
 lindness due to corneal disease is usually difficult or impossible to treat. Results of penetrating keratoplasty may be acceptable for classical indications, but in complicated situations such as profusely vascularized corneal opacities, severe ocular surface disorders, or pseudocornea, penetrating keratoplasty is either not indicated or not possible. In such cases, the only possible solution is either a central keratoprosthesis or a paralimbal scleral prosthesis. If the host cornea appears strong enough, a keratoprosthesis is implanted in the center of the corneal tissue. If the health of the host cornea appears compromised (i.e., the cornea is too thin or pseudocornea or anterior staphyloma is present), the same device is fixed to the nasal paralimbal sclera. We call this the “paralimbal scleral window,” and it was first performed by Daljit Singh in 1982.
lindness due to corneal disease is usually difficult or impossible to treat. Results of penetrating keratoplasty may be acceptable for classical indications, but in complicated situations such as profusely vascularized corneal opacities, severe ocular surface disorders, or pseudocornea, penetrating keratoplasty is either not indicated or not possible. In such cases, the only possible solution is either a central keratoprosthesis or a paralimbal scleral prosthesis. If the host cornea appears strong enough, a keratoprosthesis is implanted in the center of the corneal tissue. If the health of the host cornea appears compromised (i.e., the cornea is too thin or pseudocornea or anterior staphyloma is present), the same device is fixed to the nasal paralimbal sclera. We call this the “paralimbal scleral window,” and it was first performed by Daljit Singh in 1982.
The keratoprosthesis that we use needs some explanation (Fig. 44.1).
The Singh–Worst Keratoprosthesis
The Singh–Worst keratoprosthesis is a one-piece polycarbonate device. The anterior surface of the device is convex, with a diameter of 6.0 mm, and has a flange. The flange merges into the shaft of the keratoprosthesis, which flares out posteriorly, thus forming a “neck” at the junction of the anterior surface and the body of the prosthesis. The end of the shaft has a diameter of 4.5 mm. The diameter at the level of the neck is 3.0 mm. The flange has eight equidistant holes near the margin. Four double-armed 80-µm stainless steel sutures are passed through these holes. This prosthesis is designed as a reverse cone that provides significantly better visibility for the fundus and excellent fixation, with less risk of extrusion (Fig. 44.2).
Fixation Concept of the Singh–Worst Keratoprosthesis
The basic idea behind the Singh–Worst design is to make the prosthesis absolutely immobile and minimize any micromovements between the prosthesis and the corneal or scleral bed. This is accomplished by using the dual-fixation principle. The prosthesis is fixed to the local host tissue around the waist as well to the sclera near the equator through the eight 80-µm steel sutures. The local fixation makes the opening watertight by tightening the host tissue around the neck of the keratoprosthesis, and the distant scleral fixation gives further stability, thus making it less vulnerable to extrusion. The stability of this prosthesis results from an equilibrium between the outward push of the intraocular pressure and the inward push by the distant sutures. The more the intra-ocular pressure tries to push the prosthesis out, the more snugly the anticonical shaft fits. Because of this dual-fixation principle, this is also known as the “champagne cork” keratoprosthesis. The fitting is like the cork in the champagne bottle, fitted tightly, with an extra harness of wires in place to secure it firmly. The tight but gentle local fixation keeps the host cornea from melting and prevents infectious agents from getting into the eye.
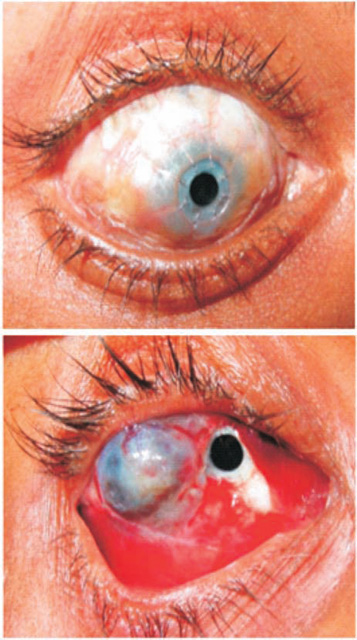
Figure 44.1. Central (top) and paralimbal (bottom) keratoprosthesis.
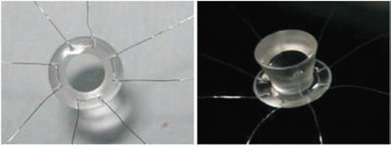
Figure 44.2. The Singh–Worst keratoprosthesis.
Indications for Surgery
All patients with corneal blindness, who are either outright unfit for keratoplasty or who have had multiple failed keratoplasty procedures, qualify for keratoprosthesis surgery. Patients with healthy corneal tissue are suitable for central keratoprosthesis, whereas a paralimbal scleral prosthesis is better suited for patients with thin corneal tissue or with pseudocornea. Common conditions include Stevens– Johnson syndrome, chemical burns, thermal burns, vascularized cornea following corneal ulcer, dry eye syndrome, and rheumatoid arthritis.
Preoperative Evaluation
The prognosis for keratoprosthesis procedures depends on the preoperative diagnosis. Keratoprostheses implanted in patients with ocular surface disorder (e.g., severe xerosis) tend to fare poorly as compared with patients with postcorneal ulcer scarring. Patients should be screened for good perception and projection of light, a normal posterior segment on B-scan ultrasonography, and normal intra-ocular pressure, checked digitally. If intraocular pressure is elevated, a filtration procedure is performed in the first stage to normalize pressure. A history of cataract surgery is important because in central keratoprosthesis, the crystalline lens or the intra-ocular lens must be removed. The patient and his or her family should be counseled regarding the possible outcome of the surgery and that prolonged postoperative care will be required.
Surgical Technique for Paralimbal Scleral Prosthesis
- Perform conjunctival peritomy and sling the rectus muscles. Before proceeding further, estimate intra-ocular pressure by indenting the cornea with the tip of a needle holder. Evaluate corneal thickness and health. Identify any previously unnoticed or hidden scleral staphylomata (Fig. 44.3).
- Spread the keratoprosthesis preloaded with sutures over the exposed eyeball. Do the preloading on a small eye drape so that steel sutures can be lifted one by one and anchored systematically in the sclera.
- Implant the paralimbal window to the nasal paralimbal sclera and perform a free tenotomy of the medial rectus muscle to direct the prosthesis forward.
- Pass all eight 80-µm steel sutures through the scleral tissue equidistant from each other, and keep the intended prosthesis implantation point at the center, at or near the equator of the globe, taking deep bites, with the sutures put in over half-sclera deep. Pass some sutures over the cornea as well. Once the scleral sutures are in place, push the keratoprosthesis aside slightly and use a 2.0-mm dermal biopsy trephine to make a mark on the paralimbal sclera.
- Use the Fugo blade to slowly ablate the sclera and the underlying ciliary body, layer by layer until you reach the vitreous. This procedure is totally bloodless. In our case, earlier we used a trephine to make the initial opening (Fig. 44.4).
- Make a full-thickness relaxing incision with the Fugo blade, parallel to the limbus in the opening thus created (Fig. 44.5). (Traditionally this has been done with Castroviejo scissors.)
- Also perform an anterior vitrectomy to reduce the chances of a retroprosthetic membrane. Use the Fugo blade to do a vitrectomy.
- Pass a preplaced 80-µm steel suture across the relaxing incision.
- Slip the posterior body of the keratoprosthesis through the opening and tighten the steel suture sufficiently to grip the prosthesis (Fig. 44.6). If need be, another suture may also be placed.
- Once you have achieved local immobilization, pull the eight 80-µm sutures together and tie adjacent sutures together with square knots to avoid slippage. Form a total of four knots. The sutures should be just tight enough to give a slightly puffed appearance to the eyeball.
- Inject fluid into the eyeball to make it just firm enough to keep the wound sealed and to buffer early postoperative intra-ocular pressure spikes.
- Suture the conjunctiva back in place.
- Pass all eight 80-µm steel sutures through the scleral tissue equidistant from each other, and keep the intended prosthesis implantation point at the center, at or near the equator of the globe, taking deep bites, with the sutures put in over half-sclera deep. Pass some sutures over the cornea as well. Once the scleral sutures are in place, push the keratoprosthesis aside slightly and use a 2.0-mm dermal biopsy trephine to make a mark on the paralimbal sclera.
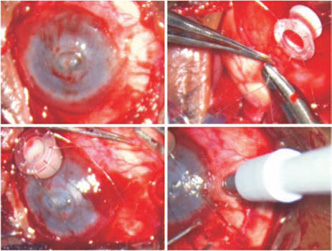
Figure 44.3. Peritomy is done, and 80-µm stainless steel sutures are passed through the sclera. A 2-mm marker is pressed on the site of paralimbal prosthesis.
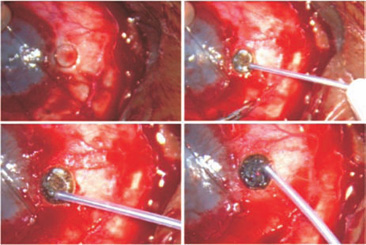
Figure 44.4. Fugo blade ablation of the marked area removes the sclera and starts ablation of the exposed ciliary body.
In patients with slightly weak corneal tissue or dry keratinized conjunctiva, a buccal mucosal graft is done around the keratoprosthesis. The graft will provide additional strength to the corneal tissue and an extra source of much-needed lubrication. Sometimes, if the anterior bulbar tissues are very weak to begin with, only a buccal mucosal transplant is done in the first stage, followed by keratoprosthesis surgery several weeks or months later.
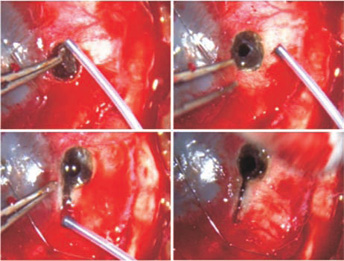
Figure 44.5. Fugo blade ablation of the ciliary body creates a hole in it. A linear relaxing incision is made in the decompressed soft eye.
When implanting a central keratoprosthesis, the basic technique remains the same except that the opening is made in the center of the corneal tissue and the crystalline/cataractous lens must be removed before implanting the prosthesis.
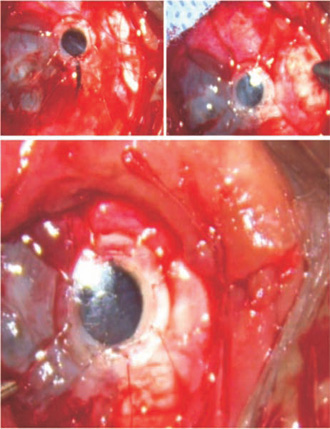
Figure 44.6. The keratoprosthesis is slipped inside. The relaxing incision is tied. The scleral sutures are tightened and tied. The conjunctiva is pulled back. The final image shows the condition of the eye at the end of the surgery.
Postoperative Management
Vision results after surgery are most surprising. Patients who undergo paralimbal scleral prosthesis surgery and have a good outcome can do nearly as well as the central-prosthesis group. They can walk around unaided and perform near-vision tasks with good dexterity. Vision does improve with the help of prescription glasses. In our series, most patients required glasses around +7.0 D spherical, which also works as a low-vision aid.
Postoperative Complications
Immediate Complications
It is not uncommon for acute glaucoma to develop in the early postoperative period, but it usually subsides in 1 to 2 weeks. The severity of postoperative inflammation varies from patient to patient. Any bleeding behind the prosthesis usually clears up in a few days.
Late Complications
Late complications after surgery can usually be handled successfully.
Glaucoma may continue perpetually after surgery in some patients. Intra-ocular pressure can only be tested digitally. The optic disc should be examined periodically whenever possible. These patients may need to continue on topical and oral antiglaucoma agents for a long time. If glaucoma is uncontrolled, transciliary filtration surgery can be performed to manage it.
Inflammatory and pigment deposits and thin retroprosthetic membrane can be cut using the yttrium– aluminum–garnet (YAG) laser; however, because of the length of the body of the device, a lot of pitting occurs. A thick retroprosthetic membrane has to be cut from the back using a 26-gauge needle. Thick vascularized membranes are cut with the Fugo blade. Most of the retroprosthetic membranes are inflammatory in nature. A course of antiinflammatory drugs usually clears them up.
Retinal detachment, if detected, can be treated by regular vitreoretinal procedures using a no-touch wide-angle viewing system.
The Singh–Worst prosthesis never extrudes completely because of the nature of its unique fixation. A loose local fixation can be reinforced with an 80-µm suture that regrips the prosthesis. In the 80-µm sutures loosen, the harness suture can be tightened by twisting the knot until it becomes tight again. If the harness suture becomes extremely loose, a new 80-µm suture can be used to stabilize it.
Endophthalmitis should be treated as with any normal patient.
Summary
Keratoprosthesis implantation surgery is a challenging surgery, in which no two eyes being operated on are the same. Keratoprosthesis presents wonderful opportunities for the prospect of restoring sight to corneal-blind individuals. The Fugo blade has contributed to this surgery by helping to make the surgery easier and safer.
Suggested Reading
Singh D. Keratoprosthesis. Indian J Ophthalmol. 1984;932: 405–407.
Singh I. Central and paracentral perforating keratoprosthesis—an experience of 200 cases. Refract Corneal Surg. 9:191–192.
Singh IR. Paralimbal scleral window. An Inst Barraquer (Barc). 2001;30:91–93.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


