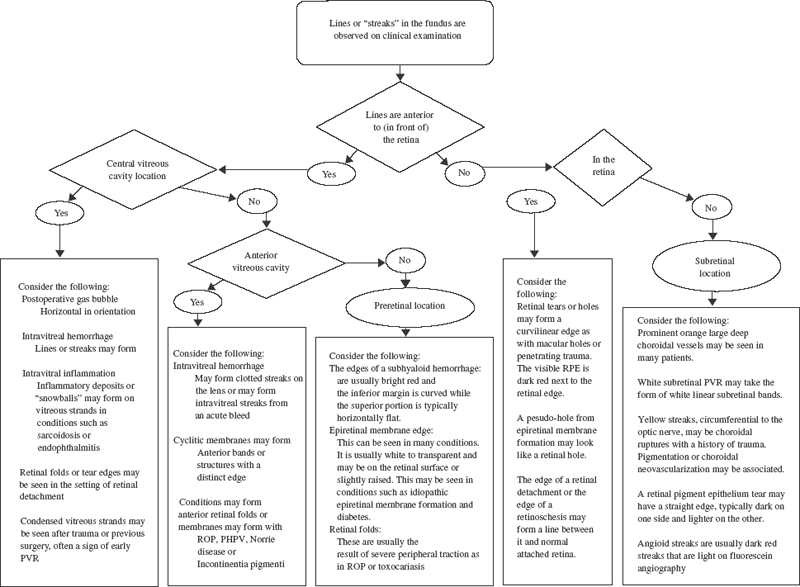
11 Hemorrhage originating in the vitreous cavity may leave blood on the posterior lens surface, either clotted with a distinct edge or in streaks. This may occur in any situation where hemorrhage is retained in the vitreous cavity. Conditions predisposing to hemorrhage include vitreous hemorrhages, which are the result of systemic disease, such as diabetes,1 retinopathy of prematurity, or sickle cell retinopathy,2 or an old traumatic injury, such as from an air bag,3 paint ball,4 or other blunt or penetrating trauma injuries.5–10 Vascular disease, such as central retinal vein occlusion11 or macroaneurysms,12 and age-related changes, like posterior vitreous detachment with retinal tears, also can lead to vitreous hemorrhage.13 Figure 11–1 shows a clinical pathway that includes a method of approaching the diagnosis of lines or streaks seen on fundus examination. Here we consider three conditions that can lead to anterior vitreous pathology in their advanced stages, manifesting as membrane formation, hemorrhage, or both. These are retinopathy of prematurity (ROP); persistent fetal vasculature (PFV), also known as persistent hyperplastic primary vitreous (PHPV); and proliferative vitreoretinopathy (PVR). The first condition, ROP, commonly involves the anterior aspect of the eye in its severe stages. First reported by Terry in 1942,14 ROP can result in both anterior retrolental membrane formation and vitreous hemorrhage as part of its spectrum of manifestations. Terry noted the formation of a V-shaped retinal detachment resulting in membrane growth behind the lens. It has since been documented that this condition is associated with premature birth, low birth weight, and neonatal oxygen use. Severe neovascular membranes grow on the retinal surface in advancing disease are thought to contribute to traction retinal detachments as well as to the development of vitreous hemorrhage. Over time, a localized vitreous hemorrhage may become dispersed within the vitreous cavity, and streaks of hemorrhage may be seen in the anterior vitreous. Cryotreatment has been used for treatment at the early signs of neovascularization and may result in an associated increase in hemorrhage shortly after treatment. It has been suggested that laser photocoagulation may produce less tissue destruction than cryotherapy and lessen the chance of vitreous hemorrhage.15,16 The second condition, PFV, is a congenital condition; when it occurs, it is most often noted during the first few days or weeks of life, usually in its anterior form.17–19 It is usually unilateral in a microphthalmic eye and generally consists of a retrolental fibrovascular mass with dragged ciliary processes and a secondary cataract. It typically forms a posterior lens attachment with a distinct or feathery edge, around which one may obtain a posterior view of the retina. In this condition, effort should be made to avoid development of glaucoma or phthisis, the most common vision-threatening result of untreated PFV. A lensectomy usually prevents econdary glaucoma, typically the result of lens–iris diaphragm-induced secondary angle-closure glaucoma. Glaucoma also can be produced from recurrent hyphema. When hyphema occurs, outflow passageways in the angle may become blocked with blood. By removing a PHPV membrane and reducing the tractional forces on the ciliary body, one can lessen the risk of phthisis.20 The third condition, PVR, is a cause of retinal membrane development, but it also may involve contracture of the vitreous. PVR is thought to result from mechanisms resulting from the presence of retinal breaks and retinal detachment. Complex retinal detachments may be caused by PVR, probably as a result of cellular changes, which occur in the vitreous cavity and on the retinal surface. These changes may include leukocytes in the vitreous and retinal pigment epithelium (RPE) cells passing through a retinal break, which can infiltrate the vitreous cavity and migrate onto the retinal surface, proliferate, and form a fibrous membrane.21 These PVR membranes can contract and apply traction at the retinal surface. PVR is a complicating factor in 5 to 7% of rhegmatogenous retinal detachment cases. A dense fibrovascular cyclitic membrane can develop after trauma22 or after inflammatory insult. It may surround the lens or cover only a portion of the lens so that an edge is seen on either surface of the lens. Cyclitic membranes develop in the setting of prolonged uncontrolled inflammation, such as in juvenile rheumatoid arthritis,23,24 trauma, or chronic proliferative conditions, such as persistent fetal vasculature.25 When hemorrhages occur in the acute setting, they may form “string-like” streaks seen by the patient. Conditions that cause vitreous hemorrhage are mentioned in the first section of this chapter and in Chapter 12. Streaks of blood in the vitreous cavity may move or remain stationary based on the status of the vitreous. Because blood may be seen after a retinal tear, one should be suspicious of an occult tear when a hemorrhage occurs in the absence of other known predisposing conditions. Close observation is required to avoid a missed break and development of a retinal detachment when a vitreous hemorrhage limits the fundus evaluation. In rare cases, blood may break through into the vitreous from the suprachoroidal space in the setting of a suprachoroidal hemorrhage.26 Inflammatory disease, such as endophthalmitis from bacterial infection or noninfectious inflammation such as sarcoidosis,27–29 may cause opacification of vitreous strands through deposition of inflammatory debris on vitreous collagen strands. Localized collections of inflammatory cells are called snowballs and typically are seen in the inferior vitreous in sarcoidosis and pars planitis.30 Intravitreal fibrin also may form under conditions of extreme inflammation. Fibrin membranes may form strands, sheets, or even encapsulating membranes. Fibrin formation can be extreme after extended vitrectomy for proliferative diabetic retinopathy or after inflammatory conditions such as endophthalmitis. When a fibrinoid syndrome occurs, fibrin deposition within the eye may produce pupillary block or even traction retinal detachment.31,32 The edge of a giant tear may be seen as a curvilinear formation in the anterior vitreous. Giant tears are tears that have a circumferential length greater than three clock hours or 90 degrees.33 Giant tear edges are seen when the tear flap inverts and it is displaced posteriorly. These tears are associated with high frequency of rhegmatogenous or complicated retinal detachment. The tear flap may be freely moving, or it may be semirigid if associated with PVR. When the flap is rolled over, either onto or close to the retinal surface, the whitened anterior rolled edge of the flap can be seen as a distinct border in relief against the dark red RPE. Also, the entire rolled flap itself may appear as a white strip or band, flanked with attached retina on one side and bare RPE on the other. In some cases of retinal detachment with retinal tears (rhegmatogenous detachment), PVR may develop and contribute to surgical treatment failure (see preceding section entitled “Retrolenticular Membranes Associated with Hereditary and Acquired Vitreous Disease.”). In such cases, it is thought that RPE cells migrate into the vitreous cavity and onto the retinal surface, where they proliferate and form contractile membranes. If the surface contraction is strong enough, these membranes may throw the retina into fixed folds.34 These folds often first form in a “star”-shaped pattern.35 With anterior PVR, circumferential retinal folds as well as peripheral radial folds may occur. The retina may take on a more curvilinear deeply folded pattern when folding is severe, often leading to a highly distorted vascular architecture. In the case of penetrating trauma, retinal incarceration into the wound site may cause deep radial folds centered at the wound site. Hemorrhage in the preretinal space, just under the posterior vitreous face (subhyaloid hemorrhage), may be seen in a variety of conditions. Some of these include diabetes, venous occlusion, retinal arterial macroaneurysm,36 sickle cell disease, shaken-baby syndrome,37 Terson syndrome,38–40 Eale’s disease,41,42 and idiopathic retinal vasculitis with arterial macroaneurysms and neuroretinitis. The inferior edge of a detachment of the vitreous may form a bright red, curved edge with hemorrhage pushing the posterior vitreous face forward. This is sometimes referred to as a “boat-shaped” hemorrhage with the inferior edge rounded and the superior edge flat horizontally. When blood is caught under the vitreous face in this way, neodymium:yttrium–garnet–aluminum (Nd:YAG) laser treatment directed at the inferior aspect of the detached vitreous surface can create a hole, possibly allowing a release of preretinal blood and clearing of the visual axis. Diabetic preretinal membranes may form as part of the end-stage process of retinal neovascularization. When membranes form, contracture occurs, which can pull at multiple points on the retinal surface and cause retinal detachment.43–48 Often, distinct edges are formed along these bands of preretinal membrane formation. They may create straight or curved structures, typically along the major vessels in the macular region. Also, organization of the vitreous may cause contracture and coalescence of its collagen fibers, which lead to linear bands within the vitreous cavity oriented either perpendicularly or obliquely to the retinal surface. These bands or membrane edges may be whitish, owing to their fibrotic nature, and they may collect blood on them if a vitreous hemorrhage is present. Retinal folds are typically the result of some type of tractional force oriented along the retinal surface. Macular pucker resulting from localized preretinal membrane formation is a folding of the retina, as its name implies.49–54 Preretinal membranes, also known as epiretinal membranes, are thought to form as the result of dehiscences in the inner limiting membrane, allowing retinal glial cells access to the retinal surface. The cells proliferate and form a membranous sheet, which contracts. A second possible mechanism of preretinal membrane formation involves contraction of vitreous cortex and proliferation of hyalocytes on the inner retinal surface (see Chapter 14). After a posterior vitreous detachment, this process may result in a contractile membrane that can cause retinal folding.55,56 Retinal folds can occur from distant points as a result of traction from peripheral granulomas in toxocariasis57,58 or with peripheral areas of fibrovascular proliferation as in retinopathy of prematurity. In addition, choroidal neovascular membranes can cause retinal strial. When proliferative vitreoretinopathy develops in the subretinal space, the result is often development of a fibrous band. These bands may develop into linear structures, forcing the retina to drape over any elevated portion, much like a sheet drapes over a clothesline.59 Angioid streaks are abnormal linear findings seen on fundus examination that may be associated with significant vision-threatening complications as well as significant systemic disease. These streaks offer a sometimes striking finding for the ophthalmologist and usually appear reddish brown and extend in a branching pattern for varying distances across the fundus. Angioid streaks are pigmented linear crack-like defects in the collagenous and elastic layers of the Bruch membrane,60 and they radiate away from the optic nerve. These streaks are named angioid because of their resemblance to vessels; however, these streaks do not follow the retinal or choroidal vasculatures. These streaks have variable appearance on both fundus examination and angiography. Clinical examination can show a range of findings from subtle pigmentary change to more dramatic large branching bands. In the peripapillary area, the streaks can branch into circumlinear cracks that connect streaks. Streaks are variably seen with fluorescein angiography. In some cases where streaks are minimally evident with fluorescein angiography, infrared indocyanine green angiography can aid in the identification of streaks, whereas in other cases it does not.61 In some cases of angioid streaks, a peau d’orange appearance occurs in the midperipheral fundus. The appearance results from multiple degenerative yellowish deposits in a mottled RPE. Histologically, calcific degeneration of the Bruch membrane often is seen along the streaks.62–65 Complications of angioid streaks result from the defects in Bruch membrane. These include subretinal or sub-RPE exudation that can decrease vision. This exudation can result from choroidal neovascular (CNV) vessels into the subretinal space. Many of the CNV vessels in this disease are type 2 CNV that can become pigmented with proliferating pigmented epithelium cells and can be visualized on clinical inspection, although occult CNV can occur. Subretinal exudation also can occur from rupture of choroidal tissue along the angioid streak. This can occur with relatively insignificant trauma, and patients should warned of this possibility.66 Fluorescein angiography can help identify CNV associated with angioid streaks. Laser photocoagulation can be used to treat CNV if the vessels spare the foveal avascular zone. The Bruch membrane in patients with angioid streaks can be regarded as brittle and prone to further defects and recurrent CNV membranes after laser therapy. If there is a CNV present, photodynamic therapy with intravenous injection of a photosensitive dye and a diffuse retinal laser application shows promise in the treatment of the type 2 CNV associated with angioid streaks.67 If a CNV is not present, spontaneous regression of the subretinal exudate is more likely. Angioid streaks may occur in association with a variety of systemic illnesses in addition to idiopathic cases and age-related degeneration of the Bruch membrane. In one study of the cause of angioid streaks, 50 consecutive patients underwent diagnostic workup; 25 patients (50%) demonstrated one of the disease associations listed subsequently.68 For the patient, loss of vision with subretinal exudation can be the first significant manifestation of these diseases. An inspection of associated physical findings, history, and review of systems should be undertaken in cases when the cause is unknown. The presence of extracellular matrix changes present in pseudoxanthoma elasticum, Paget disease, and Ehlers-Danlos syndrome results in defects in the Bruch membrane and other tissues. In sickle cell disease, calcific accumulation at the levels of the Bruch membrane are thought to result from excessive breakdown of red blood cells and hemoglobin.69,70 Related mechanisms of angioid streaks probably account for the association of angioid streaks with beta-thalassemia, hemoglobin H disease, hemoglobin C disease, hereditary spherocytosis, and abetalipoproteinemia. Of the connective tissue diseases associated with angioid streaks, pseudoxanthoma elasticum is the most common. This disease is named for the skin changes that occur with confluent yellowish papules that gives the skin a “plucked chicken” appearance. This can be seen particularly in the antecubital fossae, neck, and periumbilical areas.71–74 Vascular changes can cause gastrointestinal bleeding and calcification of larger arteries. Eye findings include orangish mottling of the RPE called peau d’orange for its orange-peel appearance and atrophic lesions from alterations in the pigmented epithelium,75,76 hyaline drusen of the optic nerve,77 crystalline bodies in the RPE,78 and pattern dystrophy-like changes in the macula.79 Inheritance is either autosomal dominant or recessive. In the case of Ehlers–Danlos, abnormal skin findings with “webbing” of the neck can be seen. Associated eye findings variably include blue sclera and corneal ectasia. Paget disease can be a disfiguring disease with variable thickening and deformity of bones. When the disease is more generalized with skull involvement and hearing loss, angioid streaks are more commonly seen. Peau d’orange can be seen in the midperipheral fundus as in pseudoxanthoma elasticum and sickle cell disease. Traumatic disruption of the choroids, Bruch membrane, and RPE can result in curvilinear yellow streaks in the posterior pole. These streaks have tapered, pointed ends and are usually concentric to the optic nerve. Although in most cases a recent history of significant ocular trauma is obvious, in some cases, such as in the presence of angioid streaks, relatively minor trauma can result in choroidal rupture.80 In most cases, acute hemorrhagic detachment of the retina obscures a portion of the choroidal and RPE rupture. Retinal changes with Berlin edema may occur initially as a result of trauma and then resolve with time. Fluorescein angiography can evaluate the extent of choroidal and RPE defects obscured by thin hemorrhage. With fluorescein angiography, larger choroidal vessels sometimes can be seen crossing the defect. Spontaneous resolution of the detachment can occur, leaving the appearance of the streaks and various RPE changes. Decreased vision can result despite resolution of the hemorrhage with hemoglobin-induced neuronal degeneration. Traumatic optic neuropathy can result in additional vision loss following trauma, with visual field defects greater than the area of hemorrhagic retina detachment. Choroidal neovascularization can develop through the defect in the RPE, resulting in the delayed reappearance of retinal exudative detachment. Treatment of CNV resulting from choroidal rupture can include focal laser photocoagulation if the fovea remains untreated. Displacement of subretinal hemorrhage with vitrectomy and pneumatic retinopexy with a meticulous (filtered room air) air–fluid exchange and face-down positioning can allow visualization for laser and can limit irreversible damage to retinal neurons resulting from hemoglobin.
Linear and Curvilinear Streaks in the Fundus
Linear or Curvilinear Streaks Observed on or Near the Posterior Lens
Retrolenticular Membranes and Hemorrhage Associated with Hereditary and Acquired Vitreous Disease
Cyclitic Membrane Edges
Linear or Curvilinear Streaks Observed in the Midvitreous
How Does a Vitreous Hemorrhagic Form Streaks?
Linear Inflammatory Debris
What Findings Are Associated with Giant Tears
Why Are Streaks Seen with Retinal Detachment?
Linear or Curvilinear Streaks Observed in the Posterior Vitreous or Preretinal Space
Can Subhyaloid Hemorrhage Form Streaks?
Do Diabetic Traction Detachment Membranes Form Linear Structures?
Linear or Curvilinear Streaks Observed in the Retina
What Causes Retinal Folds?
Linear or Curvilinear Streaks Observed in the Subretinal Space
Why Should Subretinal Proliferative Vitreoretinopathy Be Considered in the Differential of Linear Streaks?
Linear or Curvilinear Streaks Observed in the Retinal Pigment Epithelium or Bruch Membrane
What Are Angioid Streaks?
What Findings Are Associated with Choroidal Rupture?
REFERENCES
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


