24
Laryngeal Paralysis and Paresis
 Pathophysiology of the Paralyzed Vocal Fold
Pathophysiology of the Paralyzed Vocal Fold
The laryngoscopic appearance of vocal fold paralysis reflects the considerable variation in the underlying pathophysiology of the condition. Easily (and often) taken to represent a simple absence of innervation, vocal fold paralysis is in fact the product of a range of underlying neural dysfunction encompassing partial denervation, complete denervation, and variable degrees and patterns of reinnervation. An appreciation of this heterogeneity, coupled with the understanding that vocal fold paralysis is a dynamic condition that tends to evolve after onset, is at the core of interpreting the laryngoscopic examination.
A brief exposition of current knowledge regarding the relevant pathophysiology is in order, especially in contrast with long-held historical beliefs regarding the disorder. Traditionally, vocal fold paralysis has been conceptualized as an all-or-none phenomenon, with absence of motion resulting from a lack of neural input. However, both human and animal studies have shown that cases of vocal fold paralysis, unless the result of simple nerve section, differ in degree and extent of initial neural impairment. Moreover, the injury type itself is usually not uniform; most laryngeal neuropathies feature a mix of injury types among affected nerve fibers.1,2 Further, laryngeal nerves appear to have a strong propensity for reinnervation.2–4 Reinnervation appears to be the rule rather than the exception, occurring often even after deliberate recurrent nerve section, as was formerly done for spasmodic dysphonia.5,6 A paralyzed vocal fold is thus only sometimes, and perhaps quite rarely, a denervated vocal fold.
In the larynx, regeneration of the recurrent nerve is more problematic than that of most peripheral nerves because it carries a mixed population of abductor and adductor fibers. In many—possibly most—cases, vocal fold reinnervation is dysfunctional and does not yield physiologic motion. This dysfunction extends beyond the classic definition of synkinesis, in which co-contraction of adductor and abductor muscle fibers produces no net vocal fold motion; such perfectly balanced antagonism is, after all, an extremely improbable outcome of a largely random process. Dysfunctional reinnervation may also result when nerve regrowth is appropriate but inadequate, which may result in decreased force of contraction, loss of motor unit specificity, and possibly also in changes in neural organization peripherally and centrally.7 An altered afferent signal may also affect laryngeal behavior. The complex, highly specialized nature of the laryngeal neuromotor system leaves many ways in which reinnervation may miscarry.
These findings and observations have at least two important clinical implications. For most of the past century, the position of the paralyzed vocal fold was thought to reflect the site or type of lesion, a belief that has no physiologic basis and has been invalidated by careful clinical and laboratory work.8–10 It should now be clear that the position of a given paralyzed vocal fold is most likely explained by its particular degree and pattern of innervation. Thus, time-honored terms like paramedian and cadaveric carry no topognostic significance and are not useful, except perhaps as purely descriptive conventions. Similarly, notions of discrete adductor paralysis and abductor paralysis, terms that still appear in academic journals with some frequency, also have no physiologic validity. There are no grounds for believing the adductor and abductor nerve fibers have different vulnerabilities or propensity for dysfunction.
Second, the natural propensity for reinnervation most likely accounts for the tendency for the voice—and the laryngoscopic examination—to improve over time in cases of vocal fold paralysis. Reinnervation, even when it does not restore motion, acts to restore or conserve muscle bulk and tone, increasing vocal fold mass and resistance to airflow, occasionally to the point that complete glottic closure during phonation is restored and conversational voice sounds normal, although the vocal fold itself remains immobile (Fig. 24.1). This explanation for the improvement commonly observed in cases of vocal fold paralysis over time is well supported by electrophysiologic evidence and should replace the time-honored notion of gradual contralateral compensation.1,2 Gradual compensation, either as a spontaneous phenomenon or as the result of behavioral intervention, has not been convincingly documented in any study.
 Laryngoscopy of Vocal Fold Paralysis
Laryngoscopy of Vocal Fold Paralysis
Marked vocal fold hypomobility is the cardinal sign of paralysis. We deliberately avoid the term immobility because the vocal fold is rarely perfectly still, even if completely denervated. Simple in concept, hypomobility is sometimes surprisingly difficult to appreciate upon clinical examination. The examiner may be misled by small amounts of vocal fold motion caused by (1) an active interarytenoid muscle still partially innervated by the contralateral side, (2) an intact cricothyroid muscle, and/or (3) passive lateral displacement of the arytenoid cartilage of the paralyzed vocal fold by its pair during adduction. This last is known as the “jostle sign,” described by Chevalier Jackson in the 1930s. When present, it offers evidence for poor ipsilateral muscle tone and against cricoarytenoid joint dysfunction. In ambiguous cases, asking the patient to alternate sustained vowel phonation and sniffing—the so-called /ē/-sniff maneuver—usually brings motion asymmetries into stark relief. This maneuver can also help reveal a rare condition of inappropriate reinnervation resulting in adduction during sniffing or abduction during phonation. This is not to be confused with the entity called paradoxical fold motion, in which innervation is entirely intact and which has no relation to vocal fold paralysis.
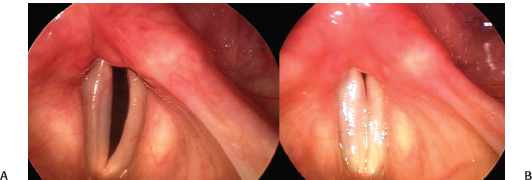
Fig. 24.1 Improvement in vocal fold paralysis: Comparison of best phonatory closure in a 76-year-old woman with left vocal fold paralysis after a carotid endarterectomy. (A) Image from an examination 1 month after injury. (B) Image from an examination 4 months after injury. In the interval, glottic closure has improved because reinnervation has restored muscle bulk and tone, although the left vocal fold itself remains immobile.
Once vocal fold hypomobility has been identified, the larynx should be examined across a variety of activities, including respiration and phonation at a range of intensity and pitch. It is useful to consider these in a logical progression, from examination under continuous light during quiet respiration to examination under continuous light during phonatory tasks to examination under stroboscopic light.
Laryngoscopy at Quiet Respiration
The position of the arytenoid mound, composed of the arytenoid cartilage and overlying cuneiforms, is best assessed during quiet respiration. It may be upright and essentially symmetric with its pair or tipped into the airway to varying degrees; this displacement is frequently referred to as prolapse (Fig. 24.2; Video Clip 30). This represents a lack of muscular support of the cartilage and should not be mistaken for cricoarytenoid subluxation. The cricoarytenoid joint facets provide very little support for the arytenoid as they are broad and shallow to accommodate the multiple degrees of freedom in arytenoid movement. The arytenoid is stabilized principally by the intrinsic laryngeal muscles, much as a tent pole is supported by guy wires. When denervation is profound, this support is lost. The predominant remaining pull is passive and anterior, due to the mass of the body of the vocal fold, including the vocal ligament and the thyroarytenoid muscle, and its attachment to the thyroid cartilage. Prolapse of the arytenoid mound displaces the vocal process, the anteromedial projection of the cartilage where the membranous vocal fold attaches, anteriorly and inferiorly. This three-dimensional displacement is easily overlooked as laryngoscopy yields the impression of predominately two-dimensional vocal fold motion.
As we have noted above, the position of the paralyzed vocal fold is not topognostic, and terms used to describe the position have not been shown to have a specific pathophysiologic correlate.
The vocal fold itself may appear to be of normal or decreased tone. The “slack” appearance in the latter condition typically results in a concave contour of the vibratory margin, often described as “bowing.” Bowing may also be caused by underlying muscle atrophy from denervation. The two conditions—hypotonia and atrophy—are not synonymous, although they are both plainly related consequences of denervation and are in some respects similar on clinical examination. Hypotonia may appear immediately, as after nerve section, and is not associated with loss of vocal fold bulk (Fig. 24.3A; Video Clip 27). Atrophy takes some time to evolve—exactly how long is not established—and produces vocal fold thinning, retraction of the ventricular fold (which shares the same innervation), and enlargement of the ventricle (Fig. 24.3B; Video Clip 29). Paradoxically, ventricular fold retraction and ventricular enlargement may yield the impression that the vocal fold is larger, because more of its superior surface, sometimes extending well lateral to the superior arcuate line, is exposed to the examining eye. Careful examination will reveal the finding for what it is.
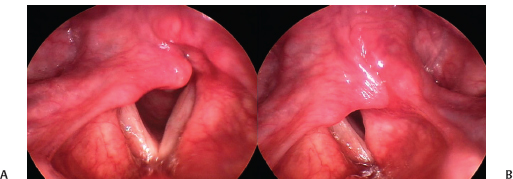
Fig. 24.2 Arytenoid prolapse: (A) In this case of idiopathic right vocal fold paralysis, the arytenoid cartilage is prolapsed into the glottis, lowering the vocal fold below the level of its partner. (B) In phonatory adduction, the prolapsed arytenoid interferes with the opposite cartilage and impedes closure. Rehabilitation without arytenoid repositioning surgery would very likely be suboptimal.
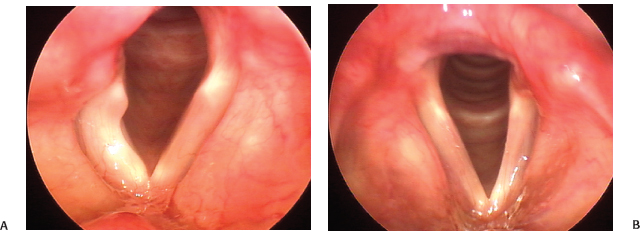
Fig. 24.3 Hypotonia versus atrophy. (A) The right vocal fold in this young woman with a metastasis at the jugular foramen is markedly hypotonic. (B) In contrast with the findings in A, the left vocal fold in this woman with long-standing idiopathic vocal fold paralysis is thin, and the ventricular fold has receded, exposing the ventricle. Both of these findings are the result of atrophy. It is common to find elements of both hypotonia and atrophy in cases of vocal fold paralysis.
As hypotonia and atrophy are both incontrovertible signs of denervation, it is intuitively compelling to think that the longer from onset they are present, the poorer the prospects for recovery. This proposition has yet to be validated by experiment but already occasionally informs our clinical decision-making in vocal fold paralysis.
Valuable signs may also be observed away from the vocal fold proper. The presence of salivary pooling, typically in the piriform sinus on the side of the paralysis, speaks to the presence of a trio of deficits resulting from a lesion of the main trunk of the vagus—a so-called high vagal injury (Fig. 24.4; Video Clip 48). These are (1) hemilaryngeal hypoesthesia or anesthesia from involvement of the superior laryngeal nerve, (2) ipsilateral constrictor muscle hypofunction, and (3) cricopharyngeal muscle hyperfunction. Of these, the last is not always present due to bilateral innervation. Inferior constrictor hypofunction manifests as piriform sinus dilatation at rest or failure to contract during a pharyngeal squeeze maneuver. Together with glottic insufficiency, these deficits form a “perfect storm” for swallowing dysfunction by creating a hypopharyngeal reservoir of secretions that are neither well-propelled nor well-drained into the esophagus upon swallowing and that the larynx cannot sense. Upon reflection, it should be obvious that medialization alone will often not remedy this situation well, as it only addresses one component of the dysfunction; yet many are often puzzled by continued aspiration after an adequate medialization laryngoplasty.
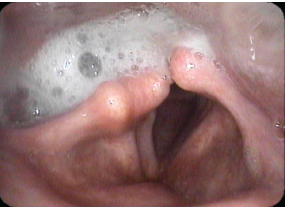
Fig. 24.4 “High vagal” injury: This examination of a 69-year-old man after surgery to remove a glomus jugulare tumor shows right vocal fold paralysis, ipsilateral salivary pooling, and dilatation of the right piriform sinus. The last two are hallmark signs of an injury involving the entire vagus rather than just the recurrent nerve. See also Figs. 24.9 and 24.10.
Laryngoscopy during Phonation
As the primary symptom of vocal fold paralysis is usually dysphonia, the importance of examination during phonation is self-evident. Clear visualization of the vocal folds may be impaired by supraglottic hyperfunction, most often manifested as contraction of the contralateral ventricular fold, a natural response to glottal insufficiency.11 The ipsilateral ventricular fold, which shares innervation with the vocal fold proper, is usually quiescent under conditions of denervation (Fig. 24.5; Video Clip 49). “Unloading the larynx,” or eliminating supraglottic hyperfunction by behavioral techniques, may be helpful to decrease supraglottic squeeze to clear the line of sight to the glottis proper.11
Dysphonia in vocal fold paralysis is principally, although not exclusively, the acoustic consequence of incomplete glottic closure and irregularities in vocal fold vibration resulting from differences in vocal fold tension and shape. Glottic insufficiency varies in both degree and configuration from case to case, and, as we have discussed above, may change in the same case over time. It is occasionally debated whether it is best evaluated by flexible or rigid laryngoscopy. Flexible transnasal laryngoscopy probably offers a more accurate impression of laryngeal function, as the tongue traction required for rigid peroral techniques introduces some artificial biomechanical factors that may be misleading. On the other hand, rigid examination, particularly when compared with fiberoptic rather than distal chip endoscopes, offers a far more precise image of the vibratory margin and may thus be the better tool for stroboscopic examination. Debate regarding the superiority of one or the other is somewhat academic, however, as there is absolutely nothing to prevent evaluation by both techniques when the clinician believes there is more information that can be obtained.
There exists no agreed-upon scale for the measurement of glottic insufficiency; most reports settle upon relative classifications such as mild, moderate, or severe. Given the variable angle and distance of the glottis from the examiner’s endoscope and all of the variations in phonation (pitch and intensity foremost among them), an absolute measurement of glottic insufficiency that is reproducible from exam to exam is probably impossible. As a generalization, however, the larger the gap, the more profound the dysphonia, and the more aggressive will be the required rehabilitation procedure; medialization laryngoplasty is superior to injection augmentation in correcting large insufficiencies.
The configuration of glottic insufficiency in unilateral paralysis most commonly takes one of two shapes. It may be essentially spindle-shaped, involving principally the membranous portion of the vocal fold, or V-shaped, marked by a distance between the vocal processes of the arytenoid cartilage (Fig. 24.6). The latter is often called a posterior glottic gap; it is important to understand that the term does not refer to the absolute distance between the vocal process or to the size of the insufficiency but rather to its configuration. The presence of a posterior glottic gap argues in favor of an arytenoid stabilization procedure for rehabilitation because implant medialization alone, or injection augmentation for that matter, is notoriously inadequate for correction of this deficit.
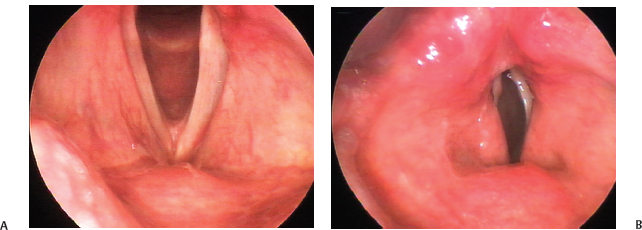
Fig. 24.5 (A) Vocal folds seen during quiet respiration in a case of left vocal fold paralysis after thyroid surgery. (B) Ventricular function in the same patient during phonation. Often, the ventricular fold of the unaffected side is engaged in effortful glottic closure in vocal fold paralysis, as in this case of left vocal fold paralysis after thyroid surgery. The resulting bulge can obscure the glottis in whole or in part.
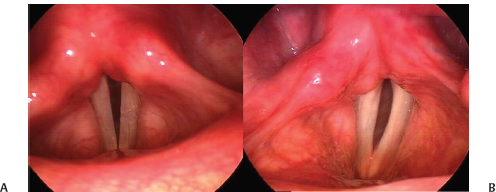
Fig. 24.6 Configuration of glottic insufficiency: (A) A case of left vocal fold paralysis after a thyroidectomy demonstrates a posterior gap. (B) A case of left vocal fold paralysis from a malignant mediastinal lymphadenopathy in which closure at the vocal process is good. Compare also with the case in Fig. 25.2, which also demonstrates a large posterior gap.
A paralyzed vocal fold may not rest in the same plane as its partner. Such three-dimensional judgments are difficult to make in laryngoscopy, particularly when the vocal folds do not lie closely juxtaposed, which is the rule in vocal fold paralysis. Nevertheless, identification of a height mismatch is clinically relevant, as simple medial displacement of the paralyzed fold may not suffice for good apposition in phonation.
The real physiologic impact of hypotonia of the paralyzed vocal fold can only be accurately assessed during phonation. When challenged with subglottic air pressure, what may have appeared to be an only mildly bowed vocal fold during quiet respiration may be revealed to be far more flaccid. Obviously, this effect will be more pronounced the louder the volume solicited from the patient. Some patients reflexively tense the vocal fold to resist this subglottic pressure. Not only does this result in a characteristic high-pitched voice known as a paralytic falsetto, but also it strongly suggests that the superior laryngeal nerve is intact to provide the cricothyroid muscle activation necessary for this response (Fig. 24.7; Video Clip 25).
Stroboscopy
Poor muscle tone and activity of the paralyzed vocal fold, besides contributing to glottic insufficiency, alters intrinsic tissue properties to yield abnormalities in phonatory vibration when compared with the normal side. These, of course, are only discernible on stroboscopy. In general, decreased vocal fold tension yields increased amplitude of vibration, increased lateral displacement of vocal fold tissue during the vibratory cycle, and mucosal wave phase shifts or outright asymmetry in frequency of vibration. In the most severe cases, mucosal vibration will be frankly aperiodic. As stroboscopic examination depends on a stable frequency of vibration to time light flashes, such cases yield the illusion of nonsequential vibration, “skipping” video frames, and other irregularities.
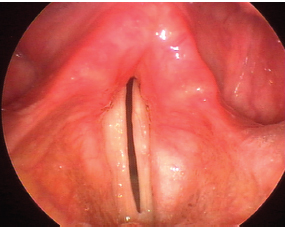
Fig. 24.7 Paralytic falsetto. This 69-year-old man with right vocal fold paralysis after cervical spine surgery uses his intact cricothyroid muscle to aid glottic resistance and improve projection. The pitch of the resulting voice is abnormally high, as would be expected.
The mucosal wave abnormalities are to be expected in vocal fold paralysis and, in and of themselves, are not particularly revealing. The real value of stroboscopy lies in more precise visualization of the configuration and degree of glottic insufficiency. Sharp delineation of the vibratory margin of the vocal fold during phonation is central to this task; only examination under stroboscopic light can reveal this adequately. Even in cases of borderline glottic insufficiency, stroboscopy may permit a more precise estimation of degree of glottic insufficiency by means of frame counting. The examiner begins review of a phonatory cycle on an image in which the glottis is unambiguously closed. Then, proceeding frame-by-frame until the cycle is completed, each image in which space is visible between the vocal folds is counted as “open.” The total number of open frames is divided by the total number of frames in a cycle to yield the open quotient. This value varies at different pitches and volumes, but as a generalization, the normal open quotient is ∼0.5 in modal phonation.
Examination after Rehabilitative Intervention
Dysphonia after rehabilitative surgery for vocal fold paralysis is not rare. It is typically due to technical factors, and surgical revision rates range from 5.4 to 14% to as high as 33% when all procedures to improve voice are considered.12–14 Stroboscopy is particularly helpful in correctly assessing glottic closure as described earlier and prescribing corrective treatment. In injection augmentation, poor voice result is usually a matter of underinjection, overinjection, or misplacement of the injectate (Fig. 24.8; Video Clip 50). Most often, this last takes the form of diffusion into the superficial lamina propria, causing focal vibratory stiffness, best identified by stroboscopy.
The most common causes of suboptimal voice outcomes after medialization laryngoplasty include persistent posterior glottic gap, usually due to inappropriate omission of an arytenoid procedure, undermedialization, or implant malposition, most often in too superior a position (Fig. 24.9; Video Clip 51). The first two of these are usually evident on careful examination. The last presents the impression of ventricular fold prominence that does not disappear with quiet respiration (Fig. 24.10; Video Clip 52).
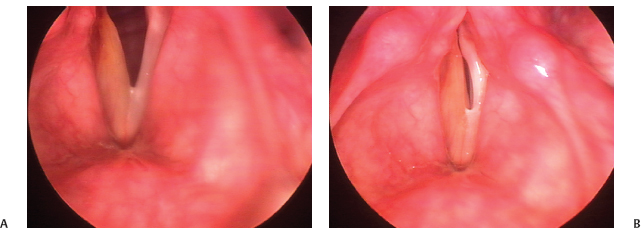
Fig. 24.8 Overinjection: This 51-year-old physician has had an injection medialization for right vocal fold paralysis after parathyroidectomy. He has a strained voice quality because of overinjection. (A) Vocal folds viewed during abduction. (B) The right vocal fold is convex and damps vibration of the left during phonation because of its bulk. It is quite stiff; its dun color represents edema.
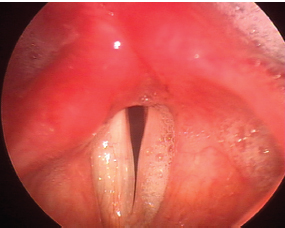
Fig. 24.9 Persistent posterior glottic gap: This 54-year-old woman with a left vocal fold paralysis after resection of a jugular foramen tumor underwent medialization laryngoplasty without an arytenoid adduction. As a result, there is a persistent gap at the vocal processes; closure overall is suboptimal. Note also the secretions overwhelming the larynx on the side of the paralysis typical of a high vagal injury.
 Laryngoscopy of Vocal Fold Paresis
Laryngoscopy of Vocal Fold Paresis
Given the heterogeneity of the neural compromise underlying vocal fold paralysis, it should come as no surprise that paresis, or incomplete paralysis in which some or most gross vocal fold mobility is preserved, exists alongside paralysis as a clinical entity. While the existence of laryngeal paresis is beyond doubt, its incidence, presentation, and significance remain debated topics. Much difficulty stems from the fact that vocal fold paresis is often diagnosed by laryngoscopy, based on mild dynamic laryngeal asymmetries that are determined to be meaningful more by clinical expectations or hypotheses rather than by physiologic evidence. It is not much of an exaggeration to say that a determined observer may find signs of paresis in virtually every larynx. Separating innocent asymmetries from significant findings is the greatest challenge in the laryngoscopic characterization of vocal fold paresis.
As in paralysis, vocal fold hypomobility is probably the most common sign of motor paresis, although mobility is retained to a far greater extent in paresis. Rubin and colleagues have described the use of repetitive phonatory tasks during examination to fatigue inadequately innervated musculature to accentuate mild vocal fold hypomobility.15 Glottic insufficiency during phonation may be difficult to discern because of its subtlety and also because of supraglottic hyperfunction, as discussed earlier. Asymmetries in vocal fold tension can be the result of both recurrent and superior laryngeal pareses and may manifest exclusively as asymmetry of mucosal wave motion, noticeable only on examination under stroboscopic light. It is worth repeating that not all such asymmetries are likely to be significant. The consistency of such findings across multiple pitches and intensities, and with patient complaints, is an important element in judging their relevance.
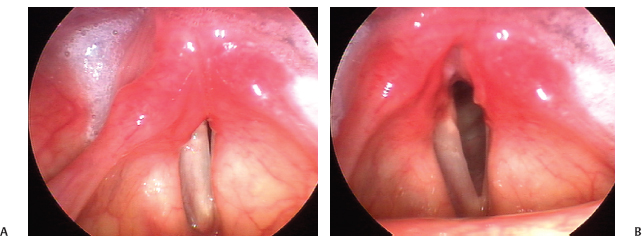
Fig. 24.10 (A) Superior implant malposition: This 27-year-old woman with a left vocal fold paralysis has a poor voice result because the implant rests above the vocal fold, medializing the ventricular fold. The ventricular fold obscures the vocal fold, which is not well adducted. (B) This is not ventricular fold hyperfunction, as the ventricular fold remains full even in respiration, at right.
Virtually every author to address the subject of vocal fold paresis has remarked on the discrepancy of clinical observations and electromyographic findings.15–18 Although electromyography is neither a perfect nor a complete means of neurologic assessment of the larynx, it comes far closer than does laryn-goscopy to an objective diagnosis of neurologic impairment. In one series, around 1 in 4 patients had electromyographic findings not predicted by the endoscopic examiner; in another, the incidence of unexpected findings was still higher, about 40%.17,19 Further, observations from blinded examinations of experimentally induced paresis demonstrate not only poor intrarater and interrater reliability but also extremely low diagnostic accuracy.20 In this context, claims of diagnostic precision using laryngoscopy alone should be treated with great caution; laryngoscopy, whether under continuous or stroboscopic light, remains a tool of uncertain utility in vocal fold paresis.
 Conclusion
Conclusion
The laryngoscopic appearance of vocal fold paralysis reflects underlying pathophysiologic processes. A thorough understanding of these and their laryngoscopic correlates allows a precise, accurate, and insightful assessment of each case, which in turn informs and individualizes clinical care in the best interest of the patient.
References
1. Bielamowicz S, Stager SV. Diagnosis of unilateral recurrent laryngeal nerve paralysis: laryngeal electromyography, subjective rating scales, acoustic and aerodynamic measures. Laryngoscope 2006; 116:359–364
2. Blitzer A, Jahn AF, Keidar A. Semon’s law revisited: an electromyographic analysis of laryngeal synkinesis. Ann Otol Rhinol Laryngol 1996;105:764–769
3. Woodson GE. Spontaneous laryngeal reinnervation after recurrent laryngeal or vagus nerve injury. Ann Otol Rhinol Laryngol 2007;116:57–65
4. Zealear DL, Hamdan AL, Rainey CL. Effects of denervation on posterior cricoarytenoid muscle physiology and histochemistry. Ann Otol Rhinol Laryngol 1994;103:780–788
5. Aronson AE, De Santo LW. Adductor spastic dysphonia: three years after recurrent laryngeal nerve resection. Laryngoscope 1983;93:1–8
6. Netterville JL, Stone RE, Rainey C, Zealear DL, Ossoff RH. Recurrent laryngeal nerve avulsion for treatment of spastic dysphonia. Ann Otol Rhinol Laryngol 1991;100:10–14
7. Zealear DL, Billante CR. Neurophysiology of vocal fold paralysis. Otolaryngol Clin North Am 2004;37:1–23, v
8. Woodson GE. Configuration of the glottis in laryngeal paralysis. I: clinical study. Laryngoscope 1993;103(11 Pt 1):1227–1234
9. Woodson GE. Configuration of the glottis in laryngeal paralysis. II: animal experiments. Laryngoscope 1993;103(11 Pt 1):1235–1241
10. Koufman JA, Walker FO, Joharji GM. The cricothyroid muscle does not influence vocal fold position in laryngeal paralysis. Laryngoscope 1995;105(4 Pt 1):368–372
11. Belafsky PC, Postma GN, Reulbach TR, Holland BW, Koufman JA. Muscle tension dysphonia as a sign of underlying glottal insufficiency. Otolaryngol Head Neck Surg 2002;127:448–451
12. Weinman EC, Maragos NE. Airway compromise in thyroplasty surgery. Laryngoscope 2000;110:1082–1085
13. Rosen CA. Complications of phonosurgery: results of a national survey. Laryngoscope 1998;108(11 Pt 1):1697–1703
14. Anderson TD, Spiegel JR, Sataloff RT. Thyroplasty revisions: frequency and predictive factors. J Voice 2003;17:442–448
15. Rubin AD, Praneetvatakul V, Heman-Ackah Y, Moyer CA, Mandel S, Sataloff RT. Repetitive phonatory tasks for identifying vocal fold paresis. J Voice 2005; 19:679–686
16. Koufman JA, Postma GN, Cummins MM, Blalock PD. Vocal fold paresis. Otolaryngol Head Neck Surg 2000;122:537–541
17. Heman-Ackah YD, Barr A. Mild vocal fold paresis: understanding clinical presentation and electromyographic findings. J Voice 2006;20:269–281
18. Altman KW. Laryngeal asymmetry on indirect laryngoscopy in a symptomatic patient should be evalu ated with electromyography. Arch Otolaryngol Head Neck Surg 2005; 131:356–359
19. Koufman JA, Postma GN, Whang CS, et al. Diagnostic laryngeal electromyography: the Wake Forest experience 1995–1999. Otolaryngol Head Neck Surg 2001;124:603–606
20. Roy N, Barton M, Smith ME, Dromey C, Merrill R, Sauder C. An in vivo model of external superior laryngeal nerve paralysis: laryngoscopic findings. Laryngoscope 2009;119:1017–1032
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


