associated biomicroscopic findings. Secondary ERM may be classified by their associated disorders.
What Is the Gass Classification?
GRADE 0: CELLOPHANE MACULOPATHY
This stage is characterized by a cellophane-like light reflex from the inner retinal surface when viewed ophthalmoscopically because the ERM may be transparent with no associated retinal distortion. Visual acuity is generally normal with minimal to no symptoms.
GRADE 1: CRINKLED CELLOPHANE MACULOPATHY
In this stage, the inner retinal surface may be contracted into small irregular folds resulting from shrinkage of the ERM. Biomicroscopically, an important sign is the presence of fine superficial radiating retinal folds extending outward from the edge of the ERM. Details of underlying retinal vessels may be obscured; the retinal vessels may show tortuosity. The visual acuity is usually better than 20/40 primarily because of distortion in the outer retinal layers with symptoms of metamorphopsia.
GRADE 2: MACULAR PUCKER
The degree of retinal distortion and wrinkling of the retinal surface is marked in this stage. Gross puckering of the macula may be present with a distinct gray membrane on the inner retinal surface. Other associated findings in this stage are intraretinal hemorrhages, retinal edema, cotton-wool spots, and localized serous retinal detachment with a PVD found in more than 90% of such cases. Visual acuity may be reduced to less than 20/200 in severe cases with prominent metamorphopsia.
What Is the Foos Classification of Epiretinal Membranes?
Simple ERM are incidental without contraction features or associated ocular disease.40 Intermediate ERM are thicker than simple ERMs and contain contraction features and pigment.41 Complex ERM are present after retinal detachment surgery or after trauma and may be secondary to other ocular conditions. Traction retinal detachments may develop as a result of contraction of such membranes.41
What Are the Risk Factors for Developing an Epiretinal Membrane?
Age is a risk factor for the development of ERMs.42,43 Idiopathic ERMs occur in eyes of patients older than 50 years of age.44 There is no sex predilection, with males and females affected equally. A PVD is present in 90% of patients.37 Idiopathic membranes have been reported in children and adolescents but are generally nonprogressive.45 Diabetes has a significant association with idiopathic ERM with or without the presence of diabetic retinopathy but with minor effects on vision.43
What Is the Risk to the Fellow Eye?
In large population-based studies such as the Beaver Dam Eye study, bilateral involvement was reported to occur in 20% of patients.42 The Blue Mountains Eye Study from Australia reported a 31% incidence of bilateral ERMs.43
What Is the Pathophysiology of Epiretinal Membranes?
The forces generated by the movement of the vitreous gel, particularly the premacular bursa, have been implicated in the formation of ERM and macular holes.46 Although stimuli for the formation of ERM are not fully understood, Gass proposed two mechanisms for the development of ERM. The first mechanism involves dehiscences in the inner limiting membrane (ILM) caused by vitreous separation from the macular surface, which allows retinal glial cells entry onto the retinal surface that then proliferate and contract. The optic-nerve head and sites along the major vessels are the most common sites of dehiscences because the ILM is noncontiguous or attenuated at these sites. A second proposed mechanism of ERM formation involves contraction of vitreous cortical remnants and proliferation of hyalocytes on the inner retinal surface after a PVD.47,48 The cortical remnants may act as a scaffold for cellular elements composing the membrane. This hypothesis may be more applicable to membranes confined to the central macular area.32,48
What Is the Histology of an Epiretinal Membrane?
In histopathologic studies, ERMs consist of a layer or multiple layers of abnormal cells with variable thickness on the retinal surface.49 These cell types have been characterized by ultrastructural studies as belonging to one of the following four morphologically distinct cell types: fibrous astrocytes (glial cells), RPE cells, fibrocytes, and macrophages.50 The collagenous component of the membranes can be derived from any of the cellular groups. Categorization of cell types is difficult because of their ability to transform into cells with similar appearance and function. The ability of each cell type to develop myofibroblast-like properties gives rise to a fifth group of cells, the myofibroblast-like cells.50 The myofibroblastic property of all the cell types mentioned is important in that it may account for the contractile ability of ERMs and allow the membranes to exert tractional forces on the retina.30,50,51
What Is the Management of an Epiretinal Membrane?
Most idiopathic ERMs remain stable both anatomically and visually over time.37,52 Surgical intervention with pars plana vitrectomy (PPV) and epiretinal membrane peeling is indicated only if vision is markedly reduced and distortion is symptomatic. Although infrequent, spontaneous resolution can occur. PPV with membrane peel has been shown to remove ERM successfully; more than 80% of eyes have visual improvement.53,54 Favorable prognostic factors for visual improvement are good preoperative vision and short duration of symptoms preoperatively.54,55 Although eyes with preoperative visions of less than 20/200 or long duration of visual symptoms have been associated with worse overall visual improvement, these eyes often improved the greatest number of lines after surgery.55
What Complications Are Associated with Surgical Management of Epiretinal Membranes?
A common complication of PPV for ERM is the development or progression of nuclear sclerotic cataract up to 31%.53,56 Although much less common, other complications are associated with surgery for ERM, including posterior and peripheral retinal breaks, retinal detachment, and phototoxic retinal burns from endoillumination.53
What Is the Chance of Recurrence after Surgery?
Recurrence of the membrane after PPV is uncommon. Reported recurrence rates are near 5%.57 Repeat vitrectomy and membrane peel can offer visual improvement in these cases.
Vitreomacular Traction Syndrome
The firmest attachment of the vitreous to the retina is at sites where the ILM is thinnest: the vitreous base, along retinal vessels, the optic-nerve head, a 1500-μ rim surrounding the fovea, and the 500-μ-diameter foveola.58 Normal aging changes in the vitreous gel lead to separation of the posterior vitreous cortex from the retinal ILM, termed a PVD.59 If this separation is incomplete with persistent vitreous attachment on the posterior retina, traction on the macula may occur resulting in the syndrome of VMT.60,61
What Are the Symptoms of Vitreomacular Traction Syndrome?
Traction on the macular region can result in decreased vision, metamorphopsia, photopsia, and micropsia.60,63,74 Symptoms in VMT syndrome may be a result of cystoid changes in the macular region.
What Are the Signs of Vitreomacular Traction Syndrome?
The clinical findings in VMT syndrome may be seen with careful slit-lamp biomicroscopy using a contact lens. The most common morphologic configuration involves a partial PVD with adherence of the posterior vitreous cortex to the macula and optic-nerve head.60,64 The site of vitreomacular adherence may have a distinct curvilinear or horizontal border in the macula, or it may be less sharply demarcated with a dense irregular pattern. In studies that evaluated intraoperative vitreoretinal relationships in VMT syndrome, two distinct patterns were found, one in which the cortical vitreous was detached in all four midperipheral quadrants with only macular adherence remaining, and the second in which one or more quadrants of vitreous remained attached in the periphery with residual attachment to the macula.60,61 Other possible findings in VMT include a thickened posterior hyaloid, radiating retinal striae, retinal distortion, and a focal traction retinal detachment. In addition, an ERM may also be present. Peripapillary vitreous traction may occur, resulting in the appearance of a ring of fibrous tissue atop the optic-nerve head, referred to as a “fleshy doughnut” sign by McDonald and associates.61
What Techniques and Diagnostic Tests Are Helpful in the Diagnosis of Vitreomacular Traction Syndrome?
The traditional method for identifying VMT syndrome remains careful slit-lamp biomicroscopy with contact lens examination. Nevertheless, even with careful biomicroscopic examination, a diagnosis of VMT syndrome may be missed. Other techniques such as FA, ultrasonography, and OCT can help to elucidate this diagnosis. FA can help to demonstrate cystoid macular leakage, retinal vascular leakage, and optic-nerve head edema. Ultrasonography is a noninterventional method for viewing vitreomacular relationships and can aid in the diagnosis of VMT syndrome. An alternate method for viewing the vitreoretinal interface is OCT, which can acquire high-resolution, cross-sectional images of the retina.65 OCT can image the partially detached posterior hyaloid exerting traction on the macula and also may show secondary cystic spaces and intraretinal thickening66,67 (Fig. 14–5).
What Is the Pathogenesis of Vitreomacular Traction Syndrome?
Direct anteroposterior traction on the retinal surface secondary to changes in the vitreous gel can lead to macular distortion. The etiology of this traction is an incomplete vitreous separation in which the vitreous maintains a focal attachment to the retinal surface. This can occur as a part of normal aging or secondary to ocular inflammation, vascular disorders, or trauma causing condensation and shrinkage of the vitreous gel.68