Purpose
To test whether comparison of the correlation between optic disc rim area and retinal nerve fiber layer thickness (rim-RNFL correlation) can differentiate eyes with nonarteritic anterior ischemic optic neuropathy (NAION) from eyes with open-angle glaucoma (OAG).
Design
Prospective cross-sectional study.
Methods
One hundred and thirteen eyes with OAG and 22 with NAION were included in this study. The rim-RNFL correlation in OAG eyes was analyzed in global and 12-clock-hour parameters using rim areas determined by Heidelberg retina tomography (HRT II) and RNFL thicknesses determined by optical coherence tomography (Cirrus OCT). The eyes with NAION were determined whether to be out of the 95% prediction interval (PI) for the rim-RNFL correlation of OAG in global and clock-hour parameters.
Results
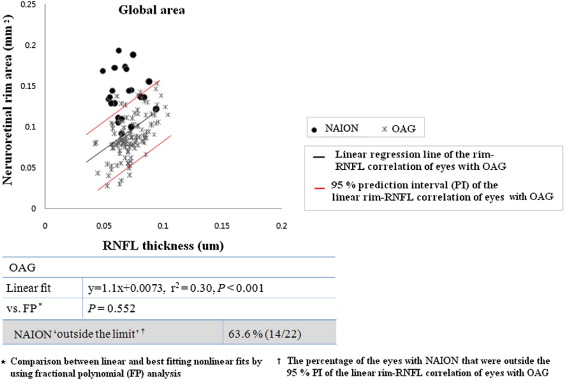
A significant linear rim-RNFL correlation was observed in global and all clock-hour sectors, except the 3-, 4-, and 9-o’clock sectors, in OAG (0.045 < r 2 < 0.64, P < .05, respectively). All eyes with NAION were outside the 95% PI of the rim-RNFL correlation of OAG in at least 1 clock-hour sector in terms of clock-hour parameters, as compared with 63.6% of eyes in terms of global parameter. All NAION eyes (n = 21) with 7- or 11-o’clock involvement had a rim-RNFL correlation outside the 95% PI of OAG for corresponding clock-hour sectors.
Conclusions
By comparison of the rim-RNFL correlation, eyes with NAION were found to be well differentiated from OAG eyes, especially in clock-hour sectors. It might be an objective approach to discriminate NAION from OAG.
Glaucomatous optic neuropathy (GON) is characterized by axonal and secondary retinal ganglion cell (RGC) loss, which results in thinning of the peripapillary retinal nerve fiber layer (RNFL) and enlargement of the optic disc cup. Because RNFL thinning is not specific for GON, it can be detected in various nonglaucomatous optic neuropathies, which are often confused with glaucoma. In this clinical situation, concomitant enlargement of the optic disc cup in proportion to RNFL thinning could provide a clue for the differential diagnosis of GON as opposed to some nonglaucomatous optic neuropathy. A small number of studies have demonstrated a disc topographic difference between GON and non-GON at a given level of RNFL loss. However, these studies did not investigate whether the correlation between the neuroretinal rim area of the optic disc and retinal nerve fiber layer thickness (rim-RNFL correlation) could be a viable criterion in terms of discriminating non-GON from GON.
Nonarteritic anterior ischemic optic neuropathy (NAION) is characterized by the sudden onset of vision loss with optic disc edema. Patients with NAION usually maintain a decreased RNFL thickness in the involved area with a relatively preserved neuroretinal rim during the chronic stage. Therefore, we considered that NAION eyes might be a good candidate to test the rim-RNFL correlation as a practical criterion for the differential diagnosis of non-GON versus GON.
Accordingly, in this study, we first analyzed the rim-RNFL correlation in global and 12-clock-hour parameters in patients with open-angle glaucoma (OAG) by using optic disc topographic value measured by Heidelberg retina tomography (HRT II) and RNFL thickness measured by Cirrus OCT. Then, we investigated whether eyes with NAION lie outside the 95% prediction interval (PI) of the rim-RNFL correlation of OAG in global and clock-hour parameters.
Method
A total of 135 patients, 113 with OAG and 22 with NAION, who visited the outpatient clinic at Seoul National University Hospital (SNUH) between April 2008 and August 2009 were enrolled. Informed consent was obtained from all subjects after providing them with an explanation of the nature and possible consequences of the study. Patients were regarded to have OAG if they had glaucomatous visual field (VF) results and RNFL defect by red-free fundus photography. A glaucomatous VF result was defined as a pattern standard deviation (PSD) outside 95% of normal confidence limits, glaucoma hemifield test results outside normal limits, and/or a cluster of at least 3 points with a P value of <.05 in the pattern deviation plot, 1 of each with P < .01 affecting the same hemifield; the cluster could not be contiguous with the blind spot and could not cross the horizontal midline on 2 consecutive Humphrey C-30-2 SITA-Standard visual field (HFA C-30-2; Carl Zeiss Meditec, Dublin, California, USA) tests. Only reliable perimetric results with less than a 20% fixation loss and fewer than 33% false-positive or false-negative results were included. Optic disc appearance was not considered to be an inclusion criterion for the OAG group to avoid selection bias in the rim-RNFL correlation.
NAION was diagnosed using the following criteria: 1) a history of sudden-onset visual loss that took place ≥4 months before study commencement, 2) a history of acute optic disc swelling and hemorrhage that had subsided at study commencement, 3) clearly delineated disc borders at study commencement, and 4) the absence of findings suggestive of arteritic AION, as described by Beck and associates, which includes systemic symptoms of giant cell arteritis, sudden marked visual loss to the level of hand motions or less, a chalky white swollen optic disc, the occlusion of one or more posterior ciliary arteries by fluorescein fundus angiography (FAG), and a high erythrocyte sedimentation rate (>50 mm/hour). Symptoms and signs of acute-phase NAION were confirmed by 1 of the authors (S.J.K.) at SNUH in 19 patients; the other 3 patients were referred from other clinics, and their symptoms and signs were reconfirmed using medical records at these referring clinics. Detailed patient interviews were also conducted to confirm sudden visual loss and other symptoms at disease onset.
The following exclusion criteria were applied: spherical refraction of greater than ±5.00 diopters (D) and astigmatism of greater than ±3.00 D, a history of ocular intervention except cataract extraction, and poor HRT II and/or Cirrus OCT image quality, as described below. For OAG patients, those with a best-corrected visual acuity (BCVA) of <20/40, an ocular or neurologic disease other than OAG, and/or a history of acute visual decrease were excluded by detailed interview about patients’ clinical history and symptoms including headache, nausea, vomiting, and acute visual decrease, and by meticulous clinical examination on ocular motility, pupillary reaction, and visual acuity, and on the optic disc. All eyes enrolled as glaucoma group did not have optic disc pallor that extends beyond the area of the cup and/or optic disc edema. For NAION patients, patients with an ocular neurologic disease other than NAION were excluded, especially those with possible evidence of glaucoma, that is, an intraocular pressure (IOP) of >21 mm Hg in either eye, a family history of glaucoma, or suspicious glaucomatous optic disc or glaucomatous visual field results in the contralateral eye. If both eyes fulfilled these inclusion and exclusion criteria, 1 eye was randomly selected. VA, refraction, and IOP by applanation tonometry were measured; anterior segments and optic discs (with a fully dilated pupil) were evaluated; and gonioscopy, Cirrus OCT, and HRT II were performed. All eyes with OAG underwent stereoscopic optic disc photography, red-free fundus photography, and HFA C-30-2. All eyes with NAION underwent stereoscopic optic disc photography and HFA C-30-2 and/or Goldmann perimetry. FAG was performed in 19 NAION patients during the acute stage, and erythrocyte sedimentation rate testing was performed in 18 patients in whom possibility of inflammatory vascular diseases or temporal arteritis could not be excluded. No patient underwent biopsy of the temporal artery.
Optical Coherence Tomography (Cirrus OCT)
RNFL thicknesses were measured by 1 technician using the Optic Disc Cube 200 × 200 program of the Cirrus HD-OCT (Model 4000; Carl Zeiss Meditec, Dublin, California, USA) after pupillary dilation to a minimum diameter of 5 mm. This program obtains a data cube of 6-mm-square grid, which was acquired as a series of 200 horizontal scan lines, each composed of 200 A-scans. For analysis, the Cirrus OCT identifies the optic disc center and automatically places a calculation circle of 3.46 mm diameter around the center. The program then extracts from the data cube 256 A-scan samples along the path of the calculation circle. Based on the delineated anterior and posterior margins of the RNFL in the extracted circle scan image, the system calculates the RNFL thickness at each point on the circle.
Satisfactory Cirrus OCT image quality was defined as an adequate signal strength (SS) ≥7 (10 = maximum) with well-focused images, as assessed by 2 experienced examiners (M.H.S., S.H.K.), who were unaware of patient information. Eyes with less-than-satisfactory Cirrus OCT image qualities were excluded.
RNFL thickness values were compared with the internal normative database of age-matched control subjects, and percentile values were calculated. Cirrus OCT software provides a color representation of RNFL thicknesses by percentiles: the top fifth percentiles as white, top 95th as green, bottom fifth as yellow, and bottom first as red, both on the RNFL thickness map and temporal, superior, nasal, inferior, temporal (TSNIT) profile ( Figure 1 , Left). The Cirrus OCT printout also provides “deviation from normal” maps, which are derived from superpixel average thickness measurements and report statistical comparisons against normative thickness values for each superpixel. These maps are overlaid on optical coherence tomography (OCT) fundus images. In these maps, age-matched normative data of superpixels whose average thicknesses fall in the 1% to 5% and <1% of normal distribution percentiles are colored yellow and red, respectively ( Figure 1 , Left).
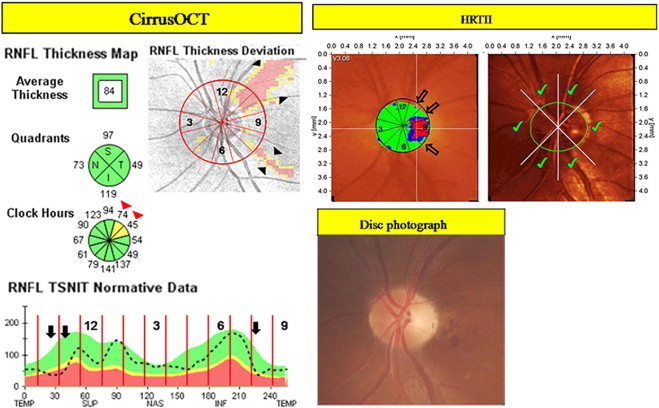
To determine “involved” clock-hour sectors on Cirrus OCT, 12 30-degree clock-hour sectors were created on “deviation from normal” maps, TSNIT profiles, and RNFL thickness maps ( Figure 1 , Left). The locations of sectors were labeled 1 through 12 starting from the superonasal sector and running clockwise in the right eye and counterclockwise in the left eye ( Figure 1 , Left). Clock-hour sectors were determined to be “involved” on Cirrus OCT when RNFLs fell to the yellow or red level on RNFL thickness maps or TSNIT profiles, or yellow- or red-colored defects radiating from the optic nerve head (ONH) of which the minimum size at a 1-disc diameter from the edge of a disc was larger than that of a major retinal vessel on the “deviation from normal” map ( Figure 1 , Left). Interpretations were performed by the 2 independent observers (S.H.K., M.H.S.), who were unaware of patient identities and other test results.
Confocal Scanning Laser Ophthalmoscopy (HRT II)
Confocal scanning laser ophthalmoscopy was performed using a Heidelberg retina tomograph (HRT) II (Heidelberg Engineering, Heidelberg, Germany) after pupillary dilation to a minimum diameter of 5 mm. Three 15-degree topographic images, centered on the optic disc, were obtained during each sitting, and these were aligned and averaged to obtain a mean topograph. Images that were not properly focused or with too much movement were excluded from further analysis, but mean topographic images of good quality (standard deviation, ≤50 μm) were included. A trained examiner (K.H.P.), unaware of patient identities and test results, outlined optic disc margins on mean images. The other 2 observers (M.H.S. and S.H.K.) reviewed ONH contours and centers on HRT II images and compared the contours with those on stereoscopic disc photographs, and compared the centers with those of the clock-face circle on the Cirrus OCT deviation map, respectively ( Figure 1 ). To reconstitute the 12-clock-hour neuroretinal rim area of optic discs, we used a user-defined segment function ( Create Segment Data ) of HRT II. The principles and applications of this function have been described elsewhere. In brief, if we defined a field segment size (degrees) limited to 0 to 90 degrees, the contour line area was subdivided into N = 360/angle sectors starting at 0 degrees (temporal side) and stereometric parameters were derived from these N sectors. We summed the 2 sectors of which the field segment size was defined as 15 degrees. For example, a rim area of the 9-o’clock clock-hour sector was the sum of 2 sectors ranging from 345 degrees to 360 degrees and from 0 degrees to 15 degrees. Global results of topographic parameters, including disc area, cup area, rim area, cup-to-disc-area ratio, cup volume, rim volume, mean cup depth, cup shape measure, and reference height were also obtained. To evaluate rim-RNFL correlations based on global area, a 360-degree global rim area value was divided by 12, so as to be in concordance with RNFL thicknesses of 30-degree clock-hour sectors for clock-hour-based analysis ( Figure 2 ).
Data Analysis
Baseline characteristics of the OAG and NAION patients were compared using the unpaired t test and Fisher exact test. To describe the rim-RNFL correlation of OAG eyes, a linear model was compared with possible best-fitting nonlinear models at each clock-hour sector and with the total average RNFL thickness parameter by fractional polynomial (FP) analysis. The concepts and applications of FP analysis have been described in detail elsewhere. In brief, FP models are defined as:
y = β 0 + β 1 x ( p 1 ) + β 2 x ( p 2 ) + … + β m x ( p m )
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


