23
Laryngeal Inflammation
Laryngitis, meaning inflammation of the larynx, is one of the most common conditions involving the larynx and occurs in both an acute and a chronic form. Acute laryngitis has an abrupt onset and is usually self-limited. The etiology of acute laryngitis includes exposure to noxious inhaled agents or infectious agents associated with upper respiratory tract infections. The infectious agents are most often viral but are sometimes bacterial (Fig. 23.1). When the etiology of acute laryngitis is infectious, white blood cells remove microorganisms during the healing process.
If a patient has symptoms of laryngitis for more than 3 weeks, the condition is classified as chronic laryngitis. Chronic laryngitis may be caused by environmental factors such as inhalation of cigarette smoke or polluted air (eg, gaseous chemicals), irritation from asthma inhalers, vocal misuse (eg, prolonged vocal use at abnormal loudness or pitch), or gastroesophageal reflux diseasse.1–5 Rarely, laryngeal inflammation results from autoimmune conditions such as rheumatoid arthritis, relapsing polychondritis, Wegener’s granulomatosis, or sarcoidosis (Fig. 23.2).
Laryngitis causes the patient to experience dysphonia or a hoarse voice. A hoarse voice is defined as one that demonstrates roughness, breathiness, and tension.6 These symptoms can result from a multitude of causes, however, and are not specific for acute or chronic laryngitis. Though usually self-limited, acute laryngeal inflammation leads to poor voice production secondary to the edema of the vocal fold tissues. Ng et al. found that fundamental frequency values were lower in patients with acute laryngitis than in patients with a normal voice.6 The lowered pitch in patients with laryngitis is a result of irregular thickening of the tissues due to swelling along the entire length of the vocal fold.7–9 Edema leads to an increase in vocal fold mass that results in a decrease in the frequency of vibration.
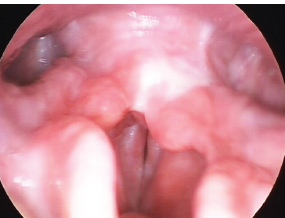
Fig. 23.1 Acute laryngitis. Note edema and erythema of the true vocal folds.
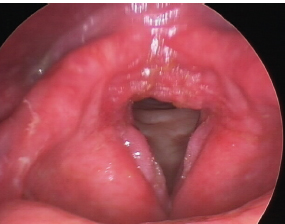
Fig. 23.2 Chronic laryngitis. Vocal folds are thickened and edematous with erythema.
The thickening and edema of the vocal folds in acute laryngitis make the vocal fold cover stiff and more difficult to set into vibration. Phonation requires greater vocal fold adduction forces or tension and subsequent increases in subglottal pressure to overcome the increase in vocal fold stiffness. The phonation threshold pressure may increase to a degree that generating adequate phonation pressures in a normal fashion becomes difficult. Frank aphonia results when a patient cannot overcome the phonation threshold pressure required to set the vocal folds into motion. Increasing vocal fold adduction forces or tension further negatively impacts voice production, causing more injury to the vocal fold tissues and ultimately delaying recovery and a return to normal phonation.
 Pathophysiology
Pathophysiology
During the early stages of inflammation, the vascular endothelium becomes “leaky.” This disruption of the blood vessels allows serum and other factors to leak into the lamina propria. The normal blood flow is altered at the site of injury. Other cells migrate to the site in response to locally released inflammatory factors. Clinically, acute inflammation results in vocal fold edema and erythema (Figs. 23.1 and 23.2).
Once acute inflammation occurs, three phases of vocal fold healing must be considered.10 Recovery from an acute inflammatory injury begins as fibroblasts secrete new materials into the tissue injury site. Fibroblasts produce various extracellular matrix components including collagen, elastin, glycosaminoglycan, and proteoglycans. The appropriate combination of these components is important to maintain the viscoelastic shear property of the vocal folds. Any changes to the amount and configuration of any of these components results in decreased vocal fold pliability, thus altering vocal fold vibration. Hyaluronic acid, which is widely distributed in the superficial layer of the lamina propria, is essential to maintaining the viscosity of the vocal fold. Studies of this molecule have shown that it may be able to modulate scarless healing, implying that it has potential as a treatment designed to reduce scarring during the early recovery stages of vocal fold injury.11,12
Deposition of scar tissue is the second stage of healing, followed by wound contracture. As with wounds in other parts of the body, vocal fold scarring is seen as the end result of both acute and chronic inflammation. In cases of excessive collagen deposition, scarring causes a severe disturbance of vocal fold vibration resulting in considerable dysphonia. Sometimes, scarring is severe enough to cause outright fixation of the mucosa to deeper tissues. Once the vocal fold is scarred, the layered structure of the lamina propria is disrupted, and the mucosal vibratory properties are changed. Altered vocal fold vibration—depending on severity and extent—can cause a range of symptoms such as hoarseness, breathy voice, increased effort to speak, and voice fatigue.
 Examination
Examination
The diagnosis of laryngeal inflammation is based on a laryngoscopy. To fully characterize the exact nature of vocal fold injury secondary to inflammation, a more detailed evaluation is necessary with the help of a videostroboscopic exam. Videostroboscopy plays an essential role in the assessment of the elasticity, viscosity, volume, and tension of the vocal folds. For periodic vibration to occur, the mechanical properties of the vocal folds and the subglottic pressure must be in equilibrium. The superficial lamina propria, a loosely packed network of fibers that functions like a ballbearing layer, allows the mucosa on top to be flexible and not tightly attached to the deeper tissues. For normal vibration to occur, the superficial lamina propria must be healthy.13–16 In inflammation, however, edema and cellular infiltration of the superficial lamina propria cause changes in the mechanical properties of the tissue that negatively affect vocal fold vibration. In general, videostroboscopic analysis of laryngitis reveals aperiodicity, vibratory asymmetry, decreased amplitude of vibration, and incomplete glottic closure with glottal gap and amplitude of vibration being most effected (Video Clips 37 and 47).
Glottic Configuration
The early stages of vocal fold inflammation are remarkable for edematous changes and thickening of the lamina propria. The inflammatory changes may compromise arytenoid approximation resulting in reduced vocal fold adduction. That, along with the irregular vocal fold margins, results in a glottic gap that allows air to escape, decreasing vocal loudness. Individuals who have a great demand for voice production will attempt to overcompensate by using the extrinsic laryngeal muscles to increase vocal fold adduction. This strategy usually results in an asymmetric approximation where only portions of the vocal folds make contact. The contact areas receive all the impact during phonation causing further localized tissue trauma and may result in the development of vocal nodules and polyps.17
Phase Characteristics and Vibration Amplitude
Inflammation of the vocal folds has been shown to increase the mean closed quotient, which is defined as the ratio of the closed phase to the pitch. The main perceptual correlate of an increase of the closed quotient is the need for increased pressure, in other words, a pressed voice. An increased closed quotient has been associated with a lower fundamental frequency, abnormal jitter and shimmer, decreased phonation time, abnormal harmonic to noise ratio, lower amplitude of vibration, and aperiodicity. Correlation analysis has demonstrated that these vocal parameters improved as the mean closed quotient decreased.18
Inflammation that is limited to the posterior part of the larynx may have the opposite effect (ie, an increase in the open quotient), which is defined as the ratio of the open phase to the pitch. The open quotient is associated with increased breathiness and a decrease in vocal intensity.19
A unilateral or, more often bilateral, decrease of vibration amplitude is also noted with inflammation of the vocal folds secondary to the increase in mass and stiffness of the tissues.20,21
Mucosal Wave
In laryngitis, the propagation of the mucosal wave is reduced. Changes in the mucosal wave occur if the inflammation extends below the level of the vocal fold basement membrane, extending into the superficial lamina propria, leading to irregular swelling of the vocal folds and reducing the mobility of the overlying vocal fold cover. Ng et al. described the mucosal wave in laryngitis as appearing to have two distinct velocities of travel. The wave travels at one speed on the surface of the vocal fold, but, at a discrete point, it changes its speed of travel.6 This finding was also described by Colton et al.22 Possible mechanisms for this phenomena are the asymmetry of vocal folds, the desynchronization of vertical and horizontal vibration within and between the vocal folds, the coupling with other laryngeal oscillators in the subglottal and supraglottal airway, and the need to alter the airflow during phonation22–25
In summary, inflammation of the vocal folds causes irregular and/or reduced vibration that is further characterized on videostroboscopy by a decreased amplitude of vibration, a decrease in the mucosal wave, asymmetric vibration, and irregular glottic configuration with a lowering of the fundamental frequency. An understanding of how inflammatory pathophysiology affects vocal folds structures explains these findings. Two studies are cited here as clinical examples of how laryngeal inflammation from various causes impacts vibratory characteristics.
In a study of laryngeal tuberculosis by Agarwal and Bais, videostroboscopy revealed asymmetry of vocal fold vibration, a decrease in the amplitude of horizontal excursion of the vocal fold, a decreased mucosal wave, and aperiodic vibration on the affected side. In cases where generalized edema and an increase in thickness of the lamina propria of both vocal folds were found, the vibratory movements were symmetric but with decreased amplitude. If the inflammatory changes resulted in irregular scarring of the vocal folds, then the amplitude of vibration was absent or significantly reduced around the lesions.26
In a study of patients with chronic laryngitis secondary to asthma inhaler use, Mirza et al. found evidence of vascular dilation including varices and small areas of hemorrhage on laryngeal examination. The margins of the vocal folds also demonstrated areas of thickening and leukoplakia. Videostroboscopy demonstrated a decrease in vibration amplitude and a decrease in the mucosal wave.5
Reinke’s Edema
Reinke’s edema of the vocal folds is a special form of chronic hyperplastic laryngitis characterized by the collection of edema fluids directly under the vocal fold epithelium (Fig. 23.3). The stroma is hyalinized and thickened. The voice, which has a pressed quality, is harsh or breathy and has an exceptionally low pitch. On stroboscopy there is phase asymmetry because of impaired closure due to the irregular vocal fold margins and a mass effect (Video Clip 36). The shape of the glottic closure is irregular because of early contact of the irregular hyperplastic mucosa. The closed phase of the vibratory cycle is relatively long. The mucosal wave shows a decrease in amplitude.27
Leukoplakia and Pachydermia
Chronic irritation from long-standing inflammation can lead to leukoplakia and pachydermia consisting of hyperplasia of the epithelial layer of the vocal folds and greater hornification in the top layers of the epithelium. The ability of the vocal folds to vibrate is directly related to the thickness of the epithelium: the thicker and stiffer the epithelium, the more restricted the mobility of the vocal fold edges, resulting in a reduction of the ability of the vocal folds to vibrate. The voice is characterized by low pitch and a harsh or breathy quality (Fig. 23.4).4
Scarring
Scarring makes the vocal folds stiff. Stiff vocal folds need the generation of greater subglottal pressure to set them into vibration. Hence, patients with scarring in one or both vocal folds usually complain of increased effort with phonation and therefore voice fatigue. The scarred area is also slower to heal with each additional inflammatory insult. Scarring at the inferior (bottom) edge of the closing (striking) surface of the vocal folds is harder to detect. Usually the superior (top) edge of the closing surface can and will compensate. With increasing scar extent or severity, loss of vocal fold pliability increases, thus hampering additional phases of the vibratory cycle—and resulting in more severe voice symptoms (Fig. 23.5; Video Clip 33).28
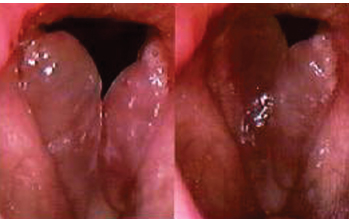
Fig. 23.3 Reinke’s edema characterized by the collection of a thickened stromal fluid within the lamina propria. The vocal fold mass increases and the voice becomes lower in pitch.
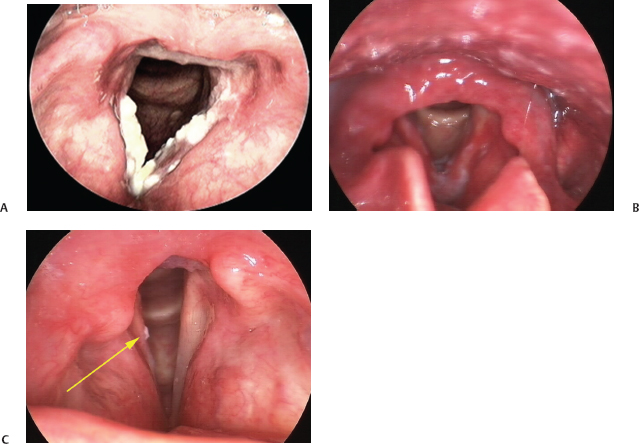
Fig. 23.4 (A) Leukoplakia of the vocal folds. The epithelium is thickened and keratinized. (B) Leukoplakia may be associated with erythema of the vocal folds. This lesion was biopsied, and histologic evaluation revealed carcinoma in situ. (C) The arrow points to a small area of leukoplakia that was identified adjacent to the resection margin of a region of carcinoma in situ. The lesion may represent recurrence at the margin of the resection.
Glottal Sulcus
The etiology of glottal sulcus is unknown but theoretically occurs at the site of a vocal fold injury or chronic inflammation. If this occurs over the medial surface of the vocal fold, it results in a groove or infolding of mucosa (Fig. 23.6). In the area of the sulcus, the mucosa is tethered down to the underlying vocal ligament, giving it a retracted, tethered appearance. The mucosal cover may fibrose to the vocal ligament and results in a diminished or absent mucosal wave at the location of the sulcus. This decreased pliability restricts the Bernoulli and vocal fold myoelastic effects, whereby transglottic airflow medializes the leading edge of the vocal fold during vibration. The overall effect usually is a higher fundamental frequency with significantly reduced harmonics and harsher voice quality. There is also a defect in the medial surface of the true vocal fold along the sulcus that may produce a glottic gap. Videostroboscopy is an important tool used by the laryngologist to reveal linear depressions or areas of incomplete closure and areas of decreased mucosal wave corresponding with the sulcus.29
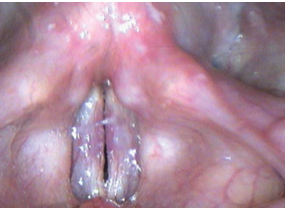
Fig. 23.5 Mild scarring of the vocal folds may be difficult to detect on examination with a constant light source. Videostroboscopy reveals a decrease in the amplitude of vibration, a decrease in the mucosal wave, and, in this case, a persistent glottic gap and irregular vocal fold margin.
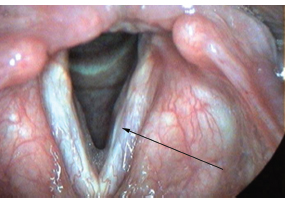
Fig. 23.6 Glottic sulcus may appear as a groove along the margin of the vocal fold as demonstrated at the tip of the arrow in this image.
Reflux
Understanding the influence of laryngopharyngeal reflux on laryngeal inflammation and hence on the vibratory characteristics of the vocal folds is complicated due to inconsistencies in diagnosis. The gold standard for making a diagnosis of laryngopharyngeal reflux (LPR) disease is 24-hour dual-channel pH monitoring, wherein one sensor is placed in the esophagus and the other is placed just above the upper esophageal sphincter (UES). The incidence and duration of acidic conditions in the pharynx during a 24-hour period can thus be determined. Although 24-hour pH probe studies are considered to be very specific for the diagnosis of reflux, they are not considered to be sensitive and may fail to identify patients suffering from reflux episodes that occur less frequently than every 24 hours. In addition, the clinical reproducibility of the test has not been established. Furthermore, this technology is still not widely available, and many clinicians continue to make a diagnosis of LPR disease based on a combination of symptoms and examination findings along with the response to empiric treatment with proton pump inhibitors. Considerable controversy exists, however, on the reliability of using the signs of laryngeal inflammation, without pH study confirmation, to make a diagnosis of LPR disease.4,30–38 Physical findings seen on laryngeal imaging that are commonly believed to be associated with LPR disease include increased interarytenoid or posterior glottic inflammation or erythema, hypertrophy of the posterior commissure (cobblestoning), vocal fold edema and erythema, vocal fold vascularity, and mucus accumulation (Figs. 23.7 to 23.9).
Complicating the issue of making a diagnosis of LPR disease on laryngeal imaging is the fact that there is poor reliability among clinicians regarding the ability to rate the visual criteria required to make a diagnosis of LPR disease. Branski et al. conducted a blinded study in which five otolaryngologists reviewed the videostroboscopy studies from 100 consecutive patients presenting with voice complaints and rated signs of reflux disease. Interrater reliability was poor, in particular with respect to the degree of pachydermia, the overall severity of LPR findings, and the likelihood that LPR was contributing to the patient’s symptoms. Raters were most likely to agree on the degree of involvement of the membranous vocal folds and were least likely to agree on the degree of involvement of the arytenoid cartilages. Only one rater was able to demonstrate consistent interrater reliability for the findings of reflux disease on examination.39 The results from a study of 105 asymptomatic volunteers by Hicks et al. were somewhat better, with good intrarater agreement found between three raters for interarytenoid bar, arytenoid medial wall erythema, and cobblestoning. Like the previous study, there was “good” intrarater agreement for the finding of true vocal fold edema. However, a strong interrater agreement was not established for any of the signs rated in the study.40
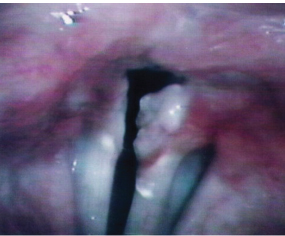
Fig. 23.7 This figure demonstrates some of the findings considered to be the result of gastroesophageal reflux disease: erythema of the mucosa over the ary-tenoid cartilages and the development of granulation tissue on the left arytenoid vocal process.
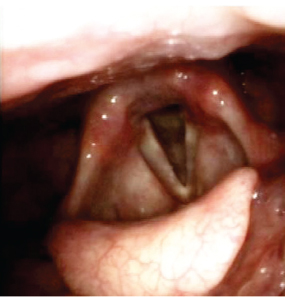
Fig. 23.8 One of the most common findings thought to be consistent with reflux disease: erythema of the mucosa overlying the arytenoid cartilages.
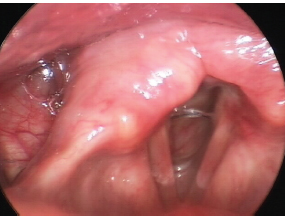
Fig. 23.9 This image demonstrates increased mucus collecting on the vocal folds and erythema of the true vocal folds in addition to erythema of the mucosa overlying the arytenoid cartilages. The arytenoid mucosa may also be edematous with evidence of edema of the tissues adjacent to the vocal process of the arytenoid cartilages. These findings are consistent with laryngeal inflammation that might be secondary to gastroesophageal reflux disease.
Recently, in a study of differences in the detection of reflux-associated laryngeal signs based on imaging modality (flexible endoscope vs rigid endoscope, with or without distal chip technology), Eller et al. reported the interrater and intrarater reliability from five examiners who retrospectively evaluated both flexible and rigid exams from 34 patients. Subjects included in the study were consecutive patients presenting with voice complaints. The examiners rated the following signs: subglottic edema, ventricular obliteration, erythema, vocal fold edema, diffuse laryngeal edema, interarytenoid thickening, granuloma, thick mucus, and posterior erythema. Interrater reliability was generally higher (Cronbach α > 0.7) when distal chip technology was used for imaging. Only the distal chip evaluation of subglottic edema and erythema had less than good intrarater reliability. Without the distal chip technology, intrarater agreement was less than 0.7 for all of the parameters except posterior erythema, which was reliable for both fiberoptic and rigid imaging. These findings might indicate that newer technologies have the potential to increase our ability to reliably evaluate these laryngeal signs. The same study, however, found that color evaluation (erythema) was more consistent when comparing flexible imaging without distal chip technology with rigid examinations than it was when comparing distal chip flexible scopes with rigid imaging.41
Even if good intrarater agreement could be developed, there remains a problem in that many of the signs considered indicative of LPR are, in fact, present in an asymptomatic population and are therefore not specific enough to make a diagnosis of reflux disease. In the Hicks study, 105 subjects without voice complaints were assessed. Individuals with a diagnosis of reflux disease were excluded from the study. Three evaluators blindly rated signs of reflux disease on videostroboscopy studies of the subject population. At least one tissue irritation finding was present on 86% of the asymptomatic study population. The most common findings were interarytenoid bar (70%), arytenoid medial wall erythema (29%), posterior pharyngeal wall cobblestoning (21%), interarytenoid bar erythema (15%), arytenoid medial wall granularity (13%), posterior cricoid wall edema (10%), arytenoid apex erythema (10%), and true vocal fold edema (10%). The findings of laryngeal irritation could not be correlated with a history of smoking or drinking alcohol or a diagnosis of asthma, thus excluding the possibility of secondary asymptomatic reflux as the reason for the examination findings.40
In another study from the same institution, the results of flexible fiberoptic examination of the larynx were compared with rigid videostroboscopy with respect to laryngeal inflammation signs in another group of asymptomatic volunteers. Unlike the study by Eller et al., this study determined that flexible laryngoscopy was more likely than rigid endoscopy to identify signs of laryngeal inflammation. The most common signs of laryngeal inflammation in this asymptomatic population were found to be abnormalities of the arytenoid complex such as erythema and edema of the arytenoid medial walls and apex (76%). Pseudosulcus, consistent with vocal fold edema, was found in 37% of the study population on flexible endoscopy.13,41
Despite the fact the laryngeal irritation signs are common in an asymptomatic population and there are significant reliability issues with the rating of these signs, many clinicians continue to believe that the improved image resolution of videostroboscopy makes it useful in the detection of subtle findings of chronic laryngeal irritation and that it can be used in the diagnosis of LPR disease. In a study at Baylor College of Medicine, 49 patients diagnosed with LPR disease by the combination of symptoms and physical examination findings were compared with 10 control subjects without a diagnosis of LPR disease. Photographs of the larynx from the LPR group were evaluated prior to treatment and again after 6 weeks of proton pump inhibitor therapy by three examiners. A rating scale was developed to determine a single severity value for the combined laryngeal irritation findings. Scores given to patients prior to treatment were significantly higher for irritation than those given to controls or to patients after treatment. Scores were based on a combination of ratings given for the following parameters: edema of the posterior supraglottis, edema of the vocal folds, edema of the subglottis, erythema of the posterior supraglottis, erythema of the vocal folds, and erythema of the subglottis. Findings that did not correlate with improvement after treatment include leukoplakia, nodules or prenodules, polyps, posterior pachydermia, web, or contact granuloma. Examiners did not tend to agree, however, on absolute scoring of each region of the larynx. Despite the fact that use of a composite scoring system was able to distinguish general improvement in laryngeal inflammation, overall intrarater agreement was only moderate for this study. Only one rater was used to evaluate interrater agreement, which was found to be poor.42
At the time of the Baylor study, the idea of using a composite scoring system to rate laryngeal irritation signs in an attempt to improve our ability to identify patients with reflux disease and obviate the need for a pH study was not new. Belafsky et al. developed a scoring system (reflux finding score; RFS) using eight laryngeal signs thought to be associated with LPR and documented a difference in the scores from 40 patients with LPR disease confirmed by pH study compared with those from 40 normal controls. This study confirmed that patients with LPR disease have signs of laryngeal irritation (95% sensitivity); it does not, however, help to differentiate those individuals with laryngeal irritation due to reflux disease from those with laryngeal irritation due to other causes or as a normal variant because such patients were not included in the study.43 In an independent evaluation of the RFS, Park et al. found that the RFS was positive in 80% of individuals with LPR disease confirmed by pH study but was negative in only 37.5% of patients demonstrating signs of laryngeal irritation but without LPR disease on pH study (sensitivity of 80.7% and specificity of 37.5%).44
In a more recent study using esophageal biopsy to establish the diagnosis of reflux disease, Pribuisiene et al. evaluated the sensitivity and specificity of their scoring system, the laryngoscopic reflux index (LRI). The study included 108 subjects. The authors excluded 19 patients from the study who presented with laryngeal inflammation but had negative esophageal biopsies, which is unfortunate because it invalidates their specificity calculations. This flaw in the study underscores the confusion regarding the usefulness of laryngeal signs in making the diagnosis of reflux disease and the importance of carefully evaluating each study that makes such claims. As in the study by Belafsky et al., the exclusion of patients with laryngeal reflux signs but no documented reflux by biopsy prevents an assessment of how often a false-positive diagnosis would be made based on the LRI alone. The authors compared the LRI from the biopsy-proven LPR subjects with those from 90 healthy controls and found a sensitivity of 96% and a specificity of 97% (falsely elevated due to patient selection factors) for the index. Although the previous studies found good interrater reliability for vocal fold erythema and edema, this study found it to be the least sensitive indicator of reflux disease.43,45 This result is not surprising given that Hicks et al. and Qadeer et al. reported a 10% and 37% incidence of vocal fold edema in an asymptomatic population, respectively.40,46
It must be kept in mind that in many of the studies described, pH probe evaluations were not performed on the study population so the symptoms and laryngeal imaging findings could not be confirmed to correlate with measured acid exposure in the hypopharynx. Those studies that do correlate laryngeal signs with the results of a pH test demonstrate a poor correlation. The study by Park et al. demonstrated a 20% false-negative rate and an 63% false-positive rate in the use of laryngeal scoring compared with pH studies to determine if reflux is the etiology of patient complaints.44 Other studies have found an even lesser degree of correlation between laryngeal findings and pH probe results. In a study of 42 patients with symptoms that were believed to be consistent with reflux disease, Noordzij et al. found only 69% to have LPR on dual-sensor pH probe study. There was no correlation between symptom severity and incidence of LPR. Signs of reflux disease were not significantly more severe in the patients with documented LPR. In fact, arytenoid edema was rated significantly worse in the group without documented LPR. None of the LPR signs correlated with LPR severity on pH study.47 In the best-designed study to date, Pawar et al. conducted a prospective, randomized, placebo-controlled study of 76 patients with complaints of “post nasal drip” (considered to be a symptom of LPR disease) without evidence of nasal disease. All patients were tested with a dual-channel 24-hour pH study. The RFS was used to document laryngeal findings. Patients were then treated with either a twice-daily proton pump inhibitor or a twice-daily placebo for 3 months. The study evaluated the response of patient symptoms and RFS depending on the results of the treatment. At the outset of the study, the RFS failed to differentiate those with reflux documented on pH study (56%) from those without. Patients with and without documented reflux had a statistically significant improvement in their symptoms if treated with a proton pump inhibitor, possibly indicative of a significant placebo effect. Paradoxically, the RFS increased in the proton pump inhibitor group and decreased in the placebo group.48
Despite the difficulties in using laryngeal signs to make an accurate diagnosis of LPR disease, once a diagnosis of LPR disease has been made using a pH study, it may be possible to follow the response to therapy using an evaluation of laryngeal inflammation signs. Building on the results of the Baylor study, which documented improvement of laryngeal inflammation signs with the antireflux treatment, Qadeer et al. blindly evaluated the laryngeal signs on flexible laryngoscopy in 10 patients before and 1 year after successful fundoplication for the treatment of reflux disease. These patients had surgery because they did not improve on twice-daily proton pump inhibitor therapy. The most common preoperative findings in these patients included medial arytenoid wall erythema and edema (60%) and interarytenoid erythema (50%). Five of the patients had resolution of laryngeal irritation findings 6 months after the surgery, and another 3 patients had resolution of laryngeal irritation signs at 12 months after the surgery. The last two patients, who were additionally diagnosed with asthma and allergies, had improvement but not complete resolution of the laryngeal signs over the course of the study. Interestingly, the symptoms of sore or burning throat, hoarseness, and cough did not improve after surgery.40,42,46
In summary, although it is a common practice, the use of findings on laryngeal imaging must be done with caution to make the diagnosis of LPR disease. Even Belafsky indicated that although the irritation due to LPR usually occurs on the posterior larynx, this area makes up <5% of the entire larynx and stressed the importance of including other laryngeal irritants in the differential diagnosis in patients with significant inflammatory changes of the vocal folds.49 Most of the emphasis regarding the signs of LPR on laryngeal imaging has been in the assessment of static signs rather than the impact of LPR on vocal fold vibratory characteristics. The degree of true vocal fold edema caused by LPR in individual cases will dictate the degree of abnormality seen on the evaluation of vocal fold vibratory characteristics. Vocal fold vibratory abnormalities caused by LPR will be consistent with the changes due to vocal fold inflammation from any cause. In a recent study, it was documented that pathologic reflux could lead to edema and hypertrophy of the laryngeal mucosa. These changes in turn interfered with phonation patterns. The most common finding was incomplete glottic closure. Other stroboscopic findings were irregularity of vocal fold vibration, asymmetry of vibration, and a reduced mucosal wave.45
 Conclusion
Conclusion
Laryngeal inflammation changes the composition and shape of the vibratory structures with a direct effect on vocal quality. Videostroboscopy and laryngeal image analysis are necessary to help in voice analysis and to provide in-depth information about the status of the vocal folds during periods of inflammation and chronic irritation. Stroboscopy has proved to be the best method to study the effects of inflammation and reflux on the vibratory patterns of the vocal folds and to monitor treatment. However, it is a valuable tool only in the presence of a thorough vocal history and physical examination.
References
1. Powitzky E. Extraesophageal reflux: the role in laryngeal disease. Curr Opin Otolaryngol Head Neck Surg 2002;10:485–491
2. Titze IR. On the mechanics of vocal-fold vibration. J Acoust Soc Am 1976;60:1366–1380
3. Behrman A. Global and local dimensions of vocal dynamics. J Acoust Soc Am 1999;105:432–443
4. Remacle M, Lawson G. Diagnosis and management of laryngopharyngeal reflux disease. Curr Opin Otolaryngol Head Neck Surg 2006;14:143–149
5. Mirza N, Kasper Schwartz S, Antin-Ozerkis D. Laryngeal findings in users of combination corticosteroid and bronchodilator therapy. Laryngoscope 2004;114:1566–1569
6. Ng ML, Gilbert HR, Lerman JW. Some aerodynamic and acoustic characteristics of acute laryngitis. J Voice 1997;11:356–363
7. Thompson L. Herpes simplex virus laryngitis. Ear Nose Throat J 2006;85:304
8. Jackson-Menaldi CA, Dzul AI, Holland RW. Allergies and vocal fold edema: a preliminary report. J Voice 1999;13:113–122
9. Spiegel JR, Hawkshaw M, Markiewicz A, Sataloff RT. Acute laryngitis. Ear Nose Throat J 2000;79:488
10. Branski RC, Verdolini K, Sandulache V, Rosen CA, Hebda PA. Vocal fold wound healing: a review for clinicians. J Voice 2006;20:432–442
11. Jiang J, Lin E, Hanson DG. Vocal fold physiology. Otolaryngol Clin North Am 2000;33:699–718
12. Ford CN, Inagi K, Khidr A, Bless DM, Gilchrist KW. Sulcus vocalis: a rational analytical approach to diagnosis and management. Ann Otol Rhinol Laryngol 1996;105:189–200
13. Milstein CF, Charbel S, Hicks DM, Abelson TI, Richter JE, Vaezi MF. Prevalence of laryngeal irritation signs associated with reflux in asymptomatic volunteers: impact of endoscopic technique (rigid vs. flexible laryngoscope). Laryngoscope 2005;115:2256–2261
14. Alberti PW. The diagnostic role of laryngeal stroboscopy. Otolaryngol Clin North Am 1978;11:347–354
15. Bless DM, Hirano M, Feder RJ. Videostroboscopic evaluation of the larynx. Ear Nose Throat J 1987;66: 289–296
16. Cantarella G. Value of flexible videolaryngoscopy in the study of laryngeal morphology and functions. J Voice 1987;1:353–358
17. Postma GN, Koufman JA. Laryngitis. In: Bailey BJ, ed. Head and Neck Surgery—Otolaryngology, 2nd ed. Philadelphia, PA: Lippincott-Raven; 1998:731–739
18. Lim JY, Choi JN, Kim KM, Choi HS. Voice analysis of patients with diverse types of Reinke’s edema and clinical use of electroglottographic measurements. Acta Otolaryngol 2006;126:62–69
19. Bouzid A, Ellouze N. Open quotient measurements based on multiscale product of speech signal wavelet transform. Research Letters in Signal Processing 2007; 62521:10.1155/2007/6252
20. Schutte HK, Svec JG, Sram F. First results of clinical application of videokymography. Laryngoscope 1998;108(8 Pt 1):1206–1210
21. Schalén L. Acute laryngitis in adults: diagnosis, etiology, treatment. Acta Otolaryngol Suppl 1988;449:31
22. Colton RH, Woo P, Brewer DW, Griffin B, Casper J. Stroboscopic signs associated with benign lesions of the vocal folds.J Voice 1995;9:312–325
23. Mergell P, Herzel H, Titze IR. Irregular vocal-fold vibration—high-speed observation and modeling. J Acoust Soc Am 2000;108:2996–3002
24. Tigges M, Mergell P, Herzel H, Wittenberg T, Eysholdt U. Observation and modeling glottal biphonation. Acustica/Acta Acustica 1997;83:707–714
25. Neubauer J, Mergell P, Eysholdt U, Herzel H. Spatiotemporal analysis of irregular vocal fold oscillations: biphonation due to desynchronization of spatial modes. J Acoust Soc Am 2001;110:3179–3192
26. Agarwal P, Bais AS. A clinical and videostroboscopic evaluation of laryngeal tuberculosis. J Laryngol Otol 1998;112:45–48
27. Altman KW. Vocal fold masses. Otolaryngol Clin North Am 2007;40:1091–1108, viii
28. Hirano S. Current treatment of vocal fold scarring. Curr Opin Otolaryngol Head Neck Surg 2005;13:143–147
29. Lee ST, Niimi S. Vocal fold sulcus. J Laryngol Otol 1990;104:876–878
30. Kendall KA. Controversies in the diagnosis and management of laryngopharyngeal reflux disease. Curr Opin Otolaryngol Head Neck Surg 2006;14:113–115
31. Bove MJ, Rosen C. Diagnosis and management of laryngopharyngeal reflux disease. Curr Opin Oto laryngol Head Neck Surg 2006;14:116–123
32. Divi V, Benninger MS. Diagnosis and management of laryngopharyngeal reflux disease. Curr Opin Otolaryngol Head Neck Surg 2006;14:124–127
33. Galli J, Cammarota G, De Corso E, et al. Biliary laryngopharyngeal reflux: a new pathological entity. Curr Opin Otolaryngol Head Neck Surg 2006;14:128–132
34. Mahieu HF, Smit CF. Diagnosis and management of laryngopharyngeal reflux disease. Curr Opin Otolaryngol Head Neck Surg 2006;14:133–137
35. Pontes P, Tiago R. Diagnosis and management of laryngopharyngeal reflux disease. Curr Opin Otolaryngol Head Neck Surg 2006;14:138–142
36. Remacle M, Lawson G. Diagnosis and management of laryngopharyngeal reflux disease. Curr Opin Otolaryngol Head Neck Surg 2006;14:143–149
37. Katz PO. Gastroesophageal reflux disease—state of the art. Rev Gastroenterol Disord 2001;1:128–138
38. Modlin IM, Moss SF, Kidd M, Lye KD. Gastroesophageal reflux disease: then and now. J Clin Gastroenterol 2004;38:390–402
39. Branski RC, Bhattacharyya N, Shapiro J. The reliability of the assessment of endoscopic laryngeal findings associated with laryngopharyngeal reflux disease. Laryngoscope 2002;112:1019–1024
40. Hicks DM, Ours TM, Abelson TI, Vaezi MF, Richter JE. The prevalence of hypopharynx findings associated with gastroesophageal reflux in normal volunteers. J Voice 2002;16:564–579
41. Eller R, Ginsburg M, Lurie D, Heman-Ackah Y, Lyons K, Sataloff R. Flexible laryngoscopy: a comparison of fiber optic and distal chip technologies—part 2: laryngopharyngeal reflux. J Voice 2009;23:389–395
42. Beaver MES, Stasney CR, Weitzel E, et al. Diagnosis of laryngopharyngeal reflux disease with digital imaging. Otolaryngol Head Neck Surg 2003;128:103–108
43. Belafsky PC, Postma GN, Koufman JA. The validity and reliability of the reflux finding score (RFS). Laryngoscope 2001;111:1313–1317
44. Park KH, Choi SM, Kwon SUK, Yoon SW, Kim SU. Diagnosis of laryngopharyngeal reflux among globus patients. Otolaryngol Head Neck Surg 2006;134:81–85
45. Pribuisiene R, Uloza V, Kupcinskas L. Diagnostic sensitivity and specificity of laryngoscopic signs of reflux laryngitis. Medicina (Kaunas) 2008;44:280–287
46. Qadeer MA, Swoger J, Milstein C, et al. Correlation between symptoms and laryngeal signs in laryngopharyngeal reflux. Laryngoscope 2005;115:1947–1952
47. Noordzij JP, Khidr A, Desper E, Meek RB, Reibel JF, Levine PA. Correlation of pH probe-measured laryngopharyngeal reflux with symptoms and signs of reflux laryngitis. Laryngoscope 2002;112:2192–2195
48. Pawar S, Lim HJ, Gill M, et al. Treatment of postnasal drip with proton pump inhibitors: a prospective, randomized, placebo-controlled study. Am J Rhinol 2007;21:695–701
49. Belafsky PC, Postma GN, Amin MR, Koufman JA. Symptoms and findings of laryngopharyngeal reflux. Ear Nose Throat J 2002;81(9, Suppl 2)10–13
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


