Purpose
To compare the anesthetic effectiveness of 3 topical agents used for intravitreal injections.
Design
Randomized, triple-armed, double-blinded, prospective, single-centered trial in patients receiving intravitreal ranibizumab for neovascular age-related macular degeneration.
Methods
Patients were randomized 1:1:1 to receive 0.5% tetracaine hydrochloride drops and a 4% lidocaine pledget (n = 31), 0.5% tetracaine hydrochloride drops alone (n = 31), or 4% cocaine (+ epinephrine 1/100 000) drops alone (n = 31). Patients were asked to score their pain experience using a visual analogue scale (VAS) immediately following and 15 minutes after their injection. The average of these scores was used as the primary outcome. The physician performing the procedure separately scored his perception of the patients’ pain using the Wong-Baker FACES scale.
Results
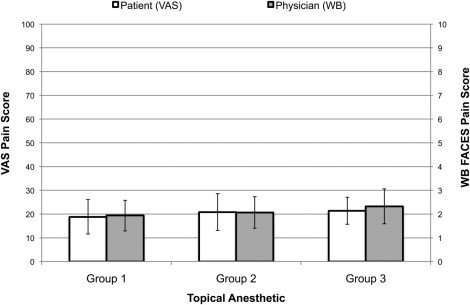
Means of the averaged VAS pain score for Groups 1, 2, and 3 were: 19 (95% confidence interval [CI] 12–26), 21 (95% CI 13–29), and 21 (95% CI 16–27) respectively. Mean Wong-Baker pain scores for Groups 1, 2, and 3 were 1.9 (95% CI 1.3–2.6), 2.1 (95% CI 1.4–2.7), and 2.3 (95% CI 1.6–3.1) respectively. There was no significant difference ( P = .549) between groups for average VAS pain score. Similarly, there was no significant difference ( P = .790) for the physician-perceived pain score between groups.
Conclusions
There was no clinical difference in patient pain experience between the 3 anesthetic options tested. The addition of a 4% lidocaine pledget offered no clinical advantage in pain relief compared to 0.5% tetracaine or 4% cocaine (+ epinephrine 1/100 000) drops alone.
Intravitreal injections represent a direct route to achieve high intraocular levels of drug into the posterior segment of the eye. This drug delivery method has enabled the successful intravitreal administration of antiviral, antibiotic, antifungal, steroidal, and, recently, anti–vascular endothelial growth factor (VEGF) drugs. With the advent of anti-VEGF injections as an effective therapy for neovascular age-related macular degeneration (AMD), intravitreal injections have the potential to become the most common intraocular procedure performed worldwide. However, to date there is no consensus for the most effective anesthetic technique or agent that should precede the injection. Patient comfort is especially important for intravitreal anti-VEGF treatments, where the patient is required to return on a regular basis for subsequent injections. If an uncomfortable procedure is experienced, the patient may refrain from this vision-saving treatment.
Aiello reviewed the available evidence and, combined with expert opinions, formulated the following guideline for topical anesthetics for intravitreal injection: 1) topical anesthetics can be applied as indicated by standard medical practice; and 2) supplemental subconjunctival (SC) anesthetic in addition to topical anesthetic can be considered. The panel of experts proceeded to offer their advice, and it seemed center-specific and physician-specific, and not based on strong evidence. Previous studies suggest that among the anesthetic techniques (including SC and peribulbar injection), topical administration is the most comfortably applied, cost-effective, and easiest to administer. However, there has yet to be a study devoted to comparing the effectiveness among different topical anesthetic agents.
This study was designed as an equivalence trial to test our hypothesis that there is no clinical difference in analgesic effectiveness between 3 common topical anesthetics: tetracaine with lidocaine pledget, tetracaine drops alone, and cocaine drops alone. The aim of this study was to provide evidence to guide existing practice as to the most appropriate topical anesthetic to use for intravitreal injections.
Patients and Methods
This prospective, randomized, triple-armed, double-blinded trial compared the anesthetic effectiveness of 1) 0.5% tetracaine hydrochloride with 4% lidocaine hydrochloride pledget, 2) 0.5% tetracaine hydrochloride, and 3) 4% cocaine (with epinephrine 1/100 000) for intravitreal injections. This study was carried out at a single center—the Ivey Eye Institute, London, Ontario, Canada—by 3 retina specialists (C.J., P.H., T.S.).
All patients greater than 50 years of age, having had at least 1 previous intravitreal injection and who were scheduled to receive intravitreal ranibizumab (Lucentis; Genentech, Inc., San Francisco, California, USA) for one eye from October 2009 to November 2009 for the treatment of neovascular AMD were evaluated for inclusion into the study. Patients were excluded if they had a history of pars plana vitrectomy, any major surgery within the prior 28 days or one planned within the next 6 months, a history of thromboembolic events, or a previous known allergic response to the topical anesthetics to be used.
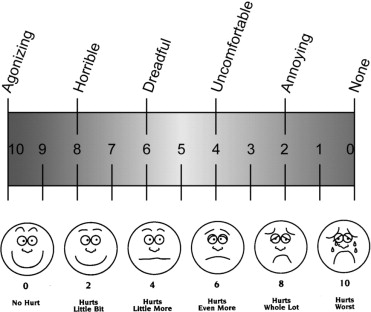
Patients approved for the study were randomized to 1 of the 3 treatment groups. The study coordinators selected a sealed envelope, arranged in sequential order, containing the treatment randomization. Physicians and patients were concealed to the allocation sequence. The order in which patients were recruited corresponded to the order with which they were scheduled in the regular clinic, and the office administration responsible for scheduling was not involved with the trial. The anesthetic was administered by 1 of 2 research assistants (A.M.P., T.J.), both having been trained in and experienced with the technique. Strictly 1 to 2 drops of topical anesthetic were applied. For Group 1, the 4% lidocaine hydrochloride pledget was applied by placing the soaked end of a Q-tip onto the injection site for 10 seconds. When complete, 1 of 3 retina specialists was invited into the treatment room, with the anesthetic concealed, and performed the intravitreal injection. Immediately following the injection, a study coordinator explained the 100-mm visual analogue scale (VAS) for pain ( Figure 1 ) and asked the subject to plot the level of pain he or she experienced specifically during the moment of injection. The VAS scale is the methodology that is most commonly used for the evaluation of pain severity and relief and has been employed in similar studies measuring ocular comfort. Those who could not adequately visualize the scale were prompted to vocalize a number from 0 to 100. Subjects were asked the same questionnaire 15 minutes later, without visualization of their first rating. A separate pain scale was employed to record physician-perceived pain. The Wong-Baker FACES scale ( Figure 1 ) was chosen primarily for its ease of use. It has been studied as an observer scale and has prior use in ophthalmic analgesia literature. The specialist recorded his perception of the subjects’ pain experience shortly after the procedure. The primary outcome of this study was the average of the 2 VAS pain scores. The secondary outcome was the physician-perceived pain score.
Sample Size Calculation
Prior studies using the VAS for pain identified that a difference of 9 to 13 (out of 100) between scores would lead to a clinically significant difference in pain experience. Thus, our sample size was calculated to detect a 13-point difference, with the level of significance set at a P value of .05 (2-sided) and a power of 80%. The standard deviation (SD) of 16 was a compromise between similar prior studies, clinical judgment, and feasibility of the sample size. A Bonferroni adjustment was made to account for the comparison of 3 means. Using the above parameters, the sample size was determined to be 31 per group, with a total of 93 subjects.
Statistical Methods
Demographic and procedural characteristics among groups were compared using descriptive statistics and univariate analysis as appropriate. One-way analysis of variance ( P < .05) was used to compare the primary and secondary outcomes among treatment groups. Log transformation was applied to normalize data. All statistical analyses were performed using the SPSS software system, version 18.0 (SPSS Inc, Chicago, Illinois, USA).
Results
Population and Procedure Characteristics
A total of 97 patients satisfied the inclusion criteria, with 93 consenting to participate in the trial. There were no deviations from protocol. All subjects allocated to a treatment group were included in the analysis. No adverse events were reported during or immediately after the intervention. The 3 specialists performing the injections were evenly distributed among the groups, as were the anesthetic operators. Baseline characteristics for each treatment group are described in Table 1 . The latency from the application of anesthetic to the injection was averaged to be 2 minutes for all 3 groups.
| Characteristic | 0.5% Tetracaine Hydrochloride With 4% Lidocaine Hydrochloride Pledget (n = 31) | 0.5% Tetracaine Hydrochloride (n = 31) | 4% Cocaine (With Epinephrine) 1/100 000 (n = 31) | P Value |
|---|---|---|---|---|
| Mean age (± SD), y | 83.6 ± 6.0 | 79.5 ± 9.9 | 82.1 ± 7.7 | .124 a |
| Mean previous intravitreal injections (± SD), y | 6.1 ± 4.6 | 5.6 ± 4.4 | 6.1 ± 6.2 | .895 a |
| % male (number) | 32% (10) | 48% (15) | 39% (12) | .430 b |
Outcomes
Means of the averaged VAS pain score for Groups 1, 2, and 3 were: 19 (95% confidence interval [CI] 12–26), 21 (95% CI 13–29), and 21 (95% CI 16–27) respectively. Mean Wong-Baker pain scores for Groups 1, 2, and 3 were 1.9 (95% CI 1.3–2.6), 2.1 (95% CI 1.4–2.7), and 2.3 (95% CI 1.6–3.1) respectively ( Figure 2 ). There was no significant difference between VAS scores at 0 and 15 minutes postinjection ( Supplemental Table , available at AJO.com ).