CHAPTER 56 Laryngeal and Pharyngeal Function
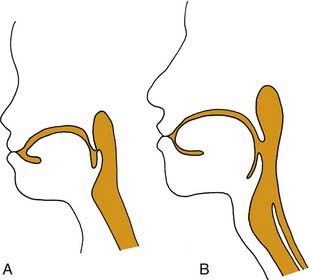
The upper aerodigestive tract serves the competing functions of respiration and swallowing. The nose is the primary respiratory orifice, and the mouth is the portal for ingestion of food. Both open into a common cavity, the pharynx. The patency of the upper airway must be actively supported during breathing, yet total and forceful collapse is required to propel food into the esophagus during swallowing. The airway must be protected during a swallow so that ingested food or water does not spill into the trachea. Aspiration of food or foreign material can lead to serious consequences, such as asphyxia or lung infection. In humans, the function of the upper aerodigestive is considerably more complex, owing to the demands of speech as well as to a significant structural difference. In infants and in all nonhuman mammals, the pharynx is functionally compartmentalized into separate passages for breathing and alimentation. The epiglottis interdigitates with the uvula to form a respiratory channel from the nose into the larynx and two lateral pathways from the mouth to the esophagus through the pyriform sinuses.1 During postnatal development in humans, enlargement of the cranium with flexion of the base of the skull results in a downward displacement of the larynx. This displacement elongates the pharynx and distracts the uvula and epiglottis so that they are no longer in contact. The result is a common pharyngeal cavity for breathing and swallowing (Fig. 56-1).2,3 The larynx begins its descent at the age of about 18 to 24 months. There are two positive outcomes. Vocal power is greater because of increased resonance, and articulatory diversity is expanded.4
This complicated and potentially hazardous configuration of the upper airway results from embryology and reflects evolution. The lower respiratory tract has evolved as an offshoot of the digestive tract, first appearing in the lungfish as a simple muscle sphincter to protect the lungs from water.5 Consequently, during embryologic development, the foregut is the common origin of the larynx, trachea, and esophagus.
Laryngeal Motion
Applied Anatomy
In the illustrations of many textbooks, the membranous vocal folds are depicted as moving solely in the axial plane, with rotational motion similar to that of a windshield washer. Details of motion of the posterior, cartilaginous portions of the larynx have been largely ignored. The reason is that early concepts of motion were based on observing laryngeal motion with a mirror, and then recording the observations with two-dimensional freehand drawings. However, with the advent of flexible endoscopy, stroboscopy, videorecording, and computerized imaging, it has become clear that laryngeal motion is more complex than previously recognized. Vocal folds move in three dimensions and undergo conformational changes in length, shape, and volume (Fig. 56-2).6 The terms cadaveric and paramedian have been commonly used to describe the position of paralyzed vocal folds. These terms are inadequate to describe the three-dimensional changes in configuration of the glottis in paralysis.7 Motion of the larynx is best understood as the net result of the interaction of its component parts.
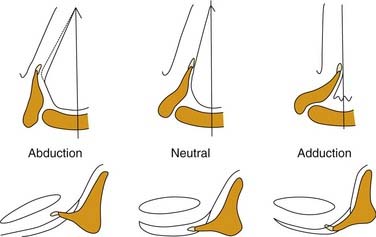
Figure 56-2. Three-dimensional motion of the arytenoid cartilage and vocal fold.
(From Hirano M. Anatomy and behavior of the vocal process. In: Baer T, Sasaki C, Harris K, eds. Laryngeal Function in Phonation and Respiration. Boston: College-Hill Press; 1987.)
The laryngeal skeleton consists of the hyoid bone and a series of cartilages. The hyoid bone is a U-shaped structure, open posteriorly, which is suspended from the base of the skull and mandible by muscles and ligaments. The thyroid cartilage, the largest cartilage in the larynx, is suspended from the hyoid bone. The word thyroid means “shield” and this name is appropriate, because the structure is not only shaped like a shield but also provides support and protection for the vocal folds. In the axial plane, the thyroid cartilage is shaped like a V, with two wings that project posteriorly. Like the hyoid bone, the thyroid cartilage is open posteriorly. The vocal folds attach to the anterior inner surface of the thyroid cartilage. The posterior ends of the vocal folds are anchored to the arytenoid cartilages, which are the chief moving parts of the larynx. The arytenoid cartilages sit atop the posterior rim of the cricoid cartilage and articulate via shallow ball-and-socket joints. The cricoid cartilage is the only complete rigid ring within the airway. It is shaped like a signet ring and is broadest posteriorly. Inferiorly, and just lateral to the cricoid joints, the inferior cornua of the thyroid cartilage articulates with the cricoid in two hingelike joints, creating a visor-like, or “bucket handle” structure, with motion that controls the space between the anterior rims of the thyroid and cricoid cartilages.8
Most intrinsic laryngeal muscles connect the arytenoid cartilages to either the cricoid or thyroid cartilages. These include the thyroarytenoid muscle, the lateral cricothyroid muscle, the interarytenoid muscle, which act to adduct the vocal folds, and the posterior cricoarytenoid muscle, which is the only active abductor of the larynx. As mentioned above, the arytenoid cartilages are the chief moving parts of the larynx. The membranous vocal folds are suspended between the thyroid cartilage and the arytenoids, and the position of each membranous vocal fold is determined by the motion of the arytenoid cartilage. The arytenoid rotates upward and outward to open the larynx, and rotates inward to close it. Contraction of the posterior cricoarytenoid muscle pulls the muscular process of the arytenoid posteriorly and caudally. The structure of the cricoarytenoid joint prevents the entire arytenoid from being pulled along this vector. Instead, the arytenoid rotates, displacing the vocal process upward and laterally, abducting the vocal fold (Fig. 56-3).9 Conversely, the lateral cricoarytenoid muscle pulls the muscular process of the arytenoid anteriorly and caudally, which rotates the arytenoid, moving the vocal process medially and adducting the vocal fold. The interarytenoid muscles pull the arytenoid cartilages together. The thyroarytenoid muscles exert some adductor force, but their major action is to shorten or tense the vocal fold and increase its cross-sectional area (Fig. 56-4).
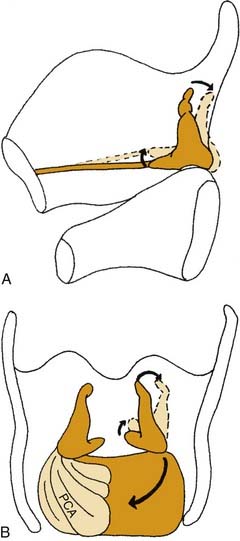
Figure 56-3. Three-dimensional effects of posterior cricoarytenoid muscle contraction. A, Sagittal view. B, Posterior view.
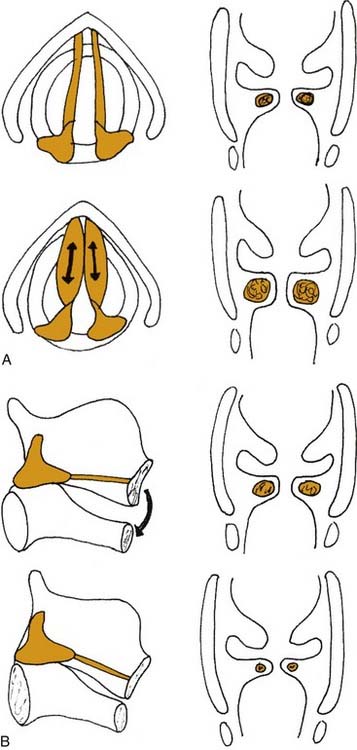
Figure 56-4. Effects of (A) thyroarytenoid and (B) cricothyroid muscle contraction on the thickness of the vocal fold.
In addition to the diverse force vectors of individual laryngeal muscles, there is also segmental compartmentalization within muscles, increasing the possibilities for fine control. For example, the human posterior cricoarytenoid muscle is divided into two compartments. These are supplied by separate nerve branches, differ in fiber type, and insert on opposing sides of the muscular process (Fig. 56-5).9,10,11 The human thyroarytenoid muscle is also compartmentalized: It has long been regarded to have a separate medial compartment, the “vocalis.”
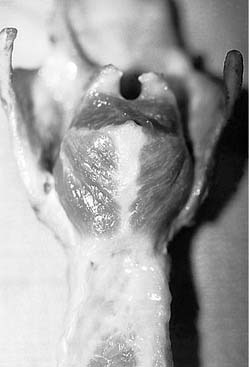
Figure 56-5. Compartmentalization of the human posterior cricoarytenoid muscle. Posterior view of a cadaver larynx.
In contrast to other intrinsic laryngeal muscles, the cricothyroid muscle does not insert on the arytenoid cartilage and therefore has no direct action on arytenoid motion. Instead, the cricothyroid muscle connects the anterior edges of the thyroid and cricoid cartilages. Contraction of this muscle pulls the two cartilages closer together, which increases the distance between the anterior commissure and the cricoid. The result is stretching of the vocal fold, increasing its length and tension. Because both vocal folds insert on the anterior commissure, contraction of either cricothyroid muscle affects both ipsilateral and contralateral vocal folds. Contraction of both right and left cricothyroid muscles results in maximal anterior traction.8
Muscles that are extrinsic to, but connect with, the larynx can also affect glottic function by exerting traction on the laryngeal cartilages. The sternohyoid, thyrohyoid, and omohyoid muscles, innervated by the ansa cervicalis, exert caudal traction on the larynx. This action, as well as downward traction on the trachea during inspiration, causes abduction of the vocal folds.12,13 Muscles that exert a cephalad force include the geniohyoid, anterior belly of the digastric, mylohyoid, and stylohyoid muscles. In patients with hyperfunctional dysphonia, excess activity can usually be palpated in the extrinsic laryngeal muscles.
Laryngeal Function in Breathing
The primary and most primitive function of the larynx is to protect the lower airway. In evolution, the larynx first appeared as a sphincter to prevent the ingress of water into the airway of the lungfish.5 Subsequently, dilator muscles evolved to permit active opening of the larynx. In more evolved animals, the larynx is not just an open or shut valve but, rather, a variable resistor, capable of regulating airflow. Other laryngeal functions are the Valsalva maneuver and coughing. The larynx is also a sensory organ, providing information about airway function and the purity of inhaled air and serving in the afferent limb of many reflexes.
Protection
When the larynx is mechanically stimulated, it closes abruptly, and respiration ceases. Apnea can also occur in response to such diverse chemical agents as ammonia, phenyl diguanide, and cigarette smoke. These are appropriate and beneficial responses that prevent the entry of foreign matter into the lower airway, although strong laryngeal stimulation may result in responses that appear to be maladaptive, such as laryngospasm and prolonged bronchoconstriction.14 These reflexes may be produced in experimental animals by electrical stimulation of the superior laryngeal nerve and probably represent an oversaturation of pathways that serve a useful function at lower levels of input.
Recurrent paroxysmal laryngospasm is occasionally encountered in clinical practice. In some patients, it is caused by gastroesophageal reflux, which responds to acid-suppressing medication. In other patients, the pathophysiology appears to be a hypersensitive laryngeal closure reflex, because such patients report some triggering event such as eating or inhaling steam or odors. The onset frequently occurs during an upper respiratory infection, but it also can occur after surgical trauma to the recurrent laryngeal nerve. Most often the condition resolves spontaneously within a few months, but it may become a permanent and debilitating problem. The laryngeal closure reflex is particularly sensitive in infants and can be elicited by a stimulus as weak as water. During early infancy, the strength of this reflex increases, then decreases, along a time course similar to that of the incidence of sudden infant death syndrome, suggesting that laryngeal reflexes may play a role in its cause.15
Cough
Another important protective reflex involving the larynx is the cough, which ejects mucus and foreign material from the lungs.16 Cough can be a voluntary action or a reflexive response to stimulation of the larynx or receptors in the lungs. The cough reflex is suppressed during sleep, so that a greater stimulus is required with progressive stages of sleep. During deep sleep, a cough cannot be elicited unless the stimulus first results in arousal to a lighter level of sleep.
Control of Ventilation
The role of the larynx as an active organ of respiration is not widely recognized. Abduction and adduction of the larynx in phase with respiration has been acknowledged for many years.17,18 It has been postulated that all respiratory motion of the larynx is passive, resulting from biomechanical coupling of the larynx to the tracheobronchial tree.12,19 There is some evidence that downward traction on the larynx dilates the glottis.13
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree