Purpose
To establish reliable criteria for diagnosing eyelid eccrine and apocrine hidrocystomas.
Design
Retrospective clinicopathologic and immunohistochemical study.
Methods
Twenty-two specimens of normal portions of eyelids were evaluated to establish the distribution and microanatomy of eccrine and apocrine glands. Immunostaining for cytokeratin 7 (CK7), gross cystic disease fluid protein-15 (GCDFP-15), alpha-smooth muscle actin (α-SMA), epithelial membrane antigen (EMA), and carcinoembryonic antigen (CEA) was performed on these tissues and on 40 lesions in 33 patients diagnosed with eccrine or apocrine hidrocystomas by unaided light microscopy.
Results
Eccrine glands were not present in the eyelid margins, the lower half of the upper eyelid pretarsal skin, or the pretarsal lower eyelid skin. Apocrine glands were restricted to the eyelid margins and canthi where the cysts were located. GCDFP-15, CK7, and α-SMA immunoreacted with the eccrine secretory coils but not their ducts; apocrine gland secretory spirals also stained positively for these markers throughout their extended courses, but not their short terminal ducts. Positivity was found in 37 of 40 hidrocystomas for α-SMA and 19 for GCDFP-15; lesions tested for CK7 displayed positivity.
Conclusions
Alpha-SMA-, CK7-, and/or GCDFP-15-positive apocrine hidrocystomas were the only type discovered in this series and arose from glandular secretory spirals within the marginal, perimarginal, or canthal skin. Three lesions did not stain for α-SMA, initially suggesting an absent myoepithelium and therefore an eccrine ductal origin; they manifested CK7 positivity, however, another characteristic of the apocrine secretory spiral but not ducts. Our findings disprove the contention that eccrine predominate over apocrine hidrocystomas in the eyelids.
After chalazia, the most common benign lesions of the eyelids are papillomas, seborrheic keratoses, melanocytic nevi, and cysts, the last encompassing epidermoid, hybrid, and Meibomian keratinous cysts, as well as ectasias of the ducts of the eccrine and apocrine glands (hidrocystomas) and mildly proliferative ectasias of the follicular infundibulum (dilated pore of Winer). Histopathologically, hidrocystomas exhibiting a deeply eosinophilic cuboidal to columnar cellular lining with oval to round basal nuclei and apical cytoplasmic decapitations (shedding of adlumenal snouts, which are incorporated into the eosinophilic secretions along with specific granules) have conventionally been designated as apocrine. In contrast, cysts lined by a low cuboidal or flattened epithelium with paler cytoplasm, no snouts, and a watery secretion without cytoplasmic contributions have been classified as eccrine. All hidrocystomas are bereft of goblet cells, which separates them from conjunctival cysts. Eccrine hidrocystomas have long been regarded as retention cysts and believed to be more common in the eyelids than apocrine cysts. The latter, by contrast, are believed to be proliferative in nature, most conspicuously when papillary formations or a solid adenoma within a stalk are found projecting from the wall into the lumen in a subset of lesions (cystadenomas).
Some dermatopathologists have totally forsaken making distinctions between supposed eccrine and apocrine hidrocystomas, positing that secondary or degenerative changes within the cysts’ linings can efface any meaningful cytologic differences. The more neutral diagnostic term “ductal hidrocystoma” has instead been recommended. It has furthermore been suggested that most if not all such cysts are truly apocrine with variably induced secondary cytologic modifications. With regard to these issues, there is no comparative study of eccrine and apocrine hidrocystomas of the eyelids that has critically assessed the validity of the standard clinicopathologic criteria that are employed in their diagnosis. This lacuna, which the present study addresses, is more urgent in view of numerous contradictions regarding hidrocystomas contained in the ophthalmic literature.
It is remarkable that the fundamental anatomic-distributional differences between apocrine and eccrine glands in the eyelids are beclouded in the minds of many ophthalmologists. Unfamiliarity also persists regarding their microscopic differences and the sources of hidrocystomas (secretory vs ductular regions). Preoperative clinical diagnostic accuracy would be considerably enhanced (particularly with reference to early basal cell carcinoma and melanoma when the cyst is pigmented) if valid differences in the locations of putative eccrine and apocrine eyelid hidrocystomas and their differential growth characteristics were discovered. At the same time, their pathologic diagnosis would have a sounder foundation if consistent immunohistochemical differences were elucidated. The accurate diagnosis of multiple periocular hidrocystomas as an ophthalmic emblem of certain systemic metabolic and rare inherited cutaneous and musculoskeletal disorders would also be facilitated. Such developments could represent a major advance since the Dutch ophthalmologist Jacob Moll (1832–1914), over 100 years ago, first described the apocrine glands of the eyelids that are named after him. Our findings confirm the primacy of Moll glands rather than eccrine glands in generating most eyelid hidrocystomas in contravention of contemporary orthodoxy.
Methods
To evaluate the distribution and normal microscopic features of eccrine and apocrine glands of the eyelid skin, hematoxylin-eosin–stained paraffin-embedded perpendicular sections of normal skin at the borders of lesions in 7 full-thickness eyelid excisions; longitudinal sections of 8 preseptal blepharoplasty skin specimens excised parallel to the eyelid margins during cosmetic surgery; and 6 exenteration specimens that included the eyelids were selected from the files of the David G. Cogan Laboratory of Ophthalmic Pathology at the Massachusetts Eye and Ear Infirmary. The eyelid resections had been performed mostly for basal cell carcinomas and occasional chalazia with undisturbed anatomic features at their medial and lateral surgical margins, and the exenterations had been undertaken for malignancies that had involved the orbital contents but had left the eyelid architecture intact. In addition, 17 vertical (sagittal) step-sections stained with Masson’s trichrome of 1 example of normal exenterated orbital contents with preserved eyelids (specimen obtained postmortem) were retrieved from the Malcolm Carpenter Teaching File Collection bequeathed to the Infirmary. All specimens were thoroughly examined with respect to any notable distributional differences of the apocrine and eccrine glands in the upper and lower eyelids.
Forty pathologic specimens obtained from 33 patients were culled from the slide files that had been diagnosed microscopically as eccrine hidrocystoma, apocrine hidrocystoma, sudoriferous cyst, ductal cyst, or adnexal cyst of the eyelids or medial and lateral canthi. The slides of the lesions were critically reviewed and rediagnosed in hematoxylin-eosin–stained sections. Cases were included in the study if 50% or more of the cystic epithelial lining could be identified, and if sufficient tissue remained in the paraffin blocks to permit deeper sections for additional immunohistochemical staining. All tissue preparations were performed in the laboratories of the Pathology Department at the Massachusetts General Hospital, Boston, Massachusetts. Well-standardized special staining included periodic acid–Schiff (PAS) with and without diastase, Perl’s reaction for cytoplasmic iron, and Masson’s trichrome. A panel of immunohistochemical probes ( Table 1 ) was employed according to methods described elsewhere: cytokeratin 7 (CK7), epithelial membrane antigen (EMA), carcinoembryonic antigen (CEA), gross cystic disease fluid protein-15 (GCDFP-15), and alpha-smooth muscle actin (α-SMA). Sections of both the normal eyelid tissues and the cysts’ epithelial linings were stained with the same probes and the immunohistochemical results were compared.
| Probe | Antigen | Cell Specificities | Staining Pattern | Source a | Dilution |
|---|---|---|---|---|---|
| Gross cystic disease fluid protein-15 (GCDFP-15) | 15 kDa glycoprotein from breast cystic fluid | Breast tissue and tumors with apocrine features; apocrine, ceruminous, Montgomery and Moll glands; secretory zones of cutaneous eccrine and apocrine glands; serous cells of acini of lacrimal, submandibular, sublingual, and minor salivary glands; serous cells of nasal and bronchial glands; apocrine tumors and cysts; Paget’s disease, prostate Ca; absent in ducts | Cytoplasmic (Golgi zone) and membranous (lumenal cuticles) | Mouse monoclonal IgG2α | 1:10 |
| Alpha-smooth muscle actin (α-SMA) | 42–45 kDa monomer polymerizes to form cytoskeletal protein subserving motility | Smooth muscle cells, myoepithelial cells, myofibroblasts, leiomyoma, leiomyosarcoma | Cytoplasmic | Mouse monoclonal IgG/κ | Prediluted |
| Cytokeratin 7 (CK7) | 54 kDa intermediate cytoskeletal protein filament subserving cellular stabilization and conformation | Epithelia of large number of simple glands; lung and breast tissue; complex and transitional (urothelial) epithelia; mesothelia; diseased but not normal corneal endothelium; secretory portions of apocrine and eccrine glands but not ducts | Cytoplasmic | Mouse monoclonal IgG1/κ | Prediluted |
| Epithelial membrane antigen (EMA) | 75 kDa transmembrane glycoprotein related to breast mucin | Glandular epithelial and mesothelial cells (epithelial differentiation supplemented by cytokeratins); breast and thyroid papillary carcinomas; cuticles of apocrine and eccrine secretory coils and ducts; plasma cells, perineural fibroblasts, notochord, chordoma, meningioma, epithelial mesothelioma; not present in hepatocellular carcinoma | Membranous | Mouse monoclonal IgG2α/κ | Prediluted |
| Carcinoembryonic antigen (CEA) | 180 kDa glycoprotein of glycocalyx | Epithelial cells of endodermal derivation or differentiation (lung, stomach, pancreas, biliary system and liver, colon); cuticles of apocrine and eccrine gland ducts; breast carcinoma | Membranous | Mouse monoclonal IgG1 | Prediluted |
a All monoclonals supplied by Ventana Medical Systems, Oro Valley, Arizona, USA.
The patients’ clinical records, including line diagrams and any clinical photographs of the lesions, were closely scrutinized to assemble the following data: age and sex; duration of any symptoms created by the lesions; unilaterality, bilaterality, and multiplicity; size, color, and any other distinguishing features; upper or lower (or both) eyelid distribution; and specific topographic location in the eyelids. Regarding the last feature, the following designations for the topography of the eyelid skin were adopted: marginal if the lesion’s epicenter involved the region of the mucocutaneous junction and gray line; perimarginal or pericilial if the lesion involved the skin adjacent to the margin, including where the cilia emerged and slightly beyond; pretarsal for the skin in front of the tarsus commencing unambiguously above (in the upper eyelid) or below (in the lower eyelid) the lashes; preseptal (supratarsal or infratarsal depending on upper or lower eyelid) or preorbital for the skin extending from the poles of the superior or inferior tarsi to the brow or inferior orbital rim, respectively; and canthal for the roughly oval portion of skin overlying the medial and lateral canthal tendons beyond the lateral and medial commissures up to the orbital rim laterally and the base of the nasal pyramid medially. The pretarsal and preseptal regions of the lower eyelid skin were less clearly separable from each other clinically than in the upper eyelid because of the smaller size of the inferior tarsus, but microscopically the distinction could be readily made.
Results
Normal Eyelid Eccrine and Apocrine Glands
Histopathologic findings
Eccrine glands were not present at the eyelid margin or in the perimarginal eyelid skin near the cilia ( Figure 1 , Top left and right); they first appeared in the pretarsal skin in small numbers at or above the midpoint of the tarsus of the upper eyelid and at the inferior pole or below the lower eyelid tarsus. Eccrine glands were much more profuse in the preseptal (preorbital) skin ( Figure 1 , Middle left). Conversely, apocrine glands of Moll were discovered only at the eyelid margin in association with the strong cilial hairs (eyelashes) of the upper and lower eyelids ( Figure 1 , Middle right) and nowhere else in the eyelid skin. No normal canthal tissues were found for inclusion in this study; uninvolved skin surrounding excised canthal cysts, however, sometimes displayed an adjacent apocrine gland, but no eccrine glands were seen. In terms of their microanatomy, eccrine and apocrine glands were strikingly different. Eccrine glands were located in the mid portion of the thin reticular dermis of the eyelid skin. (Elsewhere in the integument, they are located at the interface between the lowest portion of the reticular dermis and the subcutaneous fat or hypodermis, which is nonexistent or exiguous in the pretarsal eyelid skin.) The deepest portion of the eccrine gland, referred to as the secretory coil (a convoluted tube), appeared as a loosely organized lobular structure (smaller than elsewhere in the skin); it was invested with its own delicate fibrous adventitia containing vessels and nerves ( Figure 1 , Bottom left).
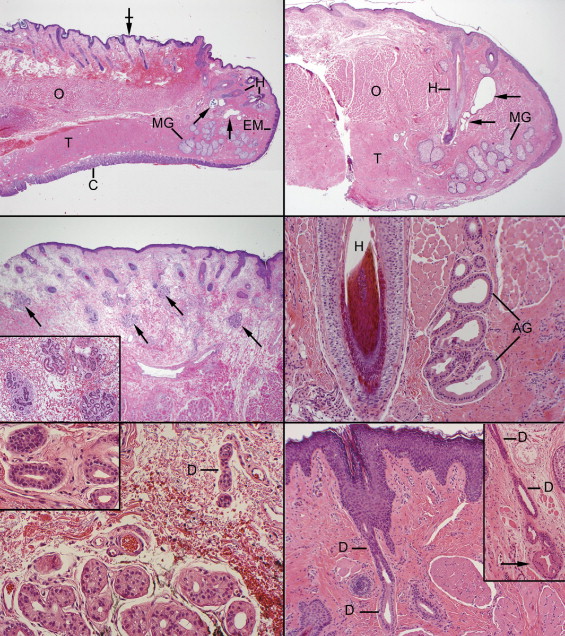
Emerging from the secretory coil after originating abruptly within its lobule, the duct (or syrinx) formed a convoluted cluster immediately next to the secretory coil ( Figure 1 , Bottom left, inset), then straightened out to vertically traverse the dermis, emptying onto the epidermis via a modified terminal intraepidermal spiral segment with a surface pore referred to as the acrosyringium. The secretory cells of the coil displayed a moderately eosinophilic cytoplasm; the constituent acini had small lumens delimited by an inner cuboidal layer and an outer, more flattened layer. Pale cells (glycogen-rich) and dark cells (endowed with scattered secretory granules that are discharged via merocrine secretion) were less readily distinguishable in the eyelids than elsewhere in the skin. Some of the inner cells had a faintly fibrillated cytoplasm attributable to overlapping lateral cell membranes that formed complicated imbrications, creating intercellular canaliculi. The ducts appeared darker because their cells had less cytoplasm. The inner adlumenal ductal cells were surrounded by an outer layer; a somewhat refractile cuticle was detectable around the lumen (attributable to microvilli and an electron microscopically demonstrable subplasmalemmal condensation of tonofilaments).
In contrast, the apocrine glands of Moll commenced deep at the eyelid margin at the level of the germinative bulbs of the cilia and near the anterior face of the tarsus. Rather than assuming a tightly coiled arrangement with a relatively long and straight duct, the secretory components appeared more like an open vertical spiral with patulous lumens that evinced variegated secretory features throughout their upward trajectory to the surface ( Figure 1 , Middle right). The adlumenal secretory cells frequently sported an apical snout of decapitation (apocrine secretion). Segments of the secretory coil were also composed of low cuboidal cells, probably indicative of a different state of metabolic and secretory activity. A distinctly separate ductal differentiation ( Figure 1 , Bottom right) was discerned only in a very short, modified segment of the most terminal region near the surface (a shorter segment than encountered in apocrine glands located elsewhere in the integument). The secretions emptied into the follicles of the cilia at approximately a right angle immediately below the level of the infundibulum through a straight apocrine acrosyringium. The secretory cells of the uppermost secretory spiral gradually converted to low cuboidal ductal cells with less eosinophilic cytoplasm; an outer layer was inconspicuous in the terminal duct and acrosyringium.
Immunohistochemical findings
The salient normal glandular reaction patterns and, in comparison, the hidrocystomas contained in this series are summarized in Table 2 . The eccrine secretory coils or lobules immunostained positively for CK7 and GCDFP-15 ( Figure 2 , Top left and inset) in the cytoplasm of the acinar cells; a thin cuticle on the apical pole or lumenal cell membrane was also observed. EMA and CEA both immunostained the cell membranes participating in the intercellular canaliculi as well as an apical adlumenal cuticle of the ducts ( Figure 2 , Top right, upper right inset); the intensities of the EMA and CEA reactions were variable from specimen to specimen. Alpha-SMA immunoreacted with the outer myoepithelial cells of the acinar secretory units ( Figure 2 , Top right, main panel and inset upper left) but not with the outer cells of the intralobular or ascending extralobular ducts ( Figure 2 , Top right, inset upper left and middle left). The eccrine ducts beginning in the secretory coils failed to express CK7, GCDFP-15, and especially α-SMA cytoplasmic reactivity; these negative staining results persisted throughout the ducts’ upward ascent in the dermis ( Figure 2 , Middle right). The negative α-SMA staining denoted the absence of a myoepithelial outer layer. EMA, CEA, and CK7 delineated a thin or faint lumenal ductal cuticle ( Figure 2 , Upper right, inset bottom right), which in the upper reaches of the ducts’ projection toward the acrosyringium became erratic, fainter, and frequently totally disappeared. No intercellular canaliculi were detected between the duct cells.
| Initial Pathologic Diagnoses | Diagnoses After Critical Review | |
|---|---|---|
| Eccrine hidrocystoma | 6 | 13 |
| Apocrine hidrocystoma | 21 | 27 |
| Ductal cyst | 5 | 0 |
| Hidrocystoma | 1 | 0 |
| Sudoriferous cyst | 7 | 0 |
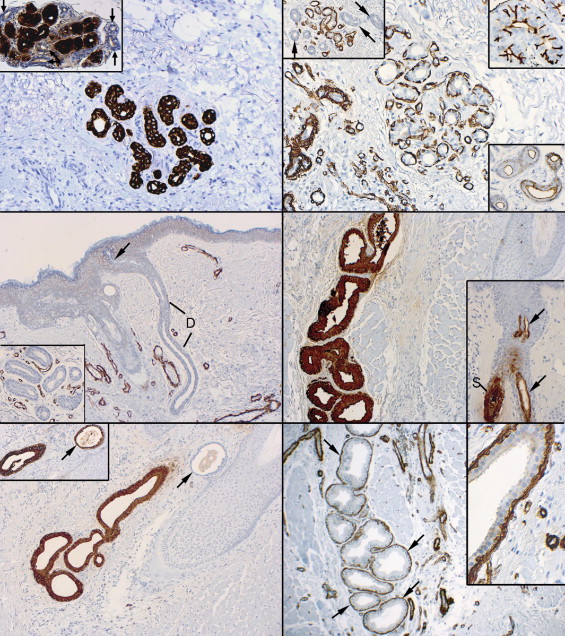
In the apocrine secretory spirals, CK7 and GCDFP-15 ( Figure 2 , Middle right, and Bottom left) diffusely stained the cytoplasm, the lumenal cuticle, and any lumenal secretory material. GCDFP-15 cytoplasmic positivity disappeared and only an episodic lumenal cuticle persisted as the uppermost spiral underwent a transition into the short terminal duct ( Figure 2 , Middle right, inset). CK7 immunostaining remained when GCFDP-15 positivity had disappeared during ductalization ( Figure 2 , Bottom left and inset). Anti-α-SMA antibodies demonstrated an outer, positively staining myoepithelial layer along the full length of the secretory spiral ( Figure 2 , Bottom right and inset); it also began to disappear just before the short terminal duct disgorged the secretions into the follicle ( Figure 3 , Top left and right). EMA lightly stained the cytoplasm of the cells of the secretory spiral and highlighted a dense lumenal cuticle, which became fainter in the upper reaches of the spiral. CEA staining was observed only as a thin lumenal cuticle in the secretory spiral and paralleled the staining distributions of EMA; it also positively stained any intralumenal secretory material. Neither EMA nor CEA revealed the presence of intercellular canaliculi in the secretory spiral. CK7 immunoreacted with the cells’ cytoplasm throughout the full stretch of the secretory spiral ( Figure 2 , Bottom left), but became fainter and eventually disappeared, transforming into a cuticle in the uppermost region of the terminal duct.
Hidrocystomas
Clinical findings
Thirty-three patients had 40 cysts. There were 17 men and 16 women with ages at presentation ranging from 42 to 90 years (mean of 64 years). Patients had been aware of their lesions usually for several months and always for less than a year. The lesions in this series involved the eyelid margins ( Figure 3 , Middle left), perimarginal and pericilial skin ( Figure 3 , Middle right, and Bottom left), or canthal regions ( Figure 3 , Bottom right panels). No lesions were discovered in the pretarsal or preseptal eyelid skin. Very lateral or medial lesions of the eyelids were distinguished from those of the canthus if they impinged on the eyelid margin. Three patients had bilateral lesions. The largest number of lesions in a single patient was 4, which occurred bilaterally in 3 eyelids. Ten of 40 lesions involved the upper eyelids and 18 the lower; 3 were located near the inferior canaliculus and required a stent to be placed during excision. When clearly mentioned among the perimarginal lesions, 9 occurred in the medial third of the lid, 5 in the lateral third, and 2 were paracentral (in the middle third). Two medial and 5 lateral lesions were situated in the canthal regions as determined after careful verification of clinical diagrams or photographs. Five other lesions were initially designated as medial (3) or lateral (2) canthal without convincing documentation as to whether they were actually situated in the outer or inner extremities of the eyelids. Our considered opinion is that they probably occurred in the innermost or outermost boundaries of the eyelids, but we have treated them as being of indeterminate location. No significant differences in the patients’ ages, sex, or symptoms were observed in the group of canthal lesions. The cysts had a smooth surface with sharp, nonelevated, and regular margins and evinced a clear or skin-colored appearance in most instances; they were uniloculated or multiloculated, and 4 were either milky (2) or bluish (2) ( Figure 3 , Bottom left). Judging from the uneven quality and thoroughness of the preoperative clinical notes, we believe a fuller description would have resulted in a greater number of discolored cysts. Sizes ranged from 1 to 5 mm in diameter, with most in the 1- to 3-mm range. No patient complained of a foreign body sensation or ocular hyperemia. The lesions were all surgically excised, often incompletely, and no recurrences were reported in the patients’ records on follow-up.
Histopathologic findings
Table 3 summarizes the initial and revised pathologic diagnoses made from hematoxylin-eosin–stained sections alone of the 40 cysts included in this study. All lesions failed to display goblet cells. Thirteen lesions were ultimately diagnosed by us as eccrine hidrocystomas because of a unicameral architecture and because their linings were composed of a nonkeratinizing, low cuboidal epithelium with a comparatively pale-staining eosinophilic cytoplasm lacking apical snouts ( Figure 4 , Top left and right); an inconspicuous flattened outer layer enveloped the adlumenal cells. In many specimens, focal or spotty snouts of the inner cells had been previously overlooked ( Figure 4 , Top left, bottom right inset). Most of the lesions appeared collapsed and thrown into folds because of release of the cysts’ fluid contents during surgical removal. No traces of an eosinophilic secretory material were discovered in the lumens of these cysts. Sometimes the inner cells were also highly compressed, presumably from intracystic pressure. A thin compact circumferential fibrous wall was distinguishable from the surrounding dermis; the latter was looser and more haphazard in the arrangement of its collagen fibers.
Twenty-five lesions were rediagnosed as apocrine cysts ( Figure 4 , Middle left) because they were multilocular (10 lesions) or exhibited a diffuse or focal lining composed of an inner layer of high cuboidal to columnar cells with glassy, opaque, or deeply eosinophilic cytoplasm manifesting at least spotty apical decapitation snouts ( Figure 4 , Middle right); there was a variably distinct outer layer composed of more flattened cells. An artifact encountered focally in 19 lesions was sloughing of the inner cells into the cysts’ lumen while the basilar cells remained attached to the fibrous capsule ( Figure 4 , Middle right). Other features favoring an apocrine diagnosis were an eosinophilic lumenal content occasionally organized into globoid bodies ( Figure 4 , Middle left, bottom left inset) (22 lesions); the discovery of adjacent, preexistent apocrine glandular epithelial units ( Figure 4 , Middle left, bottom right inset) in intimate relationship to some region of the cyst (22 lesions); or a comparatively thick, circumferentially oriented, intrinsic fibrous wall that was highlighted by Masson’s trichrome stain, which distinguished it from the surrounding looser reticular dermis (7 lesions). Papillations or focal cribriform cellular pileups or increscences of the epithelial lining were not observed in any of the lesions. Staining for cytoplasmic iron was negative in 5 tested lesions (Perl’s method); PAS staining erratically revealed small positive, diastase-resistant cytoplasmic granules in the cytoplasm of 8 tested lesions.
Immunohistochemical findings
All but 3 of the 40 cysts had an outer layer of cells that at least focally stained positively for α-SMA ( Table 2 ). The most dramatic pattern ( Figure 4 , Bottom left) was that of a virtually uninterrupted layer (13 lesions), but negative regions alternating with positive ones ( Figure 4 , Bottom right) was a more common pattern (19 lesions). The most characteristic morphology was a thin outer cellular layer displaying a straight line ( Figure 5 , Top left) or even thread-like positivity indicative of myoepithelial compression and probably some degree of accompanying atrophy ( Figure 5 , Top left, inset). In some instances only a few adlumenal cells were underlain by short linear stretches of positively immunoreacting cells (5 lesions) ( Figure 5 , Top right and bottom left inset); adjacent apocrine glandular units showed more evidence of basal cell α-SMA reactivity in 3 of these cases. Staining of occasional myofibroblasts in the fibrous wall beyond the epithelial lining had to be distinguished from the positive staining of the intrinsic myoepithelium ( Figure 5 , Top right, bottom right inset). The latter often extended small processes between the basal regions of adjacent adlumenal cells, whereas a thin mantle of collagen was interposed between the epithelium and the myofibroblasts.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


